Abstract

Keywords
A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines
Table of Contents
PREAMBLE 3
1. INTRODUCTION 5
1.1. Methodology and Evidence Review 5
1.2. Organization of the Writing Committee 5
1.3. Document Review and Approval 5
1.4. Scope of Guideline 6
2. CLINICAL ASSESSMENT FOR PAD 8
2.1. History and Physical Examination: Recommendations 8
3.1. Resting ABI for Diagnosing PAD 9
3.2. Physiological Testing 10
3.3. Imaging for Anatomic Assessment 11
4.1. Abdominal Aortic Aneurysm 12
4.2. Screening for Asymptomatic Atherosclerosis in Other Arterial Beds (Coronary, Carotid, and Renal Arteries) 12
5. MEDICAL THERAPY FOR THE PATIENT WITH PAD: RECOMMENDATIONS 13
5.1. Antiplatelet, Statin, Antihypertensive Agents, and Oral Anticoagulation 13
5.2. Smoking Cessation 13
5.3. Glycemic Control 14
5.4. Cilostazol, Pentoxifylline, and Chelation Therapy 14
5.5. Homocysteine Lowering 14
5.6. Influenza Vaccination 14
6. STRUCTURED EXERCISE THERAPY: RECOMMENDATIONS 14
7. MINIMIZING TISSUE LOSS IN PATIENTS WITH PAD: RECOMMENDATIONS 15
8. REVASCULARIZATION FOR CLAUDICATION: RECOMMENDATIONS 16
8.1. Revascularization for Claudication 16
8.1.1. Endovascular Revascularization for Claudication 16
8.1.2. Surgical Revascularization for Claudication 17
9. MANAGEMENT OF CLI: RECOMMENDATIONS 17
9.1. Revascularization for CLI 17
9.1.1. Endovascular Revascularization for CLI 18
9.1.2. Surgical Revascularization for CLI 19
9.2. Wound Healing Therapies for CLI 19
10. MANAGEMENT OF ACUTE LIMB ISCHEMIA: RECOMMENDATIONS 19
10.1. Clinical Presentation of ALI 20
10.2. Medical Therapy for ALI 20
10.3. Revascularization for ALI 20
10.4. Diagnostic Evaluation of the Cause of ALI 21
11. LONGITUDINAL FOLLOW-UP: RECOMMENDATIONS 21
12. EVIDENCE GAPS AND FUTURE RESEARCH DIRECTIONS 22
13. ADVOCACY PRIORITIES 22
REFERENCES 23
APPENDIX 1 34
Author Relationships With Industry and Other Entities
APPENDIX 2 37
Reviewer Relationships With Industry and Other Entities
APPENDIX 3 43
Abbreviations
Preamble
Since 1980, the American College of Cardiology (ACC) and American Heart Association (AHA) have translated scientific evidence into clinical practice guidelines with recommendations to improve cardiovascular health. These guidelines, based on systematic methods to evaluate and classify evidence, provide a cornerstone of quality cardiovascular care.
In response to reports from the Institute of Medicine (1,2) and a mandate to evaluate new knowledge and maintain relevance at the point of care, the ACC/AHA Task Force on Clinical Practice Guidelines (Task Force) modified its methodology (3–5). The relationships among guidelines, data standards, appropriate use criteria, and performance measures are addressed elsewhere (5).
Intended Use
Practice guidelines provide recommendations applicable to patients with or at risk of developing cardiovascular disease. The focus is on medical practice in the United States, but guidelines developed in collaboration with other organizations may have a broader target. Although guidelines may be used to inform regulatory or payer decisions, the intent is to improve quality of care and align with patients’ interests. Guidelines are intended to define practices meeting the needs of patients in most, but not all, circumstances, and should not replace clinical judgment. Guidelines are reviewed annually by the Task Force and are official policy of the ACC and AHA. Each guideline is considered current until it is updated, revised, or superseded by published addenda, statements of clarification, focused updates, or revised full-text guidelines. To ensure that guidelines remain current, new data are reviewed biannually to determine whether recommendations should be modified. In general, full revisions are posted in 5-year cycles (3–6).
Modernization
Processes have evolved to support the evolution of guidelines as “living documents” that can be dynamically updated. This process delineates a recommendation to address a specific clinical question, followed by concise text (ideally <250 words) and hyperlinked to supportive evidence. This approach accommodates time constraints on busy clinicians and facilitates easier access to recommendations via electronic search engines and other evolving technology.
Evidence Review
Writing committee members review the literature; weigh the quality of evidence for or against particular tests, treatments, or procedures; and estimate expected health outcomes. In developing recommendations, the writing committee uses evidence-based methodologies that are based on all available data (3–7). Literature searches focus on randomized controlled trials (RCTs) but also include registries, nonrandomized comparative and descriptive studies, case series, cohort studies, systematic reviews, and expert opinion. Only selected references are cited.
The Task Force recognizes the need for objective, independent Evidence Review Committees (ERCs) that include methodologists, epidemiologists, clinicians, and biostatisticians who systematically survey, abstract, and assess the evidence to address systematic review questions posed in the PICOTS format (P=population, I=intervention, C=comparator, O=outcome, T=timing, S=setting) (2,4–6). Practical considerations, including time and resource constraints, limit the ERCs to evidence that is relevant to key clinical questions and lends itself to systematic review and analysis that could affect the strength of corresponding recommendations.
Guideline-Directed Management and Treatment
The term “guideline-directed management and therapy” (GDMT) refers to care defined mainly by ACC/AHA Class I recommendations. For these and all recommended drug treatment regimens, the reader should confirm dosage with product insert material and carefully evaluate for contraindications and interactions. Recommendations are limited to treatments, drugs, and devices approved for clinical use in the United States.
Class of Recommendation and Level of Evidence
The Class of Recommendation (COR; i.e., the strength of the recommendation) encompasses the anticipated magnitude and certainty of benefit in proportion to risk. The Level of Evidence (LOE) rates evidence supporting the effect of the intervention on the basis of the type, quality, quantity, and consistency of data from clinical trials and other reports (Table 1) (3–5). Unless otherwise stated, recommendations are sequenced by COR and then by LOE. Where comparative data exist, preferred strategies take precedence. When >1 drug, strategy, or therapy exists within the same COR and LOE and no comparative data are available, options are listed alphabetically.
Applying Class of Recommendation and Level of Evidence to Clinical Strategies, Interventions, Treatments, or Diagnostic Testing in Patient Care* (Updated August 2015).

Relationships With Industry and Other Entities
The ACC and AHA sponsor the guidelines without commercial support, and members volunteer their time. The Task Force zealously avoids actual, potential, or perceived conflicts of interest that might arise through relationships with industry or other entities (RWI). All writing committee members and reviewers are required to disclose current industry relationships or personal interests, from 12 months before initiation of the writing effort. Management of RWI involves selecting a balanced writing committee and assuring that the chair and a majority of committee members have no relevant RWI (Appendix 1). Members are restricted with regard to writing or voting on sections to which their RWI apply. For transparency, members’ comprehensive disclosure information is available online. Comprehensive disclosure information for the Task Force is also available online.
The Task Force strives to avoid bias by selecting experts from a broad array of backgrounds representing different geographic regions, sexes, ethnicities, intellectual perspectives/biases, and scopes of clinical practice, and by inviting organizations and professional societies with related interests and expertise to participate as partners or collaborators.
Individualizing Care in Patients With Associated Conditions and Comorbidities
Managing patients with multiple conditions can be complex, especially when recommendations applicable to coexisting illnesses are discordant or interacting (8). The guidelines are intended to define practices meeting the needs of patients in most, but not all, circumstances. The recommendations should not replace clinical judgment.
Clinical Implementation
Management in accordance with guideline recommendations is effective only when followed. Adherence to recommendations can be enhanced by shared decision making between clinicians and patients, with patient engagement in selecting interventions on the basis of individual values, preferences, and associated conditions and comorbidities. Consequently, circumstances may arise in which deviations from these guidelines are appropriate.
The reader is encouraged to consult the full-text guideline (9) for additional guidance and details with regard to lower extremity peripheral artery disease (PAD) because the executive summary contains limited information.
Jonathan L. Halperin, MD, FACC, FAHA
Chair, ACC/AHA Task Force on Clinical Practice Guideline
1. Introduction
1.1. Methodology and Evidence Review
The recommendations listed in this guideline are, whenever possible, evidence based. An initial extensive evidence review, which included literature derived from research involving human subjects, published in English, and indexed in MEDLINE (through PubMed), EMBASE, the Cochrane Library, the Agency for Healthcare Research and Quality, and other selected databases relevant to this guideline, was conducted from January through September 2015. Key search words included but were not limited to the following: acute limb ischemia, angioplasty, ankle-brachial index, anticoagulation, antiplatelet therapy, atypical leg symptoms, blood pressure lowering/hypertension, bypass graft/bypass grafting/surgical bypass, cilostazol, claudication/intermittent claudication, critical limb ischemia/severe limb ischemia, diabetes, diagnostic testing, endovascular therapy, exercise rehabilitation/exercise therapy/exercise training/supervised exercise, lower extremity/foot wound/ulcer, peripheral artery disease/peripheral arterial disease/peripheral vascular disease/lower extremity arterial disease, smoking/smoking cessation, statin, stenting, and vascular surgery. Additional relevant studies published through September 2016, during the guideline writing process, were also considered by the writing committee, and added to the evidence tables when appropriate. The final evidence tables included in the Online Data Supplement summarize the evidence utilized by the writing committee to formulate recommendations. Additionally, the writing committee reviewed documents related to lower extremity PAD previously published by the ACC and AHA (10,11). References selected and published in this document are representative and not all-inclusive.
As stated in the Preamble, the ACC/AHA guideline methodology provides for commissioning an independent ERC to address systematic review questions (PICOTS format) to inform recommendations developed by the writing committee. All other guideline recommendations (not based on the systematic review questions) were also subjected to an extensive evidence review process. For this guideline, the writing committee in conjunction with the Task Force and ERC Chair identified the following systematic review questions: 1) Is antiplatelet therapy beneficial for prevention of cardiovascular events in the patient with symptomatic or asymptomatic lower extremity PAD? 2) What is the effect of revascularization, compared with optimal medical therapy and exercise training, on functional outcome and quality of life (QoL) among patients with claudication? Each question has been the subject of recently published, systematic evidence reviews (12–14). The quality of these evidence reviews was appraised by the ACC/AHA methodologist and a vendor contracted to support this process (Doctor Evidence [Santa Monica, CA]). Few substantive randomized or nonrandomized studies had been published after the end date of the literature searches used for the existing evidence reviews, so the ERC concluded that no additional systematic review was necessary to address either of these critical questions.
A third systematic review question was then identified: 3) Is one revascularization strategy (endovascular or surgical) associated with improved cardiovascular and limb-related outcomes in patients with critical limb ischemia (CLI)? This question had also been the subject of a high-quality systematic review that synthesized evidence from observational data and an RCT (15); additional RCTs addressing this question are ongoing (16–18). The writing committee and the Task Force decided to expand the survey to include more relevant randomized and observational studies. Based on evaluation of this additional evidence the ERC decided that further systematic review was not needed to inform the writing committee on this question. Hence, the ERC and writing committee concluded that available systematic reviews could be used to inform the development of recommendations addressing each of the 3 systematic review questions specified above. The members of the Task Force and writing committee thank the members of the ERC that began this process and their willingness to participate in this volunteer effort. They include Aruna Pradhan, MD, MPH (ERC Chair); Natalie Evans, MD; Peter Henke, MD; Dharam J. Kumbhani, MD, SM, FACC; and Tamar Polonsky, MD.
1.2. Organization of the Writing Committee
The writing committee consisted of clinicians, including noninvasive and interventional cardiologists, exercise physiologists, internists, interventional radiologists, vascular nurses, vascular medicine specialists, and vascular surgeons, as well as clinical researchers in the field of vascular disease, a nurse (in the role of patient representative), and members with experience in epidemiology and/or health services research. The writing committee included representatives from the ACC and AHA, American Association of Cardiovascular and Pulmonary Rehabilitation, Inter-Society Consensus for the Management of Peripheral Arterial Disease, Society for Cardiovascular Angiography and Interventions, Society for Clinical Vascular Surgery, Society of Interventional Radiology, Society for Vascular Medicine, Society for Vascular Nursing, Society for Vascular Surgery, and Vascular and Endovascular Surgery Society.
1.3. Document Review and Approval
This document was reviewed by 2 official reviewers nominated by the ACC and AHA; 1 to 2 reviewers each from the American Association of Cardiovascular and Pulmonary Rehabilitation, Inter-Society Consensus for the Management of Peripheral Arterial Disease, Society for Cardiovascular Angiography and Interventions, Society for Clinical Vascular Surgery, Society of Interventional Radiology, Society for Vascular Medicine, Society for Vascular Nursing, Society for Vascular Surgery, and Vascular and Endovascular Surgery Society; and 16 additional individual content reviewers. Reviewers’ RWI information was distributed to the writing committee and is published in this document (Appendix 2).
This document was approved for publication by the governing bodies of the ACC and the AHA and endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation, Inter-Society Consensus for the Management of Peripheral Arterial Disease, Society for Cardiovascular Angiography and Interventions, Society for Clinical Vascular Surgery, Society of Interventional Radiology, Society for Vascular Medicine, Society for Vascular Nursing, Society for Vascular Surgery, and Vascular and Endovascular Surgery Society.
1.4. Scope of Guideline
Lower extremity PAD is a common cardiovascular disease that is estimated to affect approximately 8.5 million Americans above the age of 40 years and is associated with significant morbidity, mortality, and QoL impairment (19). It has been estimated that 202 million people worldwide have PAD (20). The purpose of this document is to provide a contemporary guideline for diagnosis and management of patients with lower extremity PAD. This document supersedes recommendations related to lower extremity PAD in the “ACC/AHA 2005 Guidelines for the Management of Patients With Peripheral Arterial Disease” (10) and the “2011 ACCF/AHA Focused Update of the Guideline for the Management of Patients With Peripheral Artery Disease” (11). The scope of this guideline is limited to atherosclerotic disease of the lower extremity arteries (PAD) and includes disease of the aortoiliac, femoropopliteal, and infrapopliteal arterial segments. It does not address nonatherosclerotic causes of lower extremity arterial disease, such as vasculitis, fibromuscular dysplasia, physiological entrapment syndromes, cystic adventitial disease, and other entities. Future guidelines will address aneurysmal disease of the abdominal aorta and lower extremity arteries and diseases of the renal and mesenteric arteries.
For the purposes of this guideline, key terms associated with PAD are defined in Table 2.
Definition of PAD Key Terms.
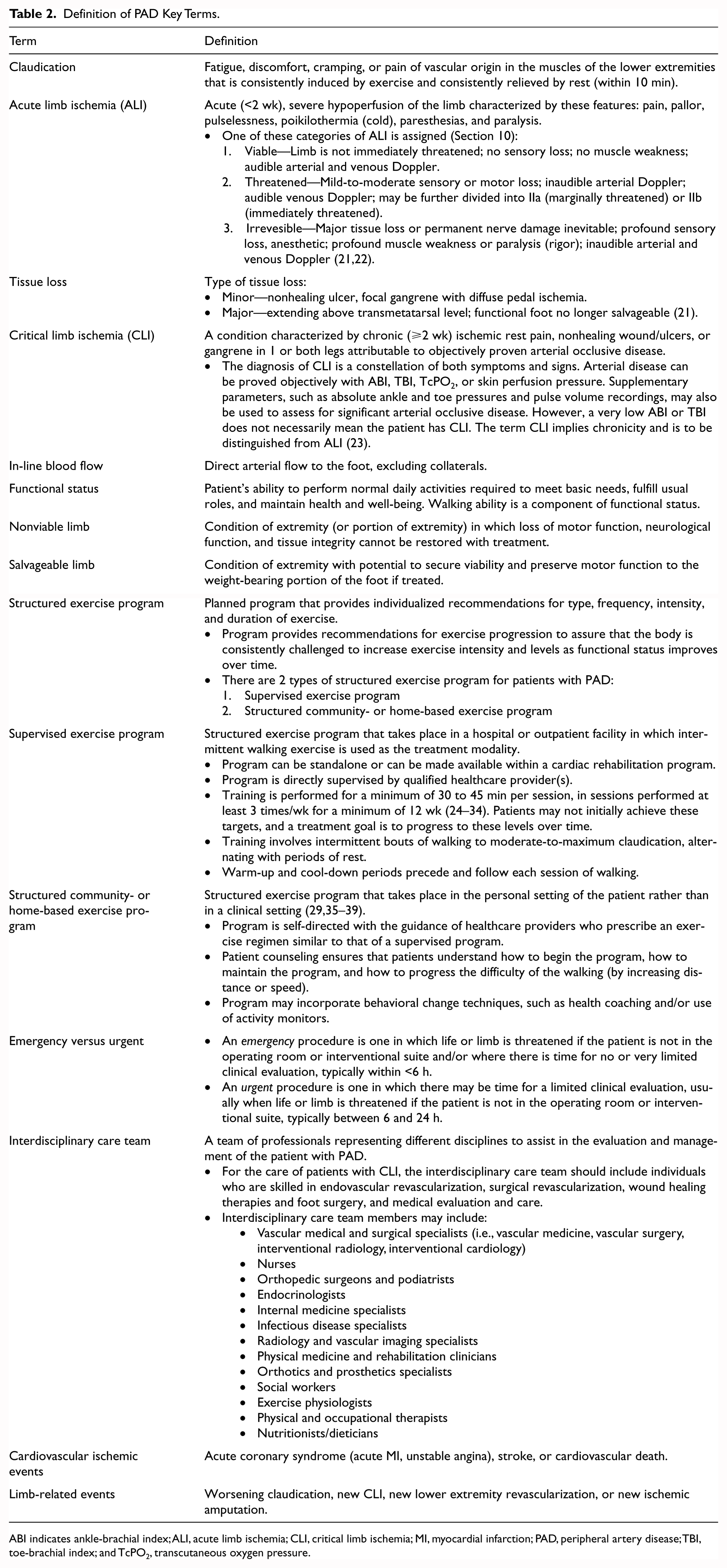
ABI indicates ankle-brachial index; ALI, acute limb ischemia; CLI, critical limb ischemia; MI, myocardial infarction; PAD, peripheral artery disease; TBI, toe-brachial index; and TcPO2, transcutaneous oxygen pressure.
2. Clinical Assessment For PAD
Evaluating the patient at increased risk of PAD (Table 3) begins with the clinical history, review of symptoms, and physical examination. The symptoms and signs of PAD are variable. Patients with PAD may experience the classic symptom of claudication or may present with advanced disease, including CLI. Studies have demonstrated that the majority of patients with confirmed PAD do not have typical claudication but have other non–joint-related limb symptoms (atypical leg symptoms) or are asymptomatic (40,41). Patients with PAD who have atypical leg symptoms or no symptoms may have functional impairment comparable to patients with claudication (42). The vascular examination for PAD includes pulse palpation, auscultation for femoral bruits, and inspection of the legs and feet. Lower extremity pulses are assessed and rated as follows: 0, absent; 1, diminished; 2, normal; or 3, bounding. See Table 4 for history and physical examination findings suggestive of PAD. To confirm the diagnosis of PAD, abnormal physical examination findings must be confirmed with diagnostic testing (Section 3), generally with the ankle-brachial index (ABI) as the initial test.
Patients at Increased Risk of PAD.

AAA indicates abdominal aortic aneurysm; PAD, peripheral artery disease.
History and/or Physical Examination Findings Suggestive of PAD.
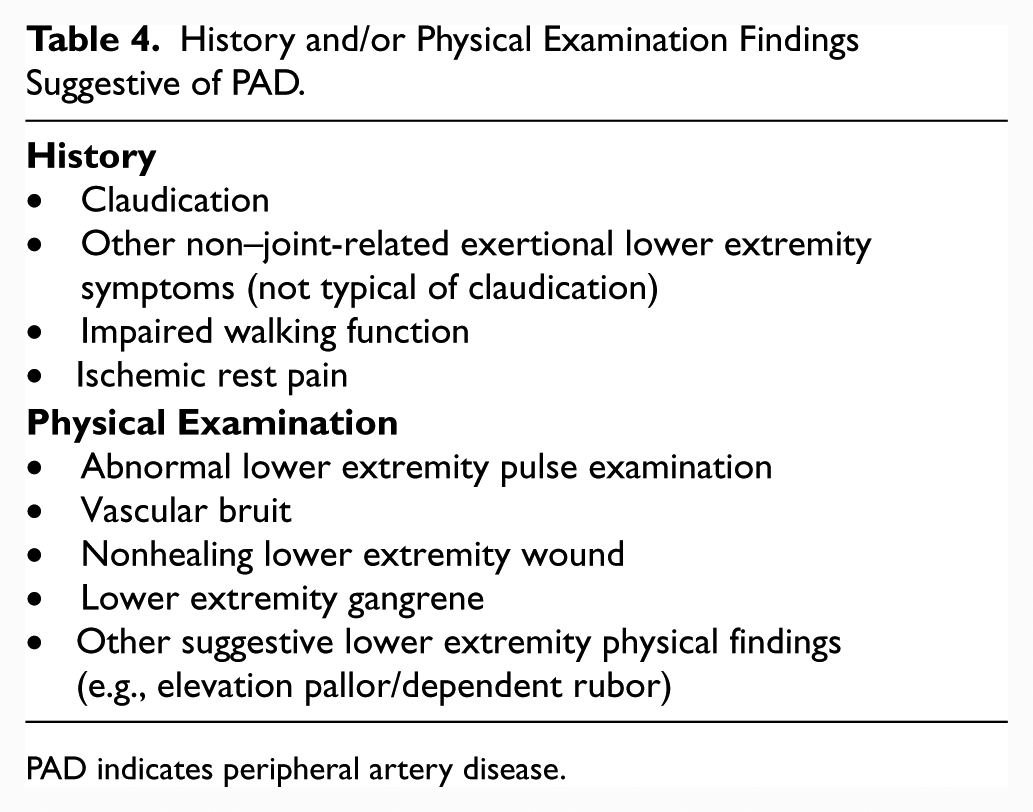
PAD indicates peripheral artery disease.
Patients with confirmed diagnosis of PAD are at increased risk for subclavian artery stenosis (43–45). An inter-arm blood pressure difference of >15 to 20 mm Hg is abnormal and suggestive of subclavian (or innominate) artery stenosis. Measuring blood pressure in both arms identifies the arm with the highest systolic pressure, a requirement for accurate measurement of the ABI (46). Identification of unequal blood pressures in the arms also allows for more accurate measurement of blood pressure in the treatment of hypertension (i.e., blood pressure is taken at the arm with higher measurements).
See Online Data Supplements 1 and 2 for data supporting Section 2.
2.1. History and Physical Examination: Recommendations
Recommendations for History and Physical Examination

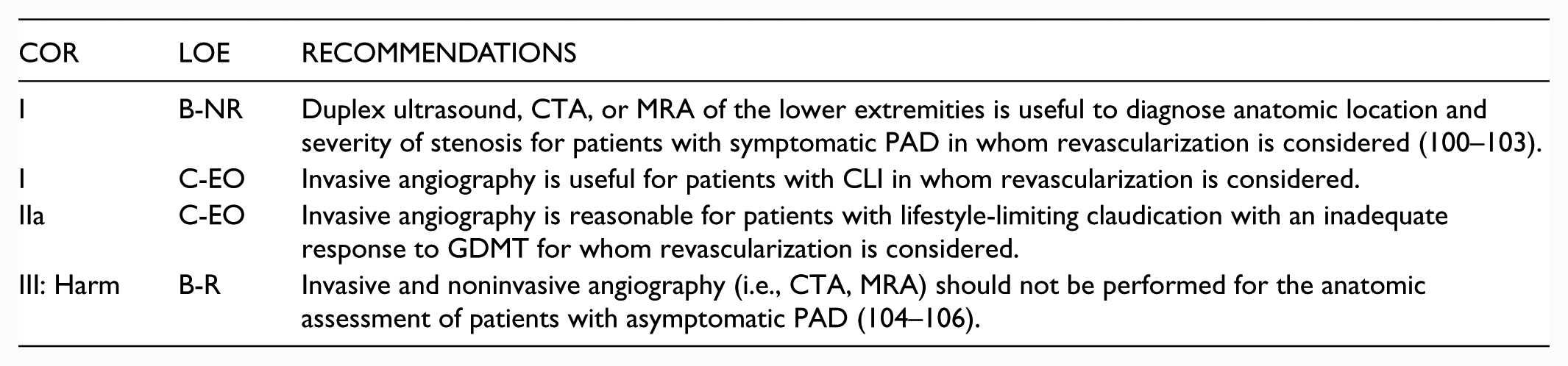
3. Diagnostic Testing For The Patient With Suspected Lower Extremity Pad (Claudication Or CLI): Recommendations
History or physical examination findings suggestive of PAD need to be confirmed with diagnostic testing. The resting ABI is the initial diagnostic test for PAD and may be the only test required to establish the diagnosis and institute GDMT. The resting ABI is a simple, noninvasive test that is obtained by measuring systolic blood pressures at the arms (brachial arteries) and ankles (dorsalis pedis and posterior tibial arteries) in the supine position by using a Doppler device. The ABI of each leg is calculated by dividing the higher of the dorsalis pedis pressure or posterior tibial pressure by the higher of the right or left arm blood pressure (46). Segmental lower extremity blood pressures and Doppler or plethysmographic waveforms (pulse volume recordings) are often performed along with the ABI and can be used to localize anatomic segments of disease (e.g., aortoiliac, femoropopliteal, infrapopliteal) (22,53,54).
Depending on the clinical presentation (e.g., claudication or CLI) and the resting ABI values, additional physiological testing studies may be indicated, including exercise treadmill ABI testing, measurement of the toe-brachial index (TBI), and additional perfusion assessment measures (e.g., transcutaneous oxygen pressure [TcPO2], or skin perfusion pressure [SPP]). Exercise treadmill ABI testing is important to objectively measure functional limitations attributable to leg symptoms and is useful in establishing the diagnosis of lower extremity PAD in the symptomatic patient when resting ABIs are normal or borderline (54–59). The TBI is used to establish the diagnosis of PAD in the setting of noncompressible arteries (ABI >1.40) and may also be used to assess perfusion in patients with suspected CLI. Studies for anatomic imaging assessment (duplex ultrasound, computed tomography angiography [CTA], or magnetic resonance angiography [MRA], invasive angiography) are generally reserved for highly symptomatic patients in whom revascularization is being considered. Depending on the modality, these studies may confer procedural risk.
See Table 5 for alternative causes of leg pain in the patient with normal ABI and physiological testing; Figure 1 for the algorithm on diagnostic testing for suspected PAD and claudication; Table 6 for alternative causes of nonhealing wounds in patients without PAD; Figure 2 for the algorithm on diagnostic testing for suspected CLI; and Online Data Supplements 3 to 7 for data supporting Section 3.
Alternative Diagnoses for Leg Pain or Claudication With Normal Physiological Testing (Not PAD-Related).
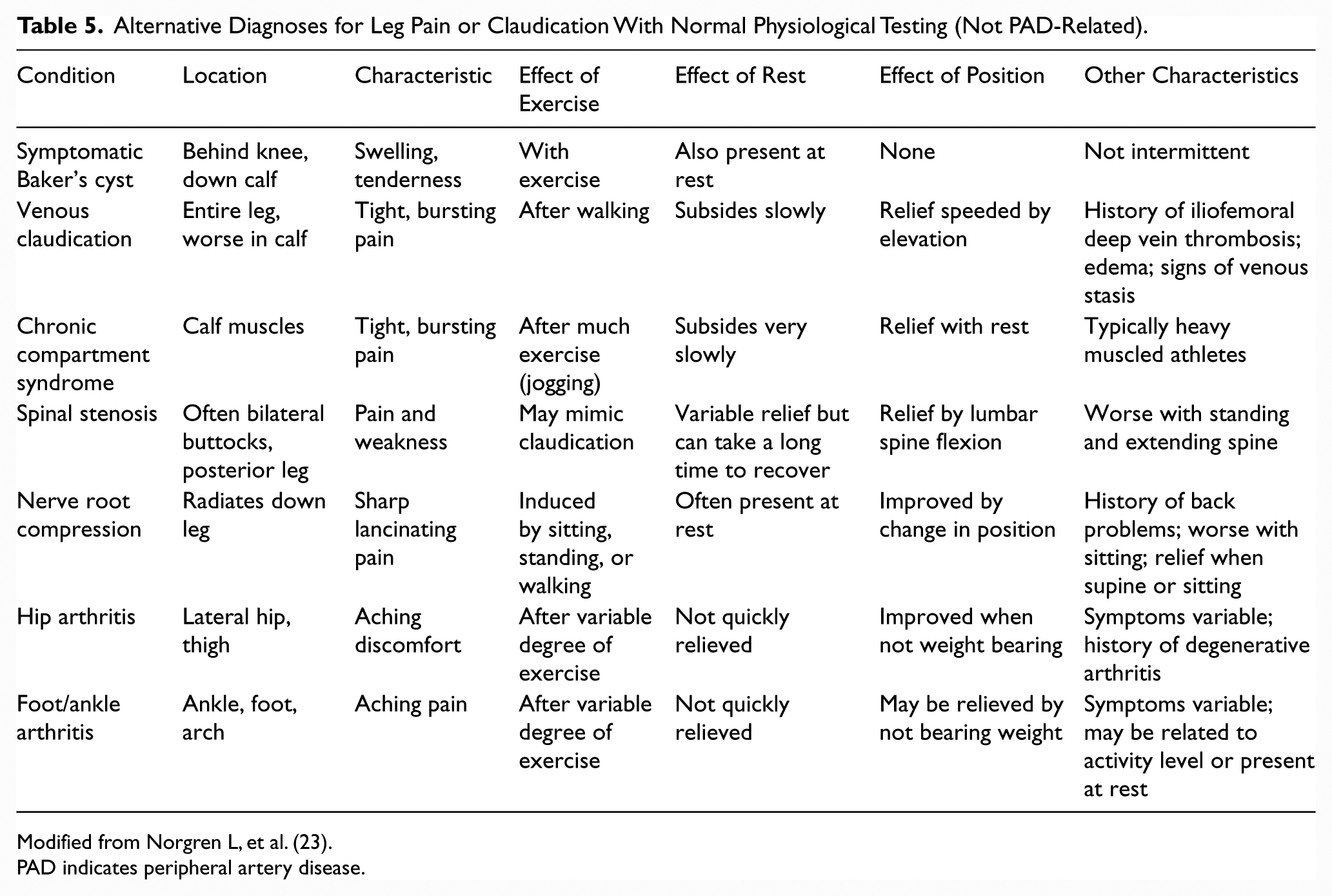
Modified from Norgren L, et al. (23).
PAD indicates peripheral artery disease.
Alternative Diagnoses for Nonhealing Wounds With Normal Physiological Testing (Not PAD-Related).
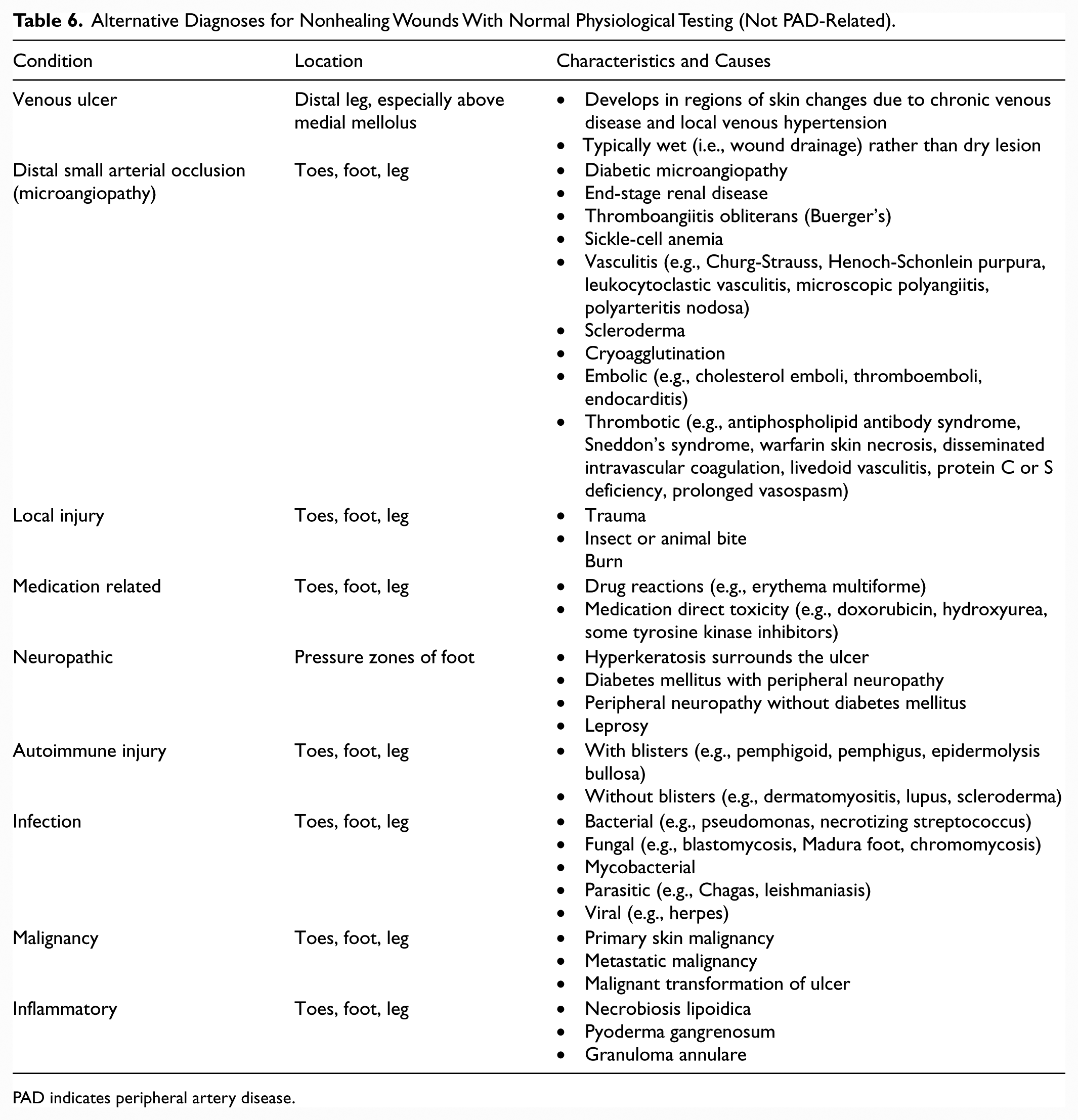
PAD indicates peripheral artery disease.
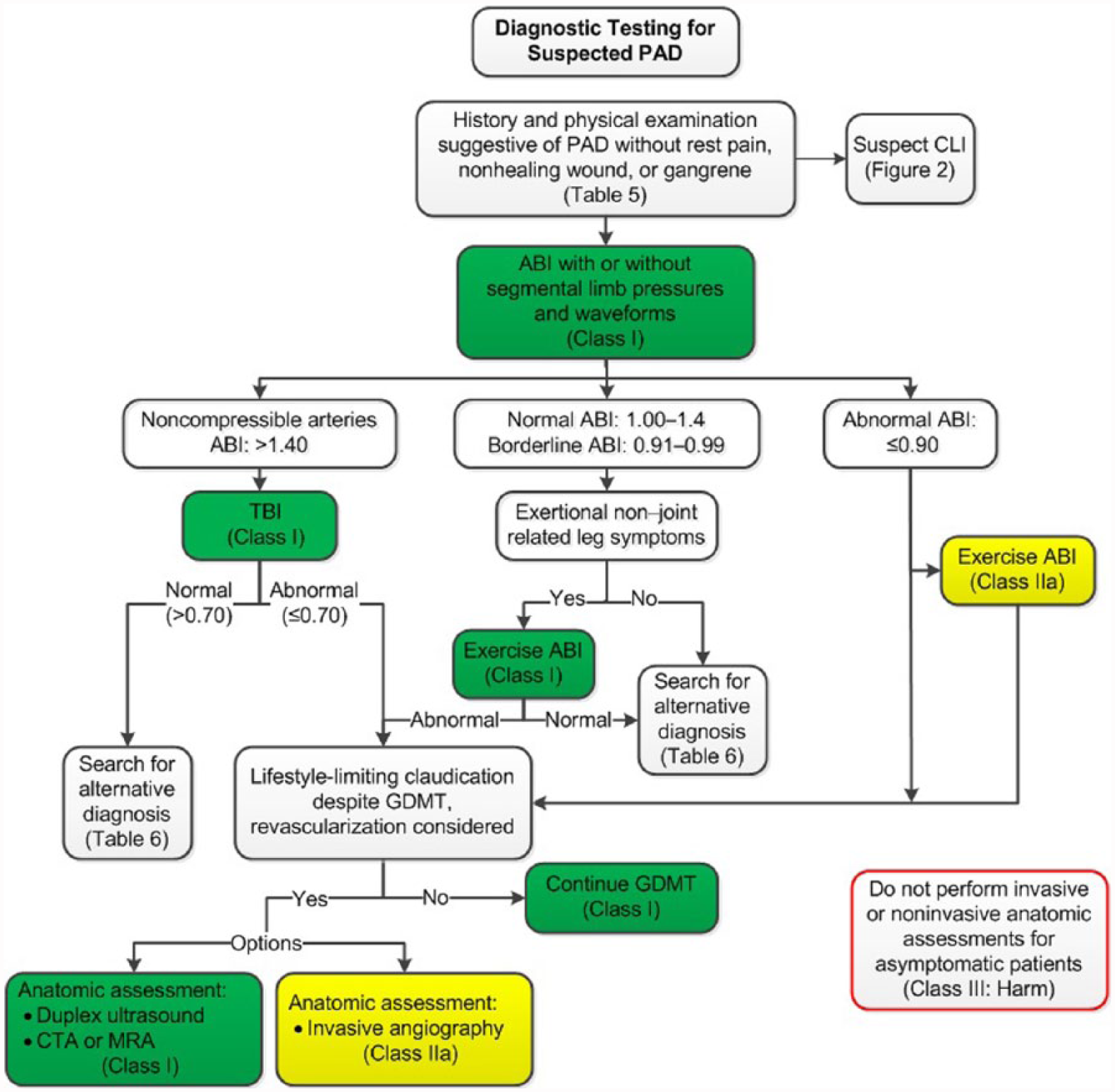
Diagnostic Testing for Suspected PAD.
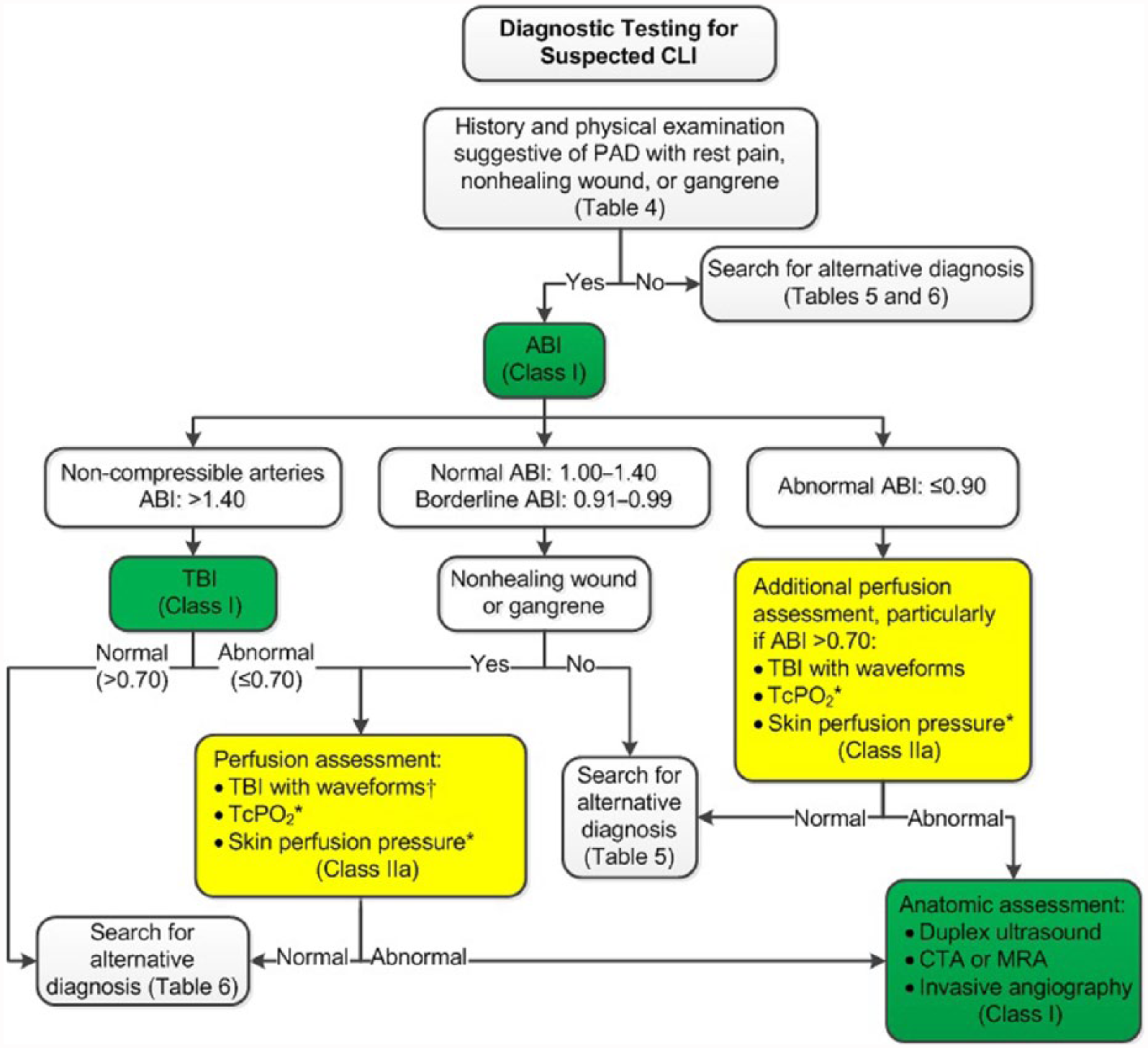
Diagnostic Testing for Suspected CLI.
3.1. Resting ABI for Diagnosing PAD
Recommendations for Resting ABI for Diagnosing PAD
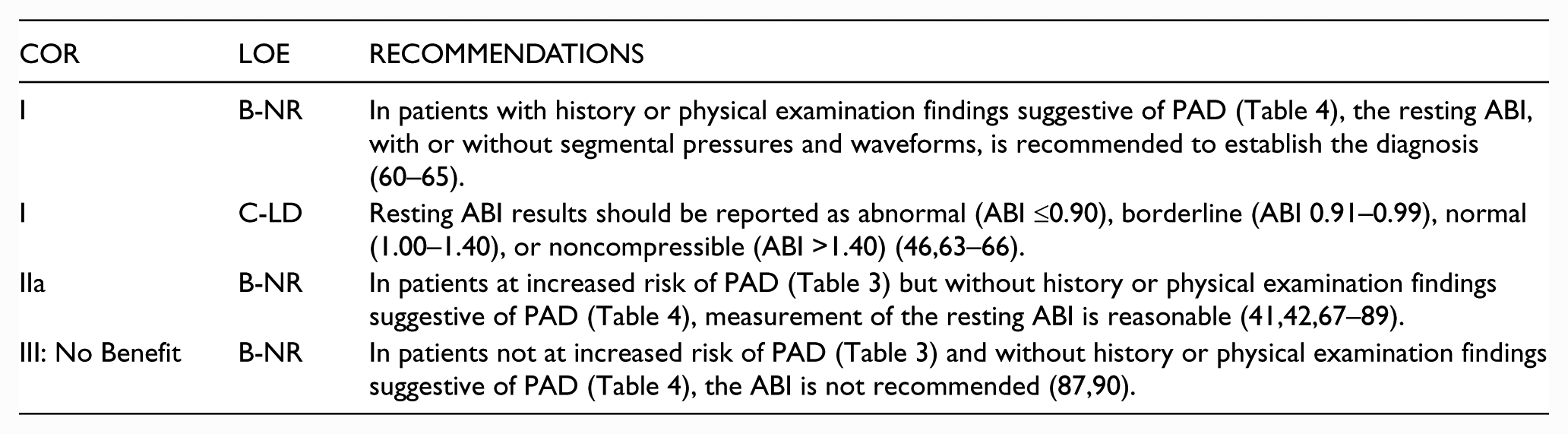
3.2. Physiological Testing
Recommendations for Physiological Testing
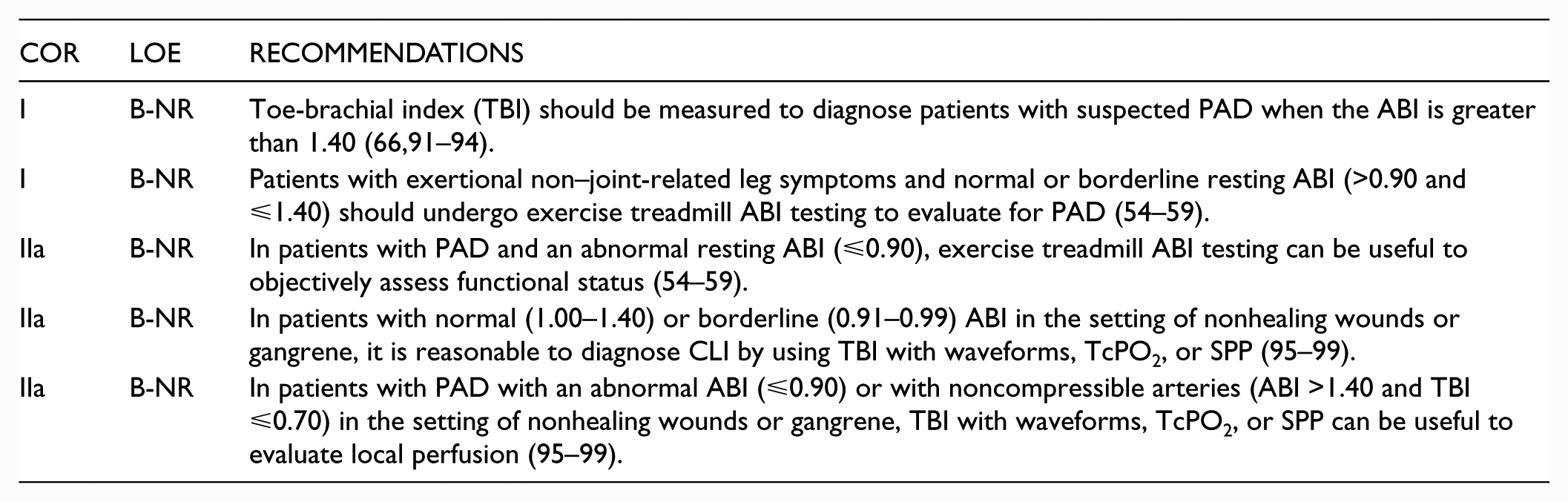
3.3. Imaging for Anatomic Assessment
Recommendations for Imaging for Anatomic Assessment
4. Screening For Atherosclerotic Disease In Other Vascular Beds For The Patient With PAD: Recommendations
See Online Data Supplement 8 for data supporting Section 4.
4.1. Abdominal Aortic Aneurysm
PAD has been recognized as a risk factor for abdominal aortic aneurysm (AAA). In observational studies, the prevalence of AAA (aortic diameter ⩾3 cm) was higher in patients with symptomatic PAD than in the general population (107,108) and in a population of patients with atherosclerotic risk factors (109). The prevalence of AAA among patients with PAD increased with age, beginning in patients ⩾55 years of age, and was highest in patients ⩾75 years of age (107). There are no data on AAA screening in patients with asymptomatic PAD. This section refers to screening patients with symptomatic PAD for AAA. Recommendations for screening the general population with risk factors for AAA (based on age, sex, smoking history, and family history) have been previously published (10).
Recommendation for Abdominal Aortic Aneurysm

4.2. Screening for Asymptomatic Atherosclerosis in Other Arterial Beds (Coronary, Carotid, and Renal Arteries)
The prevalence of atherosclerosis in the coronary, carotid, and renal arteries is higher in patients with PAD than in those without PAD (109–115). However, intensive atherosclerosis risk factor modification in patients with PAD is justified regardless of the presence of disease in other arterial beds. Thus, the only justification for screening for disease in other arterial beds is if revascularization results in a reduced risk of myocardial infarction (MI), stroke, or death, and this has never been shown. Currently, there is no evidence to demonstrate that screening all patients with PAD for asymptomatic atherosclerosis in other arterial beds improves clinical outcome. Intensive treatment of risk factors through GDMT is the principle method for preventing adverse cardiovascular ischemic events from asymptomatic disease in other arterial beds.
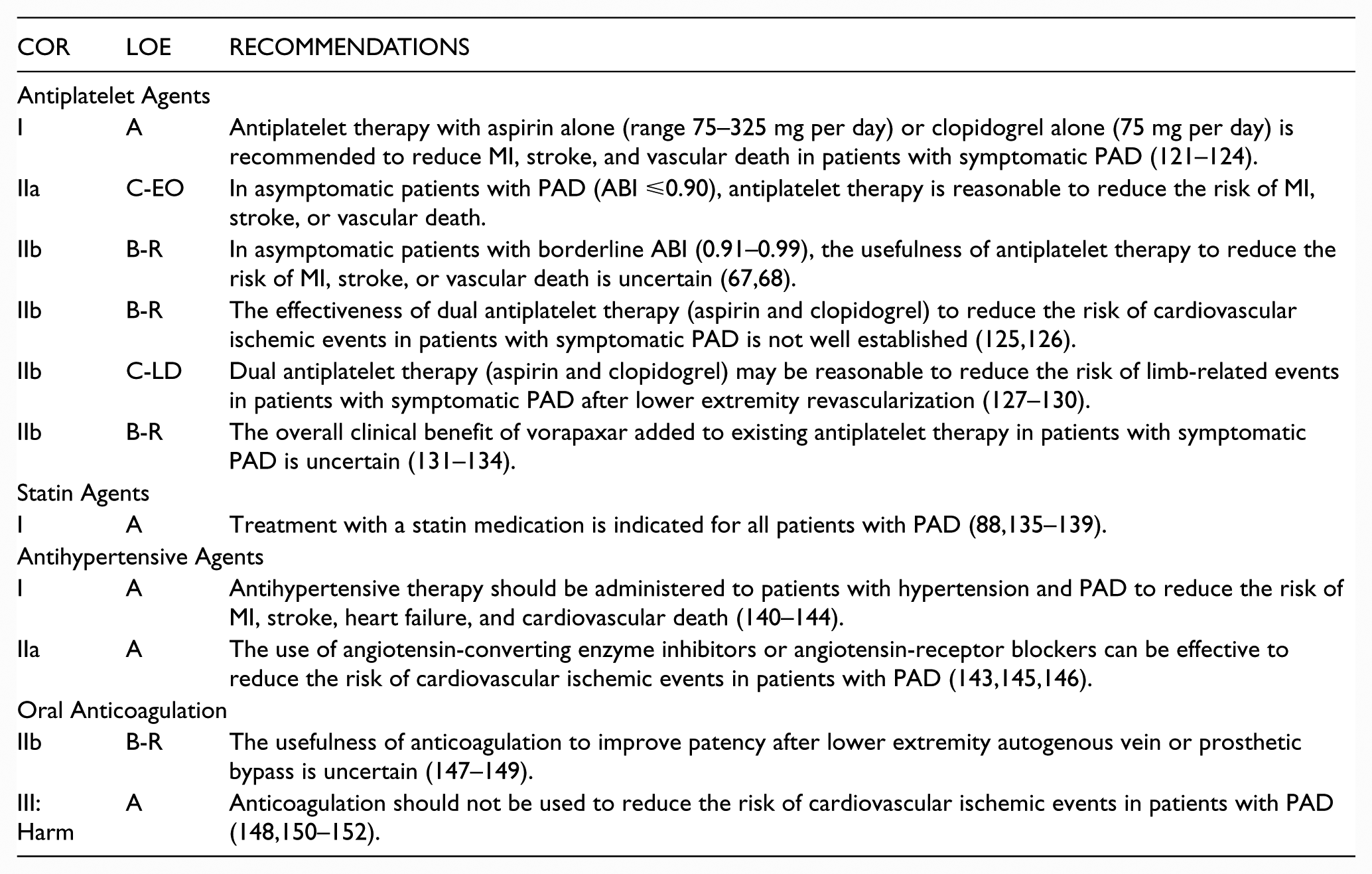
5. Medical Therapy For The Patient With PAD: Recommendations
Patients with PAD should receive a comprehensive program of GDMT, including structured exercise and lifestyle modification, to reduce cardiovascular ischemic events and improve functional status. Smoking cessation is a vital component of care for patients with PAD who continue to smoke. A guideline-based program of pharmacotherapy to reduce cardiovascular ischemic events and limb-related events should be prescribed for each patient with PAD and is customized to individual risk factors, such as whether the patient also has diabetes mellitus. Pharmacotherapy for the patient with PAD includes antiplatelet and statin agents and is customized to additional risk factors, such as whether the patient also has diabetes mellitus or hypertension. Previous studies have demonstrated that patients with PAD are less likely to receive GDMT than patients with other forms of cardiovascular disease, including coronary artery disease (116–118). Cilostazol is an effective medical therapy for treatment of leg symptoms and walking impairment due to claudication (119). However, side effects include headache, diarrhea, dizziness, and palpitations and in 1 trial, 20% of patients discontinued cilostazol within 3 months (120).
See Online Data Supplements 13 to 19 for data supporting Section 5.
5.1. Antiplatelet, Statin, Antihypertensive Agents, and Oral Anticoagulation
Recommendations for Antiplatelet, Statin, and Antihypertensive Agents
5.2. Smoking Cessation
Recommendations for Smoking Cessation

5.3. Glycemic Control
Recommendations for Glycemic Control

5.4. Cilostazol, Pentoxifylline, and Chelation Therapy
Recommendations for Cilostazol, Pentoxifylline, and Chelation Therapy

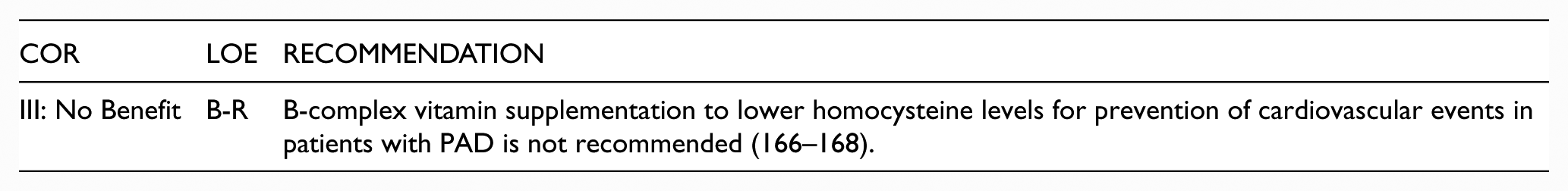
5.5. Homocysteine Lowering
Recommendation for Homocysteine Lowering
5.6. Influenza Vaccination
Recommendation for Influenza Vaccination

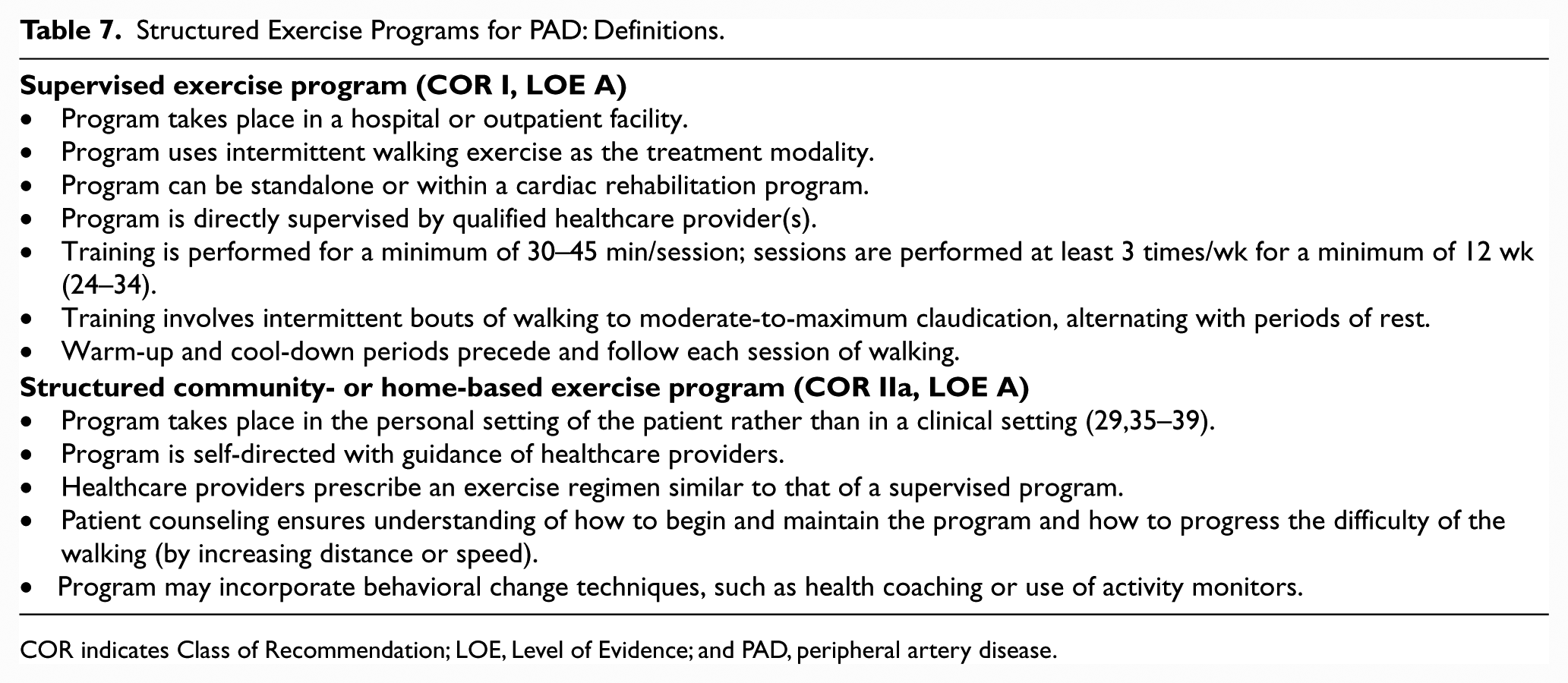
6. Structured Exercise Therapy: Recommendations
Structured exercise therapy is an important element of care for the patient with PAD. Components of structured exercise programs for PAD are outlined in Table 7. The data supporting the efficacy of supervised exercise programs as an initial treatment for claudication continue to develop and remain convincing, building on many earlier RCTs (28–34,36,169,170). Trials with long-term follow-up from 18 months (25,26) to 7 years (24) have demonstrated a persistent benefit of supervised exercise in patients with claudication. The risk–benefit ratio for supervised exercise in PAD is favorable, with an excellent safety profile in patients screened for absolute contraindications to exercise such as exercise-limiting cardiovascular disease, amputation or wheelchair confinement, and other major comorbidities that would preclude exercise (24,27,37,171–174).
Structured Exercise Programs for PAD: Definitions.
COR indicates Class of Recommendation; LOE, Level of Evidence; and PAD, peripheral artery disease.
Interdisciplinary Care Team for PAD.
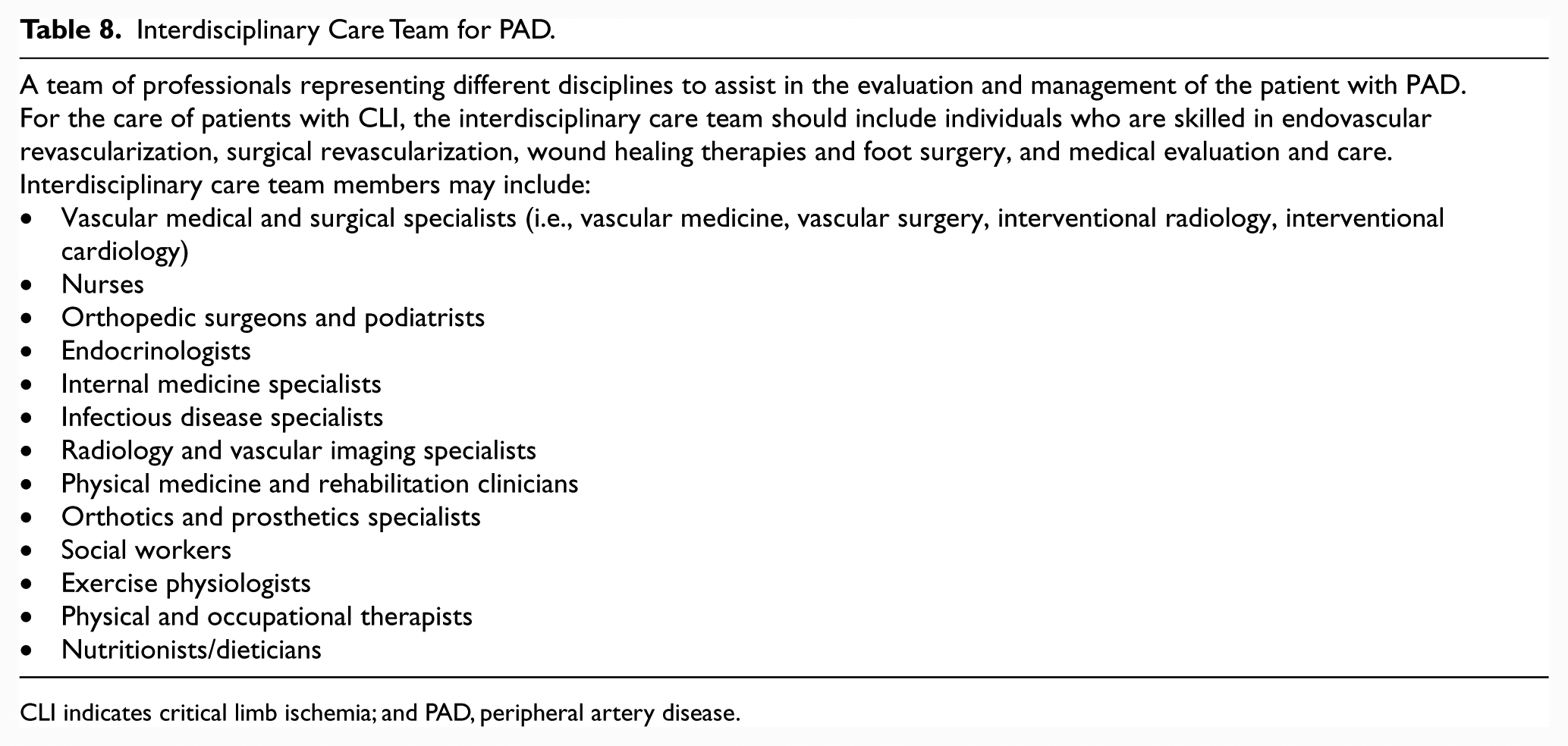
CLI indicates critical limb ischemia; and PAD, peripheral artery disease.
CLI indicates critical limb ischemia.Studies supporting structured community- or home-based programs for patients with PAD are more recent than studies supporting supervised exercise programs and have provided strong evidence in support of the community- or home-based approach (35,37,39,80,86,171). Unstructured community- or home-based walking programs that consist of providing general recommendations to patients with claudication to simply walk more are not efficacious (38).
See Online Data Supplements 32 and 33 for data supporting Section 6.
Recommendations for Structured Exercise Therapy

7. Minimizing Tissue Loss In Patients With PAD Recommendations
Prevention of wounds through patient education, foot examination, and prompt recognition of foot infection is important to minimize tissue loss among patients with PAD. Education includes teaching patients about healthy foot behaviors (e.g., daily inspection of feet, wearing of shoes and socks; avoidance of barefoot walking), the selection of proper footwear, and the importance of seeking medical attention for new foot problems (177). Educational efforts are especially important for patients with PAD who have diabetes mellitus with peripheral neuropathy.
Foot infections (infection of any of the structures distal to the malleoli) may include cellulitis, abscess, fasciitis, tenosynovitis, septic joint space infection, and osteomyelitis. Because of the consequences associated with untreated foot infection—especially in the presence of PAD—clinicians should maintain a high index of suspicion (178). Foot infection is suspected if the patient presents with local pain or tenderness; periwound erythema; periwound edema, induration, or fluctuance; pretibial edema; any discharge (especially purulent); foul odor; visible bone or a wound that probes to bone; or signs of a systemic inflammatory response (including temperature >38°C or <36°C, heart rate >90/min, respiratory rate >20/min or Pa
See Online Data Supplement 34 for data supporting Section 7.
Recommendations for Minimizing Tissue Loss in Patients With PAD

8. Revascularization for Claudication: Recommendations
A minority of patients with claudication (estimated at <10% to 15% over 5 years or more) will progress to CLI (186–189). Therefore, the role of revascularization in claudication is improvement in claudication symptoms and functional status, and consequently in QoL, rather than limb salvage. Revascularization is reasonable when the patient who is being treated with GDMT (including structured exercise therapy) presents with persistent lifestyle-limiting claudication (13,25,26,190,191). Lifestyle-limiting claudication is defined by the patient rather than by any test. It includes impairment of activities of daily living and/or vocational and/or recreational activities due to claudication. An individualized approach to revascularization for claudication is recommended for each patient to optimize outcome. Revascularization is but one component of care for the patient with claudication, inasmuch as each patient should have a customized care plan that also includes medical therapy (Section 5), structured exercise therapy (Section 6), and care to minimize tissue loss (Section 7). If a strategy of revascularization for claudication is undertaken, the revascularization strategy should be evidence based and can include endovascular revascularization, surgery, or both.
Due to the variability of ischemic limb symptoms and impact of these symptoms on functional status and QoL, patients should be selected for revascularization on the basis of severity of their symptoms. Factors to consider include a significant disability as assessed by the patient, adequacy of response to medical and structured exercise therapy, status of comorbid conditions, and a favorable risk–benefit ratio. Patient preferences and goals of care are important considerations in the evaluation for revascularization. The revascularization strategy should have a reasonable likelihood of providing durable relief of symptoms. There should be clear discussion with the patient about expected risks and benefits of revascularization, as well as discussion of the durability of proposed procedures. A general recommendation for revascularization as a treatment option for claudication is provided below followed by specific recommendations for endovascular (Section 8.1.1) and surgical (Section 8.1.2) procedures if a revascularization strategy is undertaken.
See Online Data Supplements 35 to 38 for data supporting Section 8.
8.1. Revascularization for Claudication
Recommendation for Revascularization for Claudication

8.1.1. Endovascular Revascularization for Claudication
Endovascular techniques to treat claudication include balloon dilation (angioplasty), stents, and atherectomy. These techniques continue to involve and now include covered stents, drug-eluting stents, cutting balloons, and drug-coated balloons. The technique chosen for endovascular treatment is related to lesion characteristics (e.g., anatomic location, lesion length, degree of calcification) and operator experience. Assessment of the appropriateness of specific endovascular techniques for specific lesions for the treatment of claudication is beyond the scope of this document.
Revascularization is performed on lesions that are deemed to be hemodynamically significant, and stenoses selected for endovascular treatment should have a reasonable likelihood of limiting perfusion to the distal limb. Stenoses of 50% to 75% diameter by angiography may not be hemodynamically significant, and resting or provoked intravascular pressure measurements may be used to determine whether lesions are significant (192,193). Multiple RCTs have compared endovascular procedures to various combinations of medical treatment with or without supervised or unsupervised exercise programs (13,25,26,190,191,194–206). These trials have used different endpoints and enrolled patients with anatomic disease distribution at different levels. Long-term patency is greater in the aortoiliac than in the femoropopliteal segment. Furthermore, for femoropopliteal disease, durability is diminished with greater lesion length, occlusion rather than stenosis, the presence of multiple and diffuse lesions, poor-quality runoff, diabetes mellitus, chronic kidney disease, renal failure, and smoking (207–210).
Recommendations for Endovascular Revascularization for Claudication

8.1.2. Surgical Revascularization for Claudication
Systematic reviews have concluded that surgical procedures are an effective treatment for claudication and have a positive impact on QoL and walking parameters but have identified sparse evidence supporting the effectiveness of surgery compared with other treatments (12,191,217,218). Although symptom and patency outcomes for surgical interventions may be superior to those for less invasive endovascular treatments, surgical interventions are also associated with greater risk of adverse perioperative events (219–225). Treatment selection should therefore be individualized on the basis of the patient’s goals, perioperative risk, and anticipated benefit. Surgical procedures for claudication are usually reserved for individuals who a) do not derive adequate benefit from nonsurgical therapy, b) have arterial anatomy favorable to obtaining a durable result with surgery, and c) have acceptable risk of perioperative adverse events. Acceptable risk is defined by the individual patient and provider on the basis of symptom severity, comorbid conditions, and appropriate GDMT risk evaluation.
The superficial femoral and proximal popliteal arteries are the most common anatomic sites of stenosis or occlusion among individuals with claudication. Femoral-popliteal bypass is therefore one of the most common surgical procedures for claudication. The type of conduit and site of popliteal artery anastomosis (above versus below knee) are major determinants of outcomes associated with femoral-popliteal bypass. Systematic reviews and meta-analyses have identified a clear and consistent primary patency benefit for autogenous vein versus prosthetic grafts for popliteal artery bypass (226,227).
Recommendations for Surgical Revascularization for Claudication

9. Management of CLI: Recommendations
Patients with CLI are at increased risk of amputation and major cardiovascular ischemic events. Care of the patient with CLI includes evaluation for revascularization and wound healing therapies, with the objective to minimize tissue loss, completely heal wounds, and preserve a functional foot. Medical therapy to prevent cardiovascular ischemic events is also an important component of care for the patient with CLI (Section 5).
See Online Data Supplements 39 and 40 for data supporting Section 9.
9.1. Revascularization for CLI
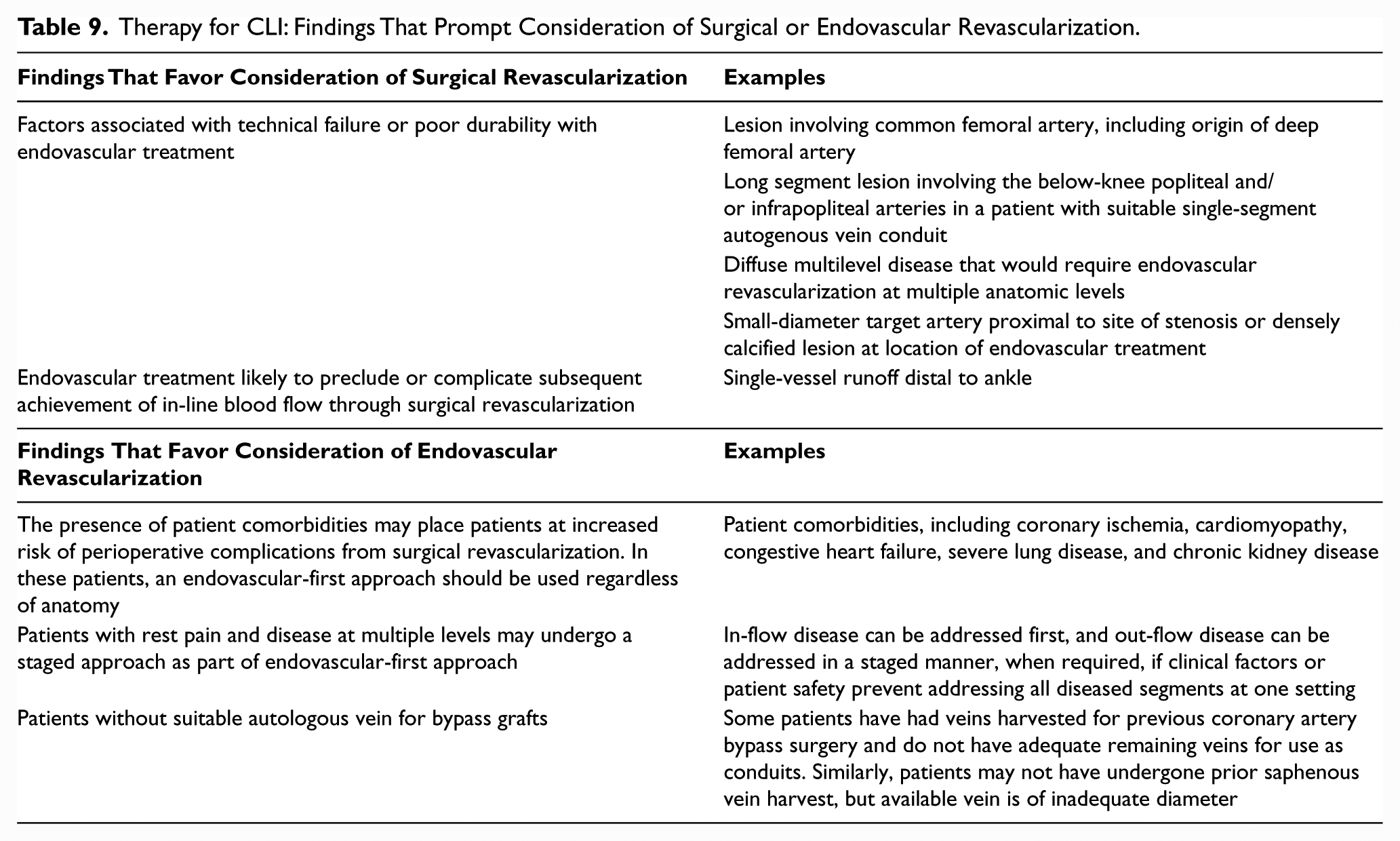
The goal of surgical or endovascular revascularization in CLI is to provide in-line blood flow to the foot through at least 1 patent artery, which will help decrease ischemic pain and allow healing of any wounds, while preserving a functional limb. The BASIL (Bypass versus Angioplasty in Severe Ischemia of the Leg) RCT (242,243) demonstrated that endovascular revascularization is an effective option for patients with CLI as compared with open surgery. The primary endpoint of amputation-free survival was the same in the endovascular and surgical arms. Of note, the endovascular arm used only percutaneous transluminal angioplasty (242,243). Multiple RCTs comparing contemporary surgical and endovascular treatment for patients with CLI are ongoing (16,17,18). Table 9 addresses factors that may prompt an endovascular versus surgical approach to the patient with CLI.
Therapy for CLI: Findings That Prompt Consideration of Surgical or Endovascular Revascularization.
The angiosome concept has been described in the literature and entails establishing direct blood flow to the infrapopliteal artery directly responsible for perfusing the region of the leg or foot with the nonhealing wound. Multiple retrospective studies and 1 small nonrandomized prospective study assessing the efficacy of this concept have been published (245–257). Meta-analyses of these studies found improved wound healing and limb salvage with angiosome-guided therapy but cautioned that the quality of the evidence was low (258,259). Although the angiosome concept is theoretically satisfying, randomized data comparing the establishment of in-line flow versus angiosome-guided therapy have yet to be published. Furthermore, there is no evidence yet to demonstrate the potential benefit of treating additional infrapopliteal arteries once in-line flow has been established in one artery, regardless of angiosome.
Recommendation for Revascularizations for CLI

9.1.1. Endovascular Revascularization for CLI
Recommendations for Endovascular Revascularization for CLI

9.1.2. Surgical Revascularization for CLI
Recommendations for Surgical Revascularization for CLI

9.2. Wound Healing Therapies for CLI
A comprehensive plan for treatment of CLI includes a plan to achieve an intact skin surface on a functional foot. The management of patients with CLI and nonhealing wounds includes coordinated efforts for both revascularization and wound healing among members of an interdisciplinary care team (Table 8). The structure and activities of interdisciplinary care teams for CLI may vary according to several factors, including the local availability of resources. Revascularization is coordinated with the efforts of clinicians who manage foot infections, provide offloading, and achieve complete wound healing, either through medical therapy, surgical options, or a combination of these options.
See Online Data Supplement 34a for a complete list of functions of the interdisciplinary care team.
Recommendations for Wound Healing Therapies for CLI

10. Management of Acute Limb Ischemia: Recommendations
Acute limb ischemia (ALI) is one of the most treatable and potentially devastating presentations of PAD. Timely recognition of arterial occlusion as the cause of an ischemic, cold, painful leg is crucial to successful treatment. The writing committee has used a standard definition of ALI in which symptom duration is <2 weeks (Table 2) (21,22). Category I refers to viable limbs that are not immediately threatened. Category II refers to threatened limbs. Category IIa limbs are marginally threatened and salvageable, if promptly treated. Category IIb are immediately threatened limbs that require immediate revascularization if salvage is to be accomplished. Category III are irreversibly damaged limbs, in which case resultant major tissue loss or permanent nerve damage is inevitable (22).
Patients with ALI should be rapidly evaluated by a vascular specialist if one is available. Depending on local clinical expertise, the vascular specialist may be a vascular surgeon, interventional radiologist, cardiologist, or a general surgeon with specialized training and experience in treating PAD. If such expertise is not locally or rapidly available, there should be strong consideration of transfer of the patient to a facility with such resources. The more advanced the degree of ischemia, the more rapidly the communication (e.g., with regard to potential patient transfer) needs to occur.
ALI is a medical emergency and must be recognized rapidly. The time constraint is due to the period that skeletal muscle will tolerate ischemia—roughly 4 to 6 hours (281). A rapid assessment of limb viability and ability to restore arterial blood flow should be performed by a clinician able to either complete the revascularization or triage the patient (282). Lower extremity symptoms in ALI can include both pain and loss of function. The longer these symptoms are present, the less likely the possibility of limb salvage (283,284). Clinical assessment must include symptom duration, pain intensity, and motor and sensory deficit severity to distinguish a threatened from a nonviable extremity (Figure 3). The bedside assessment includes arterial and venous examination with a handheld continuous-wave Doppler because of the inaccuracy of pulse palpation (22). The loss of Dopplerable arterial signal indicates that the limb is threatened. The absence of both arterial and venous Doppler signal indicates that the limb may be irreversibly damaged (nonsalvageable). Comorbidities should be investigated and managed aggressively, but this must not delay therapy. Even in the setting of rapid and effective revascularization, the 1-year morbidity and mortality rates ALI are high (283,285).
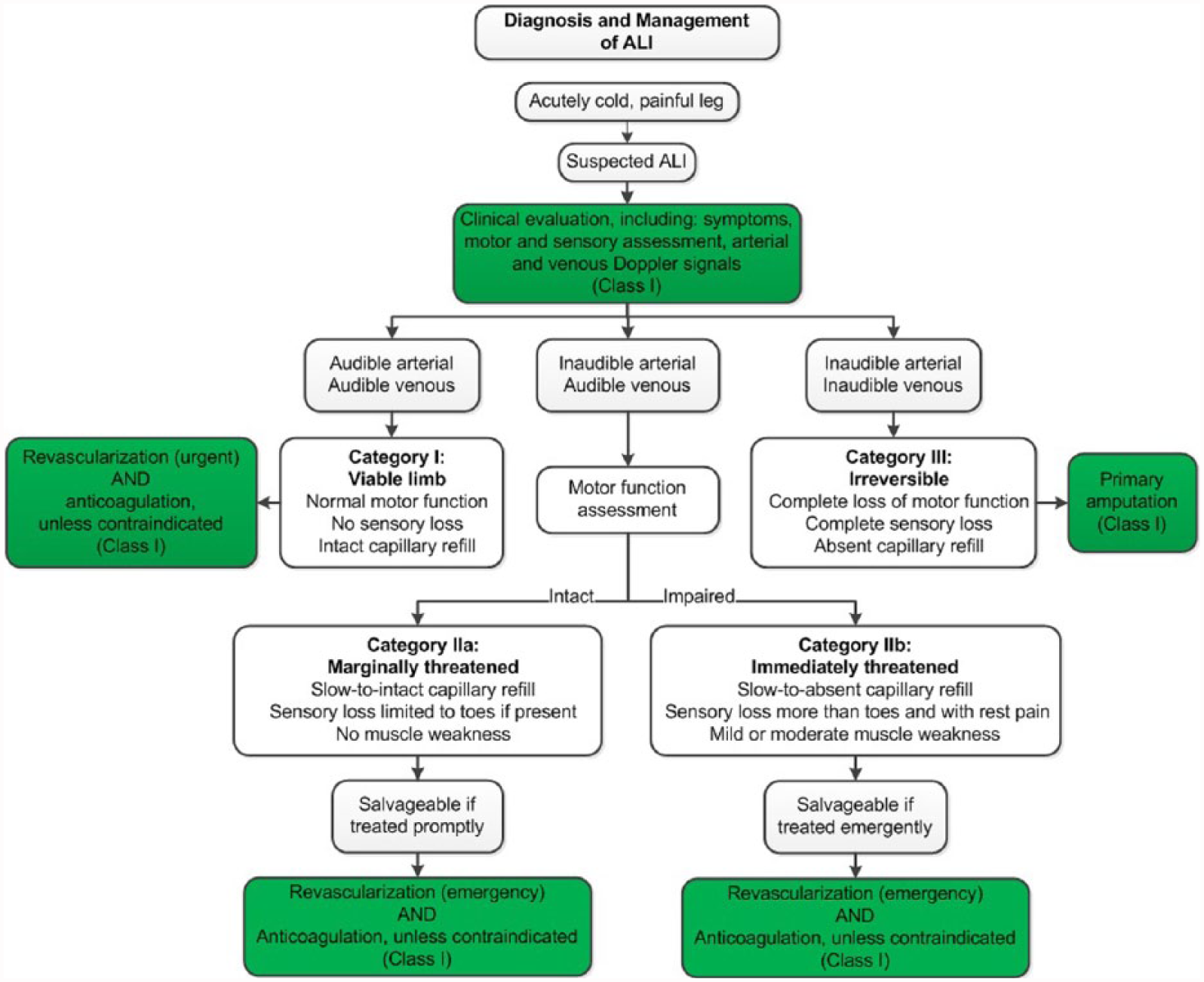
See Figure 3 for the algorithm on diagnosis and management of ALI and Online Data Supplements 45 to 50 for data supporting Section 10.
10.1. Clinical Presentation of ALI
Recommendations for Clinical Presentation of ALI

10.2. Medical Therapy for ALI
Recommendation for ALI Medical Therapy

10.3. Revascularization for ALI
For marginally or immediately threatened limbs (Category IIa and IIb ALI), revascularization should be performed emergently (within 6 hours). For viable limbs (Category I ALI), revascularization should be performed an on urgent basis (within 6–24 hours). The revascularization strategy can range from catheter-directed thrombolysis to surgical thromboembolectomy. Available facilities and clinical expertise are factors that should be considered when determining the revascularization strategy. The technique that will provide the most rapid restoration of arterial flow with the least risk to the patient should be selected. For example, catheter-directed thrombolysis can provide rapid restoration of arterial flow to a viable or marginally threatened limb, particularly in the setting of recent occlusion, thrombosis of synthetic grafts, and stent thrombosis (288). If this is not available locally, surgical options for timely revascularization should be considered, along with the feasibility of timely transfer to a facility with the necessary expertise.
Prolonged duration of ischemia is the most common factor in patients requiring amputation for treatment of ALI. The risks associated with reconstruction outweigh the potential benefit in a limb that is already insensate or immobile because of prolonged ischemia. Patients who have an insensate and immobile limb in the setting of prolonged ischemia (>6 to 8 hours) are unlikely to have potential for limb salvage with revascularization.
Recommendations for Revascularization for ALI

10.4. Diagnostic Evaluation of the Cause of ALI
ALI may be related to underlying PAD (including prior lower extremity bypass graft) or may be related to other conditions that can result in ALI through either thrombotic (e.g., hypercoagulable state) or embolic mechanisms. Treatment of ALI should not be delayed for testing for the underlying cause of the limb ischemia because delay from symptom onset to revascularization is a major determinant of outcome (283,284). The evaluation of a cardiovascular (i.e., embolic) cause for ALI is most useful in the patient without underlying PAD and can be completed after revascularization. Evaluation for cardiovascular cause includes electrocardiogram or additional heart rhythm monitoring to detect atrial fibrillation, electrocardiogram to detect evidence of MI, and echocardiography to further determine whether there is a cardiac etiology for thromboembolism, such as valvular vegetation, left atrial or left ventricular thrombus, or intracardiac shunt.
Recommendations for Diagnostic Evaluation of the Cause of ALI

11. Longitudinal Follow-Up: Recommendations
PAD is a lifelong chronic medical condition. A comprehensive care plan for patients with PAD includes periodic clinical evaluation by a healthcare provider with experience in the care of vascular patients. Ongoing care focuses on cardiovascular risk reduction with medical therapy, optimizing functional status with structured exercise, and, when indicated, revascularization. The care plan is further customized depending on whether the patient has undergone a revascularization procedure.
See Online Data Supplements 51 and 52 for data supporting Section 11.
Recommendations for Longitudinal Follow-Up

12. Evidence Gaps and Future Research Directions
In performing the evidence review and in developing the present guidelines, the writing committee identified the following critical evidence gaps and future directions for PAD-related research:
Basic science and translational studies to better understand the vascular biology of endovascular therapies and bypass grafting and to develop new methods for preventing restenosis after revascularization.
Determination of risk factors for progression from asymptomatic PAD to symptomatic disease, including CLI.
RCTs needed to determine the value of using the ABI to identify asymptomatic patients with PAD for therapies to reduce cardiovascular risk (e.g., antiplatelet agents, statins, and other therapies).
Advancement in PAD diagnostics, such as technologies for simplified yet highly accurate measurement of the ABI and tools for more reliable noninvasive perfusion assessment in CLI.
Comparative-effectiveness studies to determine the optimal antiplatelet therapy (drug or drugs and dosage) for prevention of cardiovascular and limb-related events in patients with PAD.
Development of additional medical therapies for claudication—an area of unmet medical need with a currently limited research pipeline (319).
Studies to investigate the role of dietary intervention, in addition to statin therapy, to improve outcome and modify the natural history of PAD.
Additional research to identify the best community- or home-based exercise programs for patients with PAD to maximize functional status and improve QoL, as well as the role of such exercise programs before or in addition to revascularization.
Development and validation of improved clinical classification systems for PAD that incorporate symptoms, anatomic factors, and patient-specific risk factors and can be used to predict clinical outcome and optimize treatment approach. An example of a recently developed classification system is the Society for Vascular Surgery limb classification system, based on wound, ischemia, and foot infection (WIfI), which has been validated in different populations and may permit more meaningful prognosis in patients with CLI (320–324).
Comparative- and cost-effectiveness studies of the different endovascular technologies for treatment of claudication and CLI, including drug-coated balloons and drug-eluting stents. Studies should include patient-centered endpoints, such as functional parameters, time to wound healing, and QoL, in addition to standard patency-focused outcomes. These studies could then be incorporated into value-based clinical algorithms for approach to revascularization for claudication and CLI.
Additional studies to demonstrate the impact of multisocietal registries on clinical outcomes and appropriate use. At present, these include: the Vascular Quality Initiative (VQI), the National Cardiovascular Data Registry Peripheral Vascular Intervention Registry™ (PVI Registry™), and the National Radiology Data Registry for Interventional Radiology (NRDR). These registries provide an opportunity to obtain “real-world” data on surgical and endovascular procedures for PAD and improve quality by providing feedback to participating centers. Future efforts should incorporate these registries into interventional RCTs and post-marketing studies of PAD-related devices.
13. Advocacy Priorities
The writing committee identified 3 priorities for multisocietal advocacy initiatives to improve health care for patients with PAD.
Presidents and Staff
American College of Cardiology
Richard A. Chazal, MD, FACC, President
Shalom Jacobovitz, Chief Executive Officer
William J. Oetgen, MD, MBA, FACC, Executive Vice President, Science, Education,
Quality, and Publications
Amelia Scholtz, PhD, Publications Manager, Science, Education, Quality, and Publishing
American College of Cardiology/American Heart Association
Katherine Sheehan, PhD, Director, AHA/ACC Guideline Strategy and Operations
Lisa Bradfield, CAE, Director, Guideline Methodology and Policy
Abdul R. Abdullah, MD, Associate Science and Medicine Advisor
Allison Rabinowitz, MPH, Project Manager, Clinical Practice Guidelines
American Heart Association
Steven R. Houser, PhD, FAHA, President
Nancy Brown, Chief Executive Officer
Rose Marie Robertson, MD, FAHA, Chief Science and Medicine Officer
Gayle R. Whitman, PhD, RN, FAHA, FAAN, Senior Vice President,
Office of Science Operations
Comilla Sasson, MD, PhD, FACEP, Vice President, Science and Medicine
Jody Hundley, Production Manager, Scientific Publications, Office of Science Operations
Footnotes
Appendix 3. Abbreviations.
AAA = abdominal aortic aneurysm
ABI = ankle-brachial index
ALI = acute limb ischemia
CLI = critical limb ischemia
GDMT = guideline-directed management and therapy
MRA = magnetic resonance angiography
PAD = peripheral artery disease
RCT = randomized controlled trial
SPP = skin perfusion pressure
TBI = toe-brachial index
TcPO2 = transcutaneous oxygen pressure
QoL = quality of life
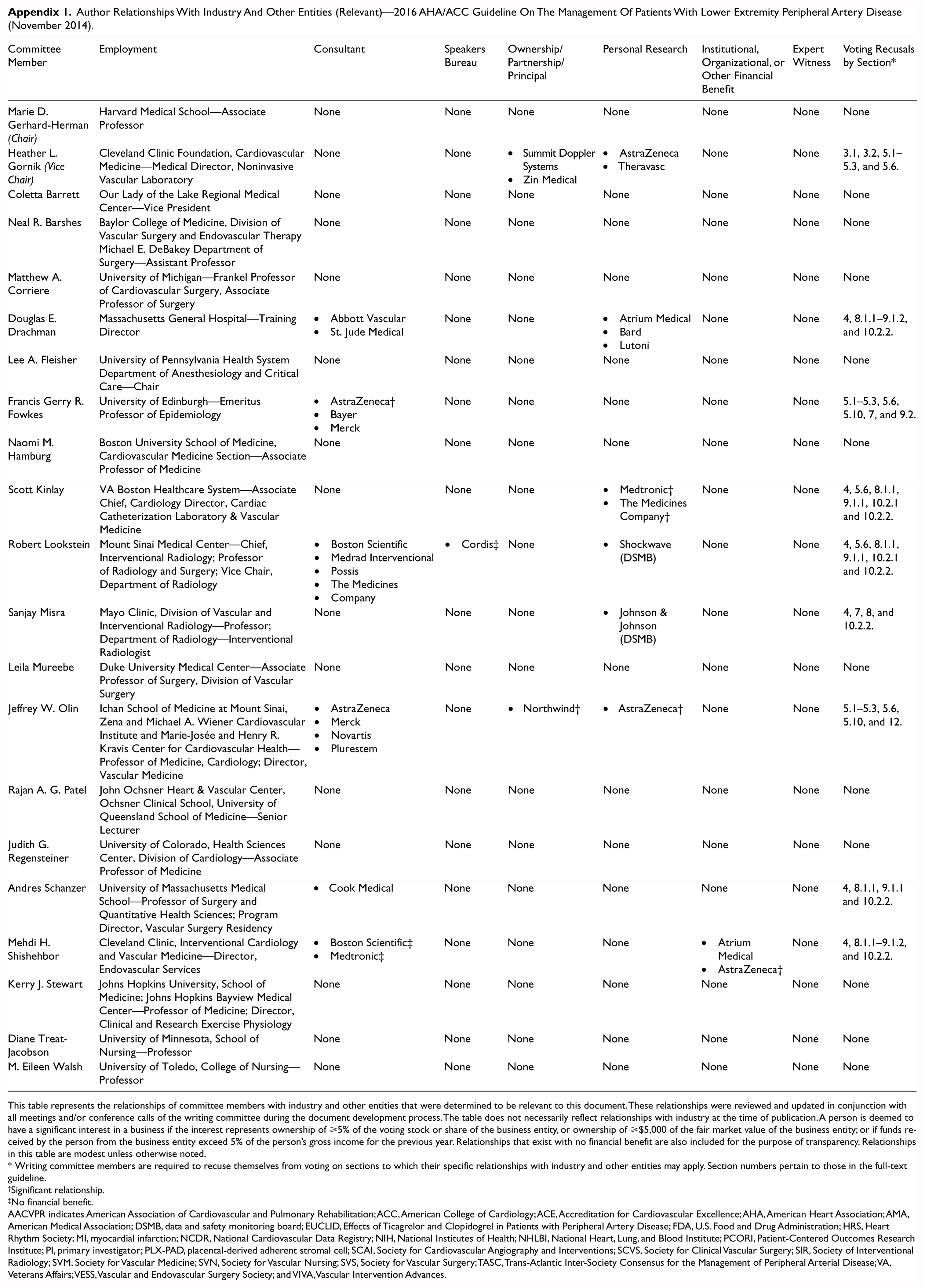
Appendix 1.
Author Relationships With Industry And Other Entities (Relevant)—2016 AHA/ACC Guideline On The Management Of Patients With Lower Extremity Peripheral Artery Disease (November 2014).
| Committee Member | Employment | Consultant | Speakers Bureau | Ownership/Partnership/Principal | Personal Research | Institutional, Organizational, or Other Financial Benefit | Expert Witness | Voting Recusals by Section* |
|---|---|---|---|---|---|---|---|---|
| Marie D. Gerhard-Herman (Chair) | Harvard Medical School—Associate Professor | None | None | None | None | None | None | None |
| Heather L. Gornik (Vice Chair) | Cleveland Clinic Foundation, Cardiovascular Medicine—Medical Director, Noninvasive Vascular Laboratory | None | None | Summit Doppler Systems Zin Medical |
• AstraZeneca Theravasc |
None | None | 3.1, 3.2, 5.1–5.3, and 5.6. |
| Coletta Barrett | Our Lady of the Lake Regional Medical Center—Vice President | None | None | None | None | None | None | None |
| Neal R. Barshes | Baylor College of Medicine, Division of Vascular Surgery and Endovascular Therapy Michael E. DeBakey Department of Surgery—Assistant Professor | None | None | None | None | None | None | None |
| Matthew A. Corriere | University of Michigan—Frankel Professor of Cardiovascular Surgery, Associate Professor of Surgery | None | None | None | None | None | None | None |
| Douglas E. Drachman | Massachusetts General Hospital—Training Director | • Abbott Vascular • St. Jude Medical |
None | None | • Atrium Medical • Bard • Lutoni |
None | None | 4, 8.1.1–9.1.2, and 10.2.2. |
| Lee A. Fleisher | University of Pennsylvania Health System Department of Anesthesiology and Critical Care—Chair | None | None | None | None | None | None | None |
| Francis Gerry R. Fowkes | University of Edinburgh—Emeritus Professor of Epidemiology | • AstraZeneca†
• Bayer • Merck |
None | None | None | None | None | 5.1–5.3, 5.6, 5.10, 7, and 9.2. |
| Naomi M. Hamburg | Boston University School of Medicine, Cardiovascular Medicine Section—Associate Professor of Medicine | None | None | None | None | None | None | None |
| Scott Kinlay | VA Boston Healthcare System—Associate Chief, Cardiology Director, Cardiac Catheterization Laboratory & Vascular Medicine | None | None | None | • Medtronic†
• The Medicines Company† |
None | None | 4, 5.6, 8.1.1, 9.1.1, 10.2.1 and 10.2.2. |
| Robert Lookstein | Mount Sinai Medical Center—Chief, Interventional Radiology; Professor of Radiology and Surgery; Vice Chair, Department of Radiology | • Boston Scientific • Medrad Interventional • Possis • The Medicines • Company |
• Cordis‡ | None | • Shockwave (DSMB) | None | None | 4, 5.6, 8.1.1, 9.1.1, 10.2.1 and 10.2.2. |
| Sanjay Misra | Mayo Clinic, Division of Vascular and Interventional Radiology—Professor; Department of Radiology—Interventional Radiologist | None | None | None | • Johnson & Johnson (DSMB) | None | None | 4, 7, 8, and 10.2.2. |
| Leila Mureebe | Duke University Medical Center—Associate Professor of Surgery, Division of Vascular Surgery | None | None | None | None | None | None | None |
| Jeffrey W. Olin | Ichan School of Medicine at Mount Sinai, Zena and Michael A. Wiener Cardiovascular Institute and Marie-Josée and Henry R. Kravis Center for Cardiovascular Health—Professor of Medicine, Cardiology; Director, Vascular Medicine | • AstraZeneca • Merck • Novartis • Plurestem |
None | Northwind† | AstraZeneca† | None | None | 5.1–5.3, 5.6, 5.10, and 12. |
| Rajan A. G. Patel | John Ochsner Heart & Vascular Center, Ochsner Clinical School, University of Queensland School of Medicine—Senior Lecturer | None | None | None | None | None | None | None |
| Judith G. Regensteiner | University of Colorado, Health Sciences Center, Division of Cardiology—Associate Professor of Medicine | None | None | None | None | None | None | None |
| Andres Schanzer | University of Massachusetts Medical School—Professor of Surgery and Quantitative Health Sciences; Program Director, Vascular Surgery Residency | Cook Medical | None | None | None | None | None | 4, 8.1.1, 9.1.1 and 10.2.2. |
| Mehdi H. Shishehbor | Cleveland Clinic, Interventional Cardiology and Vascular Medicine—Director, Endovascular Services | • Boston Scientific‡
• Medtronic‡ |
None | None | None | • Atrium Medical • AstraZeneca† |
None | 4, 8.1.1–9.1.2, and 10.2.2. |
| Kerry J. Stewart | Johns Hopkins University, School of Medicine; Johns Hopkins Bayview Medical Center—Professor of Medicine; Director, Clinical and Research Exercise Physiology | None | None | None | None | None | None | None |
| Diane Treat-Jacobson | University of Minnesota, School of Nursing—Professor | None | None | None | None | None | None | None |
| M. Eileen Walsh | University of Toledo, College of Nursing—Professor | None | None | None | None | None | None | None |
This table represents the relationships of committee members with industry and other entities that were determined to be relevant to this document. These relationships were reviewed and updated in conjunction with all meetings and/or conference calls of the writing committee during the document development process. The table does not necessarily reflect relationships with industry at the time of publication. A person is deemed to have a significant interest in a business if the interest represents ownership of ⩾5% of the voting stock or share of the business entity, or ownership of ⩾$5,000 of the fair market value of the business entity; or if funds received by the person from the business entity exceed 5% of the person’s gross income for the previous year. Relationships that exist with no financial benefit are also included for the purpose of transparency. Relationships in this table are modest unless otherwise noted.
Writing committee members are required to recuse themselves from voting on sections to which their specific relationships with industry and other entities may apply. Section numbers pertain to those in the full-text guideline.
Significant relationship.
No financial benefit.
AACVPR indicates American Association of Cardiovascular and Pulmonary Rehabilitation; ACC, American College of Cardiology; ACE, Accreditation for Cardiovascular Excellence; AHA, American Heart Association; AMA, American Medical Association; DSMB, data and safety monitoring board; EUCLID, Effects of Ticagrelor and Clopidogrel in Patients with Peripheral Artery Disease; FDA, U.S. Food and Drug Administration; HRS, Heart Rhythm Society; MI, myocardial infarction; NCDR, National Cardiovascular Data Registry; NIH, National Institutes of Health; NHLBI, National Heart, Lung, and Blood Institute; PCORI, Patient-Centered Outcomes Research Institute; PI, primary investigator; PLX-PAD, placental-derived adherent stromal cell; SCAI, Society for Cardiovascular Angiography and Interventions; SCVS, Society for Clinical Vascular Surgery; SIR, Society of Interventional Radiology; SVM, Society for Vascular Medicine; SVN, Society for Vascular Nursing; SVS, Society for Vascular Surgery; TASC, Trans-Atlantic Inter-Society Consensus for the Management of Peripheral Arterial Disease; VA, Veterans Affairs; VESS, Vascular and Endovascular Surgery Society; and VIVA, Vascular Intervention Advances.
Appendix 2.
Reviewer Relationships With Industry And Other Entities (Comprehensive)—2016 AHA/ACC Guideline On The Management Of Patients With Lower Extremity Peripheral Artery Disease (March 2016).
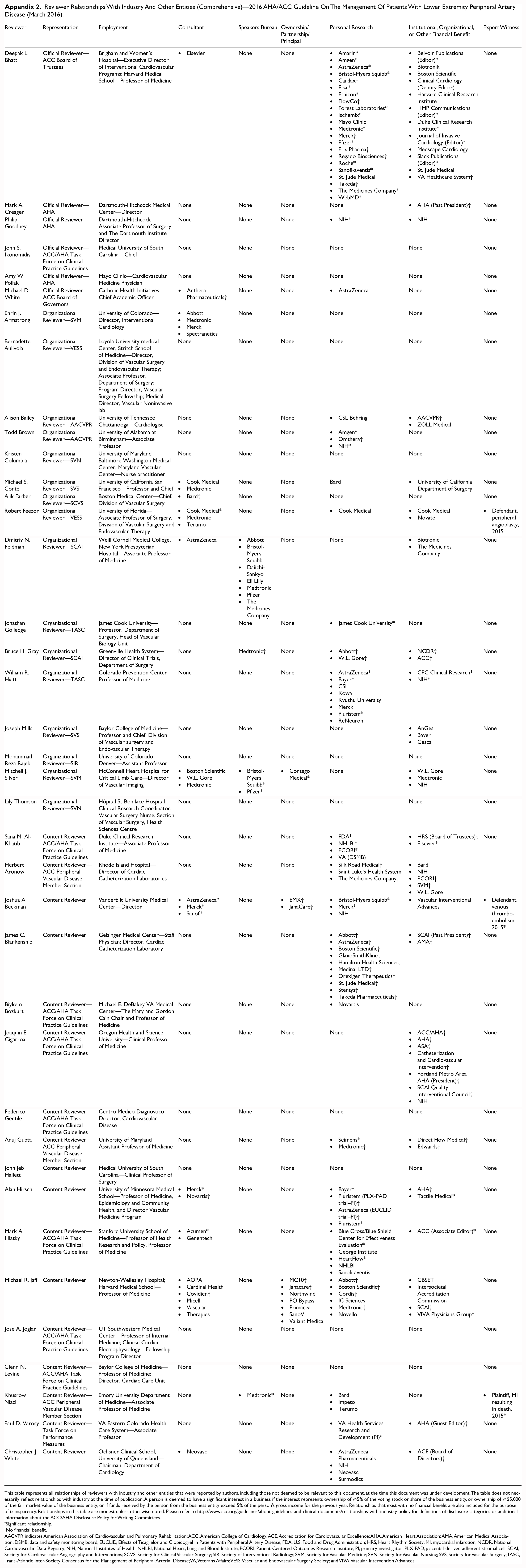
| Reviewer | Representation | Employment | Consultant | Speakers Bureau | Ownership/Partnership/Principal | Personal Research | Institutional, Organizational, or Other Financial Benefit | Expert Witness |
|---|---|---|---|---|---|---|---|---|
| Deepak L. Bhatt | Official Reviewer—ACC Board of Trustees | Brigham and Women’s Hospital—Executive Director of Interventional Cardiovascular Programs; Harvard Medical School—Professor of Medicine | Elsevier | None | None | • Amarin*
• Amgen* • AstraZeneca* • Bristol-Myers Squibb* • Cardax† • Eisai* • Ethicon* • FlowCo† • Forest Laboratories* • Ischemix* • Mayo Clinic • Medtronic* • Merck† • Pfizer* • PLx Pharma† • Regado Biosciences† • Roche* • Sanofi-aventis* • St. Jude Medical • Takeda† • The Medicines Company* • WebMD* |
• Belvoir Publications (Editor)*
• Biotronik • Boston Scientific • Clinical Cardiology (Deputy Editor)† • Harvard Clinical Research Institute • HMP Communications (Editor)* • Duke Clinical Research Institute* • Journal of Invasive Cardiology (Editor)* • Medscape Cardiology • Slack Publications (Editor)* • St. Jude Medical • VA Healthcare System† |
None |
| Mark A. Creager | Official Reviewer—AHA | Dartmouth-Hitchcock Medical Center—Director | None | None | None | None | • AHA (Past President)† | None |
| Philip Goodney | Official Reviewer—AHA | Dartmouth-Hitchcock—Associate Professor of Surgery and The Dartmouth Institute Director | None | None | None | • NIH* | • NIH | None |
| John S. Ikonomidis | Official Reviewer—ACC/AHA Task Force on Clinical Practice Guidelines | Medical University of South Carolina—Chief | None | None | None | None | None | None |
| Amy W. Pollak | Official Reviewer—AHA | Mayo Clinic—Cardiovascular Medicine Physician | None | None | None | None | None | None |
| Michael D. White | Official Reviewer—ACC Board of Governors | Catholic Health Initiatives—Chief Academic Officer | Anthera Pharmaceuticals† | None | None | AstraZeneca† | None | None |
| Ehrin J. Armstrong | Organizational Reviewer—SVM | University of Colorado—Director, Interventional Cardiology | • Abbott • Medtronic • Merck • Spectranetics |
None | None | None | None | None |
| Bernadette Aulivola | Organizational Reviewer—VESS | Loyola University medical Center, Stritch School of Medicine—Director, Division of Vascular Surgery and Endovascular Therapy; Associate Professor, Department of Surgery; Program Director, Vascular Surgery Fellowship; Medical Director, Vascular Noninvasive lab | None | None | None | None | None | None |
| Alison Bailey | Organizational Reviewer—AACVPR | University of Tennessee Chattanooga—Cardiologist | None | None | None | • CSL Behring | • AACVPR†
• ZOLL Medical |
None |
| Todd Brown | Organizational Reviewer—AACVPR | University of Alabama at Birmingham—Associate Professor | None | None | None | • Amgen*
• Omthera† • NIH* |
None | None |
| Kristen Columbia | Organizational Reviewer—SVN | University of Maryland Baltimore Washington Medical Center, Maryland Vascular Center—Nurse practitioner | None | None | None | None | None | None |
| Michael S. Conte | Organizational Reviewer—SVS | University of California San Francisco—Professor and Chief | • Cook Medical Medtronic |
None | None | Bard | • University of California Department of Surgery | None |
| Alik Farber | Organizational Reviewer—SCVS | Boston Medical Center—Chief, Division of Vascular Surgery | • Bard† | None | None | None | None | None |
| Robert Feezor | Organizational Reviewer—VESS | University of Florida—Associate Professor of Surgery, Division of Vascular Surgery and Endovascular Therapy | • Cook Medical*
• Medtronic • Terumo |
None | None | Cook Medical | • Cook Medical Novate |
• Defendant, peripheral angioplasty, 2015 |
| Dmitriy N. Feldman | Organizational Reviewer—SCAI | Weill Cornell Medical College, New York Presbyterian Hospital—Associate Professor of Medicine | • AstraZeneca | • Abbott • Bristol-Myers Squibb† • Daiichi-Sankyo • Eli Lilly • Medtronic • Pfizer • The Medicines Company |
None | None | • Biotronic • The Medicines Company |
None |
| Jonathan Golledge | Organizational Reviewer—TASC | James Cook University—Professor, Department of Surgery, Head of Vascular Biology Unit | None | None | None | James Cook University* | None | None |
| Bruce H. Gray | Organizational Reviewer—SCAI | Greenville Health System—Director of Clinical Trials, Department of Surgery | None | Medtronic† | None | • Abbott†
• W.L. Gore† |
• NCDR†
• ACC† |
None |
| William R. Hiatt | Organizational Reviewer—TASC | Colorado Prevention Center—Professor of Medicine | None | None | None | • AstraZeneca*
• Bayer* • CSI • Kowa • Kyushu University • Merck • Pluristem* • ReNeuron |
• CPC Clinical Research*
• NIH* |
None |
| Joseph Mills | Organizational Reviewer—SVS | Baylor College of Medicine—Professor and Chief, Division of Vascular surgery and Endovascular Therapy | None | None | None | None | • AnGes • Bayer • Cesca |
None |
| Mohammad Reza Rajebi | Organizational Reviewer—SIR | University of Colorado Denver—Assistant Professor | None | None | None | None | None | None |
| Mitchell J. Silver | Organizational Reviewer—SVM | McConnell Heart Hospital for Critical Limb Care—Director of Vascular Imaging | • Boston Scientific • W.L. Gore • Medtronic |
• Bristol-Myers Squibb*
• Pfizer* |
• Contego Medical* | None | • W.L. Gore • Medtronic • NIH |
None |
| Lily Thomson | Organizational Reviewer—SVN | Hôpital St-Boniface Hospital—Clinical Research Coordinator, Vascular Surgery Nurse, Section of Vascular Surgery, Health Sciences Centre | None | None | None | None | None | None |
| Sana M. Al-Khatib | Content Reviewer—ACC/AHA Task Force on Clinical Practice Guidelines | Duke Clinical Research Institute—Associate Professor of Medicine | None | None | None | • FDA*
• NHLBI* • PCORI* • VA (DSMB) |
• HRS (Board of Trustees)†
• Elsevier* |
None |
| Herbert Aronow | Content Reviewer—ACC Peripheral Vascular Disease Member Section | Rhode Island Hospital—Director of Cardiac Catheterization Laboratories | None | None | None | • Silk Road Medical†
• Saint Luke’s Health System • The Medicines Company† |
• Bard • NIH • PCORI† • SVM† • W.L. Gore |
|
| Joshua A. Beckman | Content Reviewer | Vanderbilt University Medical Center—Director | • AstraZeneca*
|
None | • EMX†
|
• Bristol-Myers Squibb*
|
• Vascular Interventional Advances | • Defendant, venous thrombo-embolism, 2015* |
| James C. Blankenship | Content Reviewer | Geisinger Medical Center—Staff Physician; Director, Cardiac Catheterization Laboratory | None | None | None | • Abbott†
|
• SCAI (Past President)†
|
None |
| Biykem Bozkurt | Content Reviewer—ACC/AHA Task Force on Clinical Practice Guidelines | Michael E. DeBakey VA Medical Center—The Mary and Gordon Cain Chair and Professor of Medicine | None | None | None | • Novartis | None | None |
| Joaquin E. Cigarroa | Content Reviewer—ACC/AHA Task Force on Clinical Practice Guidelines | Oregon Health and Science University—Clinical Professor of Medicine | None | None | None | None | • ACC/AHA†
|
None |
| Federico Gentile | Content Reviewer—ACC/AHA Task Force on Clinical Practice Guidelines | Centro Medico Diagnostico—Director, Cardiovascular Disease | None | None | None | None | None | None |
| Anuj Gupta | Content Reviewer—ACC Peripheral Vascular Disease Member Section | University of Maryland—Assistant Professor of Medicine | None | None | None | • Seimens*
|
• Direct Flow Medical†
|
None |
| John Jeb Hallett | Content Reviewer | Medical University of South Carolina—Clinical Professor of Surgery | None | None | None | None | None | None |
| Alan Hirsch | Content Reviewer | University of Minnesota Medical School—Professor of Medicine, Epidemiology and Community Health, and Director Vascular Medicine Program | • Merck*
|
None | None | • Bayer*
|
• AHA†
|
None |
| Mark A. Hlatky | Content Reviewer—ACC/AHA Task Force on Clinical Practice Guidelines | Stanford University School of Medicine—Professor of Health Research and Policy, Professor of Medicine | • Acumen*
|
None | None | • Blue Cross/Blue Shield Center for Effectiveness Evaluation*
|
• ACC (Associate Editor)* | None |
| Michael R. Jaff | Content Reviewer | Newton-Wellesley Hospital; Harvard Medical School—Professor of Medicine | • AOPA |
None | • MC10†
|
• Abbott†
|
• CBSET |
None |
| José A. Joglar | Content Reviewer—ACC/AHA Task Force on Clinical Practice Guidelines | UT Southwestern Medical Center—Professor of Internal Medicine; Clinical Cardiac Electrophysiology—Fellowship Program Director | None | None | None | None | None | None |
| Glenn N. Levine | Content Reviewer—ACC/AHA Task Force on Clinical Practice Guidelines | Baylor College of Medicine—Professor of Medicine; Director, Cardiac Care Unit | None | None | None | None | None | None |
| Khusrow Niazi | Content Reviewer—ACC Peripheral Vascular Disease Member Section | Emory University Department of Medicine—Associate Professor of Medicine | None | • Medtronic* | None | • Bard |
None | Plaintiff, MI resulting in death, 2015* |
| Paul D. Varosy | Content Reviewer—Task Force on Performance Measures | VA Eastern Colorado Health Care System—Associate Professor | None | None | None | • VA Health Services Research and Development (PI)* | • AHA (Guest Editor)† | None |
| Christopher J. White | Content Reviewer | Ochsner Clinical School, University of Queensland—Chairman, Department of Cardiology | • Neovasc | None | None | • AstraZeneca Pharmaceuticals |
• ACE (Board of Directors)† | None |
This table represents all relationships of reviewers with industry and other entities that were reported by authors, including those not deemed to be relevant to this document, at the time this document was under development. The table does not necessarily reflect relationships with industry at the time of publication. A person is deemed to have a significant interest in a business if the interest represents ownership of ⩾5% of the voting stock or share of the business entity, or ownership of ⩾$5,000 of the fair market value of the business entity; or if funds received by the person from the business entity exceed 5% of the person’s gross income for the previous year. Relationships that exist with no financial benefit are also included for the purpose of transparency. Relationships in this table are modest unless otherwise noted. Please refer to http://www.acc.org/guidelines/about-guidelines-and-clinical-documents/relationships-with-industry-policy for definitions of disclosure categories or additional information about the ACC/AHA Disclosure Policy for Writing Committees.
Significant relationship.
No financial benefit.
AACVPR indicates American Association of Cardiovascular and Pulmonary Rehabilitation; ACC, American College of Cardiology; ACE, Accreditation for Cardiovascular Excellence; AHA, American Heart Association; AMA, American Medical Association; DSMB, data and safety monitoring board; EUCLID, Effects of Ticagrelor and Clopidogrel in Patients with Peripheral Artery Disease; FDA, U.S. Food and Drug Administration; HRS, Heart Rhythm Society; MI, myocardial infarction; NCDR, National Cardiovascular Data Registry; NIH, National Institutes of Health; NHLBI, National Heart, Lung, and Blood Institute; PCORI, Patient-Centered Outcomes Research Institute; PI, primary investigator; PLX-PAD, placental-derived adherent stromal cell; SCAI, Society for Cardiovascular Angiography and Interventions; SCVS, Society for Clinical Vascular Surgery; SIR, Society of Interventional Radiology; SVM, Society for Vascular Medicine; SVN, Society for Vascular Nursing; SVS, Society for Vascular Surgery; TASC, Trans-Atlantic Inter-Society Consensus for the Management of Peripheral Arterial Disease; VA, Veterans Affairs; VESS, Vascular and Endovascular Surgery Society; and VIVA, Vascular Intervention Advances.
This document was approved by the American College of Cardiology Board of Trustees in October 2016, the American Heart Association Science Advisory and Coordinating Committee in September 2016, and the American Heart Association Executive Committee in October 2016.
The American College of Cardiology requests that this document be cited as follows: Gerhard-Herman MD, Gornik HL, Barrett C, Barshes NR, Corriere MA, Drachman DE, Fleisher LA, Fowkes FGR, Hamburg NM, Kinlay S, Lookstein R, Misra S, Mureebe L, Olin JW, Patel RAG, Regensteiner JG, Schanzer A, Shishehbor MH, Stewart KJ, Treat-Jacobson D, Walsh ME. 2016 AHA/ACC guideline on the management of patients with lower extremity peripheral artery disease: executive summary: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. J Am Coll Cardiol 2017; 69: 1465–1508.
This article has been copublished in the Journal of the American College of Cardiology and Circulation and reprinted in Vascular Medicine.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

