Abstract
Cell culture and animal work indicate that dipeptidyl peptidase-4 (DPP-4) inhibition may exert cardiovascular benefits through favorable effects on the vascular endothelium. Prior human studies evaluating DPP-4 inhibition have shown conflicting results that may in part be related to heterogeneity of background anti-diabetes therapies. No study has evaluated the acute response of the vasculature to DPP-4 inhibition in humans. We recruited 38 patients with type 2 diabetes on stable background metformin therapy for a randomized, double-blind, placebo-controlled crossover trial of DPP-4 inhibition with sitagliptin (100 mg/day). Each treatment period was 8 weeks long separated by 4 weeks of washout. Endothelial function and plasma markers of endothelial activation (intercellular adhesion molecule 1 (ICAM-1) and vascular cell adhesion molecule 1 (VCAM-1)) were measured prior to and 2 hours following acute dosing of sitagliptin or placebo, as well as following 8 weeks of intervention with each pill. Thirty subjects completed the study and were included in analyses. Neither acute nor chronic sitagliptin therapy resulted in significant changes in vascular endothelial function. While post-acute sitagliptin ICAM-1 levels were lower than that post-chronic sitagliptin, the ICAM-1 concentration was not significantly different than pre-acute sitagliptin levels or levels measured in relationship to placebo. There were no significant changes in plasma VCAM-1 levels at any time point. Acute and chronic sitagliptin therapies have neutral effects on the vascular endothelium in the setting of metformin background therapy. In conclusion, our findings suggest DPP-4 inhibition has a neutral effect on cardiovascular risk in patients without a history of heart failure or renal insufficiency.
Introduction
Endothelial dysfunction is a pathological state in which the vascular endothelium (the layer of cells lining blood vessels in direct contact with blood) is systemically inflamed and expresses pro-vasoconstrictive mediators, and develops clot forming tendencies. 1 Endothelial dysfunction is readily detectable in all parts of the arterial tree, precedes the development of atherosclerosis, and portends an increased risk of future adverse cardiovascular events. 2 Thus, endothelial dysfunction can be measured as a ‘barometer’ of future cardiovascular risk. 3 While multiple factors contribute to the development of endothelial dysfunction, the loss of endothelium-dependent nitric oxide (NO) bioavailability is a readily measurable central mechanistic contributor to the development of endothelial dysfunction.1,2 Normal homeostatic NO bioavailability limits vascular inflammation and activation, promotes vasodilation, and balances pro-thrombotic and pro-fibrinolytic forces in the coronary and peripheral vasculature.1,2 The presence of phenotypical endothelial dysfunction is well described in patients with type 2 diabetes (T2DM).4–10 The phenotype includes significant and readily measurable impairment in NO-dependent vasodilation of both coronary and peripheral arteries, as well as increased circulating endothelial adhesion molecules including E-selectin, intercellular adhesion molecule 1 (ICAM-1), and vascular cell adhesion molecule 1 (VCAM-1).4–10 The severity of insulin resistance correlates with the presence of endothelial dysfunction, and hyperglycemia exacerbates endothelial dysfunction in patients with diabetes.11–13
Overall, these data strongly suggest pharmacological interventions that both improve glycemic control and favorably impact endothelial function will have the greatest cardiovascular and metabolic benefits in patients with T2DM. In vitro and animal data on dipeptidyl peptidase-4 (DPP-4) inhibitors suggest they favorably impact endothelial function.14–23 However, human studies to date conflict on whether DPP-4 inhibition improves or impairs human endothelial function, and these data are limited by short study durations, methodological limitations, and a lack of clarity as to whether DPP-4 inhibitor administration effects on the endothelium are acute, chronic, or both.24,25 To better determine the acute and chronic impact of DPP-4 inhibition on vascular endothelial function in humans with T2DM, we performed a randomized, double-blind, placebo-controlled crossover study using sitagliptin to evaluate the effects of acute and chronic DPP-4 inhibition on endothelium-dependent vasodilation and systemic markers of endothelial activation, ICAM-1 and VCAM-1. This was performed in the setting of metformin monotherapy.
Methods
Subject selection
We enrolled consecutive subjects aged 21–70 years with T2DM on stable metformin monotherapy for at least 6 weeks with a hemoglobin A1C between 6.2% and 9.5%. We only enrolled subjects who were on a stable dose of metformin to reduce potential confounding effects of recent changes in metformin therapy on endothelial function measurements. 26 Initially, the lower range of acceptable hemoglobin A1C was 6.5%, but this was lowered to 6.2% approximately 6 months into the study to improve recruitment. Subjects with a history of atherosclerotic disease, renal insufficiency (creatinine clearance < 60 mL/min), liver disease (aspartate aminotransferase (AST) or alanine aminotransferase (ALT) levels > 2.5 times normal), illicit drug use in the past year, history of allergy to DPP-4 prior to screening, or history of pancreatitis were excluded. Pregnant women were also excluded from the study. The study protocol was approved by the Institutional Research Board at the Medical College of Wisconsin, and all subjects gave written informed consent prior to participating in any study procedures.
Study procedure
Subjects who passed a telephone screen were invited to a screening visit for study eligibility. Informed consent was obtained, relevant participant medical history was recorded – including currently prescribed medications, anthropometric measurements were taken (height, weight, and waist circumference in metric units) and blood pressure was recorded (measured in triplicate and averaged). Blood was obtained for measurements of fasting plasma glucose, glycosylated hemoglobin, insulin and lipid profiles, and blood chemistries (AST/ALT). As a measurement of insulin sensitivity, the homeostatic model assessment of insulin resistance (HOMA-IR) was calculated as [(plasma glucose concentration (mg/dL) × plasma insulin concentration)/405]. Subjects were allowed to take their blood pressure medication on the morning of their screening visit, but not the mornings of any of the other study visits to limit the acute influence of these medications on endothelial function.
Subjects who qualified for the study were randomized either to receive sitagliptin first (100 mg/day) or matching placebo. Prior to receiving either set of pills, subjects returned to the study center within approximately 1–2 weeks of the screening visit to undergo initial tests of endothelial function and receive their pills. Prior to all study visits, except screening, subjects were asked to refrain from any vigorous physical activity (weight lifting, jogging or any activity more vigorous than walking) for 24 hours. Subjects were also asked to fast 6–8 hours prior to the visit to limit acute dietary influences on vascular endothelial function.
At the initial study visit following the screening, endothelial function was determined by brachial artery reactivity testing (see below) prior to and following a single dose of 100 mg of sitagliptin or matching placebo. Blood samples were also taken at this visit for systemic measurements of endothelial cell activation/inflammation (VCAM-1 and ICAM-1) prior to and 2 hours following acute pill administration.
Endothelium-dependent vasodilation, as measured by brachial artery reactivity testing, was measured just prior to medication administration and then 2 hours following medication administration. The 2-hour time frame was chosen given that plasma levels of sitagliptin appear to peak 2 hours following dose administration. 27
At the end of this visit, subjects were given a 9-week supply of the study pills (sitagliptin or matching placebo), as dispensed by the Medical College of Wisconsin/Froedtert Hospital Investigational Pharmacy, and scheduled to return for their next visit in approximately 8 weeks for repeat measurements of endothelial function (brachial artery reactivity and measurements of plasma ICAM-1 and VCAM-1) and blood chemistries. Subjects were asked to not take any study medication for the 24 hours prior to this visit. Following this study visit, subjects underwent a 4-week washout period prior to returning to the study center to repeat their acute and chronic testing of endothelial function with the opposite set of pills in an identical manner to the prior visits. Subject adherence was determined by pill counts performed by study staff. The study compliance with medication was assessed by pill counts at the end of 8 weeks. The average compliance was 96 ± 6% with a range of compliance from 80% to 100%. No subjects were disqualified for non-compliance.
Measurement of endothelial function by brachial artery reactivity testing
The methodology to evaluate vasomotor function (flow-mediated dilation (FMD)) in the conduit brachial artery by high-frequency ultrasound has been developed and extensively validated over the past two decades and is well established in our laboratory.26,28–30 In this method, the diameter of the brachial artery was measured sonographically at baseline and the cuff was placed on the forearm below the cubital fossa and inflated to 200 mmHg to achieve occlusion of the brachial artery, which was maintained for 5 minutes and then released, starting the hyperemic phase. As previously described, 32 measurements of brachial artery were obtained in 10-second windows at 55–65, 115–125 and 175–185 seconds following cuff release. The largest average diameter of these three 10-second windows was used as the maximal brachial diameter to calculate the percentage change in FMD (FMD%) from baseline. We employ a cuff placed below the cubital fossa to maximize the NO dependence of the technique and use automatic edge-detection software to measure brachial diameters, and have excellent reproducibility with this method. 33
Non-endothelial-dependent vasodilation was measured by examining the vasodilator response to 0.4 mg sublingual nitroglycerin (NMD%) in patients without a contraindication to nitroglycerin administration (only measured at the acute dosing visits and at the chronic visits). The diameter of the brachial artery was obtained sonographically at baseline and sublingual nitroglycerin 0.4 mg was subsequently administered. Sonographic measurements of the brachial artery diameter were made in 10-second windows surrounding the 2, 3, 4 and 5-minute marks post-nitroglycerin administration. The maximum average diameter of these 10-second windows was used to compute the percentage change in non-endothelial-dependent vasodilation (NMD%)
Measurement of markers of systemic inflammation
Measurement of plasma ICAM-1 and VCAM-1 were performed using commercially available ELISA kits (R&D Systems, Inc., Minneapolis, MN, USA). The intraclass coefficients for repeated measures on the same samples were 99% and 98% for ICAM-1 and VCAM-1, respectively.
Statistical analysis
Our primary outcome measure for these studies was FMD% of the brachial artery. Secondary outcome measures included brachial diameter, resting and hyperemic shear and flow velocity in the brachial artery, nitroglycerin-mediated vasodilation of the brachial artery as our measure of endothelium-independent vasodilation, and plasma VCAM-1 and ICAM-1. The six subjects who dropped out of the study were not included in statistical analysis. Three of the 30 subjects who completed the study did not have a full set of insulin levels available for analysis. Baseline characteristics were compared between sitagliptin-first and placebo-first subjects by unpaired t-tests, Wilcoxon rank sum tests, or chi-squared tests as appropriate. Primary and secondary outcome variables were compared across measurement periods by repeated measures ANOVA, and post hoc analyses using Tukey’s test were applied if significant differences were detected. A priori sample size calculation demonstrated our study design has 80% power to detect a 1.5% absolute increase in FMD% with 30 subjects completing the entire study protocol at α=0.05. This is consistent with prior standards demonstrating the need for between 20 and 30 subjects to appropriately power crossover studies that use brachial FMD% as their primary endpoint.34,35 P-values of < 0.05 were considered statistically significant.
Results
Study subjects
A total of 60 subjects were screened for this protocol. The flow diagram of progress of these subjects through the protocol is included as Figure 1. Twenty-two subjects were excluded from participating: one for renal dysfunction, one who was unable to return for the study visits following screening, three for glycosylated hemoglobin levels above the screening limit, and 17 for glycosylated hemoglobin levels below the screening inclusion limit. Of the 38 subjects who qualified and enrolled, two were lost to follow-up, three subjects withdrew from the protocol, one subject developed a rash and was withdrawn, and two subjects were excluded for changes in diabetes medication regimens. Overall, 30 subjects completed all study visits. Their demographics, overall and broken down by which intervention was received first, are presented in Table 1. There were no significant differences between those who received placebo first and those who received sitagliptin first.
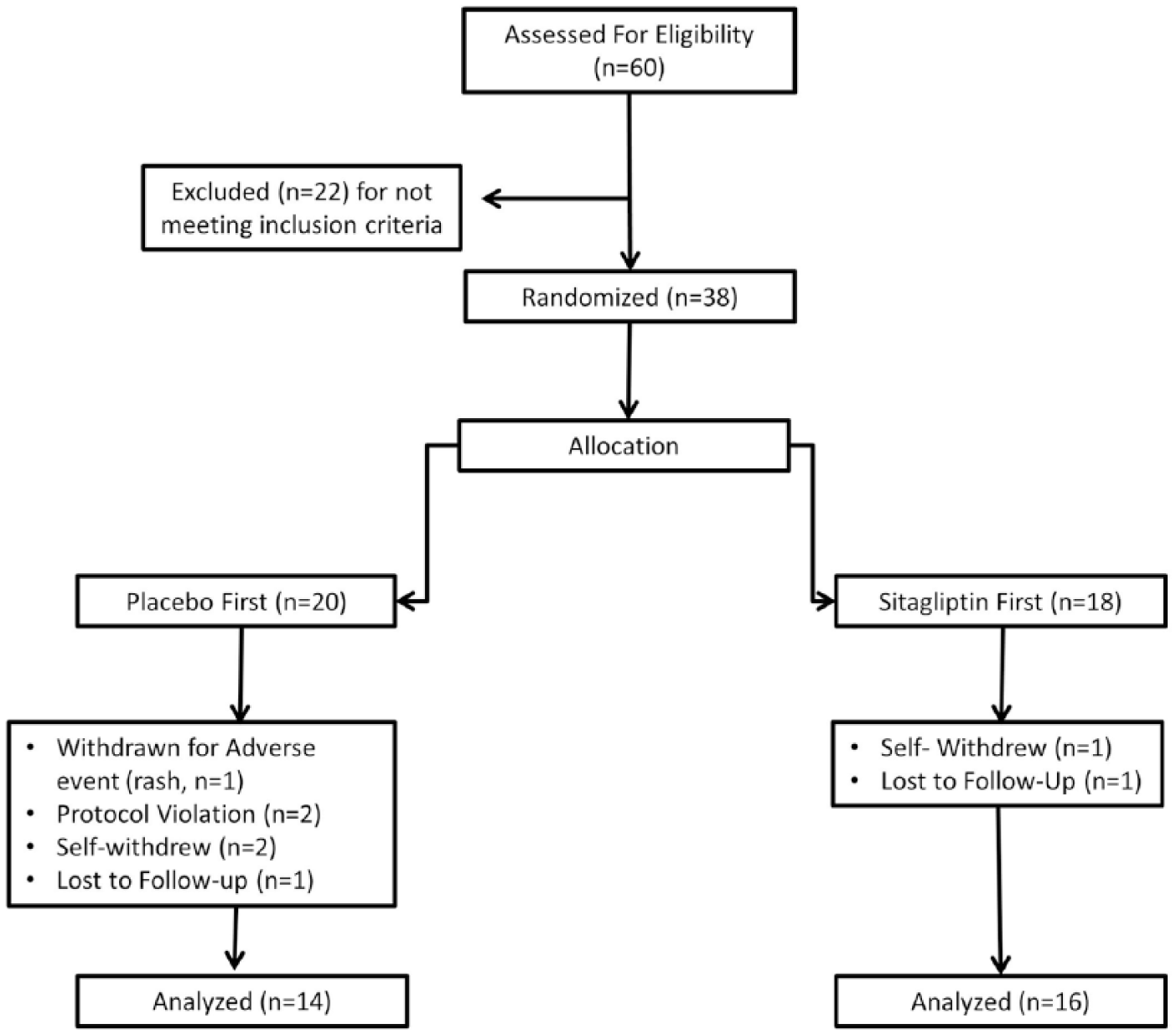
CONSORT progress diagram for study subjects.
Subject characteristics.
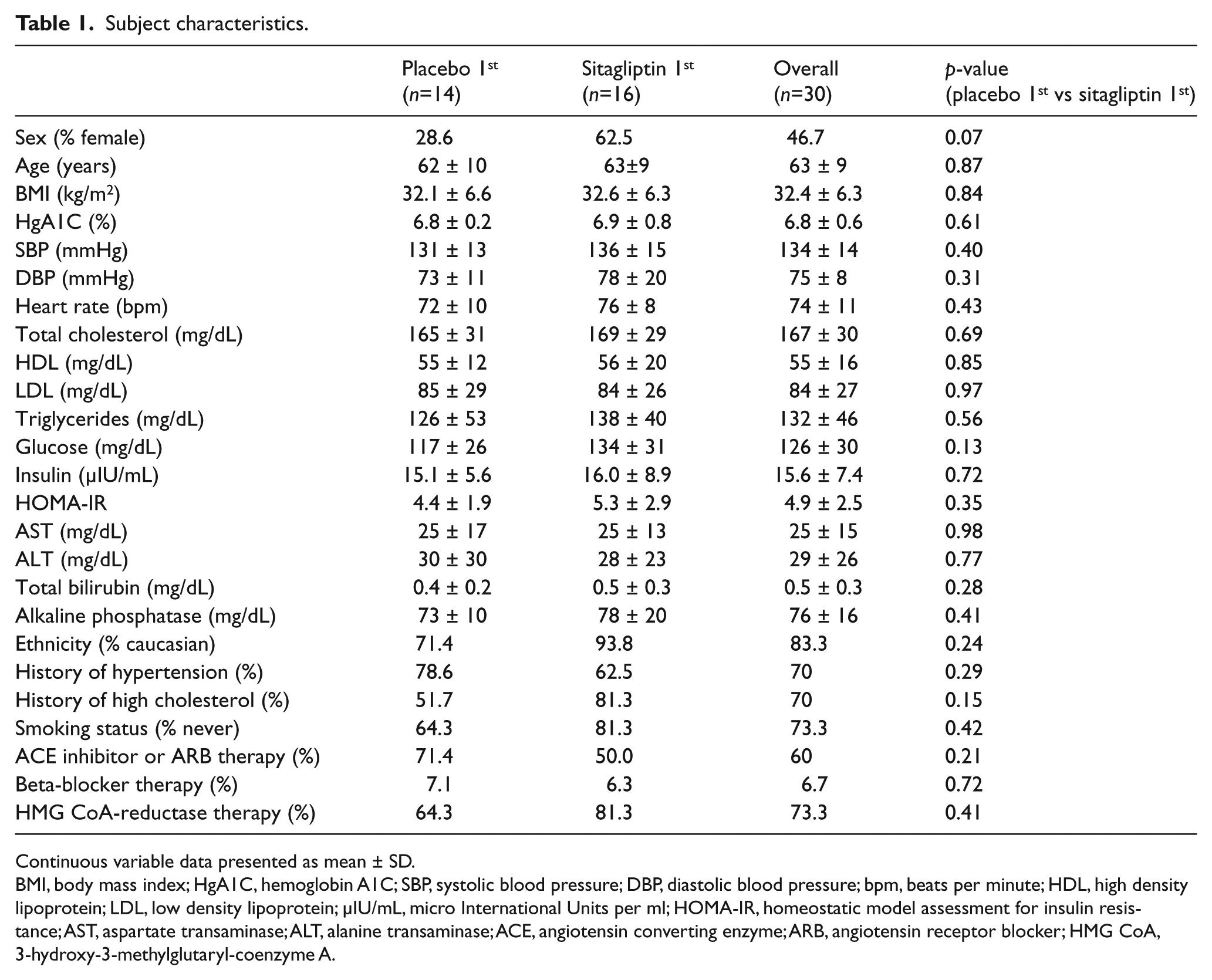
Continuous variable data presented as mean ± SD.
BMI, body mass index; HgA1C, hemoglobin A1C; SBP, systolic blood pressure; DBP, diastolic blood pressure; bpm, beats per minute; HDL, high density lipoprotein; LDL, low density lipoprotein; μIU/mL, micro International Units per ml; HOMA-IR, homeostatic model assessment for insulin resistance; AST, aspartate transaminase; ALT, alanine transaminase; ACE, angiotensin converting enzyme; ARB, angiotensin receptor blocker; HMG CoA, 3-hydroxy-3-methylglutaryl-coenzyme A.
Measures of glycemic control, lipids, and blood pressure at baseline and following each intervention arm are shown in Table 2. Eight weeks of sitagliptin therapy resulted in significant reductions in glycosylated hemoglobin and fasting plasma glucose (p=0.01 and p=0.049, respectively) relative to placebo and baseline visits. Baseline and post-placebo glucose levels and glycosylated hemoglobin percentages did not significantly differ (p=0.30 and 0.91, respectively). Diastolic blood pressure trended lower following sitagliptin but was not significantly lower than baseline or post-placebo measurements. While systolic blood pressure was overall higher post-placebo compared to baseline (p=0.01), there were no differences between systolic pressure post-placebo and post-sitagliptin (p=0.41). There were no changes in insulin sensitivity by HOMA-IR, heart rate, or cholesterol profiles between 8 weeks of sitagliptin treatment and 8 weeks of placebo. There were no differences noted in fasting insulin levels (n=27 available for analyses due to laboratory error leading to missing samples for three subjects).
Effect of sitagliptin and placebo on clinical and biochemical parameters following 8 weeks of intervention (n=30).
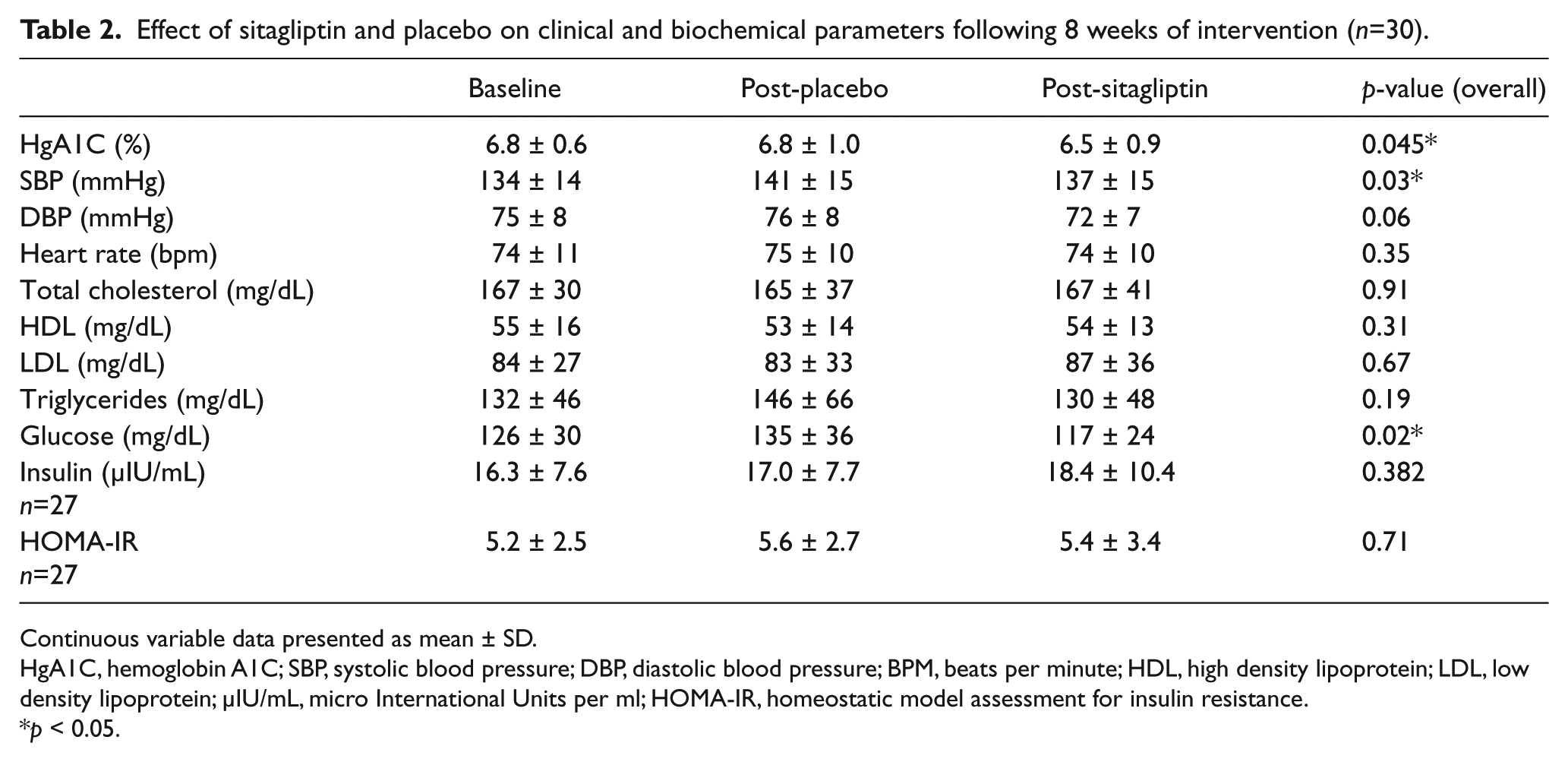
Continuous variable data presented as mean ± SD.
HgA1C, hemoglobin A1C; SBP, systolic blood pressure; DBP, diastolic blood pressure; BPM, beats per minute; HDL, high density lipoprotein; LDL, low density lipoprotein; μIU/mL, micro International Units per ml; HOMA-IR, homeostatic model assessment for insulin resistance.
p < 0.05.
Measurements of vascular function and systemic inflammation
Measurements of vascular function, including FMD% (Figure 2), baseline brachial diameter, baseline flow velocity, peak flow velocity post-cuff deflation, percent increase in hyperemic flow, and nitroglycerin-mediated dilation, did not significantly differ at any time point during the study (Table 3). FMD% did not significantly change with DPP-4 inhibition in subjects with a baseline hemoglobin A1C ⩾ 7.0% (n=10, 6.2±1.6% pre-sitagliptin to 6.5±1.8% post-sitagliptin; 6.9±2.0% pre-placebo to 7.5±2.7% post-placebo).
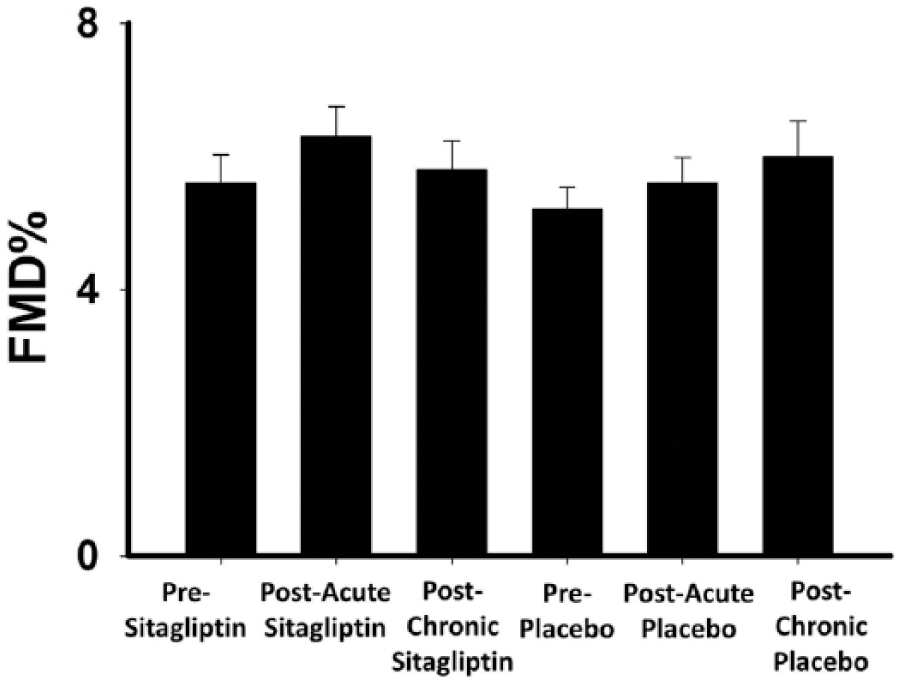
Impact of DPP-4 inhibition and placebo on acute and chronic endothelial function. Eight weeks of intervention with sitagliptin 100 mg/day and placebo had no significant effect on brachial artery flow-mediated dilation (FMD%: 5.6 ± 2.3, 6.3 ± 2.4, 5.8 ± 2.3, 5.2 ± 1.8, 5.6 ± 2.1, 6.0 ± 2.9 for pre-sitagliptin, post-acute sitagliptin, post-chronic sitagliptin, pre-placebo, post-acute placebo, and post-chronic placebo, respectively; p=0.31 by repeated measures ANOVA). Data presented as mean ± SE.
Brachial diameters and vascular measurements throughout the study (n=30).
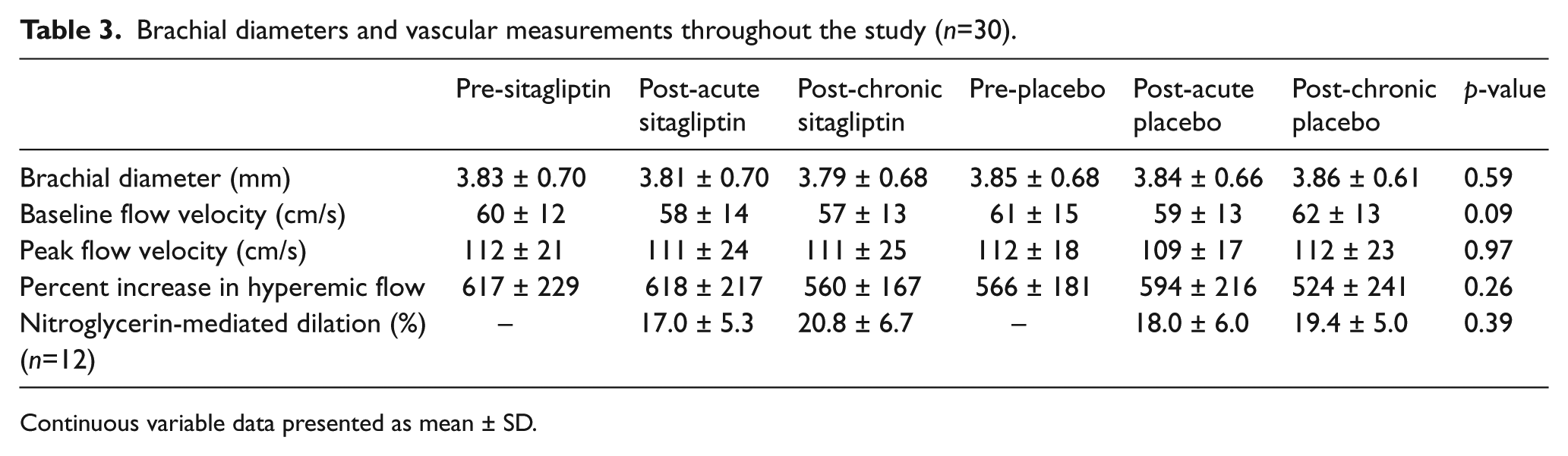
Continuous variable data presented as mean ± SD.
ICAM-1 and VCAM-1 measurements prior to and following 2 hours and 8 weeks of sitagliptin or placebo dosing are shown in Table 4. Plasma ICAM-1 levels were significantly lower post-acute dosing of sitagliptin compared to chronic sitagliptin administration (p<0.05). However, the ICAM level post-acute sitagliptin dosing was not significantly different than ICAM-1 measurements at any other time point. Plasma VCAM-1 was not significantly different at any time point (p=0.15).
Measurements of endothelial activation (n=29).
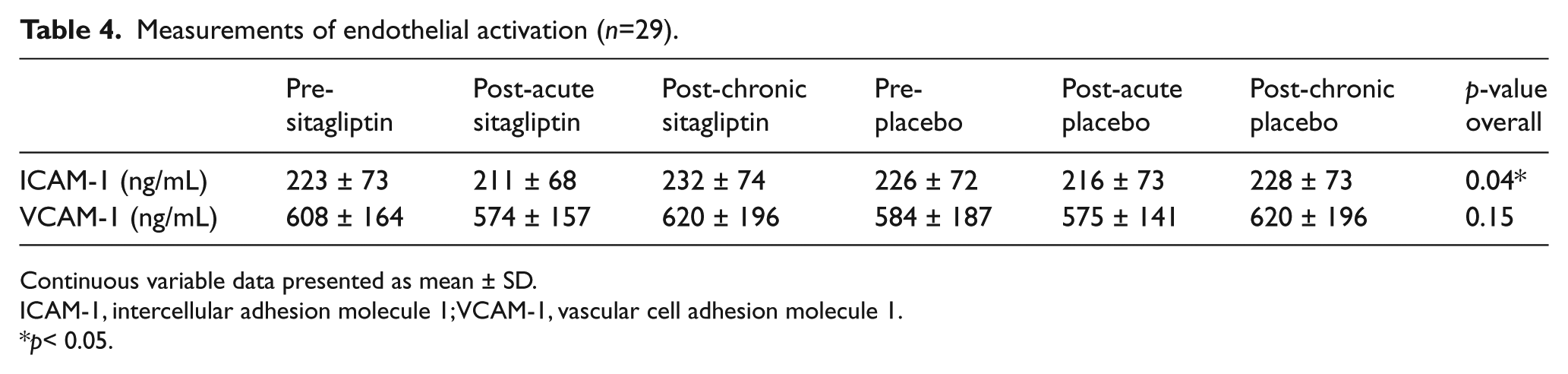
Continuous variable data presented as mean ± SD.
ICAM-1, intercellular adhesion molecule 1; VCAM-1, vascular cell adhesion molecule 1.
p< 0.05.
Discussion
In a randomized, double-blind, placebo-controlled study, DPP-4 inhibition with sitagliptin neither acutely nor chronically impacted endothelium-dependent vasodilation in humans with T2DM on concomitant metformin therapy. The observed reduction of glycosylated hemoglobin with chronic sitagliptin therapy is similar to that seen in the TECOS trial, which demonstrated no significant impact of sitagliptin on cardiovascular risk over a median follow-up of 3 years. 36 While our data suggest acute administration of a single dose of sitagliptin may reduce circulating ICAM-1 levels, this finding is not sustained with chronic therapy and the level was not significantly lower than placebo, suggesting this finding is not clinically meaningful. Taken together in the context of prior data, this work suggests DPP-4 inhibition’s impact on vascular endothelial function in humans on metformin therapy is neutral, consistent with larger clinical trials demonstrating a lack of significant impact of DPP-4 inhibition on ischemia-based cardiovascular outcomes.36–38
Two previous studies have investigated the in vivo effects of DPP-4 inhibition on vascular endothelial function.24,25 In one randomized, double-blind crossover study, vildagliptin administered to 16 subjects with T2DM for 4 weeks improved forearm blood flow responses to intra-arterial acetylcholine infusion compared to acarbose in a population in which all subjects were on metformin, and 38% were also on sulfonylurea therapy. 24 In a second randomized crossover study, both sitagliptin and alogliptin were reported to impair brachial FMD% relative to baseline following 6 weeks of therapy in a small number of subjects, of which only a minority (<35%) were on metformin therapy. 25 Glycosylated hemoglobin levels in both studies were similar compared to the current report. Differences in the findings between our study and these prior reports may relate to methods used to measure endothelial function, unmeasured differences in study populations, and differences in the use of metformin and other medications. Taken in the context of these prior data, our data support the vascular safety of using DPP-4 inhibition in the setting of metformin background therapy and extend this to suggest DPP-4 inhibition does not acutely impact vascular endothelial function. In light of the growing body of cell culture and animal data supporting favorable effects of DPP-4 inhibition of the vascular endothelium,14–23 the vascular impact of DPP-4 inhibition in the absence of metformin background therapy in humans remains unclear and merits further investigation.
Clinical trials focusing on cardiovascular risk with DPP-4 inhibition have suggested that saxagliptin and alogliptin, but not sitagliptin, may be associated with an increased risk of hospitalization for heart failure.36–38 Subgroup analyses suggest this risk occurs in patients with underlying renal insufficiency and a history of heart failure. All subjects enrolled in our study had normal renal function and no history of heart failure. Whether DPP-4 inhibition has adverse effects on the vasculature in these higher risk patient subgroups that could contribute to worsening heart failure merits future study.
Following 8 weeks of DPP-4 inhibition, we observed improvements in glycemic control but did not observe an increase in circulating insulin levels. DPP-4 inhibitor-induced increases in insulin levels depend on proximity to meals. Therefore, measurement of insulin levels following an overnight fast, as done in our protocol, may not reveal an increase in circulating insulin levels despite improved glycemic control with DPP-4 inhibition.
It is important to discuss the findings of our study of DPP-4 inhibition in the context of findings from prior studies employing glucagon-like petide (GLP)-1 agonists and their impact on endothelial function, as both classes of medications target the same pathway to improve glycemic control, and recent favorable data on the cardiovascular impact of GLP-1 agonists in high-risk patients. DPP-4 inhibitors competitively inhibit the enzyme that breaks down GLP-1, which is released in response to a meal. GLP-1 agonists are incretin mimics that bind GLP-1 receptors. Studies looking at the impact of direct GLP-1 infusion consistently showed an improvement in endothelial function.39–42 However, findings of studies employing GLP-1 receptor agonists have been mixed. A 16-week study showed an improvement in endothelial function with exenatide compared to glimepiride, 39 while a 12-week study did not show a similar benefit compared to metformin. 42
The reasons for differences in impact between DPP-4 inhibitors and GLP-1 agonists on endothelial function could be related to several factors such as differences in study design, including timing of measurements relative to meals and differences in the activity of comparator therapies.42–44 Native GLP-1 is released in response to meals. Therefore, in fasting studies, any favorable effects of DPP-4 inhibition on the vasculature related to increased native GLP-1 levels may be mitigated due to lower levels of circulating GLP-1. Conversely, GLP-1 receptor agonists bypass the need for production and release of native GLP-1 into the circulation. Future studies of DPP-4 inhibitors should consider testing the impact of DPP-4 inhibition on endothelial function after meals or a glucose challenge to better determine their effect on the vasculature via incretin-based mechanisms. Additionally, while DPP-4 inhibitors act by increasing insulin levels through increased post-prandial GLP-1 levels, the lack of a significant impact of these periodic increases in insulin levels on NO bioavailability likely, in part, relates to impaired endothelium-derived NO synthase activation due to impaired insulin receptor signaling well-described in diabetes.7,13,45
Limitations
Our data have some limitations. We did not measure markers of vascular inflammation other than ICAM-1 and VCAM-1. While it is possible other inflammatory markers could be altered by sitagliptin, the lack of change in endothelium-dependent vasomotor function with sitagliptin therapy does not support a systemic anti- or pro-inflammatory effect of sitagliptin. Overall, our study population had relatively good glycemic control at baseline (average glycosylated hemoglobin of 6.8%), similar to prior human studies examining the vascular effects of DPP-4 inhibition. We cannot determine from our work the impact of DPP-4 inhibition on endothelial function in subjects with poorer glycemic control. Balanced against these limitations are the robust design of our study and the contribution of these data to an area of controversy in the literature as well as the novel insights into the acute impact of DPP-4 inhibition on vascular endothelial function.
Conclusions
In 30 subjects with T2DM on metformin background therapy, acute and chronic DPP-4 inhibition with sitagliptin has no appreciable impact on human conduit vessel endothelial function. These data support the vascular safety regarding the use of sitagliptin in patients on metformin background therapy without renal sufficiency and a history of heart failure. Further work will be necessary to determine the impact of sitagliptin and other DPP-4 inhibitors in the absence of background metformin and in the setting of co-morbidities not represented in the current study.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding / Role of Sponsor
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by an investigator-initiated grant from Merck, Sharp and Döhme Corporation to Dr Widlansky. Dr Widlansky also receives support from NIH grants HL125409, HL081587, and HL128240. The sponsor did not contribute in the performance, analyses, or interpretation of this research study. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
