Abstract
Aortic diseases include a wide range of pathological conditions: aortic aneurysms, pseudoaneurysms, acute aortic syndromes, atherosclerotic and inflammatory conditions, genetic diseases and congenital anomalies. Acute aortic syndromes have acute onset and may be life-threatening. They include aortic dissection, intramural haematoma, penetrating aortic ulcer and traumatic aortic injury. Pain is the common denominator to all acute aortic syndromes. Pain occurs regardless of age, gender and other associated clinical conditions. In this review, we deal with the main findings in the clinical setting and the most recent indications for diagnostic imaging, which are aimed to start an appropriate treatment and improve the short- and long-term prognosis of these patients.
Keywords
Introduction
According to the most recent definitions,1,2 aortic diseases include a wide range of pathological conditions: aortic aneurysms, pseudoaneurysms, acute aortic syndromes (AAS), atherosclerotic and inflammatory conditions, genetic diseases and congenital anomalies. AAS 3 are a group of aortic diseases with acute onset; they may be life-threatening and include aortic dissection (AD), intramural haematoma (IMH), penetrating aortic ulcer (PAU) and traumatic aortic injury (TAI). The feature of AAS is laceration of the tunica media, 4 with or without intimal breach, within which the blood seeps and can contribute to the progression of dissection. Blood can remain localized (as in the case of IMH), or it can flow outside the arterial wall (as in the case of the rupture of PAU or trauma).
According to the most recent literature data, the incidence of AAS ranges from three to five cases per 100,000 inhabitants. 5 Data from the Global Burden Disease 2010 project indicate that between 1990 and 2010, the overall mortality rate for aortic aneurysms and AD increased from 2.49 to 2.78 per 100,000 inhabitants, with a male predominance.6,7 In contrast, abdominal aortic aneurysms (AAAs) showed a reduction in the prevalence and incidence in the general population, with older people and males being most at risk. 7 Despite a lower incidence of AAS in women, more frequent atypical symptoms and a greater delay in diagnosis lead to a worse prognosis compared to men. 8 If the patient is not treated promptly, the AD mortality rate can be 1.4% per hour within the first 48 hours. 4 Ascending AD carries a much higher mortality rate compared to dissection of the descending thoracic aorta. Analysing the data from the International Registry of Acute Aortic Dissection (IRAD) database 9 suggests that patients with type A AD (according to the Stanford classification explained below) without surgical intervention have a higher mortality rate (58%) compared with surgically treated patients with type A AD (26%). Whereas, patients with medically treated type B AD have the lowest mortality rate (10.7%), while mortality for surgically treated patients affected with type B AD reached 31.4%. Currently, the major predisposing factors that contribute to the onset of AAS are: arterial hypertension (75% of patients with AD or IMH have high blood pressure); cigarette smoking; dyslipidaemia; trauma from car accidents or deceleration injury; use of illicit drugs (cocaine, amphetamines); connective tissue disorders (Marfan syndrome, Loeys-Dietz syndrome, Ehlers-Danlos syndrome); congenital heart diseases such as bicuspid aortic valve or aortic coarctation; inflammatory vascular disorders (giant cell arteritis, Takayasu’s arteritis, Behçet’s disease, Ormond’s disease) or infective vascular disorders (syphilis, tuberculosis); and iatrogenic factors.9–13
In this article, we review the main findings in the clinical setting and the most recent indications for diagnostic imaging techniques, since a correct diagnosis of AAS is essential to start appropriate treatment and improve the short and long-term prognosis of these patients.1,2,14
Clinical symptoms and signs
Patients with AAS often present with symptoms that cannot be associated to a specific disorder (AD, IMH or PAU), which may lead to serious outcomes. 15 Owing to low sensitivity and specificity of the clinical AAS symptoms, physicians can experience serious difficulty in making a correct differential diagnosis. Pain is the common denominator to all AAS patients; it occurs regardless of age, gender and other associated clinical conditions.9,11,16,17
Aortic aneurysms, especially an AAA, develop without noticeable symptoms. Sometimes patients can experience a sensation of abdominal fullness or mass. Physical examination of AAAs reveals a pulsatile epigastric or peri-umbilical mass, especially in thin subjects or in subjects with large aneurysms. AAAs usually become symptomatic when they rupture, and in this case they cause intense abdominal pain, associated with cardiovascular collapse. With rupture into the retroperitoneal cavity, a newly formed peri-aortic haematoma can cause severe abdominal or back pain that may radiate to the flank or groin. AAAs may also cause embolization of thrombotic material, which is layered on their inner surface.
Thoracic aortic aneurysms (TAAs) 18 are usually asymptomatic, and their detection is often incidental during a diagnostic imaging. When symptomatic, they usually cause severe pain or a feeling of fullness in the chest, symptoms of aortic valve insufficiency, including worsening heart failure, or systemic embolism related to a mural thrombus. In other cases, they may show signs of compression of adjacent structures (trachea, bronchus, oesophagus), causing dyspnoea, bronchospasm, cough, haemoptysis, and dysphagia or haematemesis in case of formation of aorto-oesophageal or aorto-bronchial fistula. During dissection, sudden onset of sharp thoracic or lumbar pain may occur, which may be associated with hypotension in case of rupture in the pleural cavity or mediastinum.
AD is one of the most frequent AAS and is caused by the formation of an intimal tear and a false lumen within the tunica media that may propagate either anterograde or retrograde throughout the course of the artery. 19 The symptoms of AD may be variable and can mimic those of more common conditions such as acute myocardial infarction. Therefore, it is recommended that clinicians consider this diagnosis when caring for patients with chest pain to avoid diagnostic delays that may be fatal. 10 Pain is the most frequent symptom (96% of cases) 9 and occurs abruptly, reaching maximum intensity in the early stages. Patients describe pain as lacerating, rending and piercing. Sometimes the patient, driven by a sense of impending disaster, writhes, looking for a position that brings some relief. The pain is typically migrant and tends to follow the path of the aorta 9 ; it may radiate from the chest to the back or vice versa. Other clinical manifestations that can delay diagnosis and increase the rate of mortality 20 are: acute heart failure (often caused by a severe acute AD of the ascending aorta); syncope (which is frequent during AD of the ascending aorta and generally associated with haemopericardium, rupture or stroke); and severe nausea and vomiting (which is related to the involvement of the abdominal viscera). The main objective findings of AD are: hypertension (found in about 70% of patients) 9 ; hypotension (in case of cardiac tamponade, aortic rupture or acute heart failure related to acute aortic insufficiency); deficit of the peripheral pulses; neurological manifestations; and physical findings of aortic valve insufficiency (in cases of dissection in the ascending direction); acute ischaemia of the lower limbs or of the peripheral organs; and signs of acute myocardial infarction (if the dissection involves the ostium of one or more coronary arteries).
PAU and IMH are two AAS that may present with similar symptoms to AD, causing chest pain or intense low back pain. For this reason, diagnostic imaging is useful for clinicians, as it serves to distinguish them from AD.
IMH is a collection of blood within the tunica media of the aortic wall, without the presence of a clear intimal flap or false lumen. Many authors consider IMH as a result of the rupture of the vasa vasorum or the ischaemia of the tunica media in older patients. 21 The presenting symptoms and risk factors associated with IMH are similar to those observed in AD – chest pain and/or acute lumbar pain. In addition, an IMH that propagates in the ascending direction may lead to haemopericardium, rupture and, in very rare cases, aortic regurgitation. 22 Whereas there is no intimal flap or false lumen associated with IMH, stroke or visceral ischaemia would be uncommon accompanying complications.
PAU occurs with ulceration of an atherosclerotic plaque involving the aortic wall and blood penetrates through the internal elastic lamina to reach the tunica media. It is often associated with the formation of coexisting IMH. 23
PAU can be found throughout the course of the aorta, mainly at the level of the thoracic and abdominal segments. It is occasionally found incidentally during imaging; however, PAU can exhibit symptoms such as acute chest or lumbar pain, similar to AD. Physicians should be informed about the propensity to rupture of PAU, so it is important to know how to recognize it. PAU can degenerate in AD, but most patients have no associated aortic valve insufficiency, pulse deficits or visceral ischaemia.
Classifications
A temporal classification system distinguishes AD as acute if it occurs within 2 weeks, subacute for 2–6 weeks, or chronic if more than 6 weeks from the onset of pain. 10
The anatomical classification systems most commonly used in the clinical setting are those of DeBakey and Stanford, which guide the clinician in important decisions regarding surgical and non-surgical treatment (Figure 1). DeBakey’s classification 24 recognizes three types of aortic dissection: type I begins in the proximal aorta and involves the ascending aorta, the aortic arch and the descending aorta; type II is confined to the ascending aorta; type III is confined to the descending aorta, and it starts just below the origin of the left subclavian artery. Type III dissections are further divided into two groups: type IIIa dissections originate distal to the left subclavian artery but extend proximally and distally, mostly above the diaphragm; and type IIIb dissections originate distal to the left subclavian artery, extending only distally and may extend below the diaphragm. Stanford’s classification defines two types of aortic dissection: type A dissection involves the ascending aorta, and type B dissection involves the descending aorta. 25
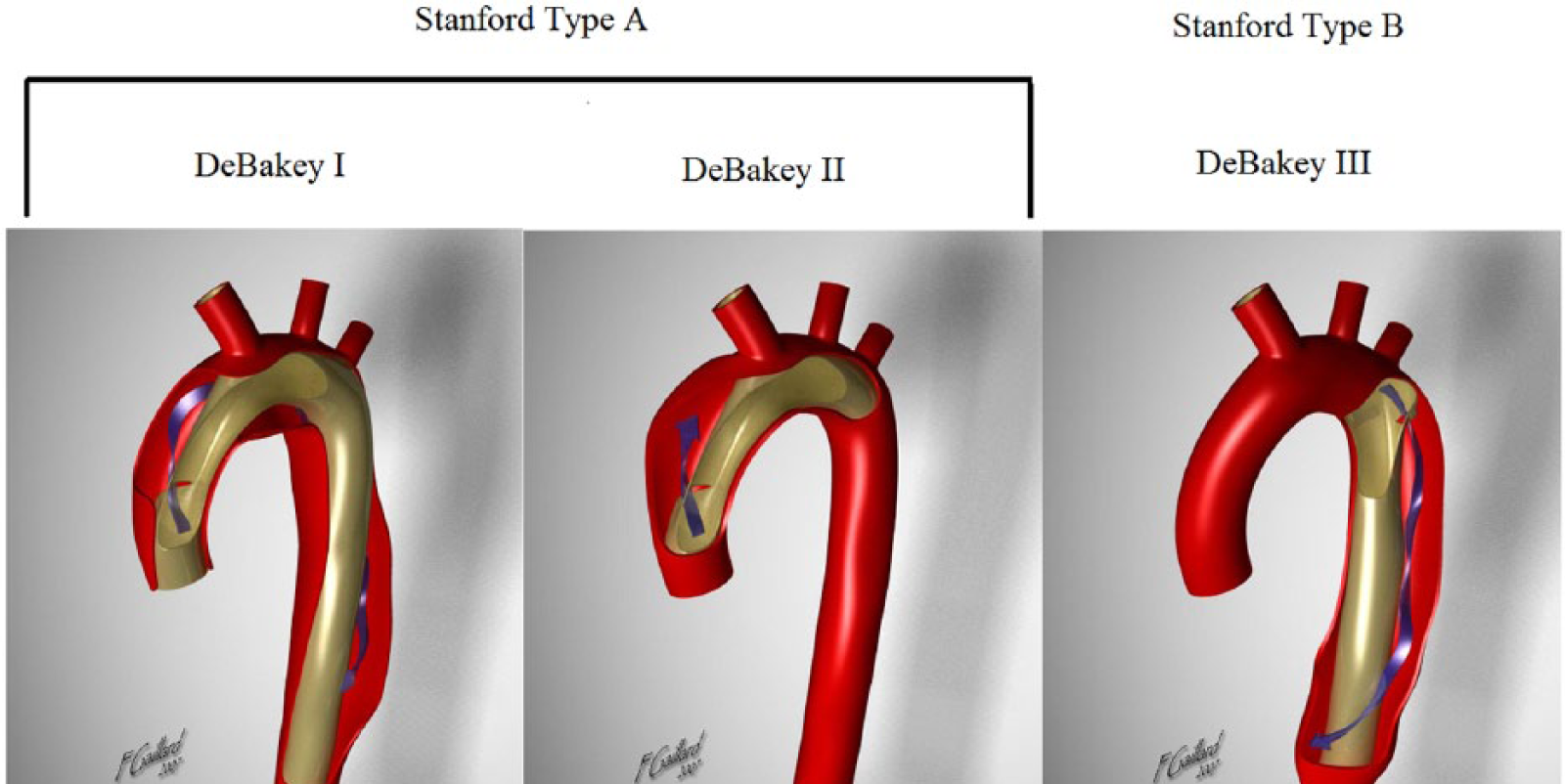
DeBakey’s and Stanford’s classification. Modified from http://radiopaedia.org/cases/aortic-dissection. Case courtesy of Associate Professor Frank Gaillard, Radiopaedia.org, rID: 7640.
Diagnostic instrumental methods
Chest radiography (Figure 2)
The role of chest radiography in patients with suspected AAS is limited, especially in patients with involvement of the ascending aorta. 26 A normal aorta profile does not exclude the presence of an ascending aorta aneurysm. Chest radiography required for other indications can detect anomalies in the profile or aortic size, and may prompt additional imaging. In addition, as suspicion of an AAS arises, chest radiography may identify other causes that can lead to the symptoms reported by the patient.
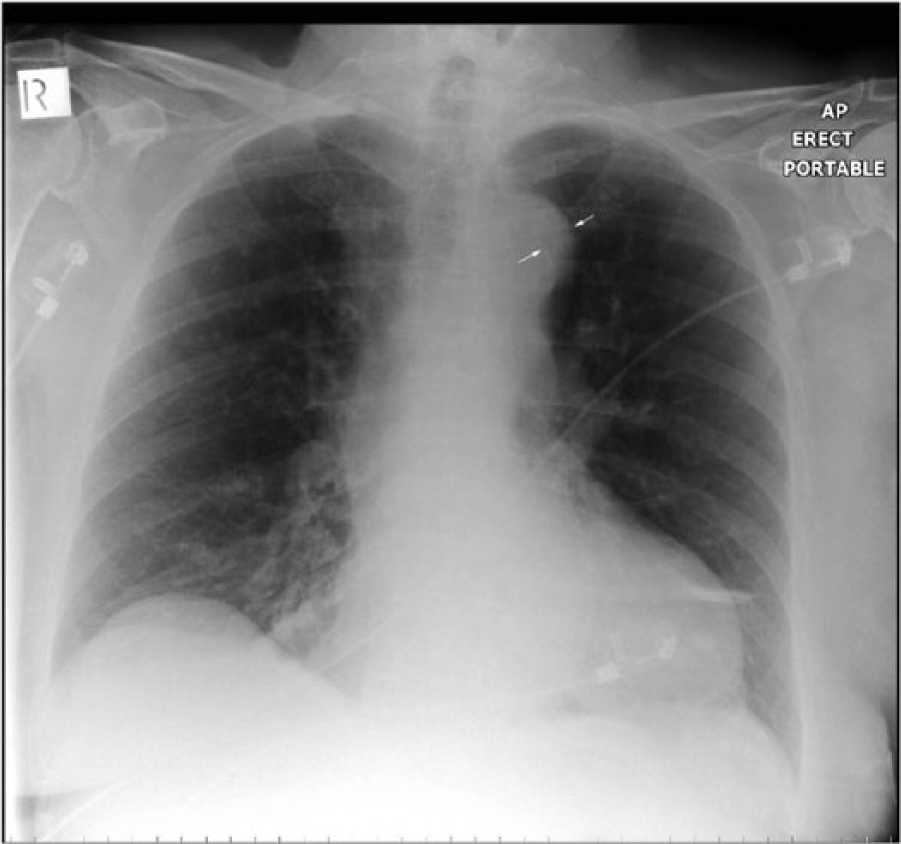
The calcium sign is a finding on a chest X-ray that suggests aortic dissection. It is the separation of the intimal calcification from the outer aortic soft tissue border by 10 mm. This is not an important sign for radiographers given that it is rarely seen. The arrowed distance represents the aneurysmal wall thickness. Figure courtesy of Dr MJ Fuller. Source: http://www.wikiradiography.net/.
Computed tomography (CT) (Figures 3–5)
Computed tomography (CT) angiography is among the most important imaging modalities for the diagnosis of aortic diseases, which is attributed to its widespread availability, rapid image acquisition, reproducibility and high spatial and temporal resolution. CT is chosen for the diagnosis of AAS as it is able to distinguish the lesions of AD, IMH and PAU. This technique also carries a sensitivity and a specificity of nearly 100% for the diagnosis of aortic diseases.27,28 CT also has an important role in the follow-up and monitoring of patients during the postoperative phase. Digital subtraction angiography may be used for congenital aortic disease.29,30 The most widely used technique is the MD-CT (multi-detector computed tomography) because it reduces image acquisition time with better spatial and temporal resolution compared to single-detector CT. Image acquisition generally follows this order: first, sequences without contrast, an arterial phase following contrast infusion, and, in some cases, delayed sequence images. CT image acquisition for aortic diseases should start at least 3 cm above the aortic arch and extend caudally to the femoral head to ensure that all aortic segments are evaluated. 31 MD-CT provides images of the entire aorta during the arterial transit of contrast bolus; 16-, 64- and even 256-slice CT scanners have replaced invasive techniques such as angiography to study medium and large vessels of the chest and abdomen. 10
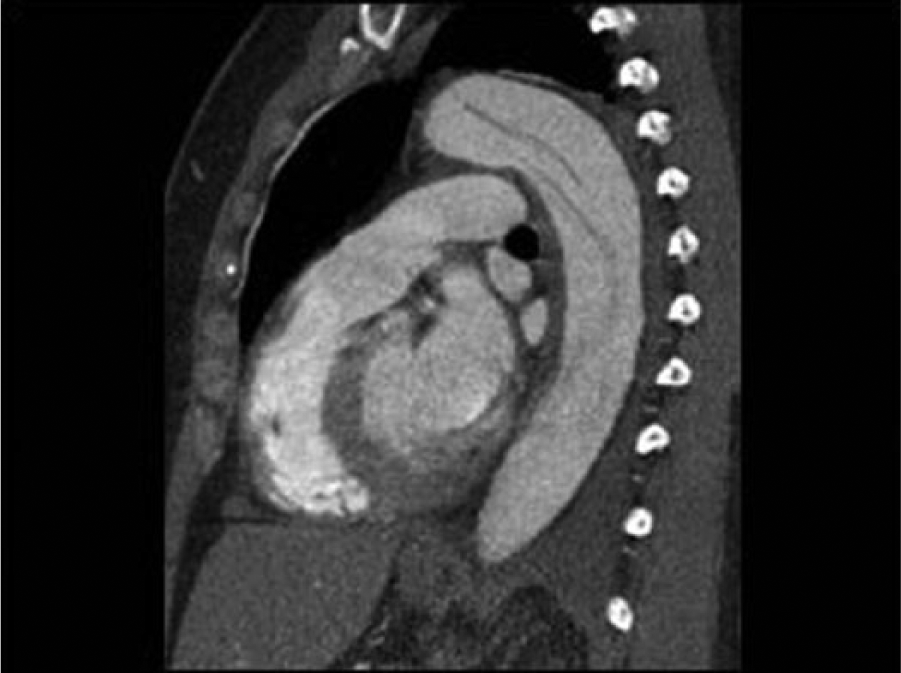
Example of an aortic dissection on a computed tomography (CT) scan. Image courtesy of Dr TSA Geertsma, Ziekenhuis Gelderse Vallei, Ede, The Netherlands. Source: http://www.ultrasoundcases.info/.
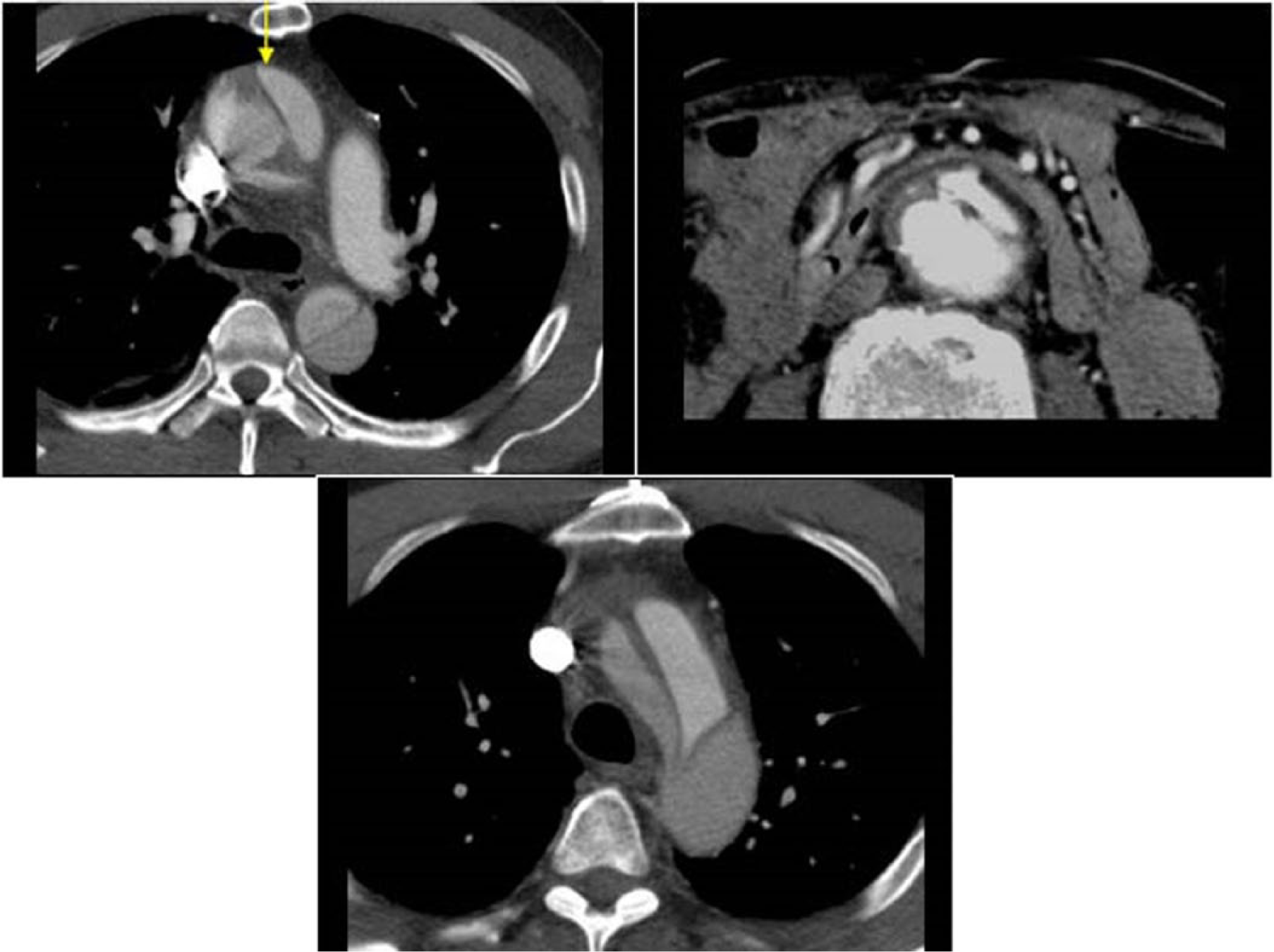
Example of an aortic dissection that originates in the ascending aorta (arrow) and continues in the aortic arch and descending aorta. CT images show dissection of the aorta with flow on both sides of the dissected intima. Images courtesy of Dr TSA Geertsma, Ziekenhuis Gelderse Vallei, Ede, The Netherlands. Source: http://www.ultrasoundcases.info/.
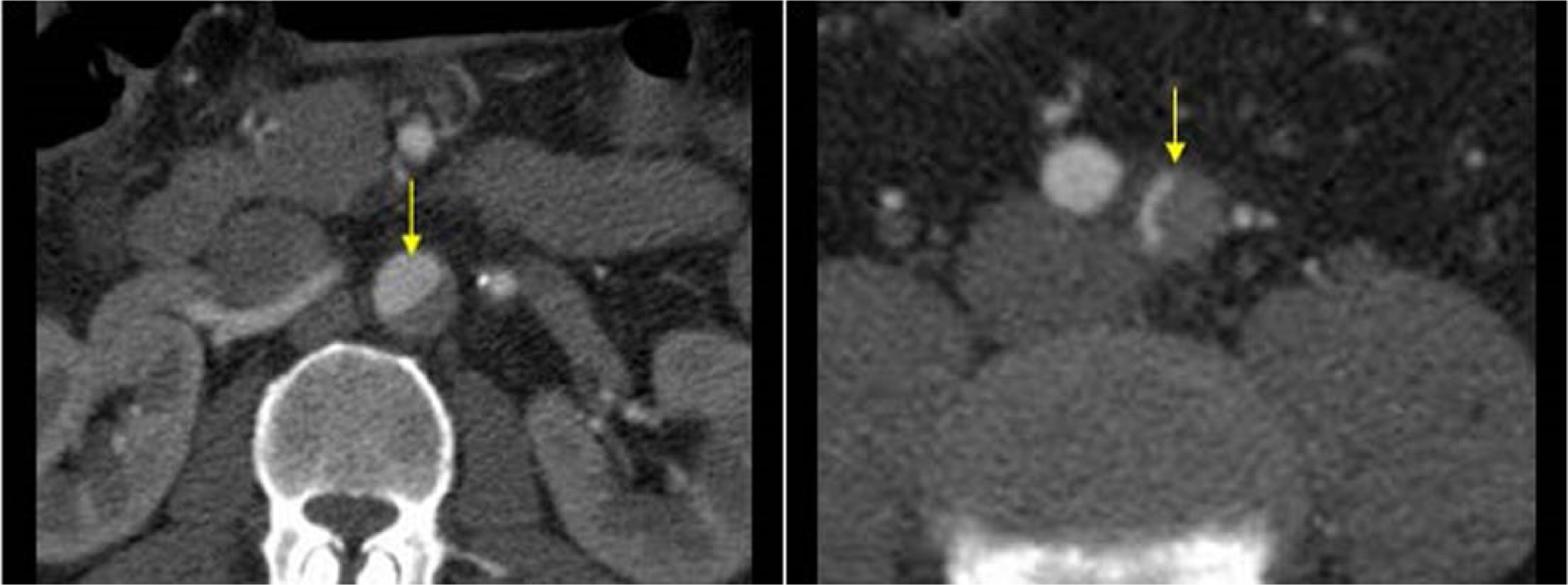
CT scan images of aortic dissection (arrows) extending in the left iliac artery. The arrows indicate the aortic dissection extending in the left iliac artery at CT scan images. Images courtesy of Dr TSA Geertsma, Ziekenhuis Gelderse Vallei, Ede, The Netherlands. Source: http://www.ultrasoundcases.info/.
A new technique involves the acquisition of electrocardiographic (ECG)-gated CT images, which allow more accurate measurements of aortic annulus, sinuses of Valsalva and proximal ascending aorta, eliminating artefacts related to the cardiac motion. ECG-gated images can be acquired in tele-systole or tele-diastole. 32
Even in a tortuous and oblique aorta, it is possible to measure the anteroposterior diameter of different anatomical points in the short axis by measuring the diameter from the external edge to the contralateral external edge of the wall, unlike echocardiography. 2 The main advantages of modern CT in emergency radiology are the speed in getting the images and rapid image reconstruction and three-dimensional (3D) processing. The 3D reconstruction technique is very useful in patients undergoing endovascular procedures. This is easier to achieve with the arterial rather than the venous phase. 33 The limitations of CT are the exposure to ionizing radiation and the use of iodinated contrast medium intravenously. The total exposure to ionizing radiation can be reduced by various methods, including: prospective ECG triggering, selection of a lower tube potential, automatic tube current modulation, and limiting the z-axis coverage. 34 The risks related to nephrotoxicity from iodinated contrast media have been reduced by the use of hypo- or iso-osmolar contrast agents, proper patient hydration and minimizing the volume of the contrast medium.
In the assessment and follow-up of an aortic aneurysm using CT images, it is important to consider the maximum diameter of the aorta, the longitudinal extent of the aneurysm, the possible involvement of the aortic valve or other arterial branches, and the presence of a peri-aortic haematoma. It is also important to look for signs of contrast extravasation and AD. 35 In patients undergoing endovascular treatment, CT plays an important role because it enables physicians to get detailed information regarding the aortic segments proximal to the origin of the aneurysm, the features of the aneurysm itself (tortuosity and angulation), and the vascular access (iliac-femoral axis) for planning endovascular stenting. In fact, some features of an aneurysm, such as a diameter of the aorta proximal to the aneurysm sac > 32 mm, an angle > 60° or calcifications > 90% of the aortic circumference, are considered unfavourable anatomical variables for an endovascular procedure. 36 In asymptomatic patients with an aortic aneurysm, the need to intervene is based on the characteristics obtained by imaging, including maximum diameter and the rate and extent of growth. It is recommended to obtain CT imaging annually when following these patients. 37 CT imaging can highlight some distinctive aspects of an aneurysm with increased risk of rupture, such as the reduction of the thrombus/lumen ratio and the presence of discontinuous areas of wall calcification. Evidence of rupture is represented by peri-aortic haematoma (which is highlighted in CT scans without contrast medium) or by visualizing the contrast in the mediastinum, pleural cavity or retro peritoneum.38,39
Magnetic resonance imaging (MRI) (Figure 6)
Both the sensitivity and the specificity of MRI are in the range of 95–100%. 40 MRI can accurately detect AD, delineate its extent, demonstrate the site of the entry tear, identify the arch vessels that are involved, and assess the renal artery involvement. Three-dimensional steady-state free precession MRI of the aorta provides aortic diameter measurements at the aortic annulus, root, and sino-tubular junction, which correlate well with measurements obtained with transthoracic echocardiography (TTE). 40 Flow dynamics can be evaluated by using four-dimensional (4D) phase contrast MRI (a technique that is, however, rarely available for clinical use) to measure the false lumen stroke volume, flow velocity, dominant entry tear position, and presence of helical flow. Therefore, MRI is a sensitive screening tool for patients with a limited sonographic window, particularly in young patients with aortic dilatation. Limitation of this imaging technique includes accessibility, difficulty monitoring unstable patients during imaging, and longer acquisition times compared to CT. However, MRI does not require ionizing radiation or iodinated contrast and is therefore highly suitable for serial follow-up studies in stable patients with known aortic disease. 41 MRI can also be used in the surveillance of medically managed type B AD. It usually begins with spin-echo black blood sequences to outline aortic shape and diameter and to identify and define the number and location of intimal flaps in the presence of AD. Gradient echo sequences are then obtained in stable patients, demonstrating changes in aortic diameters during the cardiac cycle and blood flow turbulences, distal to bicuspid valves, or in aortic regurgitation. Contrast-enhanced MRI with intravenous gadolinium can be performed rapidly to enhance aortic definition including arch vessels as a 3D angiogram without the need for ECG-gating. Gadolinium-enhanced sequences can be performed to differentiate slow flow from thrombus in the false lumen. 42 False lumen thrombosis is more accurately delineated using delayed-phase MRI with a blood pool agent compared with conventional first-pass CT or MRI. 43 MRI is also very useful for detecting the presence of pericardial effusion, aortic regurgitation, carotid artery dissection, and proximal coronary arteries, and their involvement in the dissecting process. 44
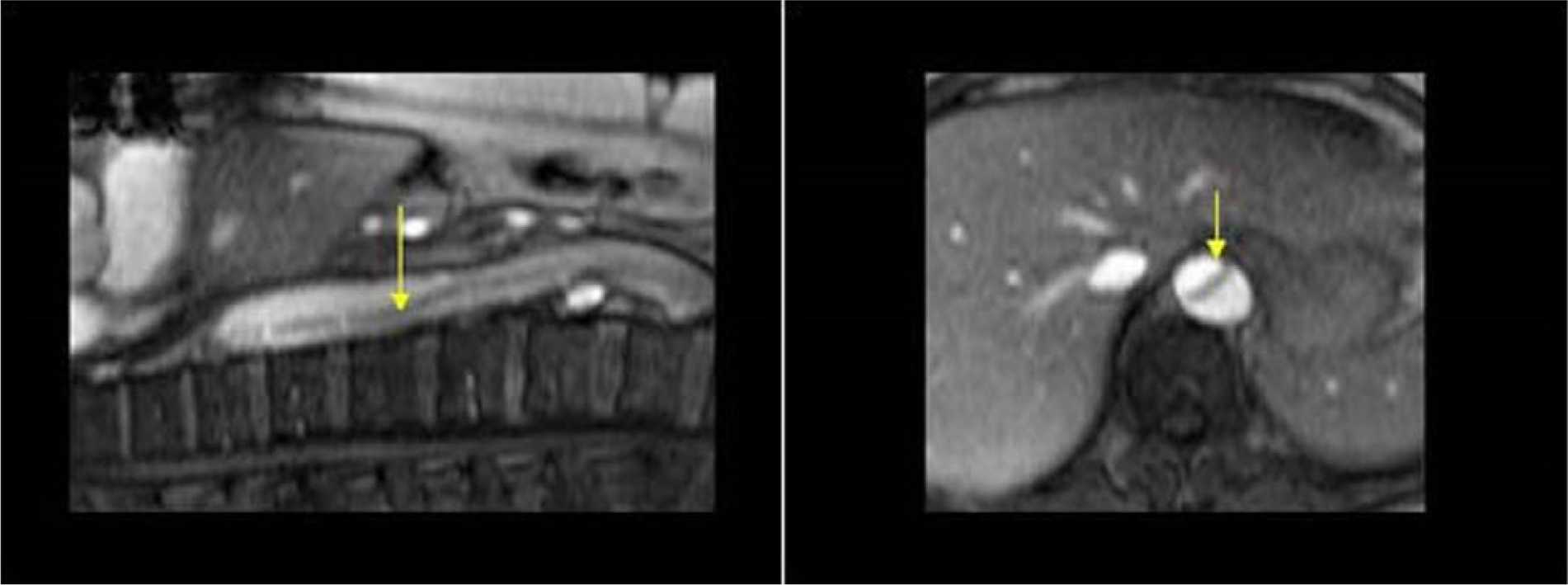
Longitudinal (left) and transverse (right) view of the dissected intima (arrows) of an aortic dissection with magnetic resonance imaging. The arrows indicate the dissected intima of an aortic dissection in longitudinal and transverse view at magnetic resonance imaging. Images courtesy of Dr TSA Geertsma, Ziekenhuis Gelderse Vallei, Ede, The Netherlands. Source: http://www.ultrasoundcases.info/.
Transthoracic echocardiography (TTE) (Figure 7)
Transthoracic echocardiography (TTE) is the standard routine in echocardiographic examination, but TTE is not the technique of choice for a full assessment of the aorta. TTE is useful for diagnosis and follow-up of some aortic segments, and is the most frequently used technique for measuring proximal aortic segments in clinical practice 1 ; however, transoesophageal echocardiography (TEE, or TOE) would be the preferred echo technique in the assessment of patients with acute AD, particularly if haemodynamically unstable. Detailed knowledge and quantification of the aortic root and aortic valve morphology is becoming crucial with the increasing use of transcatheter aortic valve implantation (TAVI) and transcatheter aortic valve replacement (TAVR) procedures. This knowledge is critically important for pre-procedural planning, intra-procedural guidance, and post-procedural assessment. 45
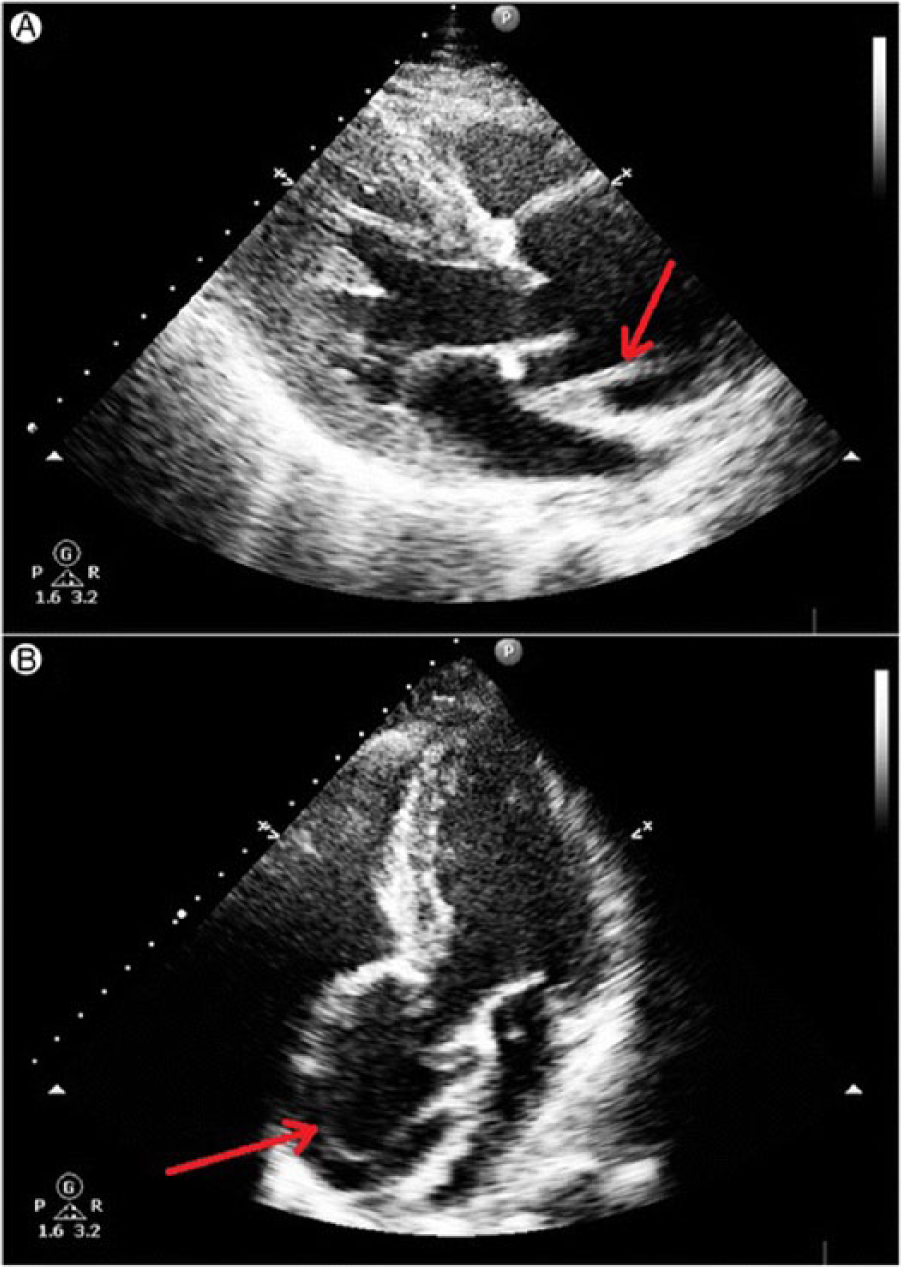
Transthoracic echocardiography showing a linear echo of an intimal tear in a dilated aortic root above aortic valve level. (A) Transthoracic echocardiography – parasternal long axis view: linear echo of an intimal tear is seen just above the aortic valve in systole. (B) Transthoracic echocardiography – apical five-chamber view: linear echo of an intimal flap is visible in the dilated aortic root above aortic valve level. Images courtesy of Dr Dorota Sobczyk. Source: Sobczyk D, Nycz K. Feasibility and accuracy of bedside transthoracic echocardiography in diagnosis of acute proximal aortic dissection. Cardiovasc Ultrasound 2015; 13: 15.
TTE is an excellent imaging modality for serial measurement of maximal aortic root diameters, evaluation of aortic regurgitation, and timing for elective surgery. 46 This technique can quickly provide clues for the diagnosis of AAS, including information about the aortic root, aortic regurgitation, pericardial effusion, and regional wall motion abnormalities, as well as left and right ventricular function. 47 According to the American Society of Echocardiography and the European Association of Cardiovascular Imaging, the aortic root measurements (maximal diameter of the sinuses of Valsalva, sino-tubular junction, and proximal ascending aorta) should be made at end-diastole in a strictly perpendicular plane to that of the long axis of the aorta. 45 Measurements of maximal diameter of the aortic root at the sinuses of Valsalva should be compared with age and body surface area (see Table 1). Three-dimensional TEE and MD-CT are emerging as reliable and preferred methods for aortic annulus measurements, particularly in patients undergoing TAVI or TAVR procedures. 45 In the suprasternal view, the aortic arch, the origin of the head and neck vessels, as well as the proximal descending aorta can be visualized. Even the abdominal aorta can be easily visualized to the left of the inferior vena cava in the sagittal subcostal view. 48 The diagnosis of acute AD by standard transthoracic M-mode and two-dimensional (2D) echocardiography is based on detecting intimal flaps in the aorta. The sensitivity and specificity of TEE range from 77% to 80% and 93% to 96%, respectively, for the ascending aorta. 1 On transthoracic M-mode echocardiography, floating intimal flaps, the enlargement of the aortic root and arch, and an increase in the aortic wall thickness were initially considered signs of AD. With the introduction of 2D echocardiography and the feasibility of taking suprasternal, subcostal, and substernal views, it has become possible to directly visualize the ascending aorta and aortic arch for floating intimal membranes, intimal tears, and false lumens. 49 The tear is defined as a disruption of flap continuity, with fluttering of the ruptured intimal borders. Smaller intimal tears can be detected with colour Doppler by visualizing jets across the flap, which also identify the spiral flow pattern within the descending aorta. TTE is restricted in patients with abnormal chest wall configuration, narrow intercostal spaces, obesity, pulmonary emphysema, and those on mechanical ventilation. 50 Furthermore, colour Doppler is of great interest in case of abdominal aorta dissection to detect perfusion of both false and true lumen and potential re-entry sites or obstruction of tributaries (e.g. the iliac arteries). 51
Aortic root dimensions in normal adults.
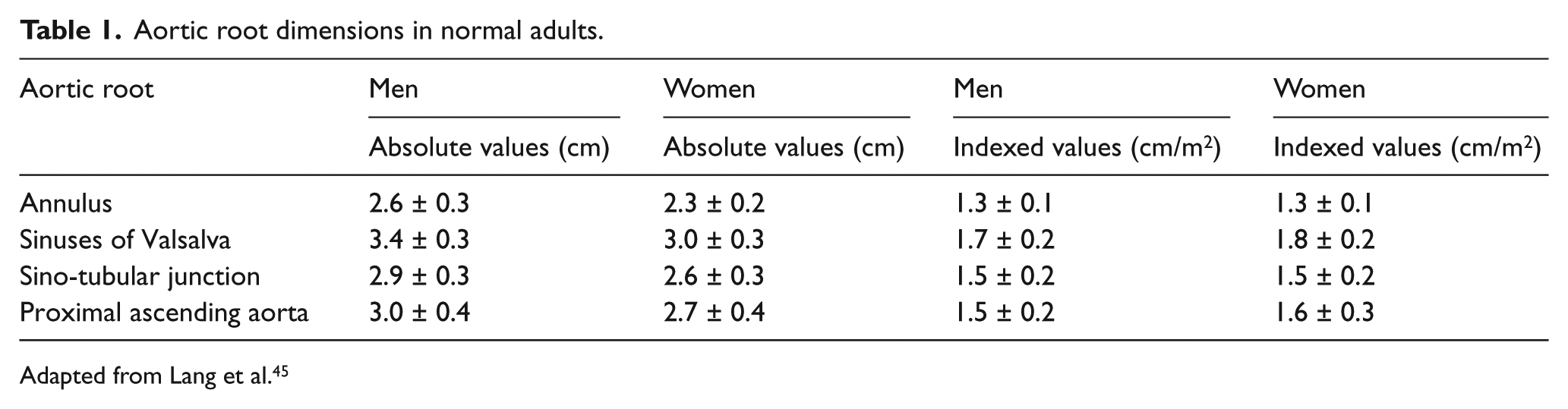
Adapted from Lang et al. 45
Transesophageal echocardiography (TEE) (Figure 8)
The role of TEE (or TOE) in aortic diseases has changed in recent years. Although TEE is a technique of choice for diagnosing thoracic aortic diseases, transthoracic echo combined with CT is more often the primary strategy in the emergency setting and follow-up of aortic diseases. TEE is a fast and readily available imaging modality with a very high diagnostic accuracy for the detection of both ascending and descending aortic diseases, and does play a crucial role in the pre-operative, intra-operative, and postoperative management of surgically treated aortic diseases. In addition to providing excellent visualization of the thoracic aorta, TEE enables detailed assessment of aortic valve function, left ventricular function, as well as the presence of pericardial or pleural effusion. 52 The thoracic aorta can be better imaged using TEE compared with TTE, whereby most aortic segments are in the near field of the TEE transducer. The aortic root and ascending aorta are best seen in the mid-oesophageal aortic valve long-axis view (three-chamber view at about 120–140°). The short-axis view of the ascending aorta is best obtained using mid-oesophageal views at about 45°. For measurements of the descending aorta, short-axis views at about 0° and long-axis views at about 90° should be obtained from the level of the diaphragm up to the aortic arch. 45 Owing to interposition of the right bronchus and trachea, a short segment of the distal ascending aorta, just before the innominate artery, remains an invisible ‘blind spot’. 1 Evangelista et al. demonstrated that using contrast-enhanced echocardiography can solve this problem. 53 In this study, contrast-enhanced TTE was especially useful for identifying AD located in the upper part of the ascending aorta, known as the ‘blind spot’ for TEE. Owing to its wide availability, safety (in experienced hands), and prompt image acquisition, which can be performed easily at the bedside, TEE has been considered a first-line imaging modality in the diagnosis of aortic dissection, mainly in patients with haemodynamic compromise. 46 This may be helpful to clarify artefacts by intraluminal reverberations, detect entry tears not identified without contrast by showing early arrival of contrast into the false lumen, identify retrograde or anterograde flow in the false lumen, and distinguish the true and false lumen in cases where systolic expansion of the true lumen is not clear. Furthermore, TEE can identify the presence of thrombus in a false lumen; the involvement of coronary and arch vessels; the presence, extent, and haemodynamic significance of pericardial effusion; and the presence and severity of aortic valve regurgitation. 52 Intra-operative TEE is highly useful during endovascular treatment in complicated descending AD. It allows for confirmation of correct guidewire insertion by identifying the true lumen in AD, and provides additional helpful information to guide correct stent-graft positioning in descending AD (there are no FDA-approved stent grafts for ascending AD in the US). TEE may also promptly identify suboptimal results and the presence of leaks and/or small re-entry tears with much higher sensitivity than angiography. This is also important for the prognosis of patients with residual patent false lumen in the descending aorta and the presence of a large proximal entry tear (>10 mm) defined by TEE, which indicates a high risk of mortality and the need for surgical or endovascular treatment during the follow-up. 54 False-positive results could occur because of reverberation echoes, fat-shift artefacts from the mediastinum, motion artefacts originating from the aneurysmal ascending aorta, calcified atheromatous plaque, and, in postoperative cases, peri-aortic haematoma. 55 TEE can also be useful in the initial diagnosis of AAS. Both TEE and MRI can detect intramural haemorrhage and haematoma accurately. The diagnostic features of IMH on TEE include the following: localized thickening of the aortic wall; intramural echo-free spaces; the absence of the dissection membrane, communication or Doppler flow signal; and the central displacement of intimal calcification. 56 Although the utility of TEE for the diagnosis of PAU has been well documented, penetrating ulcers within the echocardiographic blind spot of the distal ascending aorta and the proximal part of the aortic arch could be missed. 57
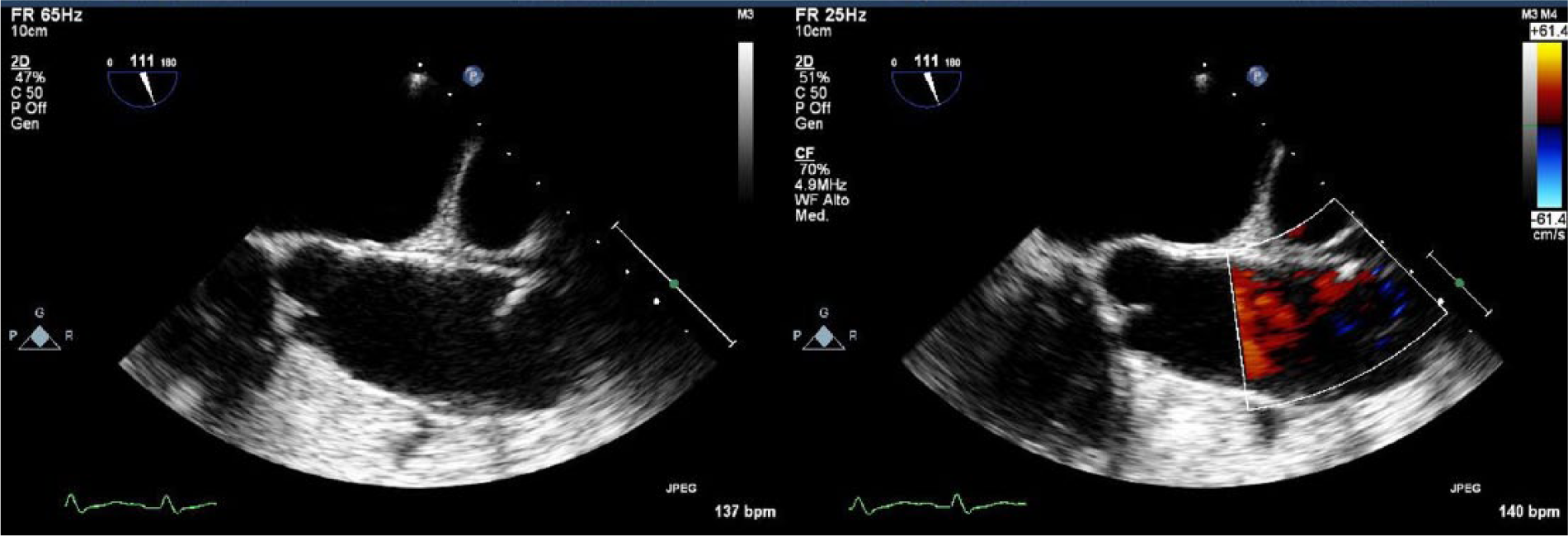
Transoesophageal echocardiography of the thoracic ascending aorta presenting a penetrating aortic ulcer that extends into the tunica media, just above the sino-tubular junction.
Intravascular ultrasound (IVUS)
Although the use of intravascular ultrasound (IVUS) is extremely operator- and experience-dependent, this technique is advocated to complement angiographic information in AD because of its ability to overcome most limitations and pitfalls of angiography. This technique enables visualization of the vessel wall from inside the aortic wall, allowing a dynamic visualization of the true and false lumen, detection of false lumen thrombosis with higher sensitivity and specificity than those of TEE, and a better definition of branch vessel involvement than that provided by TEE or CT. 58 Moreover, accurate visualization of aortic haematomas as crescent-shaped or circumferential thickening of the aortic wall is easy. 47 Although TTE and TEE are important bedside tools in dissection, both fail to provide sufficient anatomic detail to plan endovascular interventions. IVUS may emerge as a useful adjunct to endovascular procedures, but it is of little value for primary diagnosis.
Duplex ultrasound (Figures 9–10)
Duplex ultrasound is an imaging technique that combines Doppler flow and 2D imaging information, to demonstrate the direction, extent and velocity of blood flow through various vessels. In an emergency department, duplex ultrasound is a useful technique to quickly evaluate and screen for abdominal aneurysm rupture and promptly identify patients with acute aortic catastrophe in the abdomen. 59 Colour Doppler characterizes the flow within an aneurysm. With cross-section images, the true lumen of the aneurysm resembles an aspect of ‘Korean flag’ or ‘Yin-Yang’ (Figure 9). Within the aneurysmal sac, a characteristic ‘swirling’ flow pattern simultaneously demonstrates blood flow toward and away from the Doppler probe. 60 In case of rupture, it may be possible to directly demonstrate transparietal flow signals corresponding to the breach. 60
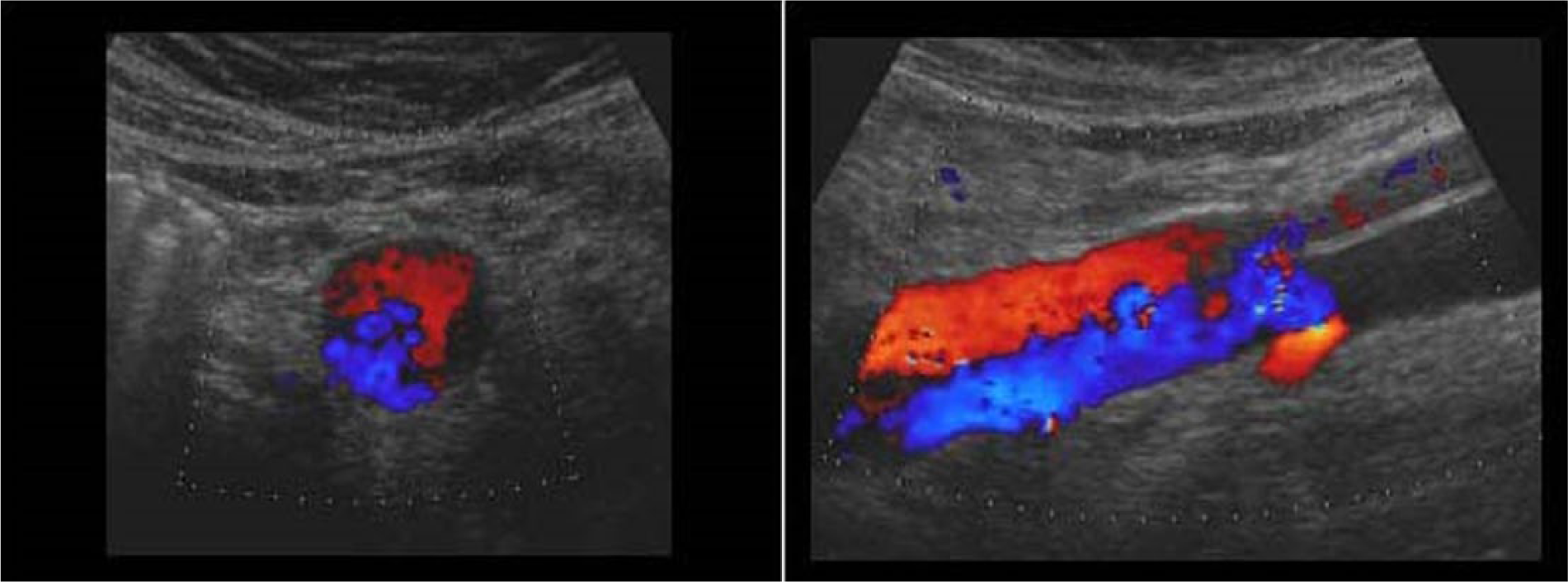
Left: transverse section of an aortic dissection with antegrade flow in the true lumen and retrograde flow in the false lumen (‘Korean flag’ or ‘Yin-Yang’ sign); right: the same image in longitudinal section. Images courtesy of Dr TSA Geertsma, Ziekenhuis Gelderse Vallei, Ede, The Netherlands. Source: http://www.ultrasoundcases.info/.
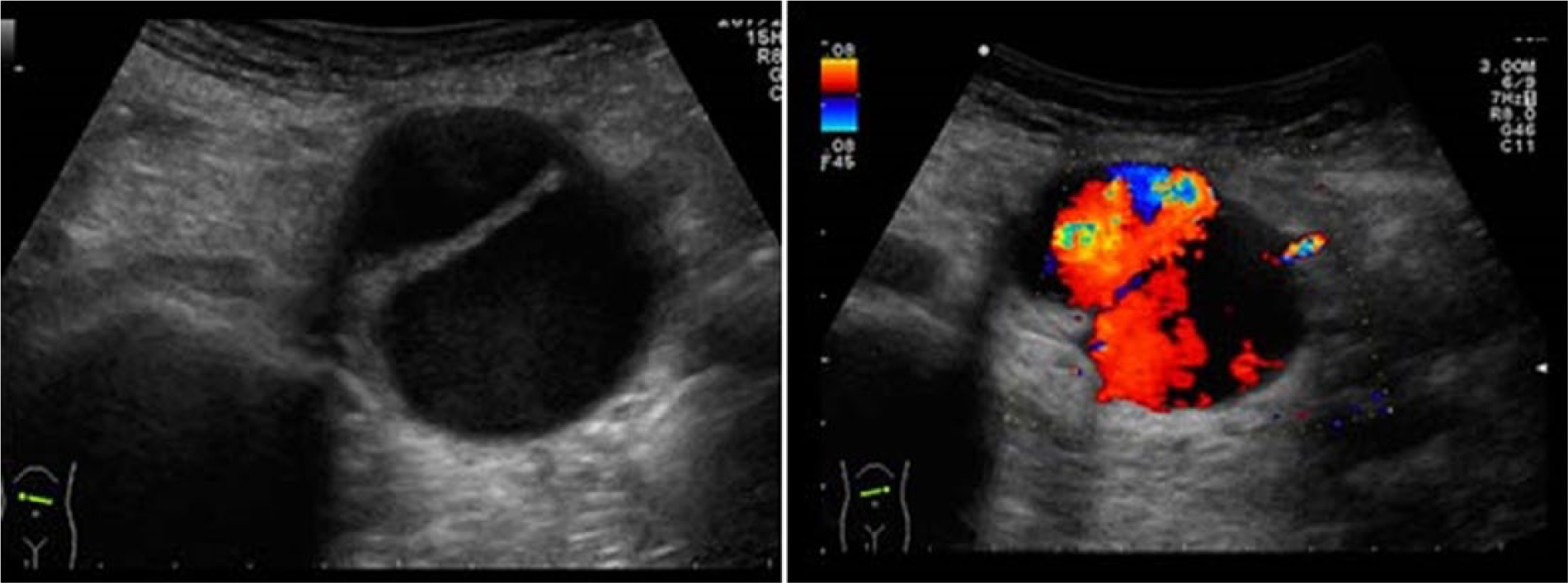
Left: transverse section of 4 cm wide aorta with dissected intima; right: flow on both sides of the dissected intima. Images courtesy of Dr TSA Geertsma, Ziekenhuis Gelderse Vallei, Ede, The Netherlands. Source: http://www.ultrasoundcases.info/.
Summary
In the context of AAS: CT-angiography, TEE, MRI-angiography, and IVUS are all available methods for the diagnosis of AD. They all have a sensitivity and specificity > 95%. 61 A meta-analysis of Shiga et al. 61 has shown that the diagnostic accuracy of aortography is comparable to that of other methods. CT-angiography is therefore preferred for the features already described. In emergency cases, CT-angiography is the imaging method of choice due to rapid image acquisition of the entire aorta and widespread availability. CT imaging can quickly interrogate the entire aorta and provide information about the collateral branches. 47 CT allows a wide spatial resolution to distinguish IMH, PAU, and AD, but requires patient transport in a radiology centre and stable haemodynamic status of the patient. The diagnosis of AD is confirmed when two lumens are evident and separated by an intimal flap inside the aorta. The true lumen may appear smaller and compressed by the false lumen, and may involve systolic expansion and diastolic collapse during the cardiac cycle. In some cases, the false lumen may be completely thrombosed. 62 In AD, CT images without contrast may reveal increased attenuation of aortic wall in axial projections, displacement of intimal calcification, an aortic aneurysm, and a line of high density in the lumen representing the intimal flap. Contrast-enhanced CT imaging of patients with AD may include the following features: a double barrel lumen, an entry tear, and expansion of the thoracic aorta. The size of the intimal tear is indicative of high-risk patients who would benefit from prompt surgery. 63 CT images without contrast can be useful for diagnosis of IMH with the demonstration of a hyper-dense mass within the aortic wall, clearly separated from the lumen that appears hypo-dense. 33 Distinguishing IMH from AD with thrombosis of the false lumen can be challenging. 63 Chin and Fleishmann have shown that IMH is often associated with a peri-aortic haematoma, while AD is associated with an increase in the mean aortic diameter at the level of its root and the sino-tubular junction, frequently extending to the abdominal aorta. 64 IMH can also present an appearance similar to an ulcer, but is easily distinguishable from PAU because it is not associated with atherosclerotic plaques. 65 Some aspects detected on CT scans indicate a worse prognosis in case of IMH, such as aneurysmal dilatation, focal contrast enhancement and an intramural thickness > 16 mm. 66 In addition, in the context of AAS, CT is a valuable aid in the diagnosis of PAU with identification of an ‘out-pouching ulcer’ of the aortic wall. PAU is associated with calcifications of the intima and a localized intramural haemorrhage in patients with severe atherosclerotic disease. 67 An increased pleural effusion, a maximum ulcer diameter of 22 mm and a depth of 14 mm are negative prognostic indicators of rapid disease progression, and are an indication to early and more aggressive intervention. 68
Conclusions
A prompt diagnosis of AAS continues to be a crucial point in the context of vascular emergencies, since the timing for proper treatment depends on it. In fact, a slight diagnostic delay can lead to the loss of the patient, since AAS are potentially life-threatening. Clinical symptoms are often unreliable and can be confounding. Therefore, the diagnostic imaging techniques presented in this review, each with its advantages and its disadvantages (Table 2), are of great help for physicians. The appropriate choice of technique is driven by the clinical context according to the emergency or urgency. In emergency cases, CT-angiography is the method of choice among the described techniques, as it is faster and readily available. In addition, it provides very high values of sensitivity and specificity. Research in this field continues to advance, especially for the prevention and timely treatment of aortic diseases; however, further studies are needed to determine the precise role of diagnostic imaging techniques in the context of AAS.
Advantages and disadvantages of imaging techniques in acute aortic syndromes.
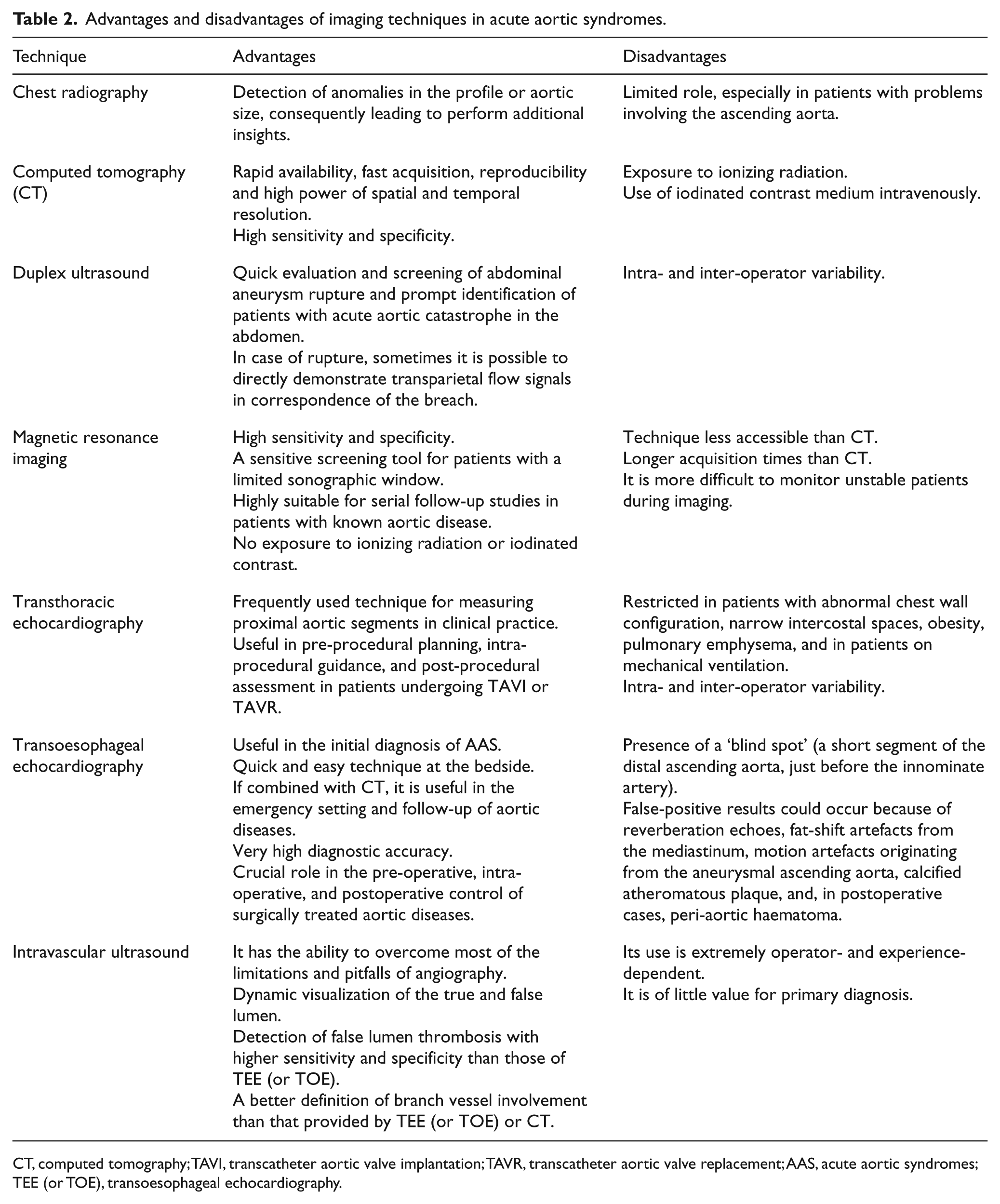
CT, computed tomography; TAVI, transcatheter aortic valve implantation; TAVR, transcatheter aortic valve replacement; AAS, acute aortic syndromes; TEE (or TOE), transoesophageal echocardiography.
Footnotes
Acknowledgements
The authors thank Dr Giuliana Benedetto for her great contribution in developing the English version of this manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
