Abstract
The aim of this study was to report the technical success, adverse events, clinical outcomes, and long-term stent patency of iliocaval stent reconstruction for naïve, non-inferior vena cava (IVC) filter-related, chronic iliocaval thrombosis. A total of 69 patients, including 47 (68%) men, with a mean age of 36 years (range: 8–71 years), underwent first-time iliocaval stent reconstruction for non-IVC filter-associated iliocaval thrombosis. The mean number of prothrombotic risk factors was 2.2 (range: 0–5), including 30 (43%) patients with IVC atresia. Upon initial presentation, the Clinical, Etiology, Anatomy, and Pathophysiology (CEAP) classification was C3 in 55 (80%) patients, C4 in four (5.8%) patients, C5 in one (1.4%) patient, and C6 in seven (10%) patients. Technical aspects of stent reconstruction, technical success, adverse events, 2-week and 6, 12, and 24-month clinical response, and 6, 12, and 24-month primary, primary-assisted, and secondary stent patency rates were recorded. Technical success was defined as recanalization and stent deployment. Adverse events were reported according to the Society of Interventional Radiology classification system. Clinical success was defined as a 1-point decrease in CEAP classification and stent patency was defined by the Cardiovascular and Interventional Radiological Society guidelines. The technical success rate was 100%. There were 352 venous stents deployed during stent reconstructions. One (1.4%) severe, four (5.8%) moderate, and four (5.8%) minor adverse events occurred and median post-procedure hospitalization was 1 day (range: 1–45 days). Clinical success at 2 weeks and 6, 12, and 24 months was 76%, 85%, 87%, and 100%, respectively. The estimated 6, 12, and 24-month primary patency rates were 91%, 88%, and 62%, respectively. The estimated 6, 12, and 24-month primary-assisted patency rates were 98%, 95%, and 81%, respectively. The estimated 6, 12, and 24-month secondary-assisted patency rates were all 100%. In conclusion, iliocaval stent reconstruction is an effective treatment for non-IVC filter-associated chronic iliocaval thrombosis with high rates of technical success, clinical responses, and stent patency.
Keywords
Introduction
Thrombosis of the inferior vena cava (IVC) is a cause of significant patient morbidity, as it is a major contributing factor to deep venous thrombosis (DVT) and chronic venous insufficiency.1,2 Patients with IVC stenosis or occlusion present with clinical symptoms of pain, varicoceles, and abdominal and pelvic collaterals as well as lower extremity varices, ulcers, and phlegmasia.2,3
Endovascular stent reconstruction is the standard of care for the treatment of chronic iliocaval thrombosis, with favorable technical success rates, clinical outcomes, and stent patency rates, 3 though reported series have focused on IVC filter-associated thrombosis 4 and primary iliofemoral thrombosis without coexisting IVC thrombosis.5–7 Several prior series have employed stent reconstruction for treatment of IVC thrombosis with good results.8,9
The purpose of this report was to describe the technical success, clinical outcomes, adverse events, and stent patency of iliocaval stent reconstruction for non-IVC filter-related chronic iliocaval lesions.
Materials and methods
Patient identification and selection
This study was conducted with Institutional Review Board approval (HUM00107111, HUM00120211, and 18-3199) and complied with the Health Insurance Portability and Accountability Act. Informed consent was not required for this retrospective study, which followed the Strengthening the Reporting of Observational Studies (STROBE) statement in its reporting.
A retrospective search of electronic medical records was performed from April 1999 to October 2016 (186 months) using the electronic medical record search engine EMERSE (Ann Arbor, MI, USA) (and manual review) with the search terms ‘iliocaval reconstruction,’ ‘iliocaval recanalization,’ ‘iliocaval occlusion,’ ‘iliocaval thrombosis,’ ‘venous reconstruction,’ ‘iliocaval stenting,’ ‘inferior vena cava stent,’ and ‘iliac vein stent.’ 10 This yielded 1154 patients.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Inclusion and exclusion criteria
A total of 1154 patients were identified as meeting the initial search criteria and additional manual review that were performed. A total of 69 (6.0%) patients were identified that met the inclusion criteria of having undergone naïve (first-time) recanalization and stent reconstruction for chronic or acute-on-chronic non-IVC filter-associated iliocaval occlusion. Patients underwent venous recanalization and stent reconstruction if they had symptoms referable to a venous occlusion, which was confirmed on noninvasive or invasive venography, were medically stable for recanalization attempts, and did not have an uncorrectable coagulopathy.
Patient demographic data
Demographics are shown in Table 1. There were 69 patients who met the inclusion criteria: 47/69 (68%) patients were men; the mean age was 36.4 years (range: 8–71 years); 58/69 (84%) were Caucasian, seven (10%) were African American, two (2.8%) were of mixed or unknown ethnicity, one (1.4%) was Asian, and one (1.4%) was Middle-Eastern.
Patient demographics, including prothrombotic risk factors and description and acuity of presenting clinical symptoms.
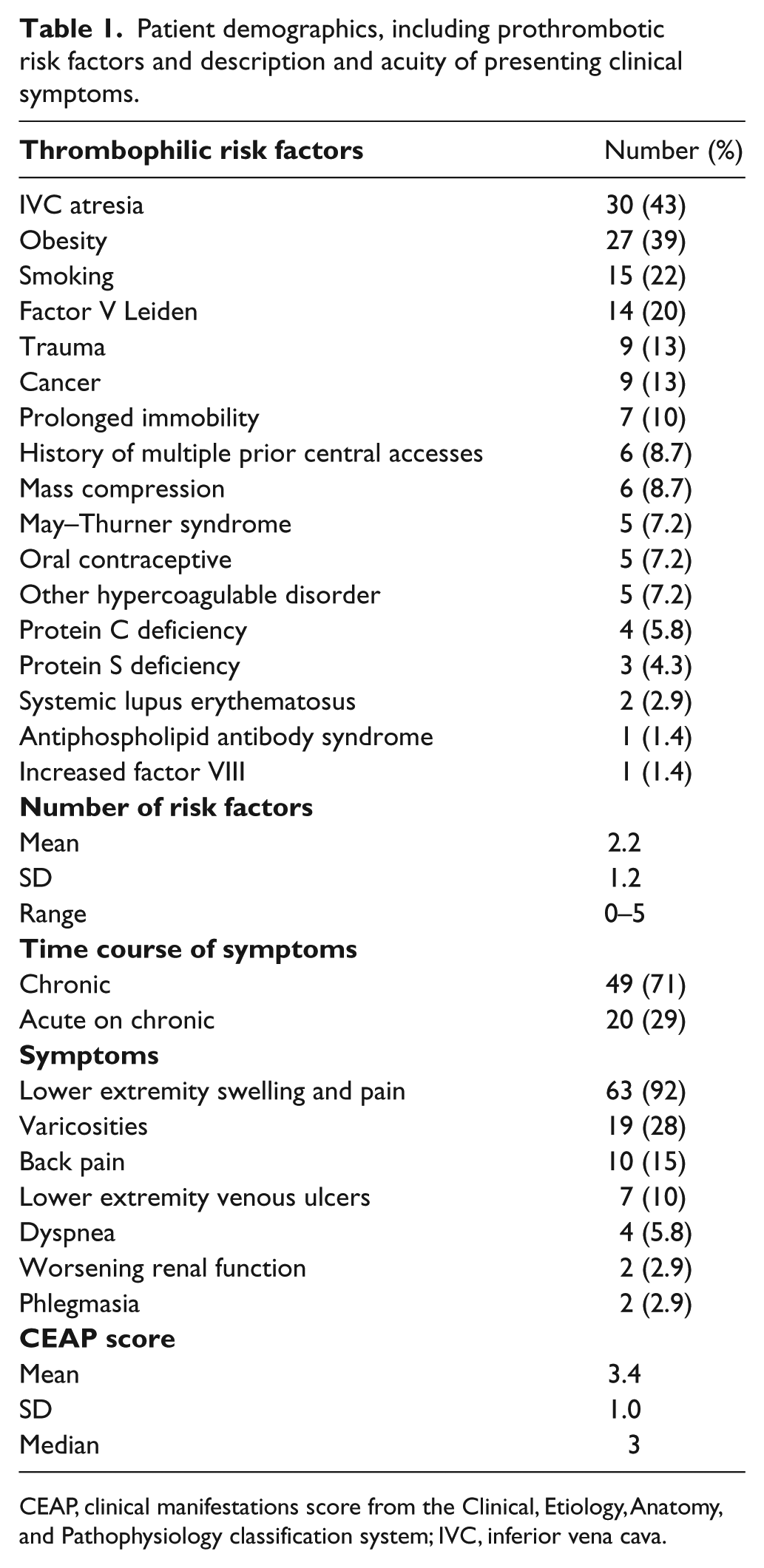
CEAP, clinical manifestations score from the Clinical, Etiology, Anatomy, and Pathophysiology classification system; IVC, inferior vena cava.
The mean number of prothrombotic risk factors was 2.2 ± 1.2 (range: 0–5), the most common of which were congenital atresia of the IVC (n = 30; 43%) and obesity (n = 27; 39%). At the time of presentation, 43 (62%) patients were chronically anticoagulated.
The most common presenting symptom was lower extremity edema and pain (n = 63; 91%), with less common presenting symptoms including varicosities (n = 19; 28%) and back pain (n = 10; 15%). Clinical symptoms were classified in accordance with the clinical manifestations score from the Clinical, Etiology, Anatomy, and Pathophysiology (CEAP) classification system. 11 The distribution of CEAP scores was: C1 (n = 1; 1.4%), C2 (n = 1; 1.4%), C3 (n = 55; 80%), C4 (n = 4; 5.8%), C5 (n = 1; 1.4%), and C6 (n = 7; 10%).
The time course of presenting symptoms included chronic (> 4 weeks) (n = 49; 71%) and acute worsening of chronic symptoms (n = 20; 29%).
Pre-procedural imaging included venous duplex ultrasound (n = 44; 64%), computed tomography venography (n = 41; 59%), magnetic resonance venography (n = 16; 23%), and conventional catheter-based venography (n = 5; 7.2%) to confirm the diagnosis of iliocaval occlusion and for planning of venous recanalization and stent reconstruction. Some patients had multiple imaging modalities employed.
Procedural technique
All patients were seen in consultation by an attending interventional radiologist in the clinic or hospital prior to the procedure. Most venous thrombolysis procedures were performed using moderate sedation with intravenous midazolam (Novell Pharmaceutical Laboratories; Jakarta Barat, Indonesia) and fentanyl (Janssen Pharmaceuticals; Beerse, Belgium); most venous recanalization and reconstruction procedures were performed under general anesthesia with endotracheal intubation. Ultrasound-guided access was achieved, and catheter-based venography was performed to document the sites of venous occlusion.
Catheter-directed thrombolysis was performed in 20 (29%) patients, as described previously,5,12,13 without or with mechanical and pharmacomechanical techniques, based on the Society of Interventional Radiology’s ‘quality improvement guidelines for the treatment of lower-extremity DVT,’ 14 the extent of thrombus, and operator preference.
Iliocaval recanalization and stent reconstruction was then performed as described previously.2–4,12,15–18 Representative clinical cases are shown in Figures 1, 2, and 3. Briefly, venous access was obtained. Blunt recanalization with a vertebral-tip catheter (Terumo; Tokyo, Japan) and a straight, stiff tip, Terumo Glidewire was initially attempted in all patients. In cases in which blunt recanalization was unsuccessful, sharp recanalization with an 18-gauge BRK transseptal needle (St Jude Medical; St Paul, MN, USA) and a loop snare was performed. Once occlusions were crossed, exchange-length wires were placed to establish through-and-through venous access. Subsequently, angioplasty with a 4-mm balloon was performed and contrast venography without or with intravascular ultrasound was performed to evaluate wire placement and presence of extravasation. If extravasation was noted, a 3-minute prolonged balloon inflation was performed followed by repeat intravascular ultrasound and contrast venography evaluation. After the possibility of extravascular recanalization and extravasation had been excluded, patients were heparinized, with a targeted activated clotting time > 250 seconds, and sequential angioplasty and stent reconstruction was performed to a goal diameter depending on the age of the patient and the specific vein in order to maintain patency and establish adequate flow. In general, goal diameters were 18-22 mm in the IVC, 16 mm in the common iliac veins, and 14 mm in the external iliac or common femoral veins.
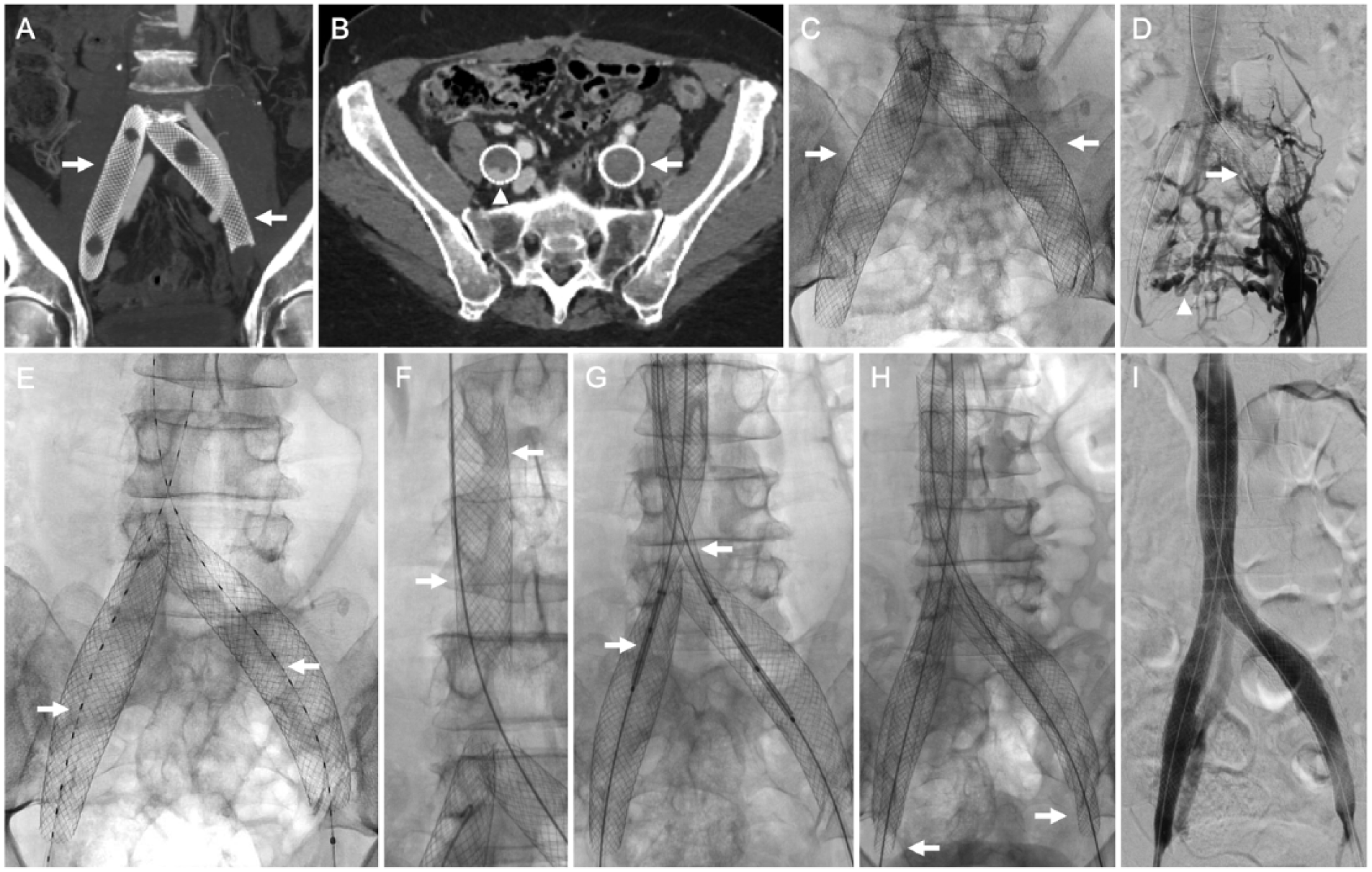
A 68-year-old-female with prior bilateral iliac venous stent-associated iliocaval occlusion. (A) Coronal computed tomography of the pelvis with contrast showing oversized bilateral iliac venous stents (arrows) placed at another institution. (B) Axial computed tomography with venographic phase imaging demonstrating complete occlusion of the left iliac venous stent (arrow). There was partial flow through the dependent portion of the right iliac venous stent (arrowhead). (C) Fluoroscopic image showing the malpositioned and oversized iliac venous stents (arrows). (D) Ascending venography, performed from a left groin approach, showing complete left iliac venous stent occlusion (arrow). Multiple cross-pelvic venous collaterals are seen (arrowhead). (E) Bilateral iliocaval lysis was initiated (arrows) with tissue plasminogen activator administered at 1 mg/hr. (F) Following overnight lysis, a 22-mm Wallstent was placed in the infrarenal IVC (arrows). (G) ‘Kissing’ 18-mm Wallstents were deployed in a ‘double barrel’ fashion (arrows) extending from the IVC into the common iliac veins. (H) Afterwards, 16-mm Wallstents were extended into the bilateral external iliac veins (arrows). (I) Completion bilateral iliocaval venography showing brisk in-line flow from both common femoral veins to the IVC.
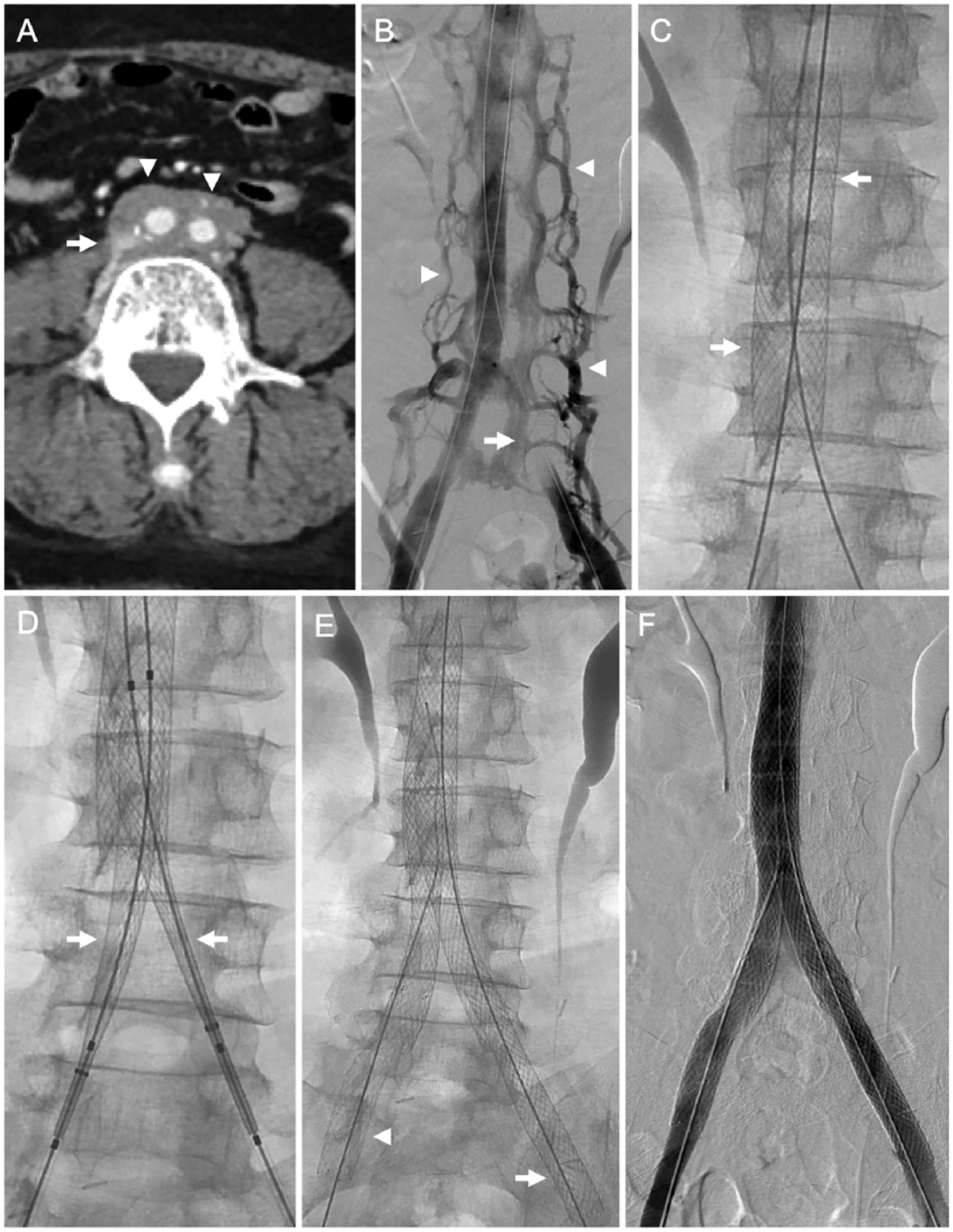
A 67-year-old female with retroperitoneal fibrosis-associated iliocaval occlusion. (A) Axial computed tomography of the pelvis after contrast administration showing complete obliteration of the IVC (arrow) by the circumferential soft tissue (arrowheads) of retroperitoneal fibrosis. (B) Ascending venography, performed by contrast injection from both groins, showing occlusion of the left iliac venous system (arrow). Multiple ascending lumbar venous collaterals are visualized (arrowheads). (C) Venous stent reconstruction was performed with a 22-mm Wallstent (arrows) placed in the IVC. (D) ‘Kissing’ 16-mm Wallstents were deployed in a ‘double barrel’ fashion (arrows) extending from the IVC into the common iliac veins. (E) A 14-mm Wallstent (arrow) was extended into the left external iliac vein and a 12-mm Protégé stent was extended into the right external iliac vein (arrowhead). (F) Completion bilateral iliocaval venography demonstrating brisk in-line flow from both common femoral veins to the IVC.
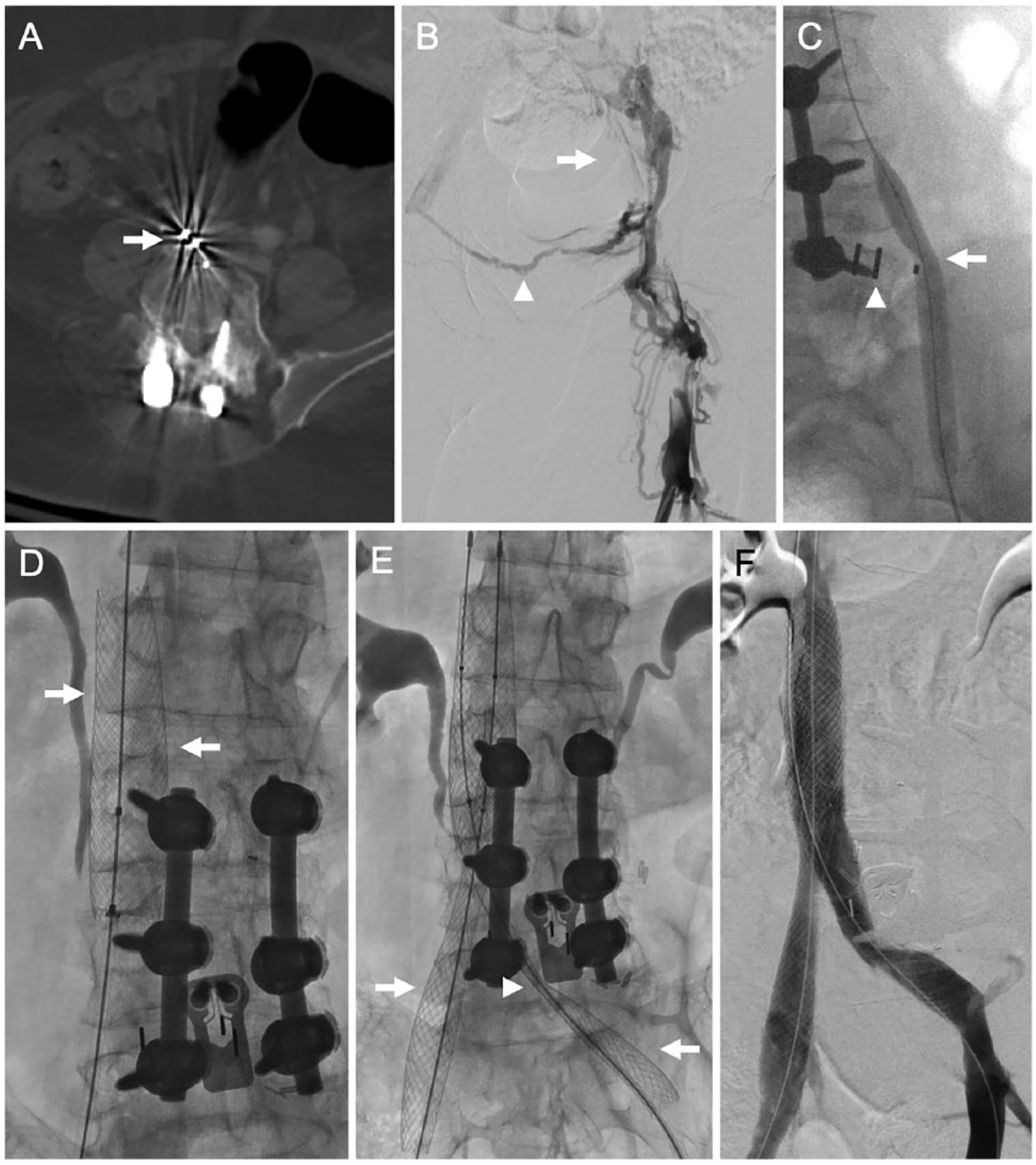
A 58-year-old female with spinal fusion hardware-associated iliocaval occlusion. (A) Computed tomography of the pelvis without contrast showing spinal fusion hardware (arrow) encroaching on the IVC. (B) Ascending venography, performed from a left groin approach, showing left iliocaval occlusion (arrow) with cross-pelvic venous collaterals (arrowhead). There was right iliocaval occlusion as well (not shown). (C) Angioplasty of the left common iliac venous system showed impingement of the fusion hardware (arrowhead) on the left common iliac vein (arrow). (D) Venous stent reconstruction was performed with placement of a 22-mm Wallstent (arrows) in the IVC. (E) ‘Kissing’ 16-mm Wallstents were deployed in a ‘double barrel’ fashion (arrows) extending from the IVC into the common iliac veins. The site of impingement by the spinal hardware is seen (arrowhead). Post-deployment angioplasty was performed using 16-mm balloons. (F) Completion bilateral iliocaval venography showing brisk in-line flow from both common femoral veins to the IVC.
Post-procedural anticoagulation and antiplatelet agents
Patients were initiated on enoxaparin 1 mg/kg twice daily and transitioned to warfarin or novel oral anticoagulant 4 weeks after the procedure. Patients were also discharged on antiplatelet therapy consisting of clopidogrel 300 mg loading dose, then 75 mg daily, and aspirin 81 mg daily. Warfarin or novel oral anticoagulant were continued for at least 1 year. Clopidogrel was discontinued 2 months after the procedure and restarted only if future stent placement was required. Aspirin was prescribed indefinitely as long as there was no contraindication.
Clinical follow-up evaluation
Clinical response was assessed at 2 weeks as well as at 6, 12, and 24 months after the procedure. Venous reconstruction patency was assessed at 6, 12, and 24 months with contrast venography (or noninvasive imaging).
Recorded variables
Technical aspects of stent reconstruction, technical success, complications, the 2-week and 6, 12, and 24-month clinical response, and the 6, 12, and 24-month primary, primary-assisted, and secondary stent patency rates were recorded.
Outcomes defined
Technical success was defined as successful recanalization with angioplasty, stent placement, restoration of in-line flow, and less than 20% residual stenosis, as described previously as per the Cardiovascular and Interventional Radiologic Society guidelines. 19 Adverse events were prospectively recorded and retrospectively classified according to the Society of Interventional Radiology adverse event classification system. 20
Clinical success was defined as at least 1-point improvement in the clinical manifestations score of the CEAP classification system. 11 The rate of clinical success at each time interval was calculated among the patients with the requisite follow-up duration. Venography (or noninvasive imaging) was performed at 6, 12 and 24 months, according to institutional practices, and primary, primary-assisted, and secondary patency were assessed.
Statistical analyses
Calculations of means, medians, ranges, and frequency were performed with statistical spreadsheet software (Microsoft Excel; Microsoft; Redmond, WA, USA). Kaplan–Meier curves for estimation of stent patency duration and estimated stent patency rates at 6, 12, and 24 months were performed using SPSS 24 statistical software (IBM Corp., Armonk, NY, USA).
Results
Sites of venous occlusive disease
Sites of venous occlusive disease are shown in Table 2. Most common sites of occlusion were the infrarenal IVC (n = 62; 90%), right common iliac vein (n = 56; 81%), and left common iliac vein (n = 56; 81%).
Sites of thrombosis.
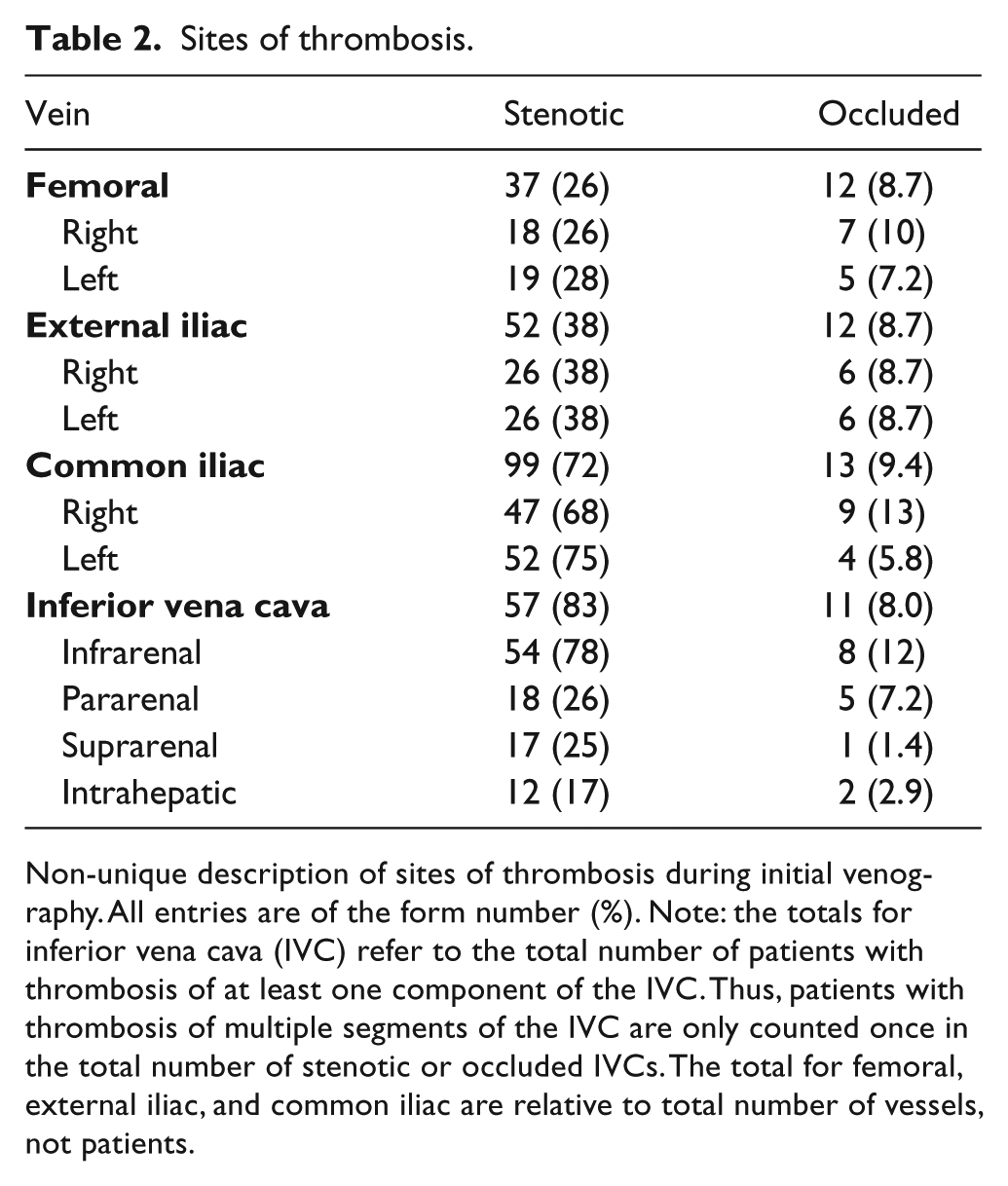
Non-unique description of sites of thrombosis during initial venography. All entries are of the form number (%). Note: the totals for inferior vena cava (IVC) refer to the total number of patients with thrombosis of at least one component of the IVC. Thus, patients with thrombosis of multiple segments of the IVC are only counted once in the total number of stenotic or occluded IVCs. The total for femoral, external iliac, and common iliac are relative to total number of vessels, not patients.
Thrombolysis
Twenty out of 69 (29%) patients underwent technically successful, uncomplicated, thrombolysis, including 16 (23%) patients with acute-on-chronic symptoms and four (5.8%) patients with chronic symptoms.
Technical aspects of venous recanalization and stent reconstruction
The most frequent number of access sites was three (range: 1–5), with the complete description of access sites in Table 3. Recanalization was performed using blunt recanalization techniques in 59/69 (86%) and sharp recanalization techniques in 10 (14%) patients. Contrast venography was employed in 69 (100%) procedures and intravascular ultrasound was employed in 49 (71%) interventions.
Access sites.
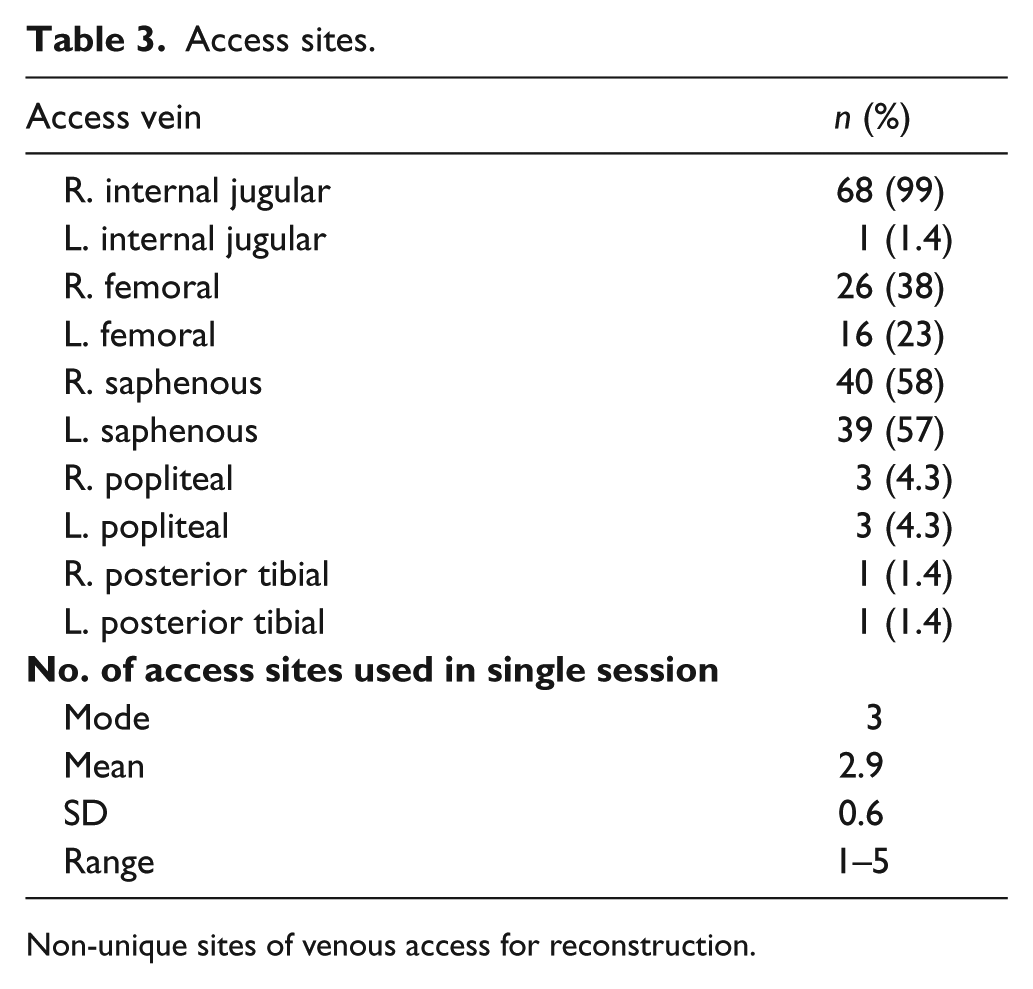
Non-unique sites of venous access for reconstruction.
All 69 patients underwent stent reconstruction, with a mean number of five stents placed per patient (range: 1–11). The total number of stents deployed was 352. Stent combinations included Wallstent endoprosthesis only (Boston Scientific; Marlborough, MA, USA) (n = 53; 77%), Wallstent and Z-Stent (Cook Medical; Bloomington, IN, USA) endoprostheses (n = 9; 13%), Z-Stent only (n = 2; 2.9%), AneuRx (Medtronic; Minneapolis, MN, USA) (n = 1; 1.4%), Palmaz only (Cordis; Milpitas, CA, USA) (n = 1; 1.4%), SMART CONTROL (Cordis) (n = 1; 1.4%), Wallstent and VIABAHN (W.L. Gore & Associates; Flagstaff, AZ, USA) endoprostheses (n = 1, 1.4%), and Z-Stent and Palmaz (n = 1, 1.4%) stents.
A total of 45 (65%) patients underwent bilateral iliocaval stent reconstruction, 10 (14%) underwent unilateral iliocaval stent reconstruction, and 14 (20%) underwent reconstruction of the IVC only. Technical success of venous recanalization and stent reconstruction was obtained in 69 (100%) patients.
Peri-procedural data, adverse events, and hospital stay
The mean contrast volume was 123 ± 72 mL (range: 10–350 mL). The mean fluoroscopy time was 125 ± 52 minutes (range: 34–231 minutes). The mean radiation dose was 3.2 ± 2.3 Gy (range: 0.69–9.8 Gy). The mean estimated blood loss was 54 ± 34 mL (range: 10–150 mL).
Per the Society of Interventional Radiology adverse event classification system, 20 there was one (1.4%) severe adverse event (stent migration into the heart requiring open surgical retrieval). There were four (5.8%) moderate adverse events, including two (2.9%) cases of post-angioplasty IVC pseudoaneurysm requiring covered-stent placement (one of which was associated with hemorrhage requiring blood transfusion), one (1.4%) case of distal stent migration requiring additional stent placement, and one (1.4%) case of residual narrowing of the left external iliac vein requiring additional stenting on post-procedure day two. There were four (5.8%) minor adverse events, including three (4.4%) access-site hematomas that did not require intervention and one (1.4%) mild extravasation at the IVC bifurcation without hemodynamic change or additional intervention.
The median length of post-procedural hospital stay was 1 day (range: 0–45 days). The patient with a 45-day length of stay required iliocaval stent reconstruction due to chronic IVC thrombosis after hepatic lobectomy. The patient had multiple other postoperative complications unrelated to IVC reconstruction which were responsible for the extended hospital stay.
Clinical outcomes and long-term stent patency of iliocaval reconstruction
Clinical outcomes at 2 weeks
There were 58/69 (84%) patients who completed a 2-week follow-up clinic visit, which occurred at a mean of 18 days after the procedure (range: 7–43 days). Clinical success at 2 weeks was obtained in 44/58 (76%) patients. Specifically, three (5.2%) had a 4-point improvement in their CEAP score relative to baseline, 32 (55%) had a 3-point improvement, one (1.7%) had a 2-point improvement, and eight (14%) had a 1-point improvement.
Clinical outcomes at 6 months
There were 41/69 (59%) patients who completed the 6-month follow-up clinic visit at a mean of 187 days after the procedure (range: 80–269 days). Clinical success at 6 months was obtained in 35/41 (85%) patients. Specifically, 2/35 (5.1%) patients had a 4-point improvement in CEAP score relative to baseline, 24 (69%) had a 3-point improvement, one (2.9%) had a 2-point improvement, and eight (23%) had a 1-point improvement.
Clinical outcomes at 12 months
There were 31/69 (45%) patients who completed the 12-month follow-up clinic visit at a mean of 418 days after the procedure (range: 288–571 days). Clinical success at 12 months was obtained in 27/31 (87%) patients. Specifically, 2/27 (7.4%) patients demonstrated a 4-point improvement in CEAP score relative to baseline, 20 (64%) demonstrated a 3-point improvement, one (3.2%) demonstrated a 2-point improvement, and four (13%) demonstrated a 1-point improvement.
Clinical outcomes at 24 months
There were 17/69 (25%) patients who completed the 24-month follow-up clinic visit at a mean of 753 days after the procedure (range: 536–1241 days). Clinical success at 24 months was obtained in 17/17 (100%) patients. Specifically, 1/17 (5.9%) patient demonstrated a 4-point improvement in CEAP score relative to baseline, 13 (76%) demonstrated a 3-point improvement in CEAP score relative to baseline, and three (18%) demonstrated a 1-point improvement.
The 6-month stent patency and repeat interventions
There were 41/69 (59%) patients who completed a 6-month catheter-based venography. Thirty-nine of 41 (95%) reconstructions were patent at 6 months, with 2/41 (4.9%) occlusions that required intervention. Additionally, three (7.3%) patients had in-stent stenosis requiring angioplasty and additional stent placement.
The 12-month stent patency and repeat interventions
There were 31/69 (45%) patients who completed a 12-month catheter-based venography. Patent reconstructions were demonstrated by 31/31 (100%) patients. One (3.2%) patient required repeat stenting for in-stent stenosis. All five patients who required repeat intervention at 6 months post-procedure demonstrated patent reconstructions at 12 months and did not require further intervention.
The 24-month stent patency and repeat interventions
There were 17/69 (25%) patients who completed a 24-month catheter-based venography. Fifteen out of 17 (88%) patients demonstrated patent reconstructions; one (5.8%) patient required thrombolysis and recanalization of the IVC stent; and one (5.8%) required recanalization and stenting of the right femoral, external iliac, and common iliac stents. Three (18%) required repeat stenting for in-stent stenosis. No patients that required repeat stenting during prior venography required further intervention to maintain patency.
Duration of patency
The mean estimated duration of primary patency was 50.7 ± 7.0 months (95% CI: 37.0–64.6 months) (Figure 4). The mean estimated duration of primary-assisted patency was 62.0 ± 7.3 months (95% CI: 47.8–76.3 months) (Figure 5).
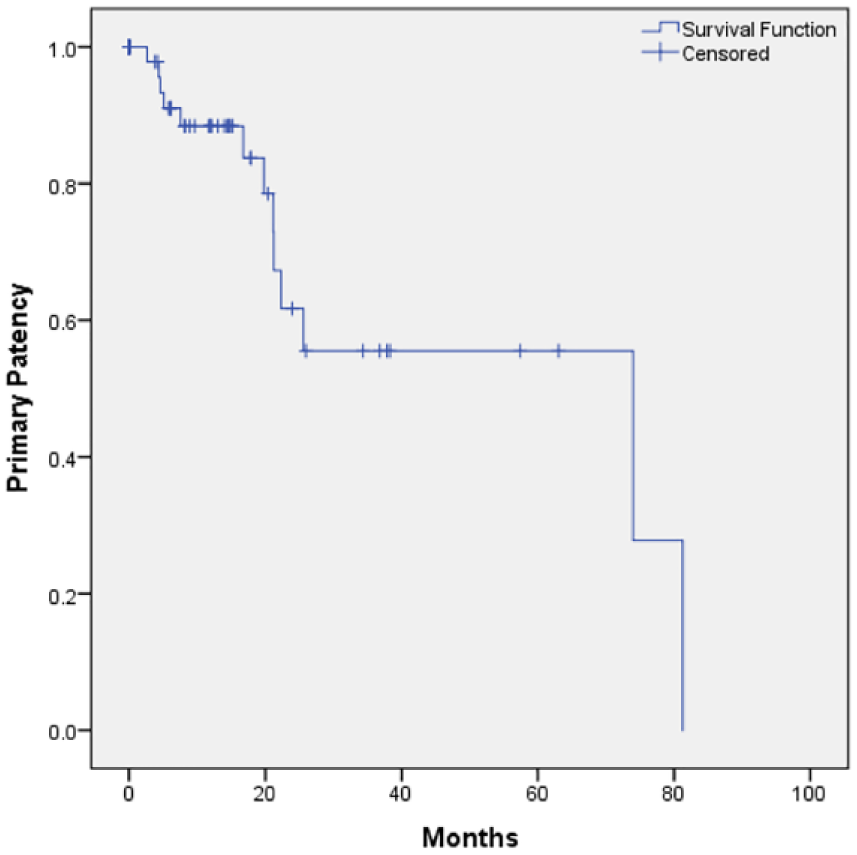
Kaplan–Meier curve demonstrating primary patency of reconstruction over time.
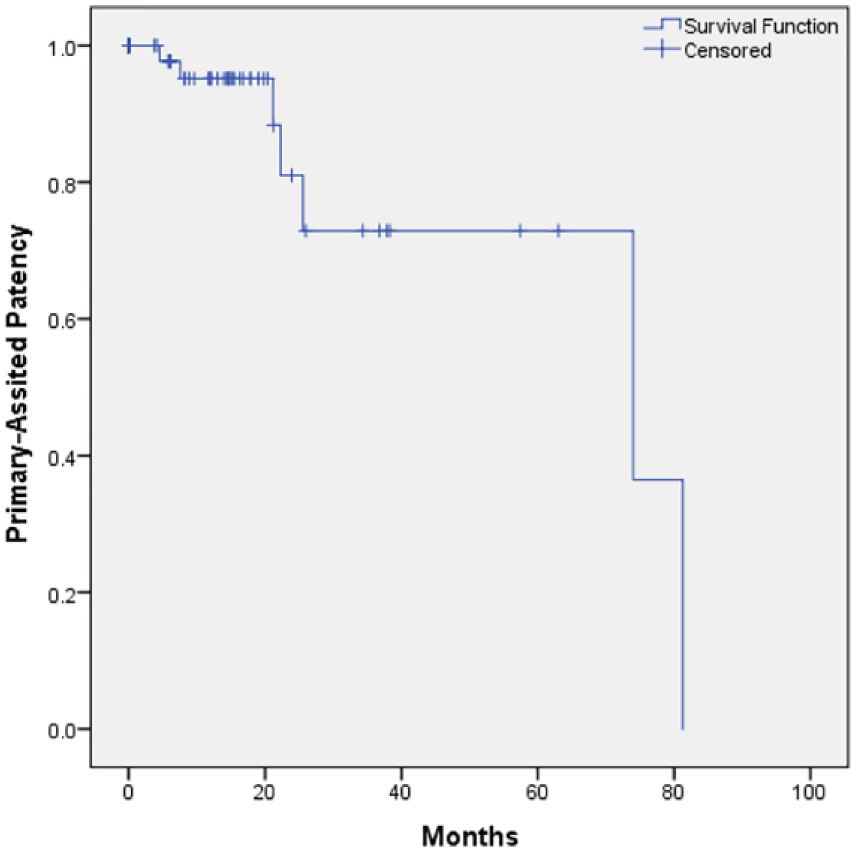
Kaplan–Meier curve demonstrating primary-assisted patency of reconstruction over time.
Estimated patency metrics
Estimated primary patency at 6, 12, and 24 months was 91%, 88%, and 62%, respectively (Figure 4). Estimated primary-assisted patency at 6, 12, and 24 months was 98%, 95%, and 81% (Figure 5). As there were no cases of stents that were not secondarily patent, estimated secondary patency at 6, 12, and 24 months were all 100%.
Mortality
The 30-day mortality was zero; the 90-day mortality was 1.4%, as one patient died of hepatic failure unrelated to his recanalization. Seven (10%) patients had died as of the final follow-up, none of which was related to iliocaval stent reconstruction.
Discussion
This series demonstrates that endovascular iliocaval stent reconstruction is technically feasible with good clinical outcomes and stent patency rates in patients with non-IVC filter-related chronic iliocaval occlusion. Endovascular reconstruction for iliocaval thrombosis has been established in large series for adults with IVC filter-related or iliac vein-associated occlusions.4,6,7 This report differs from prior reports in its focus on primary IVC or bilateral iliocaval thrombosis, its high incidence of congenital IVC abnormalities, and its absence of IVC filters.
The 2-week, 6-month, 12-month, and 24-month clinical success rates in this series were 76%, 85%, 87%, and 100%. Etiology of the improvement over time cannot be established with certainty, but may be due to augmentation of collaterals. Raju et al. reported a series of 97 patients with IVC obstruction (some of which were IVC filter-related), with relief of pain (per the Visual Analog Scale) in 74% and relief of swelling in 51% at the final median follow-up of 11 months, lower than the clinical success rate in this study. 9 The 12-month clinical success rate in the present series is similar to that reported in a series of 115 patients with bilateral iliocaval confluence, but not proximal IVC obstruction, by Neglen et al., in which the final clinical success measured by pain relief was achieved in 85% of patients at the final median follow-up of 12 months. 6 The use of various clinical success definitions makes detailed comparison difficult.
Rollo et al. reported a series in which 42% of patients had filter-associated thrombosis, reporting a decrease in CEAP classification in 65% of patients at the 1-year follow-up, 21 a lower clinical success rate than the current study. Chick et al. also defined clinical success as a 1-point decrease in CEAP classification in a 120-patient series of patients with filter-associated IVC thrombosis, finding clinical success in 84% at 2 weeks and 96% at 6 months, higher than the current study. 4 Relative to Chick et al., in which patients had, on average, 1.5 non-filter-related thrombotic risk factors, patients in this study had more non-IVC filter-related thrombotic risk factors and were much more likely to have anatomic IVC anomalies, which may explain the slightly lower rates of clinical success in the present series. 4 Specifically, filter removal during reconstruction is a modifiable risk factor that has been shown to be associated with a decreased risk of subsequent reconstruction thrombosis. 21 Overall, the rates of clinical successes in the present study are on the high end of the 60–85% range of clinical success reported in other iliocaval stent reconstructions exclusively focused on patients with filter-bearing IVC thrombosis.22,23
In the present study, the estimated 6, 12, and 24-month primary patency rates were 91%, 88%, and 62%, respectively; 6, 12, and 24-month primary-assisted patency rates were 98%, 95%, and 81%, respectively; and 6, 12, and 24-month secondary-assisted patency rates were all 100%. The primary-assisted patency rates in this study are similar to those reported by Raju et al., who reported an 82% primary-assisted patency rate at 2 years. 9 Raju et al. employed only aspirin as post-procedural anticoagulation in the majority of patients, which is less aggressive than the regimen employed in the present series, in which both anticoagulation and antiplatelet therapy were employed. The primary and primary-assisted patency rates in this study are also similar to those reported by de Graaf et al., who reported 82% primary patency at 12 months and 72% at 36 months. 8 Of note, 38% of the patients in the de Graaf et al. series had adjunctive open phlebectomy and creation of arteriovenous fistula to improve venous inflow, and that group employed at least 6 months of post-procedural warfarin, but did not routinely prescribe life-long aspirin as was employed in this series. 8 Hartung et al. reported 94% primary-assisted patency at 12 months, very similar to this series, though the patients in that series differed from the current series in that only 9% of patients had IVC thrombosis that extended past the confluence, a much lower proportion of IVC involvement than in the current study. 5 Rollo et al., who reported a 31-patient series of iliocaval thrombosis (42% of whom had indwelling IVC filters), reported a 75% primary-assisted patency at 12 months, lower than this study. 21 The lower patency rates in Rollo et al. may also be due to use of single-agent anticoagulation (either anticoagulation with warfarin or antiplatelet therapy, depending on patient characteristics). 21 Sebastian et al. reported a 62-patient series of reconstructions for non-filter associated IVC thrombosis, showing a similar (57%) 24-month primary patency, but a lower (76%) 24-month primary-assisted patency. 24 Similarly, Grøtta et al. reported a 20-patient series that had a similar (67%) 24-month primary patency and a lower 24-month secondary patency (83%). 25
Relative to patency rates reported for IVC filter-associated thrombosis, Chick et al. reported a primary-assisted patency of 95% and 87% at 12 and 24 months, respectively, in a series of 120 patients, identical at 12 months and slightly better than the current series at 24 months. 4 Chick et al. employed the same post-procedural anticoagulation regimen as this series, likely accounting for the similar rates of patency, and the higher rates relative to prior series who employed less aggressive antithrombotic medical therapy. 9
There was one (1.4%) severe, four (5.8%) moderate, and four (5.8%) minor adverse events. Moderate and severe adverse events consisted of stent migrations and IVC pseudoaneurysms requiring additional stenting. The reported range of major bleeding during iliocaval stent reconstruction is 0–7.5%,8,9,21 which includes the rate in the current series. Additionally, stent migrations have been reported previously with similar frequency. 5 Minor adverse events in this series consisted of access site bleeding and hematomas, which are expected to occur in up to 14% of cases.5,6,12,13
Limitations
The present study has several limitations. The study was retrospective, and thus, evaluation and documentation of clinical outcomes and complications were not standardized and limited to what was documented in the medical record. The extent of stent placement was determined by each individual operator, so operator-specific biases, as well as changes in institutional practice patterns, may confound patency rates. This study employed follow-up venography in many patients to assess patency, even in asymptomatic patients, which was an institutional practice that differs from the standard of care and may impact the generalizability of the patency measures. Additionally, all procedures were performed at a tertiary referral center, which may have an adverse impact on follow-up length for patients not located near the medical center, and some patients were lost to follow-up, introducing potential bias into the accuracy of follow-up clinical and patency data, as well as mortality.
Conclusion
Iliocaval stent reconstruction is an effective treatment for non-IVC filter-related chronic iliocaval thrombosis with high rates of technical success, 6, 12, and 24-month clinical responses, and 6, 12, and 24-month primary and secondary stent patency rates, particularly with triple-agent post-procedural antithrombotic medical therapy.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
