Abstract
Transcatheter aortic valve replacement (TAVR) is now an accepted pathway for aortic valve replacement for patients who are at prohibitive, severe and intermediate risk for traditional aortic valve surgery. However, with this rising uptrend and adaptation of this new technology, vascular complications and their management remain an Achilles heel for percutaneous aortic valve replacement. The vascular complications are an independent predictor of mortality for patients undergoing TAVR. Early recognition of these complications and appropriate management is paramount. In this article, we review the most commonly encountered vascular complications associated with currently approved TAVR devices and their optimal percutaneous management techniques.
CME Accreditation Statement
The University of Virginia School of Medicine designates this journal-based CME activity for a maximum of 1 AMA PRA Category 1 Credit™ per article. Physicians should claim only the credit commensurate with the extent of their participation in the activity. Learners are expected to read this article along with any references and supporting material as appropriate, and complete the online post-test questions with an 80% pass rate to receive credit. Post-test questions are accessed through the member portal on the Society for Vascular Medicine (SVM) website (www.vascularmed.org). Please note that CME credits are only available to members of SVM with a journal subscription. This activity expires 2 years after the publication date, on June 1, 2019.
Heather Gornik has disclosed research support from AstraZeneca, intellectual property rights from Summit Doppler Systems, Inc, and intellectual property rights and stock/ownership from FlexLife Health (proceeds donated). Aditya Sharma has disclosed research support from National Institute of Health Sciences, AstraZeneca, Biomet Biologics, Portola Pharmaceuticals, and Pedra Technology. Valerie Clark has no financial affiliations to disclose.
Introduction
In the past decade, the growth of transcatheter aortic valve replacement (TAVR) has been exponential; approximately 200,000 procedures have been performed worldwide, and in the US over 26,000 were performed between 2012 and 2014.1,2 TAVR remains an important option for patients with severe aortic stenosis who are inoperable 3 or at high surgical risk,4–6 and the procedure was recently approved for those at intermediate surgical risk. 7 Despite evolution in device technology, including smaller delivery sheath profiles, vascular complications remain relevant and are reported in 4.2–5.6% of TAVRs. 1 In this article, we review optimal access techniques and the incidence, prognosis, predictors, prevention and management of TAVR-associated vascular complications.
Procedural technique
TAVR devices
Currently there are two US Food and Drug Administration (FDA)-approved TAVR platforms: the balloon-expandable Edwards SAPIEN S3 and Edwards SAPIEN XT™ valves (Edwards Lifesciences Inc., Irvine, CA, USA) and the self-expanding Medtronic CoreValve® Evolut™ R System valves (Medtronic, Minneapolis, MN, USA). The Evolut R (23, 26, 29, 31-mm valve sizes) and Edwards S3 valves (20, 23, 26- mm valve sizes) require a 14-Fr sheath while the Edwards 29-mm valves require a 16-Fr sheath. The Edwards SAPIEN XT and S3 valves are approved for treating severe native valve aortic stenosis in patients at inoperable, high or intermediate risk for death or complications with surgical aortic valve replacement (SAVR), whereas the Edwards SAPIEN XT valve is also approved for severe stenosis of bioprosthetic aortic valves or valve deployment through transapical route. The Medtronic CoreValve Evolut R is only approved in the inoperable and high risk cohorts. 8 Refer to the online appendix (Supplementary Tables A and B; approved TAVR sheaths and guidewires).
Vascular access
Transfemoral (TF) access
The common femoral artery (CFA) is the access site of choice for most TAVR procedures, employed in nearly 89%. 9 The transfemoral approach is most often utilized because it provides direct arterial access, is the least invasive approach for doing so and is associated with the lowest morbidity relative to other approaches. Transapical (TA) TAVR was the earliest developed alternative access route for a TAVR and is still performed occasionally when other access routes are not feasible.1,10,11 Mortality is lower with TF-TAVR compared with TA-TAVR7,12,13 (Figure 1).
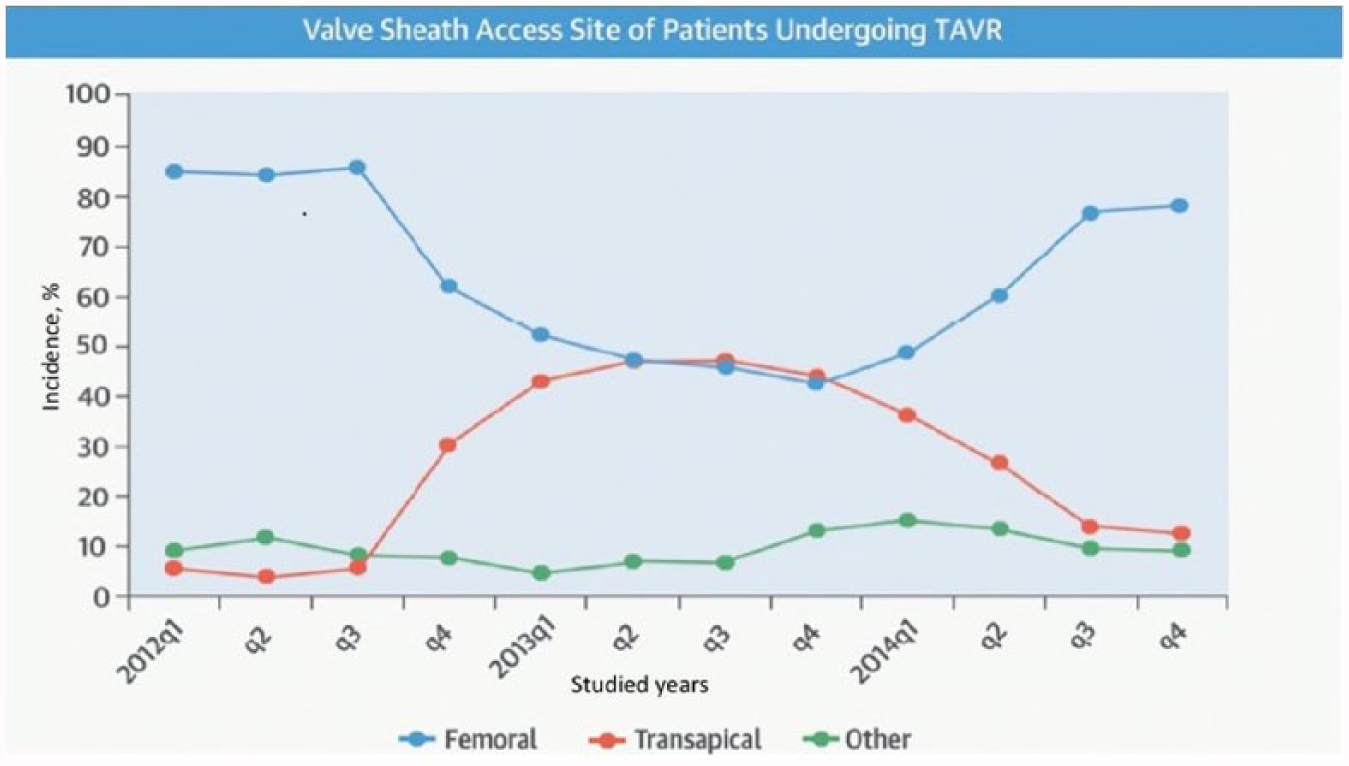
Transcatheter aortic valve replacement (TAVR) vascular access site in the Transcatheter Valve Therapy (TVT) Registry, 2012–2014. The changing valve sheath access site over time has resulted from multiple factors, including FDA instructions for use and changing technology. Republished with permission of Elsevier, from ref. 1; permission conveyed through Copyright Clearance Center, Inc. Copyright © 2015 American College of Cardiology Foundation and The Society of Thoracic Surgeons.
Non-femoral/alternate access for TAVR
Severe aortic stenosis patients with femoral arteries less than or equal to 5 mm should be evaluated for TAVR through non-femoral accesses or, if possible, surgical cut down to access iliac arteries. Acceptable alternate accesses for TAVR include transapical, direct aortic, carotid artery, 14 and subclavian/axillary; 15 in some centers, use of the subclavian artery for vascular access approaches 20% of cases performed. 16 Transcaval access is reserved for patients who have no viable access, where the abdominal aorta is accessed from the inferior vena cava followed by advancement of the TAVR delivery system through the aortic puncture site, and on completing the procedure, closing of the iatrogenic arteriovenous (AV) fistula with a nitinol vascular plug. 17
Regardless of the site accessed, programmatic success hinges on applying a consistent, if not algorithmic, approach to vascular access site selection, transit and closure (Figure 2).
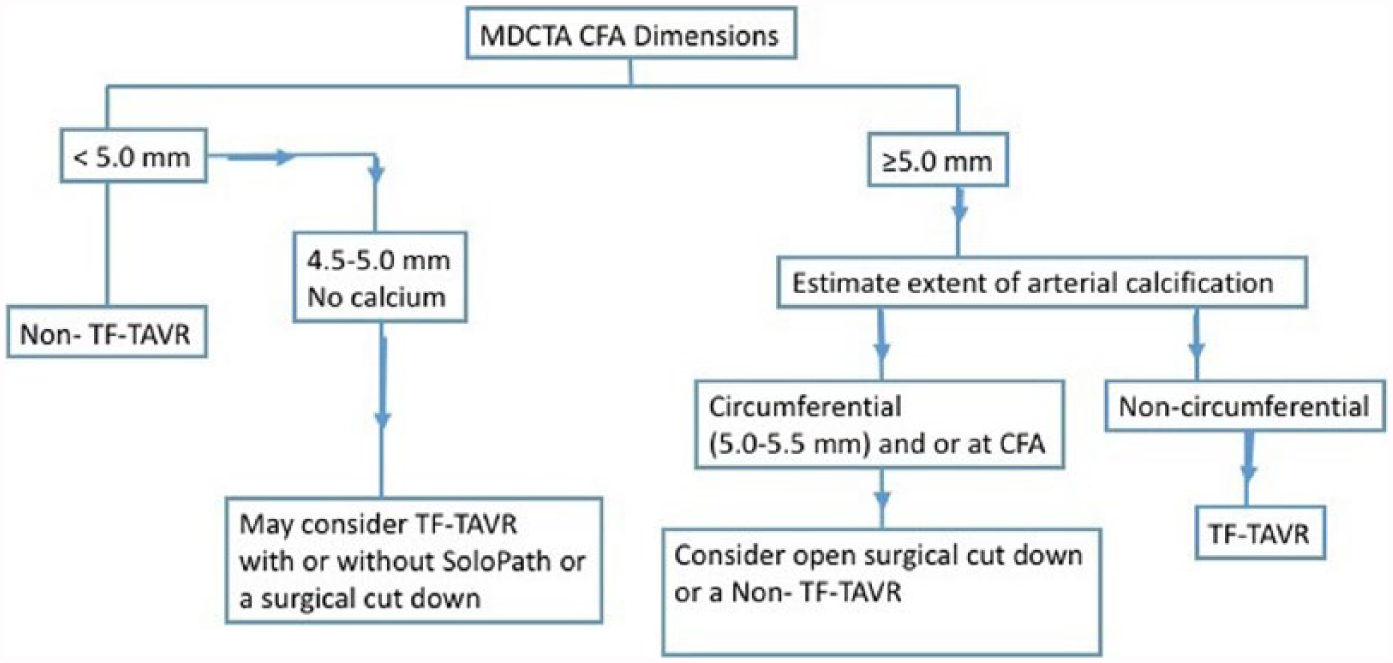
Flow diagram for patient selection to undergo transfemoral or non-transfemoral access TAVR. This is based on expert opinions, device recommended usage guidelines, and the standard operating practice of our institute. (MDCTA, multidetector computed tomography angiography; CFA, common femoral artery; TF-TAVR, transfemoral transcatheter aortic valve replacement.)
Hemostasis techniques
Hemostasis after sheath removal has traditionally involved simultaneous use of two suture-based devices: the Prostar XL 10 Fr and Perclose ProGlide (Abbott Vascular Devices, Redwood City, CA, USA). Suture-based devices have had high success with very low vascular complication rates after coronary revascularization. 18 The Prostar XL, which is commercially available in the United States, is approved for closure of up to 10 Fr, and the 6-Fr ProGlide can be used for arteriotomies of up to 8 Fr. However, neither of these devices have been approved in the US for use in a ‘preclose’ fashion, where the devices are deployed at the time of sheath insertion rather than sheath removal; doing so allows the operator to gain hemostasis more safely and reliably. 19 If bleeding persists despite use of two closure devices, a third ProGlide device or an 8-Fr Angio-Seal (Abbott Vascular, Redwood City, California) can be utilized; both methods appear to be equally effective and safe. 20
Another adjunctive means of hemostasis involves deploying a guidewire from the contralateral femoral artery across the TAVR sheath access site before insertion of the TAVR sheath and then performing the crossover balloon occlusion technique (CBOT).21,22 This technique can be used selectively in higher risk patients and has been associated with a lower risk of Valve Academic Research Consortium-2 (VARC-2) major vascular and bleeding complications. 23
Vascular complications
Incidence
Historically, the reported incidence of TF-TAVR-associated vascular complications has ranged from 1.9% to 30.7%,24–28 depending on the bleeding definitions, devices employed and vascular access sites chosen. The relationship between larger sheath diameter and greater vascular access site bleeding risk is well established, 29 and with reductions in TAVR device diameters, there has been a decline in the rate of bleeding complications over time. The VARC has standardized definitions for use in clinical research, 30 and these have been updated in the VARC-2 document 31 (Table 1).
Valve Academic Research Consortium-2 classification of vascular access site and access-related complications.
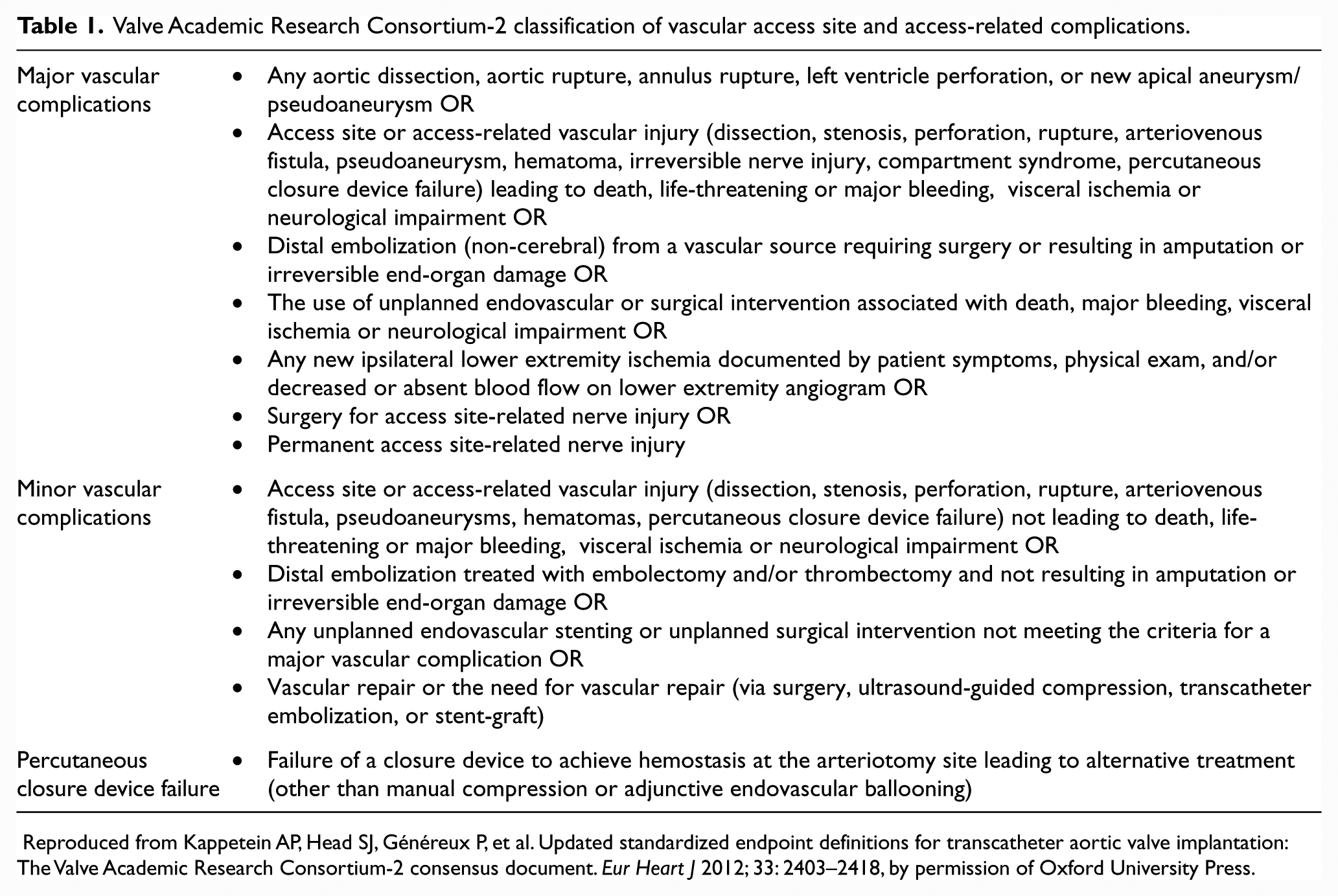
Reproduced from Kappetein AP, Head SJ, Généreux P, et al. Updated standardized endpoint definitions for transcatheter aortic valve implantation: The Valve Academic Research Consortium-2 consensus document. Eur Heart J 2012; 33: 2403–2418, by permission of Oxford University Press.
In a meta-analysis of 16 TAVR trials (n=3519 patients) employing VARC-1 definitions, the incidence (95% confidence interval) of major, minor and all vascular complications was 11.9% (8.6–16.4%), 9.7% (6.7–14%) and 18.8% (14.5–24.3%), respectively; life-threatening, major, minor and all bleeding occurred in 15.6% (11.7–20.7%), 22.3% (17.8–28.3%), 9.9% (6.9–14.3%) and 41.4% (35.5–47.6%), respectively. 32 Transfusion of ⩾1 unit occurred in 42.6% of patients; 32 however, with recent advances in technology it has decreased to as low as 11.1% in TF-TAVR. 33 The SAPIEN XT (18, 19 Fr) was compared with the SAPIEN S3 valve (22, 24 Fr) in the PARTNER 2 inoperable cohort; vascular complications occurred less frequently with the newer SAPIEN XT (9.4%) than with the SAPIEN S3 -treated patients (15.5%) (p=0.04). 34 Similar trends were observed in intermediate-risk patients with severe aortic stenosis in PARTNER 2A. 7 More recent data from the Society of Thoracic Surgeons/American College of Cardiology Transcatheter Valve Therapy (STS/ACC TVT) Registry suggest that the frequency of overall vascular complications is declining over time. In a registry analysis of 26,414 patients comparing outcomes in patients who underwent TAVR between 2012 and 2013 to those who did so in 2014, the incidence of major bleeding, life-threatening or disabling bleeding and vascular complications declined from 5.5% to 4.2%, 6.4% to 4.3%, and 5.6% to 4.2%, respectively. 1 Vascular complications are significantly lower with newer generation TAVR devices that require smaller delivery sheaths (major vascular complications were reduced from 8% (22–24 Fr sheaths) to 1% (18–19 Fr sheaths) 35 ) and with greater accrual of operator experience.
Small diameter internal jugular or femoral venous access is usually obtained for transvenous pacemaker placement, but no significant venous access complications have been reported.
Prognosis
Vascular complications associated with TAVR portend significantly worse clinical outcomes, 36 including increased mortality, 37 longer length of hospital stay, greater cost of care and poorer quality of life.24–27 The severity of vascular complications impacts outcome as well. Patients who suffer minor vascular complications using either the VARC-1 or VARC-2 definitions have similar rates of mortality and other complications when compared with those without vascular complications. 38 However, both VARC-1 38 and VARC-2 major vascular complications are associated with increased length of hospital stay, longer procedural time38,39 and mortality; VARC-2 definitions better predict survival than VARC-1 definitions. 40
Predictors
Traditional risk calculators, such as the STS score and the EuroSCORE, do not specifically predict the risk of vascular complications. 41 A number of patient, procedural and operator/center factors independently predict vascular complications associated with TF-TAVR. Patient-related variables include female sex, 24 renal insufficiency, iliofemoral arterial calcification (especially when circumferential) and concomitant peripheral vascular disease.24,25,35,37,42,43 Tortuous iliofemoral arteries do not predict TF-TAVR-associated vascular complications.25,42 Procedural factors include larger sheath to femoral artery ratio (SFAR) ⩾1.05. 25 In studies comparing the older generation Edwards SAPIEN device (requiring a larger sheath) with the Medtronic CoreValve, fewer vascular complications were observed with the latter. 35 Similarly, the incidence of vascular complications has declined with newer (i.e. Edwards SAPIEN XT and S3) compared with older Edwards devices.44,45 However, no statistically significant reduction in the incidence of vascular complications has been observed when comparing the Edwards S3 versus SAPIEN XT devices (0% vs 7.7%, p=0.15) 45 or when comparing the Edwards SAPIEN XT with the Medtronic CoreValve (2.8% vs 3.3%, p=0.66). 46 The real-world incidence of TF-TAVR-associated vascular complications should continue to decline with the advent of new, smaller diameter devices. Operator and center experience play an important role as well, and greater experience is associated with fewer such complications.47,48
While greater sheath-to-artery diameter ratio49,50 and sheath-to-artery area ratio 49 independently predict incident vascular complications, the latter metric appears to be more sensitive. The subclavian approach is safe and feasible with procedural success rates, safety end points, including major vascular complications, and life-threatening bleeding similar to those seen with TF-TAVR. 16 Caval-aortic fistula formation is the most common complication of caval-aortic access, but there are few published data describing its severity and incidence.
Prevention
Many factors that predict vascular complications are not modifiable, and mitigation of risk with respect to these can only be accomplished through careful patient selection. In other circumstances, procedural modification may facilitate risk reduction.
Vascular access
Careful review of pre-operative imaging in the context of a multi-specialty heart team has been associated with fewer vascular complications.35,51 The selection of vascular access requires detailed and accurate assessment of vascular anatomy, primarily using multidetector computed tomography (MDCT) or invasive angiography. MDCT provides clear and complete three-dimensional assessment of the iliofemoral system, revealing vessel tortuosity, extent of vessel calcification, and identifying atheroma and dissection, if present. 52 Real-time ultrasound guidance use can facilitate optimal common femoral artery access and visualization of calcification, which permits deployment of a stitch in a calcium-free zone using the preclose technique described above. Use of ultrasound has been shown to improve first-pass success rate, 53 reduce the number of attempts needed, reduce time to vascular access, and simplify simultaneous venous access by marking the artery’s location, and may reduce vascular complications. 54 A propensity-matched comparison of open surgical versus percutaneous access for TF-TAVR suggested no difference in the incidence of vascular complications but shorter length of stay with the latter. 50
Vascular closure
There are limited and conflicting data from studies comparing the Prostar XL and ProGlide hemostasis devices in the setting of TAVR.55,56 Vascular closure device failure is not uncommon; its reported incidence is highly variable, ranging from 2.7% to12% and it is an independent predictor of vascular complications.22,25,57,58 In addition to patient-specific factors cited above, an operator learning curve for device deployment may contribute. 59 One single-center study evaluated hemostasis using the Angio-Seal device as an adjunct to dual Perclose ProGlide devices in this setting. It demonstrated that Angio-Seal use was feasible and effective and suggested that it may be considered as a bail-out or an alternative strategy when the dual Perclose closure device technique fails to obtain complete hemostasis. 20
Management
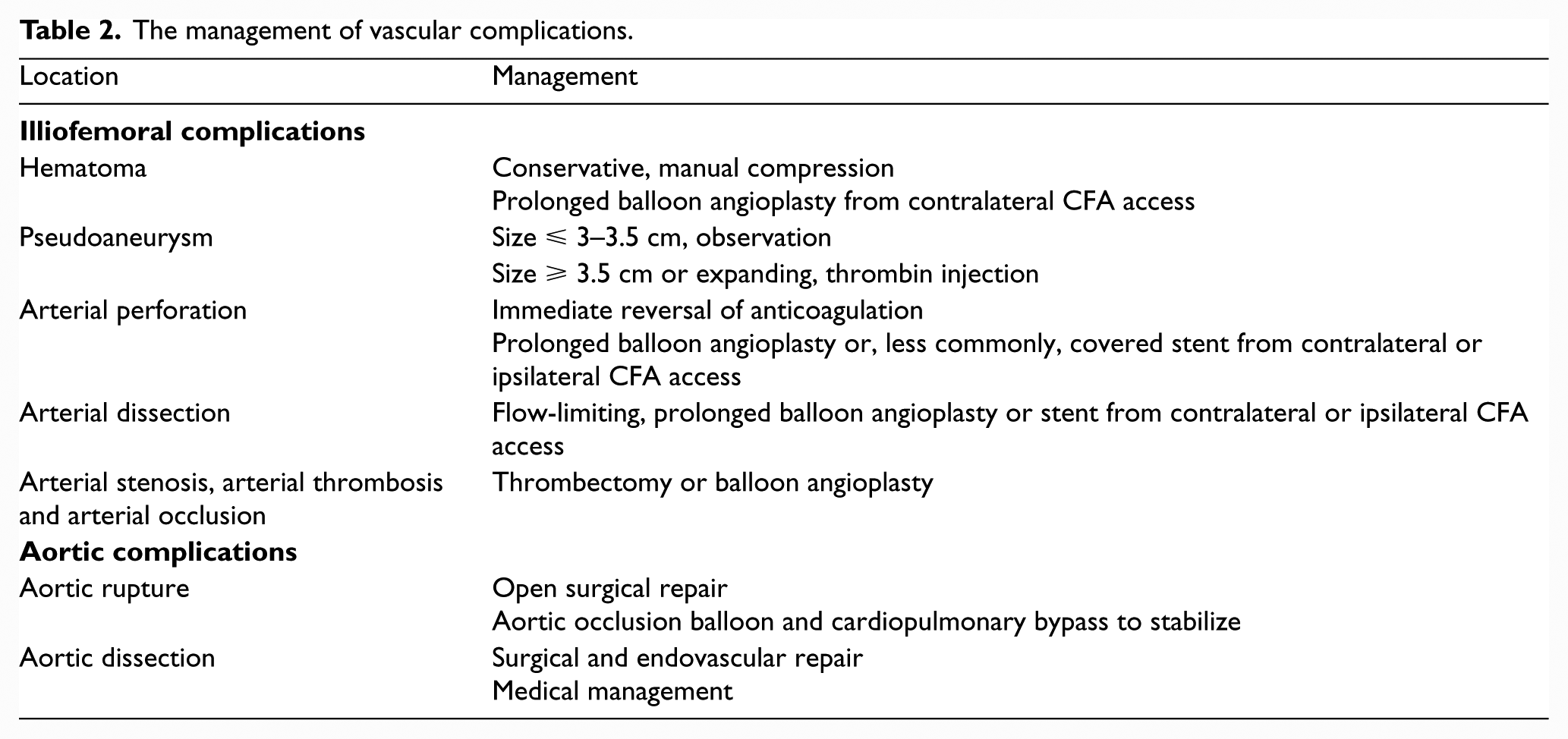
The management of vascular complications is outlined in the text below and summarized in Table 2.
The management of vascular complications.
Iliofemoral
Illiofemoral complications are the most commonly encountered complications of TF-TAVR. 28 Traditionally, TF-TAVR-associated vascular complications have been addressed surgically; 60 however, this approach is often associated with poor wound healing and higher infection rates61,62 with significant resultant morbidity and mortality.60,63 Consequently, a number of catheter-based approaches have been developed and tested.
Access site hematoma
Access site bleeding and hematoma formation after TAVR is common (11–18%) 26 and associated with increased hospital length of stay, secondary infection, blood transfusion and mortality.64–66 The incidence is declining, in part due to smaller required sheath sizes, a move from surgical cut down to percutaneous access, and increased operator experience and familiarity with suture-mediated closure devices. Most hematomas can be managed conservatively with manual digital compression and reversal of anticoagulation. However, rapidly expanding hematomas can compress surrounding structures. Blood transfusion during surgery is itself associated with excess mortality, potentially resulting from transfused blood’s prothrombotic effects, impaired oxygen delivery of transfused erythrocytes, microvascular obstruction, and transfusion reactions.67,68 An actively expanding hematoma can often be treated percutaneously by gaining contralateral common femoral arterial access, advancing a balloon catheter to the site of bleeding and then performing prolonged balloon inflation using traditional peripheral angioplasty balloons. A 2015 Danish study of 48 TAVR vascular access complications suggested that Viabahn covered stents (WL Gore & Associates, Newark, DE, USA) are a safe and effective therapy with a success rate of 98% and 1-year patency of 100% by ultrasound; 69 these devices are most appropriate when the site of hemorrhage is not adjacent to a major arterial branch (e.g. internal iliac or profunda femoris).
Pseudoaneurysm
Pseudoaneurysm may occur in the setting of incomplete hemostasis at the arteriotomy, resulting in a pulsatile hematoma that communicates with the arterial lumen. As a preventive measure, it is our standard practice to obtain digital subtraction imaging of the ipsilateral illiofemoral system either using a 6-Fr pigtail catheter in the distal abdominal aorta or a 5-Fr internal mammary artery (IMA) catheter introduced from the contralateral CFA to confirm hemostasis. Continuous post-procedural pain and access site swelling with or without an associated bruit or thrill suggest the need for diagnostic duplex ultrasound. Risk factors for pseudoaneurysm include calcified vessels, advanced age, female sex, obesity, larger sheaths, cannulation of an artery other than the CFA, combined arterial and venous puncture and percutaneous hemostatic device failure. 70 Pseudoaneurysms of less than 3.0–3.5 cm close spontaneously in more than 50% of cases and, if not painful, may only require observation using serial ultrasound exam.71–73 Ultrasound- guided thrombin injection is nearly 100% effective and recommended in cases of larger pseudoaneurysms so long as there is an adequate length neck present to minimize the risk of arterial thrombosis. Other modalities of pseudoaneurysm management include ultrasound-guided compression, surgical repair, percutaneous coil embolization and exclusion with covered stents 70 (Figure 3).
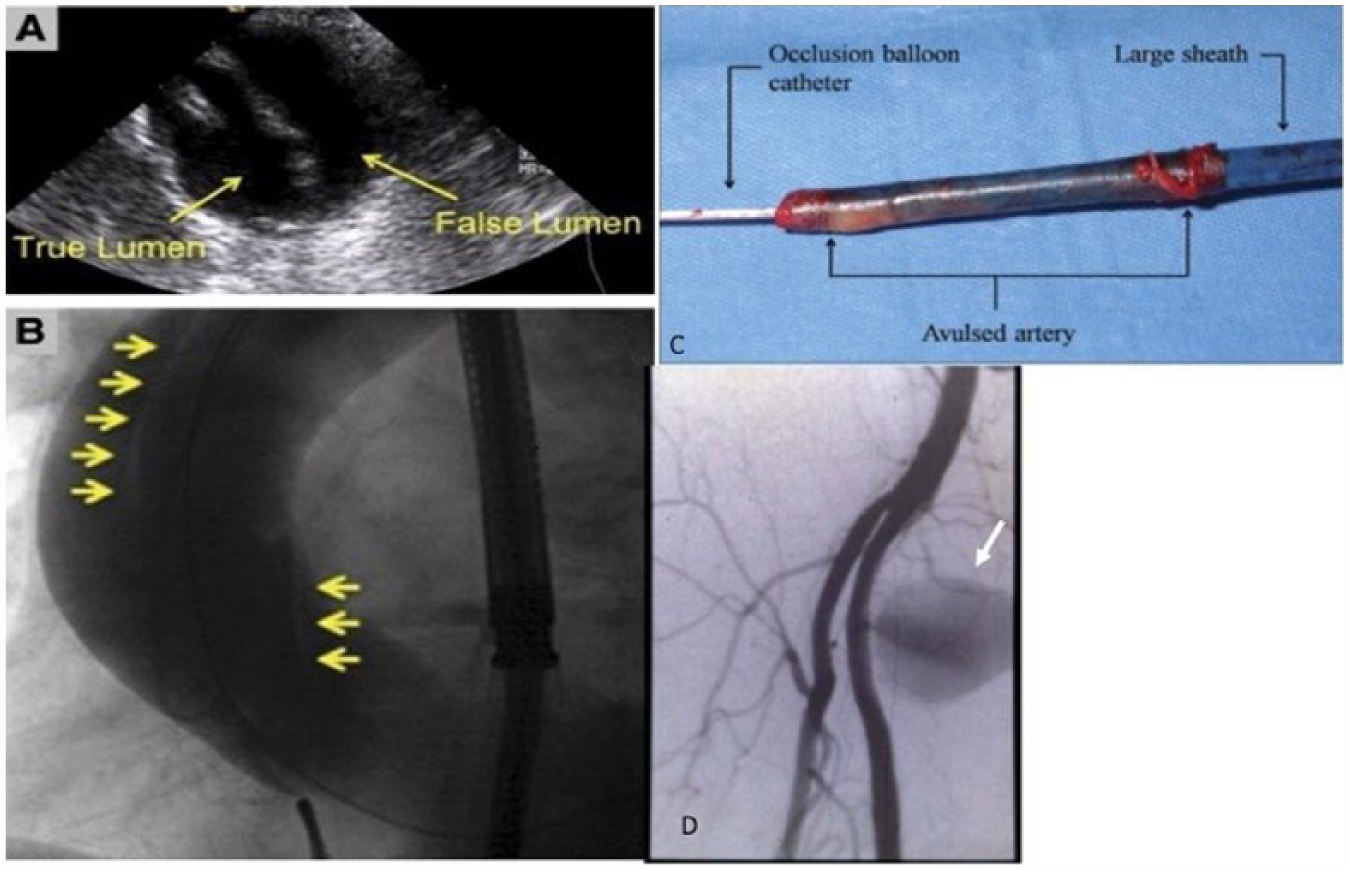
(A) Transesophageal echocardiographic and (B) angiographic images (arrows delineate a spiral dissection). (C) Complete avulsion of the iliac artery on sheath removal. (D) Pseudoaneurysm (arrow). Panels A-C reprinted from ref. 18, with permission from Elsevier. Copyright © 2009 American College of Cardiology Foundation.
Arterial perforation
Vascular perforation with or without retroperitoneal bleeding may lead to acute hemodynamic compromise and occurs in approximately 3.5% of patients undergoing TF-TAVR. 74 Its incidence may have decreased with the advent of lower-profile delivery systems. Digital subtraction angiography (DSA) of the iliofemoral arteries before and after knot closure of suture-mediated devices is generally recommended. Perforations are usually manifest once the TAVR sheath is withdrawn, as the sheath often tamponades the site of perforation while in place. For this reason, hemodynamics should be closely monitored at the time of sheath withdrawal. If a perforation occurs, immediate reversal of anticoagulation is advisable with reinsertion of the sheath over its dilator to temporarily seal the defect. This should be immediately followed by advancement of a balloon catheter from the contralateral CFA so that tamponade can be performed using prolonged balloon inflation. If prolonged balloon inflation is unsuccessful, consideration must be given to open surgical repair versus an endovascular covered stent. In the event that deployment of a covered stent is feasible, it should be noted that most covered stents require 7-Fr or larger sheaths; consequently, some operators place a 7-Fr sheath in the contralateral groin at the outset of the procedure.
Arterial dissection
Iliofemoral artery dissection is the most common TAVR-associated vascular complication, reported in ~ 6.5%, 24 usually occurring due to traumatic sheath insertion into a fragile, calcified artery, creating a tear in the arterial wall. Medial staining is most obvious using unsubtracted angiographic images. Most dissections are limited, non-occlusive and non-flow-limiting: with retrograde dissections, antegrade flow is usually preserved as the dissection flap is forced against the arterial wall by the upstream blood pressure. If there is no evidence of neurovascular compromise, the patient can be managed conservatively with serial leg examinations, with or without duplex ultrasound, to monitor for vessel patency. There is no uniform consensus available to treat acute non-flow-limiting, small arterial dissections after TAVR, and treatment strategies are highly variable.
Extensive dissection may compromise flow to the limb due to superimposed thrombosis/intramural hematoma or obstructive dissection flap, resulting in acute limb ischemia. Once identified, extensive flow-limiting dissections require urgent attention, and most can be treated with prolonged angioplasty with or without deployment of a self-expanding stent advanced from the contralateral groin.
Careful review of fluoroscopy at the time of sheath insertion may predict development of a dissection. If movement of arterial calcification is observed during sheath insertion, it is advisable to stop the advancement of the sheath.
Arterial avulsion
Rarely, large sheaths may become adherent to the vascular endothelium in highly atheromatous, calcified vessels. 18 Excessive resistance at the time the sheath is withdrawn may signal significant risk for avulsion. Once suspected, an occlusion balloon must be rapidly placed proximal to the site of resistance, ideally in the abdominal aorta below the renal arteries, and the patient should undergo immediate surgical exploration18,75 (Figure 3).
Arterial stenosis, arterial thrombosis and arterial occlusion
The CFA may develop stenosis after the deployment of a vascular closure device. This is usually manifest as a puckered appearance on DSA and is most often mild, without vascular compromise, 35 not requiring further treatment. In cases of severe stenosis with compromised distal flow, balloon dilation may help; stents are rarely required21,22 (Figure 4). Thrombosis at the site of the arteriotomy is rare, but can occur with prolonged inflation of a crossover hemostasis balloon. Arterial thrombosis may require thrombectomy and/or balloon angioplasty to re-establish patency21,76 (Figure 5).
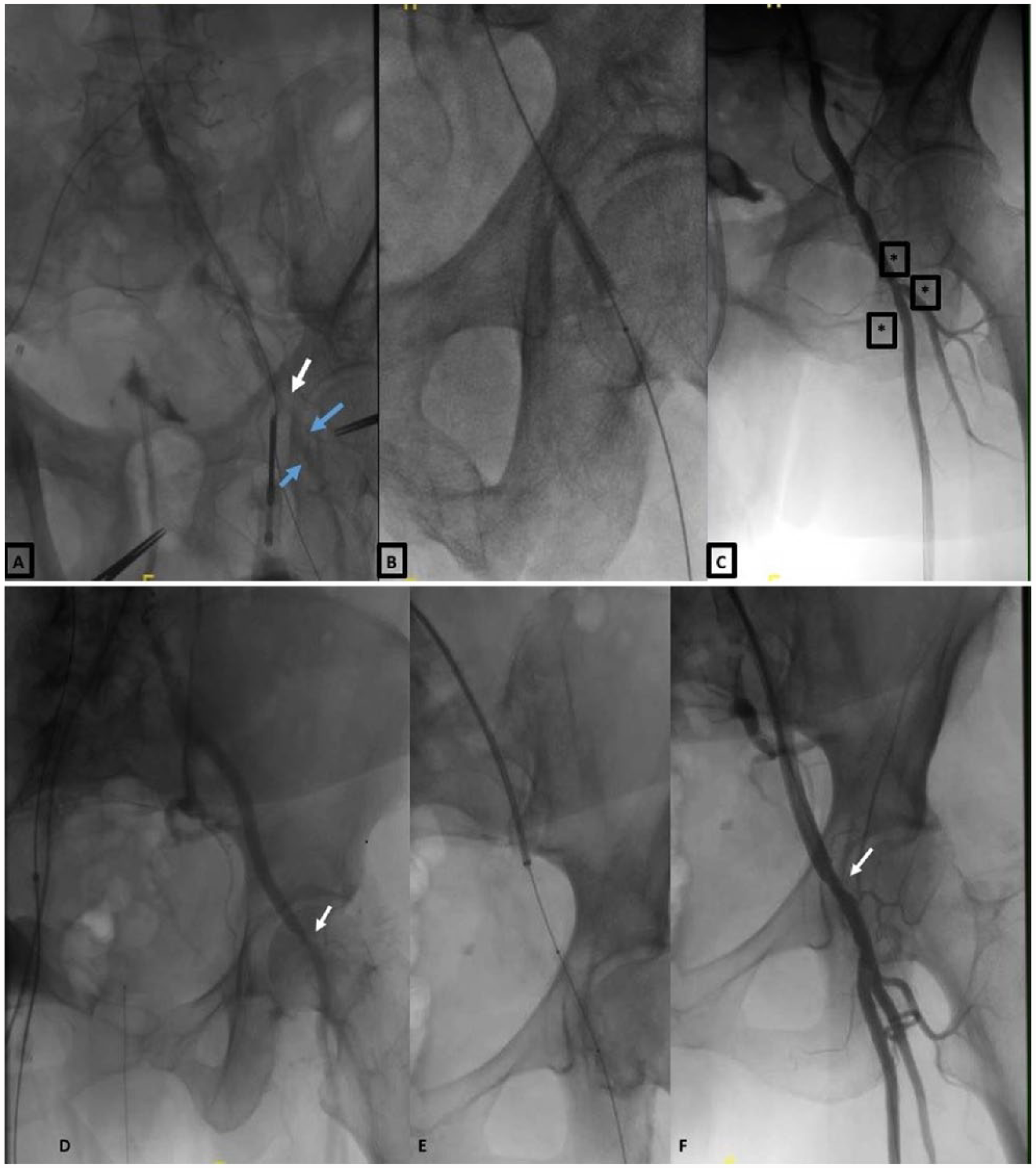
(A) Focal dissection of the common femoral artery (CFA) (white arrow) and slow distal superficial femoral (SFA) and profunda femoris artery (PFA) flow (blue arrows) treated with (B) a 7×40-mm balloon resulting in (C) acceptable antegrade distal flow in the CFA, SFA and PFA (asterisks). (D, E) Stenosis of the CFA (arrow) with a compromised antegrade flow (E, F) result following successful treatment with balloon angioplasty.
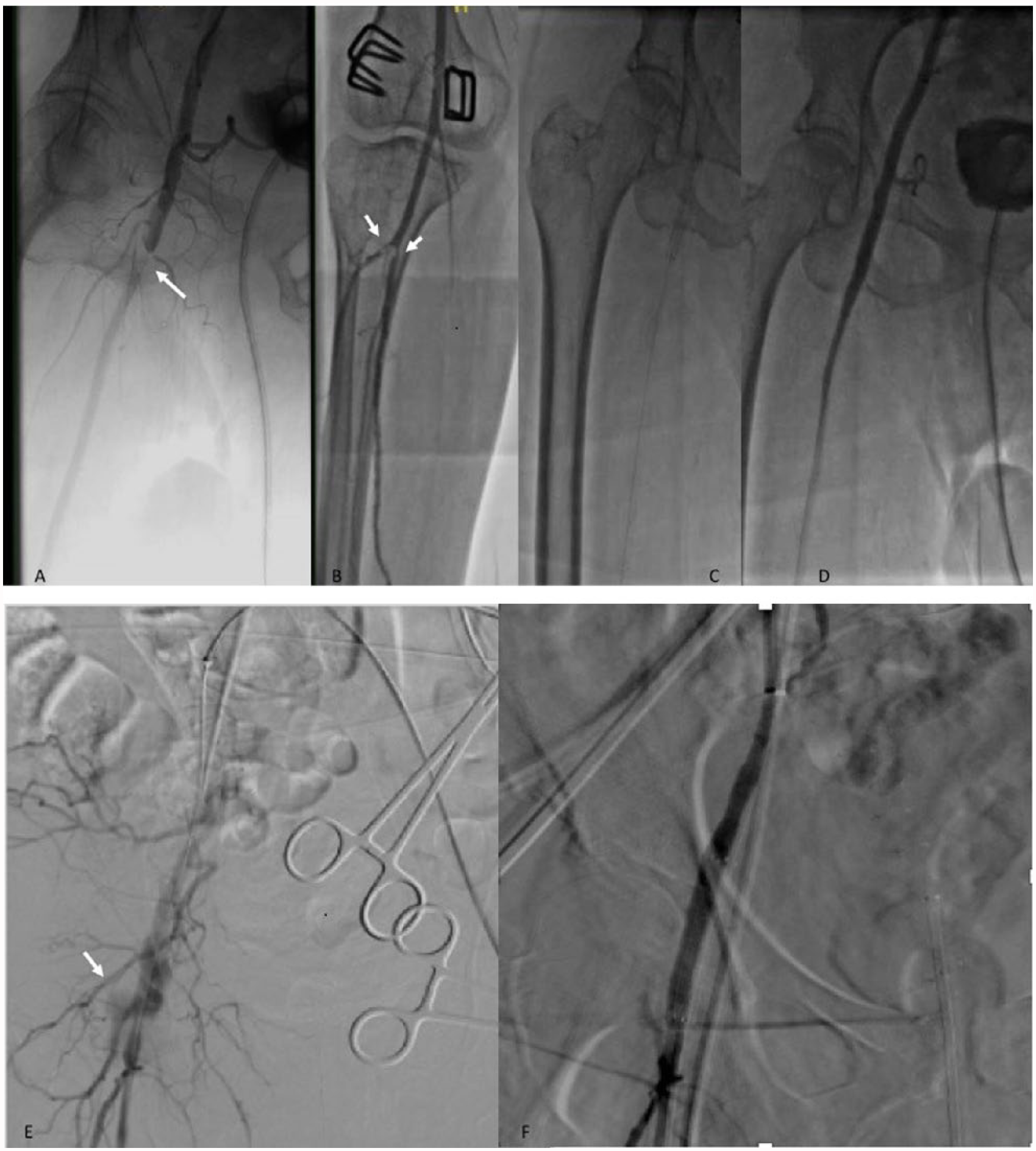
(A) Thrombus at the common femoral artery (CFA) (arrow) after deployment of a Perclose device. (B) Distal thrombus at the level of tibioperoneal trunk trifurcation (white arrows), treated with aspiration thrombectomy. (C, D) Self-expanding stent at the CFA, resulting in good antegrade flow. (E, F) Perforation after transcatheter aortic valve replacement (arrow) treated successfully with a covered stent.
Aortic
Aortic complications with TAVR are rare, especially in the era of lower-profile delivery systems; however, when present these may be associated with grave outcomes.
Aortic rupture
Acute or sub-acute aortic rupture is rare (<1%) but catastrophic. 77 Rupture may occur due to trauma from the valve delivery system, especially if it is inadvertently advanced without a wire in an atheromatous and/or tortuous portion of the aorta. Angiographic findings may be subtle and can require a high index of suspicion, as an acute rupture can rapidly lead to hemorrhagic shock. The preferred treatment is surgical; however, even with prompt surgery, this complication still carries a very high mortality. An aortic occlusion balloon can be used to stabilize the patient until surgical repair. Endovascular repair with covered stent grafts may be an option, depending on the location of the tear. 78
Aortic dissection
Aortic dissection after TAVR is rare, occurring in 0.6–1.9%79,80,81 and can involve any segment of the aorta. When TAVR is performed with transesophageal echocardiographic (TEE) guidance, aortic dissection may be incidentally detected. As the signs and symptoms may not manifest until after the procedure, it is prudent to screen for iatrogenic dissection on TEE after completion of the procedure (Figure 3). Recommended surgical, endovascular and/or medical management of aortic dissection with TAVR is similar to that following spontaneous aortic dissection. 82
Device evolution and vascular complications
Newer generation valves (see below) require 14–16-Fr sheaths, and the new Edwards CENTERA aortic valve (self-expanding valve, ultra-low-profile and repositionable) is expected to be compatible with a 12-Fr (catheter outer diameter (OD)) sheath for all valve sizes. 83 Likewise, the SoloPath sheath (Terumo Medical Corporation, Irvine, CA, USA) is a balloon expandable and re-collapsible sheath, available in internal and outer diameters as small as 14 and 17 Fr. The sheath is inserted into the vessel in a folded state over a balloon-expandable dilator. Once the sheath is in the desired position, the dilator is inflated, the sheath expands, and later the dilator is removed. On completion of the procedure, the balloon is deflated and the sheath shrinks and returns to its original OD. The safety and efficacy of the 19-Fr SoloPath sheath was investigated for TF-TAVR in a single-arm study of 90 patients. When patients were dichotomized into those with a SFAR of ⩽ 1.05 vs > 1.05, the 19-Fr SoloPath sheath appeared feasible and safe even in patients with a SFAR > 1.05 (a traditional indicator of increased vascular risk) and there was no difference in technical or procedural success, total vascular complications, or total bleeding rates between groups. 84 The safety of the SoloPath access sheath was confirmed in a recent multicenter study of patients with ⩽ 5.0-mm illiofemoral access undergoing TF-TAVR using the CoreValve device. 85 Vascular complication rates will likely continue to fall with the advent of smaller diameter valve platforms and delivery sheaths.
Summary
TAVR has undergone rapid evolution since it was first described in 2002. Vascular complications remain one of the most serious associated adverse events, but their overall incidence is declining, likely due to improved vascular access and closure technique, maturation 1 of the required technology, including smaller access sheaths and delivery systems, growing operator experience, and improved patient selection. Patient selection algorithms should be employed to form institutional-level standard operating procedures (Table 2). It is of paramount importance that operators become familiar with the range of potential TF-TAVR-associated vascular complications and facile in detecting and managing these when they occur.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
