Abstract

Pulmonary embolism (PE) is the world’s third leading cardiovascular (CV) cause of death. Care for myocardial infarction and stroke, the two leading causes of CV death, has been characterized by the proliferation of comprehensive centers able to provide urgent revascularization to patients suffering from these conditions. Historically, there has been little effort dedicated to transformative healthcare delivery initiatives for the care of acute PE.
Part of the reason for the discrepancy in how PE is handled is due to a difference in the pathophysiology of the disease process. In myocardial infarction and stroke, time is myocardium or brain, respectively. Infarction of these tissues leads to largely irreversible damage that has an impact on both short and long-term clinical outcomes. With PE, time is not lung. The most important pathophysiologic effect of PE that drives symptoms is due to an indirect effect on the right ventricle from acutely increased afterload. Observational data suggest that the majority of patients who present with PE will improve dramatically with anticoagulation alone and will not suffer long-term cardiopulmonary consequences.1,2 However, important subsets of patients experience an increase in short-term mortality or long-term sequelae of PE with development of dyspnea, chest pain syndromes or clinically significant chronic thromboembolic pulmonary hypertension. 3
The variability in prognosis of the acute PE event leads to difficult decision making, a complexity that is often heightened in the face of a variety of patient comorbidities. And all of this occurs in a setting of relatively little high-quality comparative data regarding differing acute management strategies, rapidly advancing technology for device therapy, and a number of evidence-based clinical practice guidelines with varying and sometimes conflicting recommendations based on a paucity of high-level evidence. It is in this context that the ‘pulmonary embolism response team’ (PERT) model of care has emerged over the past 5 years with a stated aim of urgent multidisciplinary care provision to patients suffering from intermediate or high-risk pulmonary embolism.4,5 Widespread adoption of the PERT concept has been predicated on the promise of: (1) reduced heterogeneity of PE management both within individual medical centers and across healthcare systems; (2) greater access to advanced therapies for those with an increased risk of adverse outcomes; (3) more appropriate use of interventional therapies for PE; (4) improved clinical outcomes; and (5) a reduction in the length of stay and costs associated with PE care. Skeptics of the PERT model have raised concerns about such a strategy encouraging greater utilization of interventional therapies for PE in the absence of supportive high-quality randomized controlled trial data and evidence-based guideline recommendations.
Despite the rapid proliferation of PERTs across the nation, and indeed internationally, there has been remarkably little reported data regarding their structure and real-world function. In fact, a search of ‘pulmonary embolism response teams’ in PubMed recovers only 23 publications. In this issue of Vascular Medicine, Sista et al. add to the sparsely reported literature on PERTs with a description of the management of intermediate and high-risk PE at Weill Cornell Hospital in New York City over a 20-month period after the initiation of their PERT. 6 The authors describe the management of 87 consecutive patients with PE, comprised of 79 intermediate and eight high-risk patients. Fifty-four (62%) of the patients were managed with isolated anticoagulation while 31 (36%) received thrombolysis, the vast majority of which was done via an invasive catheter-directed technique (25 of the 31 cases). Twenty-two (25%) patients received inferior vena cava filters. The rates of escalation of therapy beyond anticoagulation are similar to the only other institutional report of PERT practice and outcomes, though the degree of invasive therapies administered to intermediate-risk PE patients in both reports may give some pause for thought. 7
While it is somewhat reassuring that the Weill Cornell PERT did not serve as a mechanism to algorithmically feed all cases into the procedure suite, there remains an open question about what proportion of intermediate-risk PE patients should be treated aggressively, and what the appropriate role of invasive therapies (as opposed to systemic thrombolysis) should be in this population. As the PERT system of care continues to develop, we must be highly cognizant of the fact that its promulgation is occurring in a setting of several factors that incentivize performing procedures. These include hospital administrative pressures to continually grow catheterization laboratory and interventional radiology suite volumes, fairly robust productivity-based compensation associated with the catheter-directed therapies (as judged by the substantial associated relative value units assigned to these cases), and industry influences on procedural growth and associated use of related products.
In addition, we must recognize another potential pitfall of the widespread adoption of a PERT strategy: many medical centers in the US and worldwide are expending considerable resources to institute PERTs without high-quality data proving that such an approach standardizes PE care, improves outcomes, and is cost-effective (Table 1). Of the 23 papers in PubMed focused on a multidisciplinary team approach to acute PE, the vast majority are either descriptions of the initial experience with PERT models or reviews on the implementation of such strategies.7–9 As of yet, none has advanced our understanding of the impact of the PERT model past that of unfulfilled potential. While a multidisciplinary team approach is fundamental to improved outcomes in myocardial infarction, stroke, and transcatheter aortic valve replacement, for example, we must be circumspect about extrapolating these observations to PE care.
Pulmonary embolism response team (PERT) midterm report card.
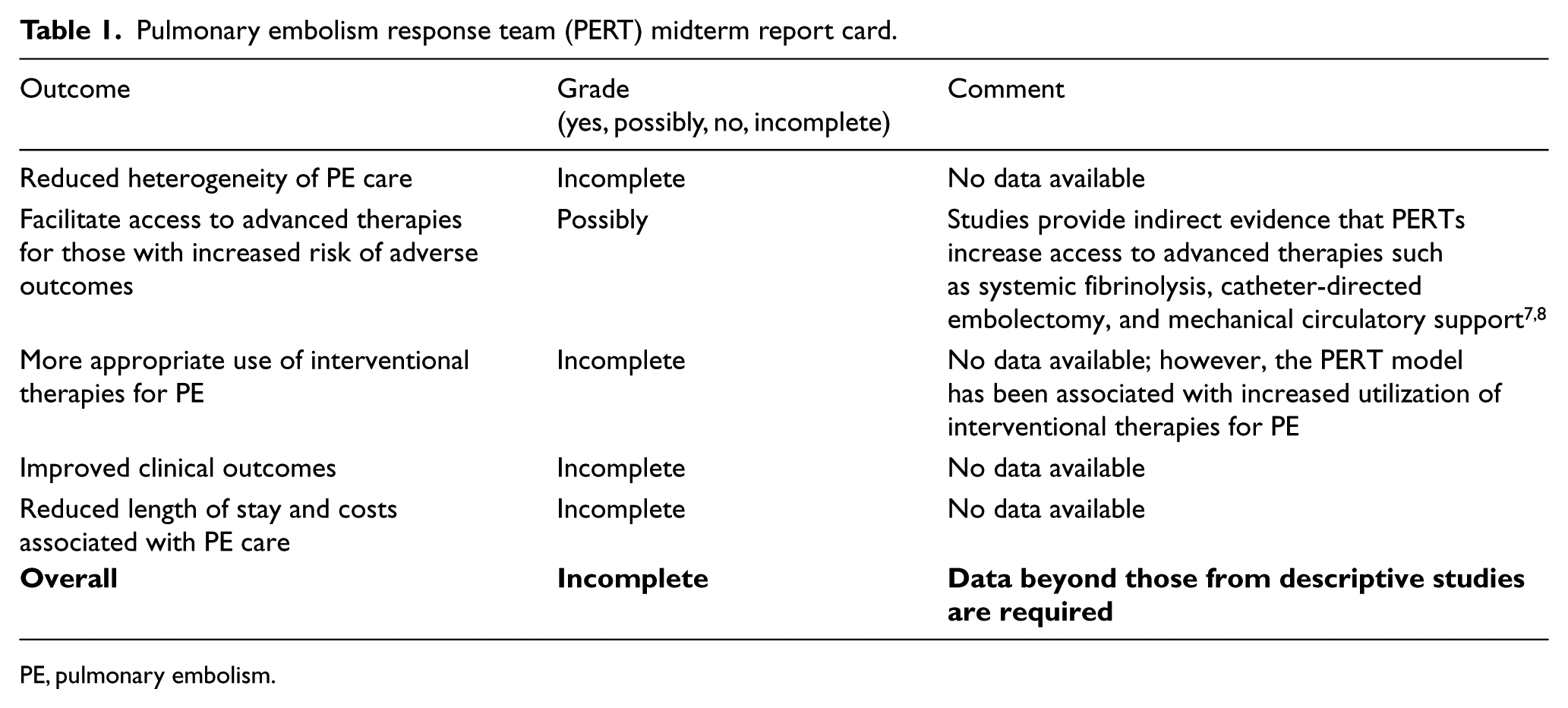
PE, pulmonary embolism.
While a multidisciplinary rapid response approach to PE has a great appeal to providers on the frontlines and hospital administrators focused on quality improvement and cost containment, the rationale for adopting PERTs should be held to the same scientific rigor as other aspects of PE care. Just as we demand clinical outcome-driven trials and the highest quality of evidence from the manufacturers of drugs and devices used to treat PE, we must be consistent and require a sufficient foundation of data to justify the implementation of PERTs. Furthermore, we must recognize the potential, and possibly unwanted, downstream consequences of the PERT model on resource and personnel utilization as well as interventional and surgical volumes. Sista et al. provide us with a glimpse of the ramifications of the widespread adoption of PERTs without the data required to develop a balanced understanding of its impact.
Reports such as that by Sista et al. serve an important purpose in describing the implementation and function of a PERT strategy. While there is much to be learned from such reports, it is time to advance our understanding of the implementation science of the PERT model and take a critical look at how such a strategy can impact patient outcomes as well as resource utilization. Research consortiums (pertconsortium.org) and prospective outcome-focused studies will inform us on how best to integrate a PERT model into our care of PE patients and how to maximize the benefits of such a strategy while minimizing any possible unwanted consequences. Only through critical appraisals of the PERT model that emphasize continuous quality improvement can we truly tap into its promise of improved care of our patients with PE.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Giri is a member of the Board of Directors for the PERT Consortium, a 501(c)(3) non-profit organization; Dr Piazza has received research grant support from EKOS, a BTG International group company, Bristol-Myers Squibb, Daiichi-Sankyo, and Janssen.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
