Abstract
The purpose of this study is to characterize the plaque morphology of severe stenoses in the superficial femoral artery (SFA) employing combined near-infrared spectroscopy and intravascular ultrasound (NIRS-IVUS). Atherosclerosis is the most common cause of symptomatic peripheral arterial disease. Plaque composition of SFA stenoses has been characterized as primarily fibrous or fibrocalcific by non-invasive and autopsy studies. NIRS has been validated to detect lipid-core plaque (LCP) in the coronary circulation. We imaged severe SFA stenoses with NIRS-IVUS prior to revascularization in 31 patients (46 stenoses) with Rutherford claudication ⩾ class 3. Angiographic parameters included lesion location and stenosis severity. IVUS parameters included plaque burden and presence of calcium. NIRS images were analyzed for LCP and maximum lipid-core burden index in a 4-mm length of artery (maxLCBI4mm). By angiography, 38 (82.6%) lesions were calcified and 9 (19.6%) were chronic total occlusions. Baseline stenosis severity and lesion length were 86.0 ± 11.0% and 36.5 ± 46.5 mm, respectively. NIRS-IVUS identified calcium in 45 (97.8%) lesions and LCP in 17 (37.0%) lesions. MaxLCBI4mm was 433 ± 244. All lesions with LCP also contained calcium; there were no non-calcified lesions with LCP. In conclusion, this is the first study of combined NIRS-IVUS in patients with PAD. NIRS-IVUS demonstrates that nearly all patients with symptomatic severe SFA disease have fibrocalcific plaque, and one-third of such lesions contain LCP. These findings contrast with those in patients with acute coronary syndromes, and may have implications regarding the pathophysiology of atherosclerosis in different vascular beds.
Keywords
Introduction
Peripheral arterial disease (PAD) afflicts 8–12 million patients in the United States, 1 with the greatest prevalence in patients with age > 65 years, diabetes, chronic kidney disease, and history of smoking. PAD commonly co-exists with atherosclerotic coronary artery disease (CAD) and cerebrovascular disease (CVD), and it is presumed that atherosclerosis in different vascular territories reflects common underlying pathophysiological mechanisms. 2
The availability of invasive vascular imaging techniques has led to advances in our understanding of the pathogenesis of CAD. Recent studies of near-infrared spectroscopy (NIRS) suggest that plaque composition has important implications for plaque progression, plaque rupture and thrombosis, and risks during percutaneous coronary intervention.3–5 Combined NIRS and intravascular ultrasound (IVUS) is a new multi-modality invasive imaging technique that provides co-registered data on plaque architecture by IVUS and lipid composition by NIRS. 6 The present study was designed to characterize plaque morphology employing NIRS-IVUS in patients with symptomatic severe superficial femoral artery (SFA) stenoses prior to percutaneous revascularization.
Methods
Between December 2012 and April 2015, 31 patients with Rutherford claudication ⩾ class 3 underwent invasive angiography and NIRS-IVUS imaging prior to percutaneous revascularization, according to a protocol approved by the Human Investigations Committee at Beaumont Health System. Clinical, angiographic, NIRS, and IVUS analyses were performed on all target lesions. Clinical information included age, gender, Rutherford class, and co-morbidities.
Angiographic technique
Peripheral angiography was performed according to standard methods employing digital subtraction angiography (DSA) and 6-French vascular sheaths. A radiopaque ruler was placed adjacent to the thigh to guide NIRS-IVUS localization. Quantitative angiography (QA) was performed with Medis QAngio XA software (Rotterdam, The Netherlands) to assess lesion length, reference vessel diameter (RVD), minimum lumen diameter (MLD), and diameter stenosis (DS). Angiography was analyzed qualitatively to identify the number of calcified lesions.
NIRS-IVUS (near-infrared spectroscopyintravascular ultrasound) technique
NIRS and IVUS were performed over a 0.014-inch guidewire using the single-catheter InfraRedX TVC Imaging System (Burlington, MA, USA). Five lesions required pre-dilation with a 2.0-mm balloon to facilitate passage of the NIRS-IVUS catheter. The system consists of a 3.2-French rapid-exchange catheter that acquires 1000 NIRS measurements/12.5 mm of artery scanned. Each measurement interrogates 1–2 mm2 of the lumen surface area perpendicular to the long axis of the catheter, and rotational pullback is performed at 0.5 mm/s and 960 RPM. The measurement of the probability of lipid-core plaque (LCP) is displayed as a map, with the x-axis indicating the pullback position in millimeters and the y-axis the circumferential position of measurement in degrees. The map of probability of LCP is displayed in a chemogram using a false color scale. Yellow indicates a high probability of LCP, while red indicates a low probability. A summary of the presence of LCP at 2-mm intervals in four probability categories is presented as a block chemogram in (Figure 1). 3
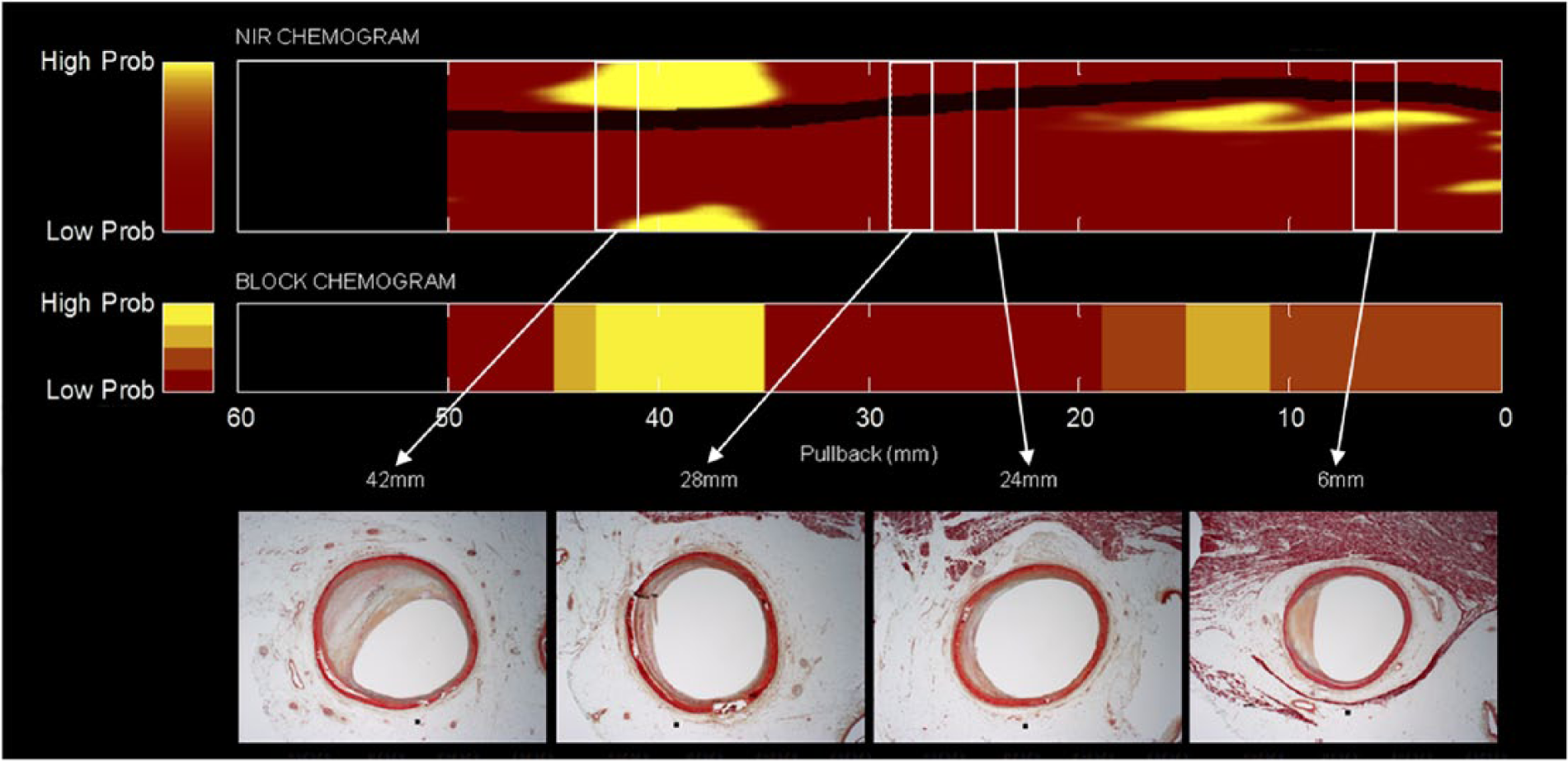
Near-infrared spectroscopy (NIRS) scan from a coronary artery from an autopsy study with histologic correlation. Upper panel: Chemogram depicting a false color map of the artery wall indicating the probability of LCP along the length (x-axis, in mm) and circumference (y-axis, in degrees) of the scanned artery (red = low probability, yellow = high probability). Middle panel: Block chemogram summarizing the presence of LCP at 2-mm intervals in four probability categories (yellow, tan, orange, red). Lower panel: Russell-Movat’s pentachrome-stained cross-sections (5-μm thick) at 42, 28, 24, and 6 mm, respectively, along the scanned artery (white rectangles). Image interpretation: In the chemogram, a prominent LCP signal is detected from 35 to 45 mm, occupying approximately 160° circumference. The block chemogram shows the highest probability LCP signals extend from 35 to 43 mm. The NIRS signals at 28, 24, and 6 mm demonstrate diffuse plaque with low probability of LCP. NIRS signals are confirmed by histology. (NIRS, near-infrared spectroscopy; LCP, lipid-core plaque.) Image courtesy of InfraRedX, Inc.
IVUS images were acquired via a rotating transducer operating at 40 MHz. Automatic pullback was initiated 1 cm distal to the lesion, and terminated 1 cm proximal to the target lesion. Registration of NIRS-IVUS and angiographic data was obtained by the following process (Figure 2). First, the proximal and distal edges of the target lesion were identified by angiography. Second, the distance from the MLD to the proximal and distal lesion edges was measured. Third, the MLD was identified on the co-registered NIRS-IVUS image. Fourth, the lesion on the NIRS-IVUS image was defined using the measurements obtained by QA from the MLD-proximal lesion edge and MLD-distal lesion edge. Once the lesion was defined by NIRS-IVUS, a frame-by-frame analysis of the entire lesion length was performed to identify the presence of calcium and LCP. The calcium and lipid-core burden at the MLD was highlighted in the final analysis.
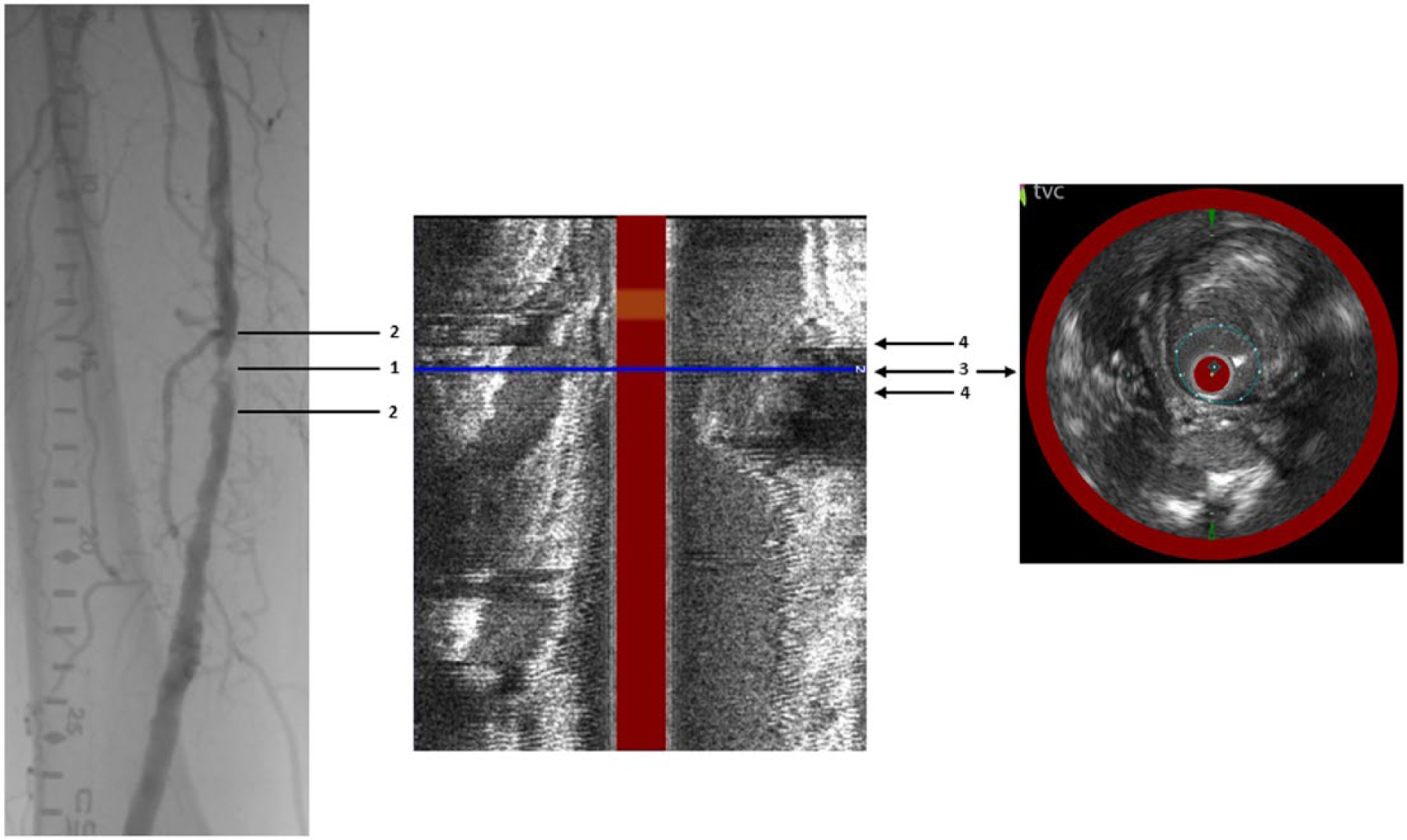
Methodology used to identify SFA lesions on angiography and NIRS-IVUS. Left panel: The MLD (1) was identified by angiography. The distance from the MLD to the edges of the proximal and distal lesions was calculated (2). Middle panel: The MLD was then identified on the co-registered NIRS-IVUS image longitudinal view (3). Right panel: The NIRS-IVUS lesion was defined by the distance from the MLD to the proximal and distal edges based on measurements from angiography (4). (SFA, superficial femoral artery; NIRS-IVUS, near-infrared spectroscopy – intravascular ultrasound; MLD, minimum luminal diameter.)
IVUS analysis
IVUS images were analyzed as previously described. 7 The lesion length, RVD, reference vessel area, MLD, and minimum lumen area (MLA) were measured by IVUS. The plaque burden and eccentricity index (EI) were calculated as previously described. 8 The extent of calcification was assessed by measuring the circumferential arc of calcium in degrees (0–360°) using manual calipers, the number of quadrants that the calcium occupied (0–4) at the MLD, the length of calcium in the lesion, and the percentage of lesion length that was calcified. Quadrants of calcium were defined in relation to the circumference of a circle within a given cross-section of plaque, as follows: one quadrant (< 91°); two quadrants (91–180°); three quadrants (181–270°); and four quadrants (> 270°).
NIRS analysis
Assessment of LCP was performed as previously reported for CAD by identifying at least one bright yellow block (high probability) on the NIRS block chemogram.3–5 The amount of LCP was quantitated by the lipid-core burden index (LCBI), the maximum LCBI in any 4-mm region (maxLCBI4mm), the circumferential arc of LCP (0–360°), the number of quadrants occupied by LCP (0–4), the length of LCP, and the percentage of lesion length that contained LCP. Since the combination of IVUS and NIRS permits assessment of fibrotic plaque, calcification, and LCP, we classified lesions into four groups according to the presence or absence of lipid and/or calcium.
Statistical analysis
Mean and standard deviation (mean ± SD) were calculated for continuous variables, and frequency (%) was calculated for dichotomous variables. SAS for Windows (version 9.3; Cary, NC, USA) was used for statistical analysis.
Results
Baseline characteristics were notable for advanced age, and a high incidence of diabetes, smoking, chronic kidney disease, and concomitant CAD (Table 1). Target lesions were evenly distributed between the left and right SFA; 89.1% were located above the adductor canal (Table 2). Procedural success was achieved in all lesions, including balloon angioplasty and stenting in 44 lesions and atherectomy in 4 lesions.
Baseline clinical characteristics of 31 patients with symptomatic superficial femoral artery disease.

Angiographic characteristics of 46 superficial femoral artery stenoses.

SFA, superficial femoral artery; QA, quantitative angiography; CTO, chronic total occlusion.
NIRS-IVUS
IVUS identified calcified plaque in 45 (97.8%) lesions in 30 (96.8%) patients; only 1 (2.2%) lesion did not contain calcium (Table 3). The circumferential distribution of calcium was one quadrant in 30.4%, two quadrants in 28.2%, three quadrants in 26.0%, and four quadrants in 13.0% of lesions (Figure 3). The circumferential arc of calcium at the MLD was 127 ± 84°, and the longitudinal extent of calcification was 77.6 ± 29.3% of lesion length. By NIRS, 17 (37.0%) lesions contained LCP, and in such lesions the maxLCBI4mm was 433 ± 244 (Table 3). LCP was identified at the MLD of 14 (82.3%) lesions. The circumferential distribution of LCP was one quadrant in 41.2%, two quadrants in 29.4%, and three quadrants in 29.4% (Figure 3). There were no lesions with four quadrants of LCP.
NIRS-IVUS (near-infrared spectroscopy-intravascular ultrasound) characteristics of 46 superficial femoral artery stenoses.
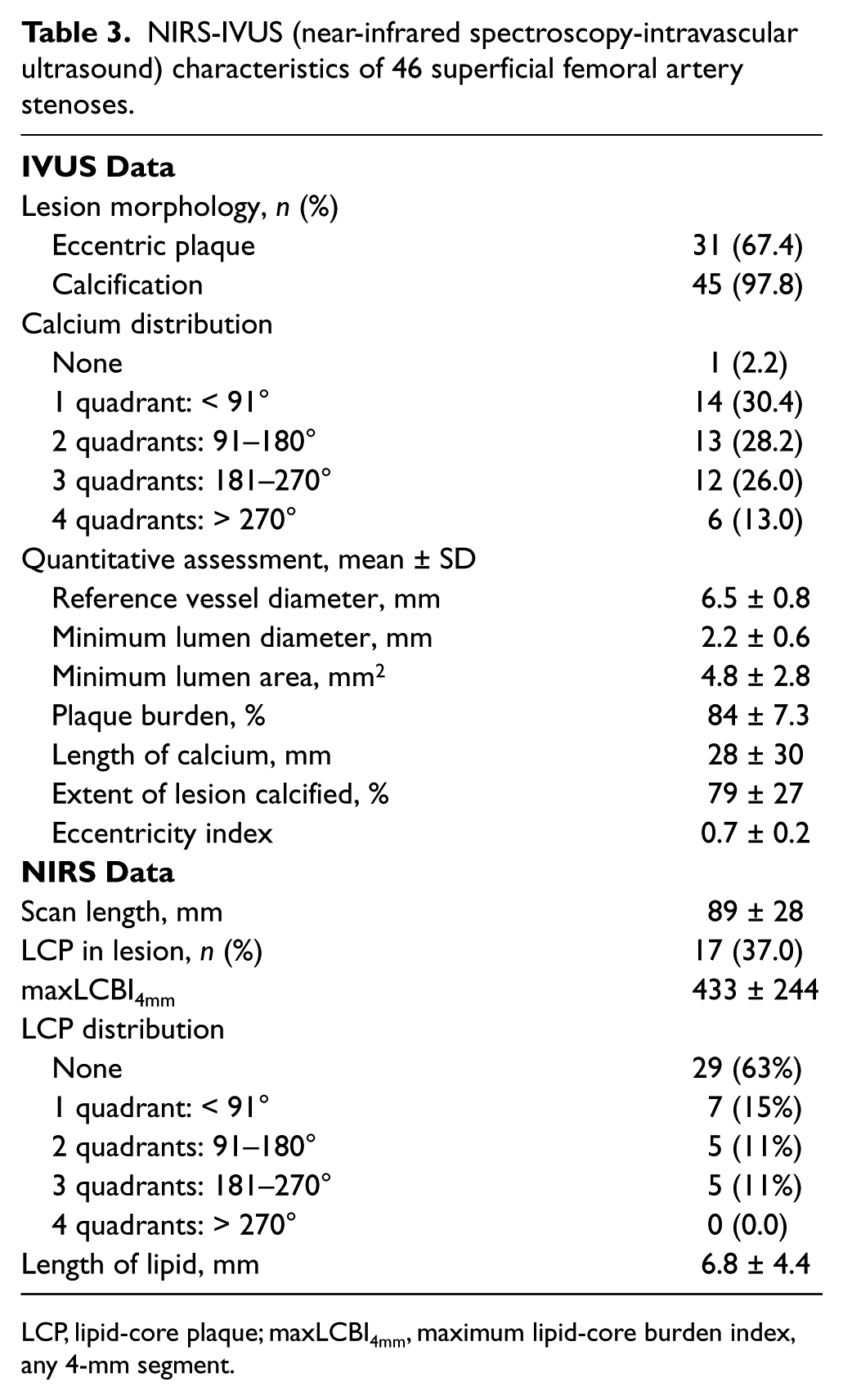
LCP, lipid-core plaque; maxLCBI4mm, maximum lipid-core burden index, any 4-mm segment.
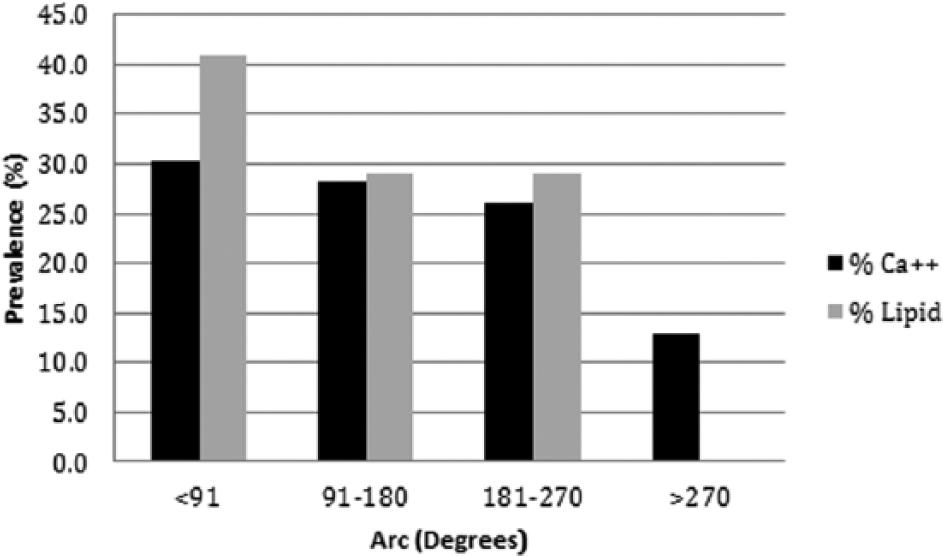
Distribution of calcified and lipid-core plaque burden in 46 superficial femoral artery lesions.
Lesions were classified into four types depending on the presence of calcium and/or LCP (Figure 4). The most common plaque composition was fibrocalcific plaque without LCP in 28 (60.1%) lesions. In contrast, fibrocalcific plaque with LCP was identified in 17 (37.0%) lesions, and fibrous plaque without calcium or LCP was identified in 1 (2.2%) lesion. All lesions with LCP also contained calcium; there were no non-calcified lesions with LCP.
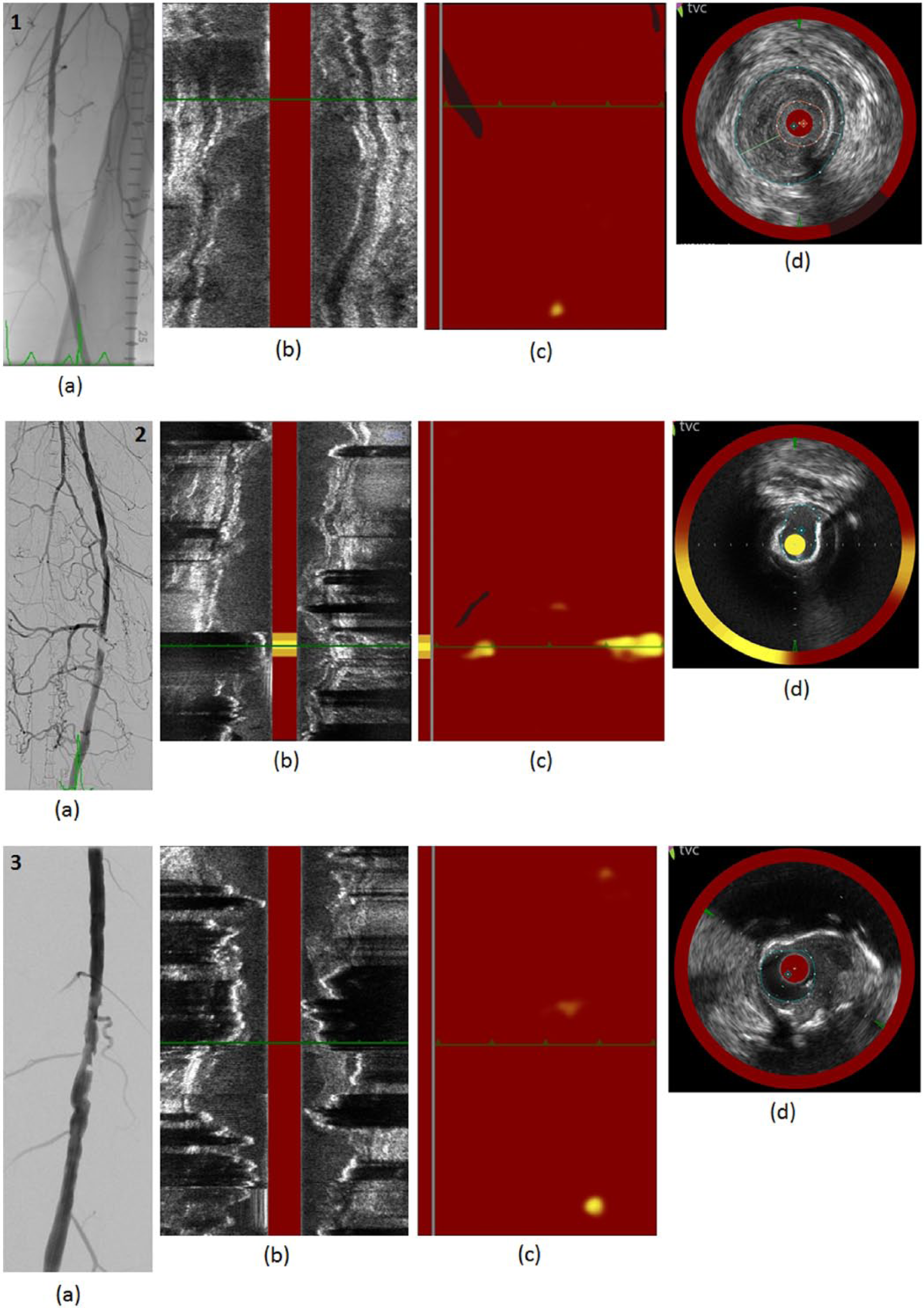
Co-registered angiographic and NIRS-IVUS images. Top (1): Fibrous plaque without LCP. (a) Angiogram shows a severe focal stenosis. (b) IVUS long axis shows irregular stenosis without calcification. (c) NIRS chemogram and block chemogram show absence of LCP in target lesion. (d) Co-registered NIRS-IVUS short axis shows dense fibrous plaque at the MLD. Middle (2): Fibrocalcific plaque with LCP. (a) Angiogram shows a severe focal stenosis. (b) IVUS long axis shows irregular stenosis with calcification. (c) NIRS chemogram and block chemogram show LCP in target lesion. (d) Co-registered NIRS-IVUS short axis shows dense fibrocalcific plaque with LCP at the MLD. Bottom (3): Fibrocalcific plaque without LCP. (a) Angiogram shows a severe focal stenosis. (b) IVUS long axis shows irregular stenosis with calcification. (c) NIRS chemogram and block chemogram show absence of LCP in target lesion. (d) Co-registered NIRS-IVUS short axis shows dense fibrocalcific plaque without LCP at the MLD. (NIRS-IVUS, near-infrared spectroscopy – intravascular ultrasound; LCP, lipid-core plaque; MLD, minimum luminal diameter.)
Discussion
Combined NIRS-IVUS has been previously utilized to examine plaque composition in the coronary circulation in patients with acute and chronic coronary syndromes.5,9 To our knowledge, this is the first study employing combined NIRS-IVUS for atherosclerotic plaque characterization in the peripheral circulation. Observations from the present study demonstrate that nearly all patients with symptomatic severe SFA disease have fibrocalcific plaque, and approximately one-third of such lesions contain LCP.
Atherosclerosis is the most common cause of CAD, CVD, and PAD. Autopsy and surgical studies typically describe atherosclerosis with varying degrees of fibrous, fibrocalcific, and lipid-laden plaque. When characterized in this fashion, atherosclerosis is qualitatively similar in many vascular beds. However, as we develop a more sophisticated understanding of atherosclerosis, it is apparent that some arterial beds are relatively resistant to atherosclerosis (e.g. the left internal mammary artery); atherosclerotic risk factors may have different impact in different arterial beds; and phenotypic expressions of atherosclerotic plaque may vary in different arteries. 10 In fact, comprehensive morphological classifications of atherosclerotic lesions are still under debate and modification.2,11 While contrast angiography has allowed us to describe arterial ‘luminology’, the availability of newer invasive imaging techniques has expanded our ability to characterize in vivo atherosclerosis.
SFA atherosclerosis
Recent autopsy and magnetic resonance imaging (MRI) studies report a high prevalence of fibrous plaque in the SFA.8,12 According to the most recent AHA classification of atherosclerosis, 52% of SFA lesions at autopsy are classified as Type VIII (fibrocalcific plaque without LCP, in which < 50% of plaque area is calcified).2,10
Invasive imaging for PAD
Currently available intravascular imaging techniques include IVUS, optical coherence tomography (OCT), and NIRS. IVUS may be subdivided into greyscale-IVUS and virtual histology-IVUS (VH-IVUS). These intravascular modalities have been validated in the coronary circulation, and facilitate percutaneous coronary intervention (PCI) planning and stent implantation. Intracoronary imaging can define lumen dimensions of the target vessel, assess lesions of intermediate severity, and assess the risk for peri-procedural myocardial infarction and angiographic no-reflow. In comparison to greyscale-IVUS, NIRS-IVUS provides identical IVUS information, plus information about LCP. While VH-IVUS provides a color-coded map of the atherosclerotic plaque components, including fibrous (dark green), fatty (yellow/light-green), white (calcified), and red (necrotic LCP) components, the accuracy of VH-IVUS is limited in the presence of heavy calcification.13,14 A positive relationship has been shown between VH-derived % necrotic core plaque and NIRS-derived LCBI in non-calcified plaques, but not in calcified plaques. 15 OCT is much less practical for imaging long segments of SFA disease because of limited penetration depth and the need for large volumes of contrast.
As is true for coronary angiography and CAD, peripheral angiography underestimates the extent of atherosclerosis in patients with PAD. 16 In general, IVUS applications for PAD are similar to those for CAD, including accurate assessment of lumen and vessel dimensions, enhanced assessment of lesion eccentricity and calcification, and clinical utility for ambiguous lesions before and after intervention. 17 The difference in resolution and penetration depth of ultrasound waves emitted at 40 MHz and 20 MHz frequencies (present in different commercial IVUS transducers) is negligible and did not have a significant effect on image acquisition in the SFA. 18
In a comparative analysis of angiography and IVUS in SFA disease, both provided similar information on diameter stenosis; however, RVD was larger by IVUS, and the presence and severity of calcification was better assessed with IVUS. 19 By VH-IVUS, 63% of SFA stenoses had primarily fibrous plaque, 14% had necrotic plaque, 9% had calcific plaque, and 14% had mixed plaque. Another IVUS study in the ileofemoral arteries reported that 42% of patients had plaque rupture, which was strongly associated with recent acute coronary syndromes. 20 Compared to PAD patients with popliteal disease, VH-IVUS showed that patients with tibial disease had less fibrous and fibrofatty plaque, more calcified plaque, and similar LCP. 21
The present study is the first to use NIRS-IVUS to characterize the composition of atherosclerotic plaque in patients with symptomatic PAD. Although the observed prevalence of fibrocalcific plaque (97.8%) is much higher than prior autopsy and MRI studies, the differences are probably due to the presence of severe PAD in all of our patients. Another unique observation was that 37.0% of SFA lesions contained LCP, and LCP was localized to the arc of calcified plaque. These NIRS observations in the SFA contrast sharply with those in the coronary circulation for several reasons. First, LCP by NIRS is an invasive marker for acute coronary syndromes, plaque rupture, and thrombosis5,9; in the SFA, the role of LCP remains unclear. Second, while large LCP identifies patients at higher risk for embolization and no-reflow after PCI, 4 LCP was not associated with any complications after endovascular intervention in the SFA. Third, the presence of echo-attenuated plaque is considered a reliable IVUS signature for unstable CAD 22 ; while such findings are unreliable in heavily calcified lesions, identification of LCP by NIRS is not impaired by heavy calcification.
Clinical implications
It is well known that the burden of atherosclerosis in multiple vascular beds is correlated with cardiovascular death; SFA atherosclerosis indicates a generalized susceptibility to atherosclerosis, CAD, and cardiac death. 12 It is also known that IVUS is superior to angiography for the assessment of plaque burden in patients with CAD and PAD, but the differential impact of IVUS versus angiographic plaque burden on long-term prognosis has not been determined. Finally, while the extent of LCP seems to have a clear relationship among patients with unstable acute coronary syndromes, 23 22% of patients with mild SFA disease have LCP 24 ; further study is needed to determine if LCP burden has prognostic importance in PAD patients. It is unknown whether LCP prevalence in CAD versus PAD is due to alternative pathways for atherosclerotic plaque initiation and progression, structural differences in the arterial wall, or differential mechanical and shear forces.10,25
Applications of NIRS-IVUS in PAD
Although it is clear from previous studies in the coronary circulation that different clinical syndromes have different NIRS signatures,4,9,23 this has yet to be defined in the SFA. Potential applications for NIRS-IVUS are similar to IVUS, including accurate assessment of vessel and lumen dimensions, the extent of vessel and lesion calcification, and enhanced characterization of ambiguous lesions. Whether identification of these characteristics will permit more informed selection of endovascular devices (i.e. angioplasty, bare and drug-eluting stents, atherectomy, drug-eluting balloon) is not known at this time. While identification of LCP appears to have value in the coronary arteries for predicting no-reflow, we did not identify flow disturbances in this study. It is possible, but unproven, that LCP in the carotid or renal arteries could have a predictive role for distal embolization; further study is needed.
Limitations
The primary limitation of this study is the lack of histologic correlation with SFA atherosclerosis by NIRS, since no such studies have been performed. This is in contrast to the coronary circulation, in which NIRS has been validated with histology. Nevertheless, in an appropriately-sized lumen (⩽ 4 mm), there is no reason to expect signals for LCP to be different in PAD patients than in CAD patients. However, other investigators have reported correlations between PAD atherosclerosis and IVUS in the femoral arteries, 26 and IVUS has been reported in PAD patients for identifying plaque rupture. 20 Other studies have compared IVUS and angiography for assessment of vessel and lumen dimensions and for plaque morphology in PAD patients. 19 Two major reviews discussed the applications of IVUS in PAD patients, specifically for assessment of vessel dimensions and plaque morphology.17,27 Second, the findings in our study are based on 46 lesions at a single center, so larger studies are needed to confirm these findings. Third, our study only evaluated patients with severe PAD who were referred for revascularization for lifestyle-limiting claudication (Rutherford ⩾ 3) refractory to medical therapy. Therefore, we were unable to evaluate the lipid-core burden in patients with milder disease. Finally, the depth of penetration from the center of the catheter is 3.5 mm for NIRS and 4 mm for IVUS with the 40-MHz transducer, assuming the catheter is in the center of the vessel. Although the SFA RVD was 6.5 mm, the MLD was 2.3 mm, which permitted adequate penetration and consistent images in all lesions.
Conclusions
NIRS-IVUS demonstrates that nearly all patients with symptomatic severe SFA disease have fibrocalcific plaque, and one-third of such lesions contain LCP. These findings contrast with those in patients with acute coronary syndromes, and may have implications regarding the pathophysiology of atherosclerosis in different vascular beds.
Footnotes
Acknowledgements
The authors would like to thank Paul A Poronto, RN, BAS for image acquisition and Judith Boura, MS for statistical analysis.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Dr Goldstein is a consultant for, and has equity in, InfraRedX, Inc.; Dr Madder receives research support from InfraRedX, Inc.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
