Abstract
Patients with inferior vena cava (IVC) filter-associated deep venous thrombosis (DVT) are a challenging subset of patients for endovascular intervention. Given the lack of available data pertaining to this clinical scenario, the purpose of this study was to evaluate the authors’ experience with the use of endovascular treatment for DVT in patients with IVC filters. Primary aims included assessing the technical and clinical success, complications, and clinical patency in these patients. This was a retrospective single-center review of adult patients with IVC filters undergoing endovascular treatment of DVT between 1/2005 and 4/2014. Patient electronic medical records were reviewed for demographic data, anticoagulation status, symptoms, symptomatic extremities, extent of thrombosis, therapies received, technical and clinical success, and complications. Query yielded 82 patients (mean 53 years, range 18–96; 66% male), all of whom were included in our analysis. The majority of patients presented with lower extremity pain and swelling, with extensive clot burden despite the use of anticoagulant medication. Treatment elements utilized included pharmacologic lysis in 92%, mechanical thrombectomy in 77%, angioplasty in 63% and stent placement in 50% of patients. Interventions were technically successful in restoring flow in 87% of patients, and clinically successful in improving presenting symptoms in 79% of patients. By SIR criteria, 24% of patients experienced complications (categorized as 10% minor and 14% major). There were two deaths from intracranial hemorrhage. The probability of thrombosis-free survival at 1, 3, 6, 9 and 12 months was 0.85 (CI 0.74–0.93), 0.81 (CI 0.69–0.89), 0.74 (CI 0.62–0.83), 0.70 (CI 0.57–0.8) and 0.70 (CI 0.57–0.8), respectively. Endovascular interventions are usually effective in relieving symptoms in patients with DVT and pre-existing IVC filters. However, these outcomes are achieved with significant complication rates that may exceed those observed when endovascular therapy is provided for other DVT populations.
Keywords
Introduction
The technical and clinical success rates as well as complications of endovascular therapy for deep venous thrombosis (DVT) have been extensively documented in the literature.1–9 The authors of the multicenter randomized CaVenT trial reported substantial benefits in the prevention of post-thrombotic syndrome (PTS) after catheter-directed treatment at 2 years (26% relative risk reduction) and 5 years (39% relative risk reduction).4,10
Currently, an NIH sponsored multicenter, randomized controlled trial (ATTRACT 11 ) is underway to compare rates of PTS in patients with acute proximal DVT who receive endovascular therapy with conventional medical therapy compared to conventional medical therapy alone.
A subgroup of patients at particular risk for poor DVT outcomes and potentially higher complications is those who receive inferior vena cava (IVC) filters. 12 In over 20 years since the development of percutaneous interventions for DVT treatment, there have been few reports detailing the benefits and safety profile of endovascular therapy for DVT in the subpopulation of patients who have a pre-existing IVC filter.13–15 This group may be hypothesized to carry a higher risk for recurrent DVT following endovascular intervention given their prior history of DVT, the likely presence of thrombus of varying age within the filter in many patients, hypercoagulability, the greater likelihood of having contraindications to anticoagulation, and the presence of a venous foreign body. When bilateral DVT prompts use of an increased dose or duration of thrombolytic drugs, peri-procedural bleeding could also be more frequent. However, with greater clinical severity due to more extensive thrombus involving iliac vessels and IVC, there may be more to gain from therapy and such treatment may be worth the increased risks. Currently, there is little evidence to help guide interventionalists in making difficult risk/benefit decisions in these patients. Furthermore, advanced age, bilateral acute iliac DVT, active cancer and limb threatening thrombosis are often present in this cohort, but may be excluded from randomized clinical trials such as the ATTRACT trial. 11
The purpose of this study was to examine the authors’ experience with endovascular DVT treatment in patients with IVC filters in order to aid therapeutic decision-making, guide informed consent discussions, and help establish risk/benefit profiles in similar patients for both the interventionalist and referring providers.
Materials and methods
Patients
See Table 1 for patient demographics. This study was approved by our institutional review board. Informed consent was obtained prior to the procedures. Declaration of Helsinki principles were adhered to in all cases. Computerized radiology reports from January 2005 through April 2014 were retrospectively queried to identify all patients with existing IVC filters presenting for DVT endovascular treatment. Patients were excluded if a caval filter was placed in preparation for endovascular treatment and/or were younger than 18 years old. From this group, 82 patients with DVT were identified as having undergone therapy in the treatment of 134 limbs.
Patient demographics.
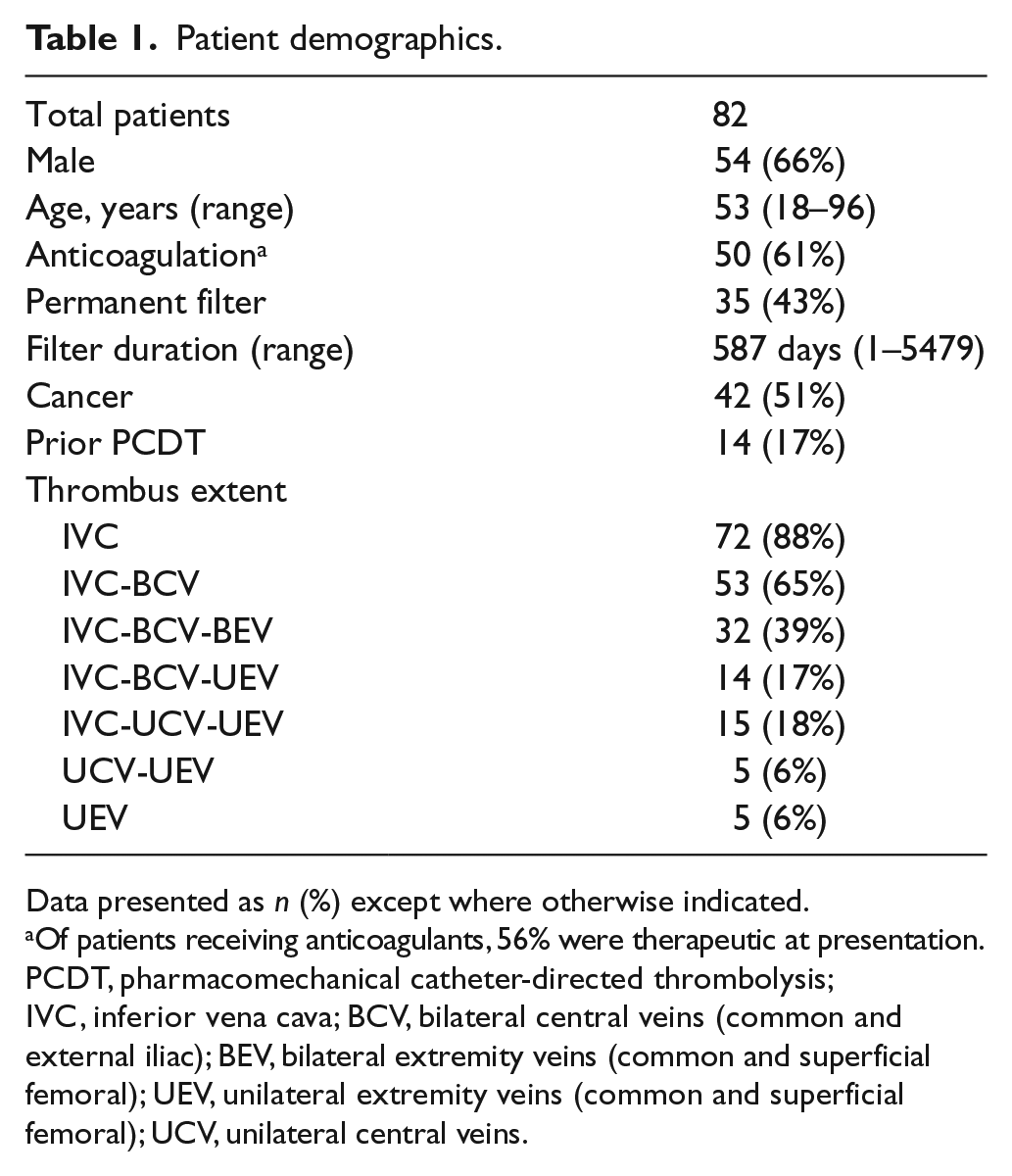
Data presented as n (%) except where otherwise indicated.
Of patients receiving anticoagulants, 56% were therapeutic at presentation.
PCDT, pharmacomechanical catheter-directed thrombolysis; IVC, inferior vena cava; BCV, bilateral central veins (common and external iliac); BEV, bilateral extremity veins (common and superficial femoral); UEV, unilateral extremity veins (common and superficial femoral); UCV, unilateral central veins.
Medical record evaluation
The patients’ electronic medical records were reviewed by a single author (who was not involved in the patients’ treatment) for demographic data, type of IVC filter placed as well as indication and duration, anticoagulation type, whether the patient was therapeutically anticoagulated at the time of presentation, hypercoagulable condition, clinical presentation, prior and subsequent endovascular treatment, modality used to discover the thrombus (all were confirmed by venography), fibrinogen levels, intersession lysis infusion changes, lytic dosages, clinical outcomes, complications, and clinical/radiological patency. When exact filter placement date was not available, dates were rounded up to the nearest month or year. Follow-up intervals were defined by last available clinical records including radiology examinations, clinical notes and autopsy reports with pertinent information about the clinical issue in question.
Definitions
Technical success was defined as restoration of venous flow through the treated vessels, as judged by the operator. If imminent re-thrombosis (vessel patency with poor inflow) was reported, then the case was treated as a technical failure. Sessions were defined as the patient being brought back to the interventional radiology suite for venography and evaluation. Specific endovascular interventions were defined according to the reporting standards proposed by Vedantham et al. 3 Thrombosis-free survival is defined by absence of clinical or imaging findings of thrombosis in a treated or untreated limb. Complications were defined by Society for Interventional Radiology criteria (SIR): minor (A) and (B) being no therapy or consequence, and nominal therapy and no consequence, respectively. SIR major complications included: (C) requiring therapy, hospitalization less than 48 hours; (D) requiring major therapy, unplanned increase in level of care, prolonged hospitalization greater than 48 hours; (E) permanent adverse sequelae; or (F) resulting in death. 16
Materials and interventions
The specific method and extent of intervention was at the discretion of the treating interventionalist for each individual patient. Figures 1–3 outline the various methods used by our interventionalists for endovascular treatment. Patients who received thrombolytic infusions were monitored overnight in a step-down or intensive care unit.
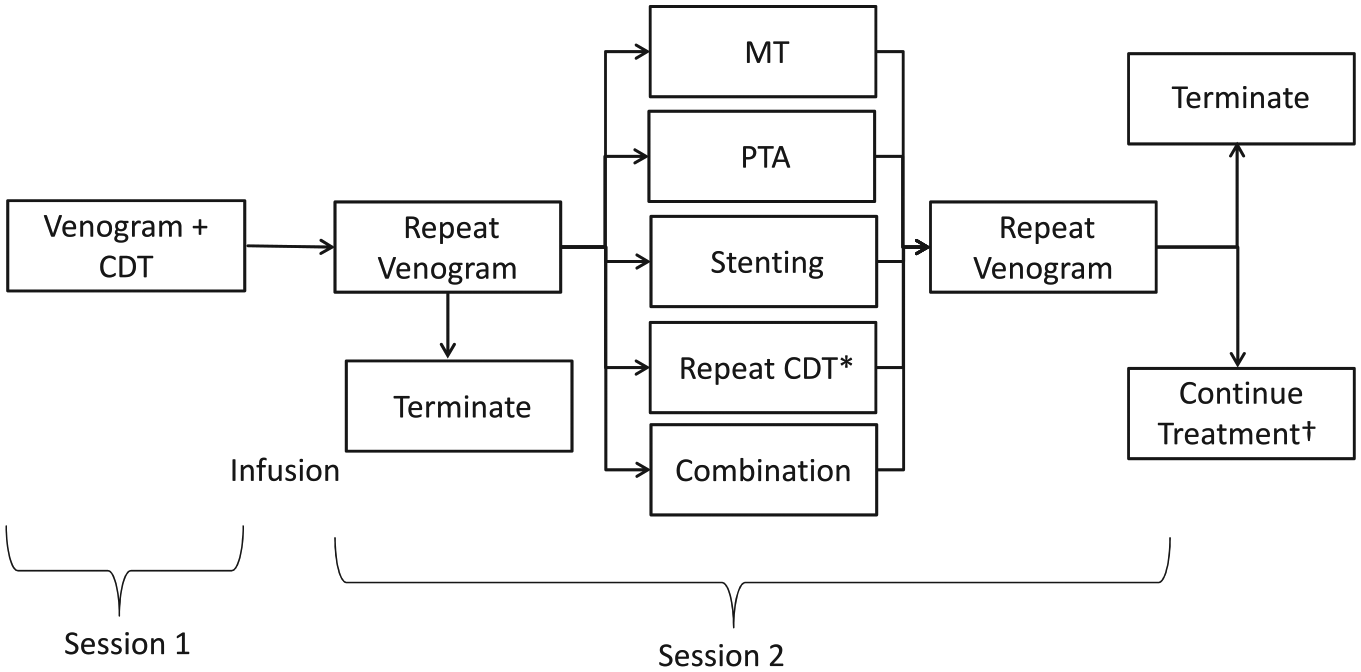
The extended infusion protocol at our institution began with popliteal venous access using ultrasound guidance, followed by placement of multi-sidehole infusion catheters and infusion of a lytic agent. Patients were subsequently monitored overnight in the intensive care unit and brought back to the department for follow-up venography at which point the interventionalist terminated the procedure, performed MT, angioplasty, and/or stenting or continued the infusion. Venography was then performed and the procedure was terminated or treatment continued dependent on the findings. This method was employed in 46 (56%) patients. *With repeat CDT, the catheters were replaced after the venogram and lytic agent was administered while the patient was observed either in the department or in the intensive care unit. As such, the repeat venogram occurred in session 3. †Continued treatment included any of the listed treatments during the same session or over the course of several sessions. CDT, catheter-directed intrathrombus thrombolysis; MT, mechanical thrombectomy; PTA, percutaneous transluminal angioplasty.
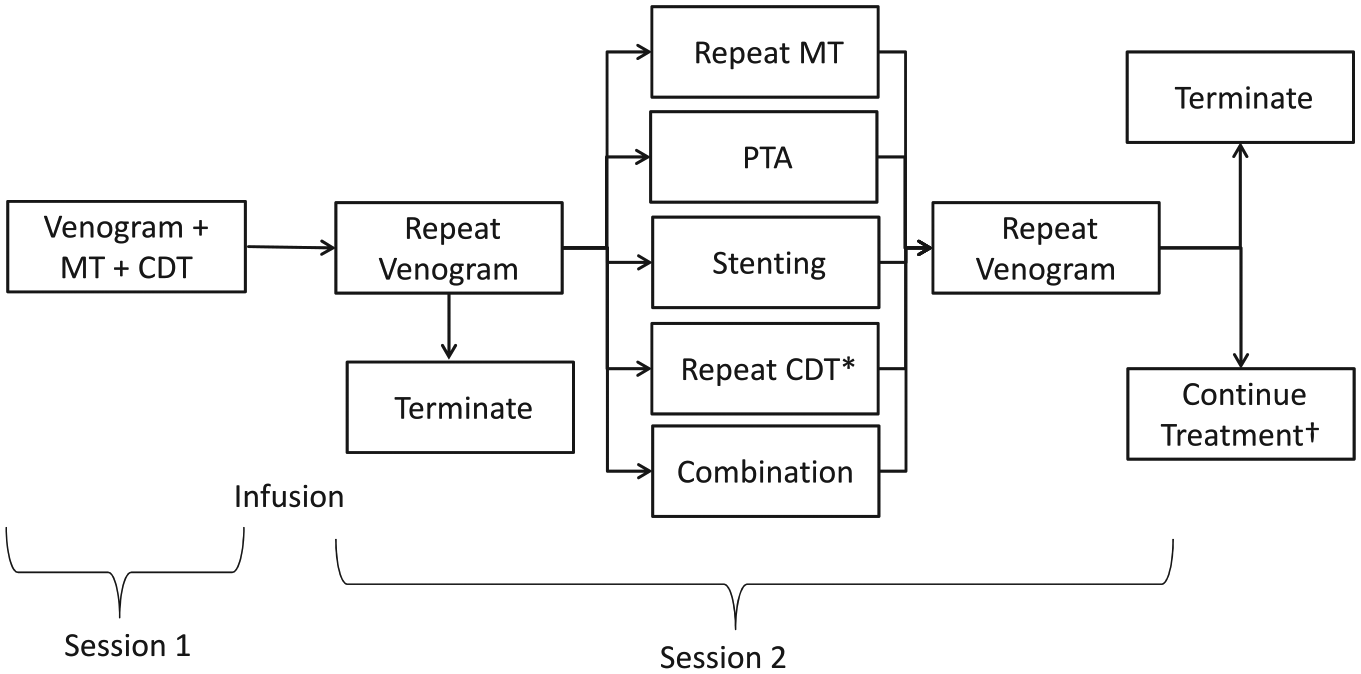
In 13 (16%) patients, mechanical thrombectomy was used during the initial session and the catheters were left in place for extended infusions with subsequent venography to document response and need for further intervention. *With repeat CDT, the catheters were replaced after the venogram and lytic agent was administered while the patient was observed either in the department or in the intensive care unit. †Continued treatment included any of the listed treatments during the same session or over the course of several sessions. CDT, catheter-directed intrathrombus thrombolysis; MT, mechanical thrombectomy; PTA, percutaneous transluminal angioplasty.
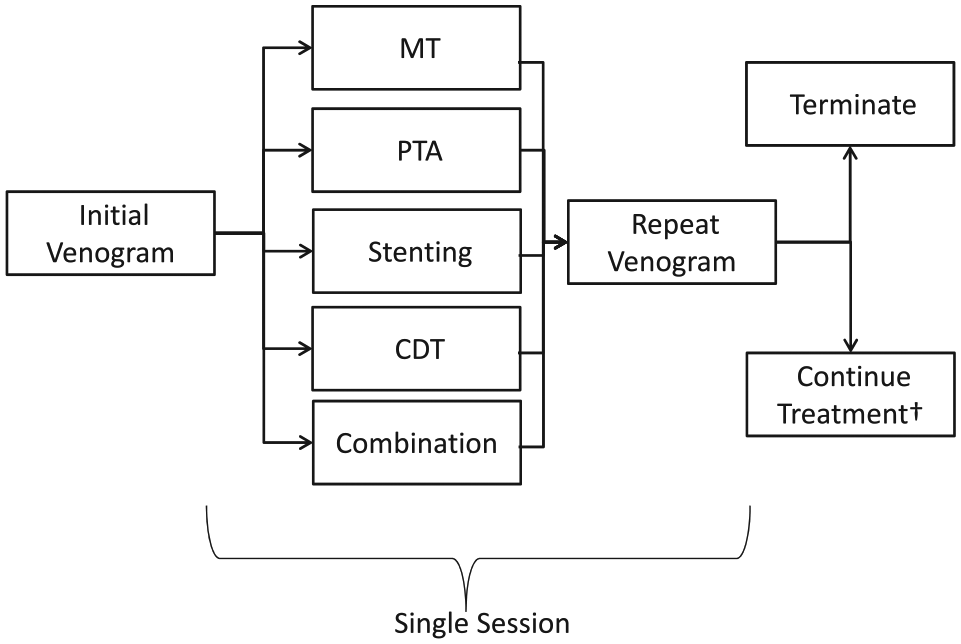
There were 12 (15%) patients who received procedural lytic agents alone or in combination with MT without extended infusion. There were seven patients who were not administered lytic agents who received only MT, angioplasty or stenting alone due to comorbidities, coagulopathy, recent operation, patient agitation leading to procedure termination, stented veins in which lysis would have a minimal effect and in one case the reason was unspecified. CDT, catheter-directed intrathrombus thrombolysis; MT, mechanical thrombectomy; PTA, percutaneous transluminal angioplasty.
The mechanical thrombectomy devices were advanced typically through a 7–9 French vascular sheath. Devices used for thrombectomy can be found in Table 2. Multiple devices were used in 30 of 63 patients. The thrombolytic agents used were urokinase (Abbott Laboratories, North Chicago, IL, USA) in 10 patients and alteplase (Genentech, San Francisco, CA, USA) in 63 patients. The dosing and infusion parameters for administration were guided by physician preference and extent of thrombus (typical alteplase dose was 0.01 mg/kg/h with a maximum of 1 mg/h). A single case exceeded a total of 1 mg/h in 2008 with 4 mg/h administered for a total of 6 hours. The stents used were 10–20 mm self-expandable WALLSTENT™ (Boston Scientific, Natick, MA, USA). Systemic anticoagulation during endovascular treatment varied by treating physician. Systemic intravenous heparin with a target activated partial thromboplastin time (aPTT) of 1.5–2.5 times control was used in 26% of patients and a subtherapeutic target was used in 43% of patients. Procedural heparin bolus alone without systemic heparin administration post-procedure was used in 5% of patients. Low-molecular weight heparin (weight-based therapeutic dosing) was used in 11% of patients. Non-heparin systemic anticoagulation was used in 10% (therapeutic ranges for INR or aPTT with Warfarin®, argatroban and bivalirudin). Heparin dosages were unspecified in 3%, and 2% did not receive systemic anticoagulation.
Mechanical thrombectomy devices utilized.

Microvena, White Bear Lake, MN, USA.
Possis, Mineapolis, MN, USA.
Rex Medical, Conshohocken, PA, USA.
Covidien, Mansfield, MA, USA.
Not including balloon use for angioplasty purposes.
Radiology report evaluation
Procedure reports were reviewed by a single author for extent of thrombosis, access, filter positioning, type of intervention, number of sessions, administration of heparin, lytic agent, use and type of mechanical thrombectomy (MT), use of angioplasty, use of stenting, technical success, post-procedural anatomic filter positioning and complications. Venographic results were extracted from the radiologic reports. Procedural venographic images were not independently reviewed. Technical success was defined as restoration of venous flow. IVC filter anatomic positioning was extracted from the reports with special attention to the patients who received stents in the IVC; when uncertain, the images were reviewed. When the type of filter was not evident by the prior procedure note or medical record, the images were reviewed to specify the type.
Data analysis
Statistical analyses were performed in conjunction with the biostatistics department at Washington University in St Louis. All the analyses were conducted using SAS 9.3 (SAS Institute, Cary, NC, USA). Continuous variables were summarized by means, medians, standard deviations, and ranges. Categorical variables were summarized by contingency tables. For continuous variables, group difference was examined via Wilcoxon Rank-Sum tests. For categorical variables, chi-squared tests were used to compare across groups.
Results
Clinical presentation
See Table 1 for patient demographics. There were 82 patients who met the criteria for entry into the study. The mean age was 53 (18–96; SD 15.8) years. Fifty-four patients (66%) were male. There were 134 limbs at risk (54 patients had bilateral, 18 left, and 8 right lower extremity symptoms). There were two patients without extremity symptoms: one patient had symptomatic pulmonary embolism with clot above the filter and the second was a young patient with IVC thrombus despite therapeutic anticoagulation. The most common symptom complex was lower extremity pain and swelling alone (68%). Documented pulmonary embolism was identified with or without additional findings in 12%. More severe manifestations of disease were seen in 16%, and included phlegmasia alba dolens, phlegmasia cerulea dolens, ulceration or compartment syndrome. A total of 51% of patients had cancer, 13% had lupus anticoagulant, factor V Leiden was present in 8% and prothrombin 20210 mutation was present in 5%. Of the 82 patients, 50 (61%) were on anticoagulation at presentation with the most common being warfarin (58%) followed by low-molecular weight heparin (18%) and unfractionated heparin (14%). Dalteparin, dabigatran and rivaroxaban accounted for the remainder, each under 5%.
The exact or approximate filter placement times were known in 74/82 patients with 26/74 requiring approximation. The mean filter duration was 1.5 years (range: 1 day to 15 years) in the 74 patients where these data were available. Indications for filter placement were: failed anticoagulation (40%), bleeding on anticoagulation or at risk of bleeding on anticoagulation (22%), pre-operatively for prophylaxis or cessation of anticoagulation in preparation for an operation (22%), trauma (2%), high clot burden at risk for fatal pulmonary embolus (2%) and unspecified or unknown (11%). See Table 3 for information regarding IVC filter types.
Inferior vena cava (IVC) filters.

Cordis, Warren, NJ, USA.
Cook Medical, Bloomington, IN, USA.
Rex Medical, Conshohocken, PA, USA.
Bard, Tempe, AZ, USA.
Braun, Evanston, IL, USA.
Boston Scientific, Natick, MA, USA.
Previous endovascular therapy for DVT was performed in 14 (17%) patients either at institutions outside our hospital system or preceding the January 2005 inclusion date.
Treatments
Data regarding interventions can be found within Figures 1–3. An illustrative successful treatment case is shown in Figure 4. Pharmacologic thrombolysis was used in 75 (92%), MT in 63 (77%), angioplasty in 63%, and stent placement in 50% of patients. Most patients received a combination of interventions, with the most common being thrombolysis, MT, angioplasty and stenting in 23 (28%) patients. Lysis, MT and angioplasty were performed in 16 (20%) patients and lysis with MT in 14 (17%). Lysis, MT and stenting alone occurred in less than 10 patients and no patients received only angioplasty. Of the patients undergoing extended infusion, the alteplase dose was changed in eight patients with six patients having lysis stopped (six patients for low fibrinogen levels, one patient’s dose was increased and one patient for bleeding at the access site). The filters remained anatomically intact in 58/82 (71%) of patients with 11 (13%) having their filters removed and 13 (16%) deemed non-functional due to angioplasty or stenting through the filter.
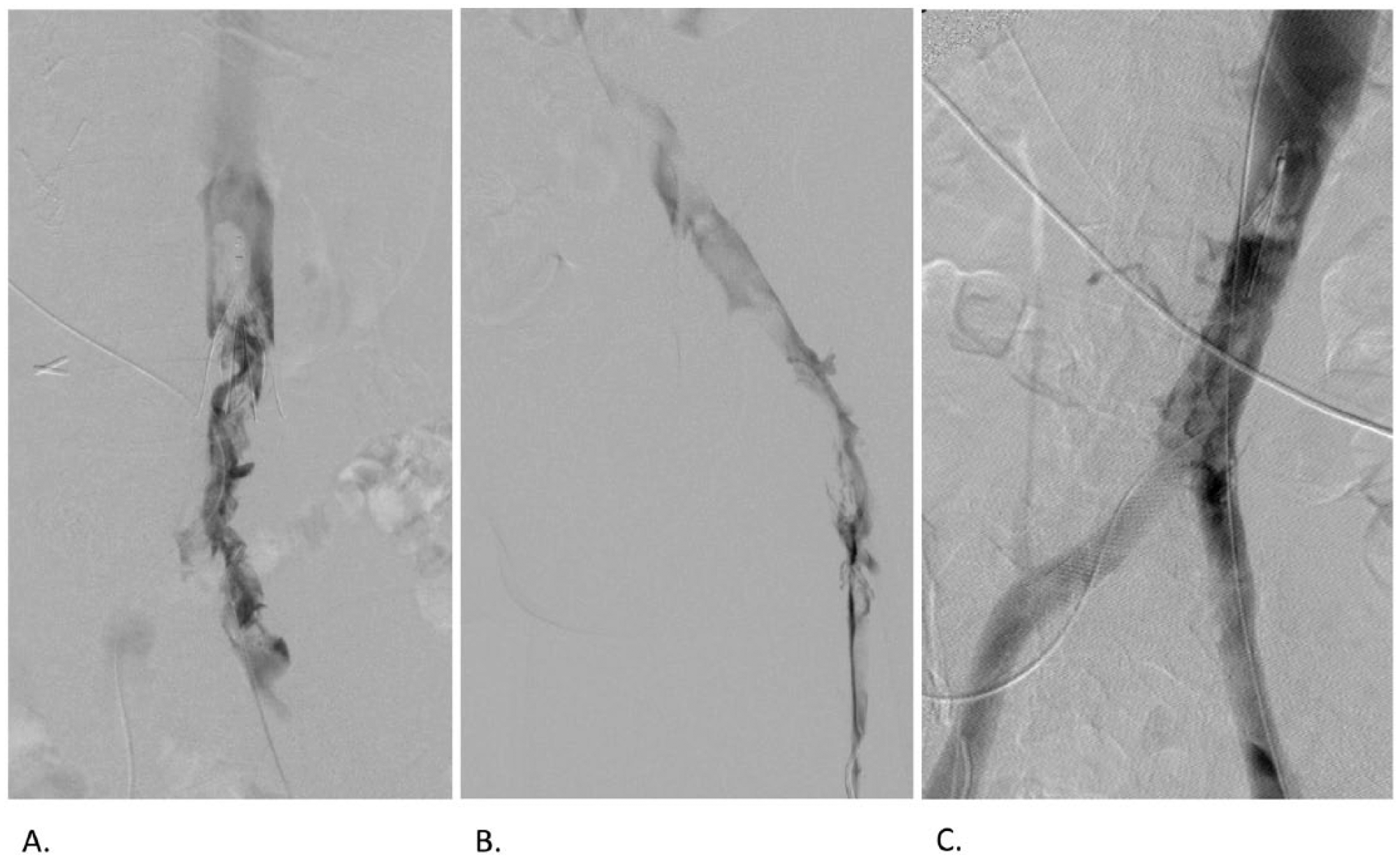
A. Patient in prone position with venography demonstrating acute thrombus in the cava, filter and external iliac veins without collateral vessels. The thrombus extends above the filter. B. Acute thrombus is present in the right common iliac vein extending into the femoral vein. C. After thrombolysis bilaterally and angioplasty and stenting of a May-Thurner lesion in the left common iliac vein (patient is prone), there is restored flow with minimal residual thrombus in the filter tip.
Records for dosage of urokinase were incomplete due to limitations of the electronic medical record at the time the procedures were performed. There was complete dosage data for 42/49 patients who received alteplase. The mean procedural dose in patients alone or with subsequent infusion was 8.9 (SD 5.4) mg without a significant difference in the two groups. The mean extended infusion dose was 20.0 (SD 10.9) mg of alteplase. The mean total dose for extended and procedural plus extended infusion was 25.0 (SD 13.0) mg. The average infusion time was 22.0 (range 1.0–64.1) hours. The mean number of treatment sessions was 1.9 (SD 0.7; range 1–4). Besides the use of alteplase instead of urokinase, there was no other major change in practice between 2005 and 2014.
Outcomes
Overall technical success was achieved in 71 patients (87%). Resolution or improvement of symptoms in the immediate post-procedural period was achieved in 65 patients (79%), with unknown clinical outcome due to immediate facility transfer in two patients. Procedural complications were observed in 20 (24%) patients. Eight patients (10%) had SIR minor A and B complications and 12 patients (14%) had major C/D/F complications. Two patients died (category F) from intracerebral hemorrhage (ICH). One of the patients with ICH who subsequently died received a similar amount of alteplase (23.0 mg) and adjunctive therapies as the rest of the cohort. The second death from ICH was a patient with an INR of 3.0, heparin-induced thrombocytopenia (HIT), metastatic adenocarcinoma (no evidence of intracranial metastases either on pre-procedure imaging or on subsequent autopsy), recent pericardial window and gangrene of bilateral lower extremities. The patient was deemed a poor candidate for amputation due to his co-morbidities, but wanted to proceed to improve his quality of life having accepted the high risk of intracranial or pericardial hemorrhage. The patient received similar alteplase (20.0 mg) dosing and adjunct therapy and was not administered heparin due to HIT. Of the three category D patients, one had common iliac vein extravasation requiring placement of a stent, one had respiratory distress requiring medical intensive care unit admission and one patient had an angioplasty balloon burst with a retained fragment in the filter managed with stented exclusion of the filter. The seven category C patients had excessive bleeding from the catheter site, a pre-existing ostomy, hematuria or epistaxis, all requiring blood transfusion. Temporal differences in clinical outcomes were not identified during the project period. There was no significant difference in bleeding complications when accounting for the systemic anticoagulation agent, method of delivery (i.e. bolus, pre-determined rate, therapeutic target, etc.), aPTT (greater than the upper limit of prescribed target range), or plasma fibrinogen nadir less than 150 mg/dL.
The mean follow-up time was 17.7 (range 0–99.0, SD 21.7) months. Of the 71 patients who had technically successful studies, one patient died of intracranial hemorrhage (included above) prior to evaluation for DVT, yielding 70 patients at risk for re-thrombosis. Kaplan–Meier analysis of the probability of thrombosis-free survival (Figure 5) at 1, 3, 6, 9 and 12 months was 0.85 (CI 0.74–0.93), 0.81 (CI 0.69–0.89), 0.74 (CI 0.62–0.83), 0.70 (CI 0.57–0.8) and 0.70 (CI 0.57–0.8). The patients lost to follow-up were as follows: two patients at 1 month, seven patients at 3 months, five patients at 6 months, five patients at 9 months and two patients at 12 months. There was no correlation between thrombosis-free survival and extent of thrombosis at presentation, although this analysis may be limited given that a large proportion of the group presented with extensive disease. Overall, 17 patients (21%) underwent subsequent endovascular DVT intervention (within our hospital system) whether planned (seven patients) or unplanned (10 patients).
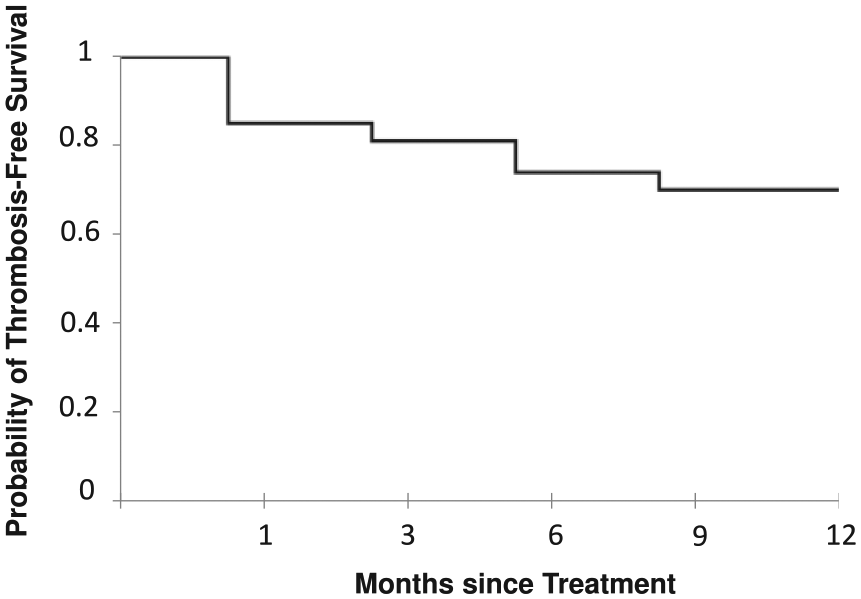
Kaplan-Meier analysis of the probability of thrombosis-free survival. Of the 70 successfully treated patients, the probability of thrombosis-free survival was 85% at 1 month (CI 0.74–0.93), 81% at 3 months (CI 0.69–0.89), 74% at 6 months (CI 0.62–0.83) and 70% at 9 and 12 months (CI 0.57–0.8).
Discussion
To our knowledge, this study is distinctive in providing outcome and complication information following endovascular treatment of DVT in a large cohort of patients with pre-existing IVC filters. Most patients had extensive clot burden with IVC and iliac involvement bilaterally. While pain and swelling were the most common presenting symptoms, documented pulmonary embolism and physical exam findings of venous compromise combined were present in 28%. These factors underscore the clinical severity of disease in this challenging population and may in part explain results that differ in some aspects to the DVT populations described in most other studies.
Our data suggest that despite often significant disease burden, endovascular therapy in patients with IVC filters can be effective, with similar technical and early clinical success rates published for patients without IVC filters undergoing similar treatment.4,15,17 Most published studies focused on treatment of first-time acute iliofemoral DVT, but in contrast a majority of our cohort had caval and bilateral iliofemoral involvement.4–6,8,18–24 Additionally, half of our patient population had cancer and almost a fifth had prior endovascular treatment for deep venous thrombosis. With these factors in mind, thrombosis-free survival was greater than 80% in the first 3 months and gradually plateaued to 70% at 9 and 12 months. While published follow-up and reporting intervals varied widely, a comprehensive literature review by Vedantham et al. reported 91% freedom from early re-thrombosis of treated segment within 1–3 months. 17 The long-term efficacy of catheter-directed thrombolysis (CDT) has not been rigorously studied, except for one multicenter randomized controlled trial that focuses on the development of PTS in a general population with high-proximal DVT (not necessarily involving the iliac vein or IVC). 4 Given the retrospective nature of this study spanning nearly 10 years, the available medical records did not allow for a proper assessment of PTS. The more objective available data focused on documented clinical recurrence of DVT and symptomatology which makes comparison of long-term outcomes with current available studies limited.
Patients with indwelling IVC filters represent a unique group of patients with increased risk of thrombosis. Virchow’s triad of stasis (flow alteration at the filter), trauma (local endothelial damage from implantation) and hypercoagulability (prior DVT, cancer, clotting disorders) all apply to this group. The presence of a permanent IVC filter has been shown to be associated with higher rates of DVT and a reduced rate of pulmonary embolism. 12 Additionally, patients who receive IVC filters are more likely to have contraindications to or failure of anticoagulants. Our study highlights the large thrombus burden in this population, most with extension of clot from the filter through both iliofemoral segments and in a quarter of patients extending above the filter.
In our study, 12% of patients experienced a major bleed (including two intracranial bleeds). This event rate is higher than that reported in the available multicenter randomized controlled trial (3% major bleeds, none intracranial, but the study utilized restrictive entry criteria), and near the upper end of reports from registries representing experiences closer to real-world care. In 1999, Mewissen et al. reported on a 473-patient prospective multicenter registry of patients receiving urokinase CDT. In that study, major bleeds were observed in 11.4% of patients. 2 In 2006, Vedantham et al. reviewed the available published literature to that time and identified an 8% pooled rate of major bleeding in patients undergoing CDT. 3 In 2014, Bashir et al. examined a large cohort of 3649 patients from the National Inpatient Sample database who had undergone CDT, finding that 11.5% received a blood transfusion during their hospital stay (compared with a 6.5% transfusion rate in a propensity-matched but non-randomized cohort of DVT patients who did not undergo CDT). 25
After therapy, 70% of indwelling IVC filters remained anatomically intact. Of the remaining filters, half were removed and the other half were stented and thus were non-functional. Most of the filters were left in place due to the long duration (average 1.6 years) and because 43% were permanent filters. The number of removed filters may be underestimated as patients may have had their filters removed outside of our hospital system at a later date. Only retrievable filters were removed (11 patients) within our cohort, while five retrievable filters were excluded by a stent. All non-functional permanent filters were excluded via stent. Neglén et al. investigated the safety and efficacy of angioplasty and stenting across an obstructed IVC filter compared with below the filter obstruction and in non-filter groups receiving venous stenting. 26 In their study, stenting through an obstructed filter did not yield significant differences in early or late stent occlusion when accounting for degree of occlusion. Additionally, there were no cases of IVC rupture or hemorrhage in their cohort, which was the case in our study as well. 26
Limitations
Limitations of this study include small sample size, retrospective design, heterogeneous endovascular treatment techniques, insufficient data to assess long-term clinical outcomes such as PTS, lack of independent venographic image review, lack of a control group of patients who did not receive endovascular therapy, and significant losses to follow-up.
Conclusion
Our analysis, although limited, demonstrates that the presence of an IVC filter should not necessarily be considered a contraindication to endovascular therapy for DVT. Rather, the use of endovascular therapy in this high-risk patient population may carry a unique risk/benefit profile that should be factored into clinical decisions regarding therapy. Our results suggest that technical success rates are acceptable in the short term, and while there may be lasting benefits to treatment for some patients, this comes at an increased risk of complications. Prospective studies are needed to further evaluate the risks and benefits of performing these procedures in this patient population.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
