Abstract
Antiplatelet agents are recommended for the reduction of major adverse cardiovascular events among all patients with symptomatic peripheral artery disease. However, the optimal antiplatelet regimen and duration of therapy in peripheral artery disease (PAD) remains unclear, largely due to limited and conflicting data in this patient population. This article reviews current data on antithrombotic therapy in PAD and discusses the implications of this data for current practice and future research.
Keywords
Introduction
Peripheral artery disease (PAD) comprises atherosclerosis of the major vessels supplying the lower extremities. While the majority of patients with PAD are asymptomatic, others develop lifestyle-limiting claudication or limb ischemia. An estimated 8 to 12 million Americans have PAD, with a worldwide prevalence exceeding 200 million. 1 Patients with PAD are at increased risk of cardiovascular mortality and major adverse cardiovascular events (MACE) regardless of whether they are symptomatic and experience a diminished quality of life due to limitations in physical functioning.2,3 Despite its prevalence and association with adverse outcomes, PAD is underdiagnosed and undertreated.4,5
Two of the primary objectives in treating patients with PAD are to attenuate the progression of lower limb atherosclerosis and prevent future cardiovascular events in other vascular beds, including myocardial infarction (MI) and stroke. This is best achieved through an approach that integrates aggressive risk factor modification (e.g. smoking cessation) with optimization of secondary prevention medications including the initiation of statins and ACE inhibitors. Antiplatelet therapy has also been shown to reduce the occurrence of MACE in patients with PAD, particularly atherothrombotic events occurring in the coronary and cerebral vasculature.6–8
ACCF/AHA guidelines currently recommend aspirin as first-line therapy in the prevention of atherothrombotic events in patients with PAD. 9 However, the optimal antiplatelet regimen and duration of therapy in PAD remains unclear, largely due to limited and conflicting data in this patient population. Furthermore, the application of novel oral anticoagulant agents to patients with PAD is an area of active clinical investigation. This article reviews current data on antithrombotic therapy in PAD and discusses the implications of this data for current practice and future research.
Platelets in atherothrombosis and PAD
Atherosclerosis is a chronic inflammatory process characterized by the accumulation of lipids, immune cells and connective tissue elements in the arterial wall.10,11 Platelet adhesion to sites of arterial injury, mediated by interactions between sub-endothelial proteins and receptors on the platelet surface, contributes to the initiation and progression of chronic atherosclerotic lesions and is central to the acute ischemic complications of atherosclerosis, including MI and acute limb ischemia (ALI).12–14
Activated platelets adhere to each other and bind to leukocytes through a variety of processes mediated by adhesion molecules expressed on the platelet surface, as depicted in Figure 1. Levels of platelet cell adhesion molecules (e.g. P-selectin) and bound fibrinogen are increased in patients with PAD when compared to healthy controls.15,16 Studies have also shown a linear relationship between the severity of PAD and degree of platelet activation when assessed by aggregation and flow cytometry.15,17 Patients with subcritical limb ischemia demonstrate higher levels of adenosine diphosphate (ADP)-stimulated P-selectin expression and increased fibrinogen binding when compared to patients with intermittent claudication. 15 Higher levels of both spontaneous and agonist-induced platelet aggregation have been demonstrated in patients with lower limb PAD taking aspirin when compared to otherwise healthy subjects taking aspirin. 18
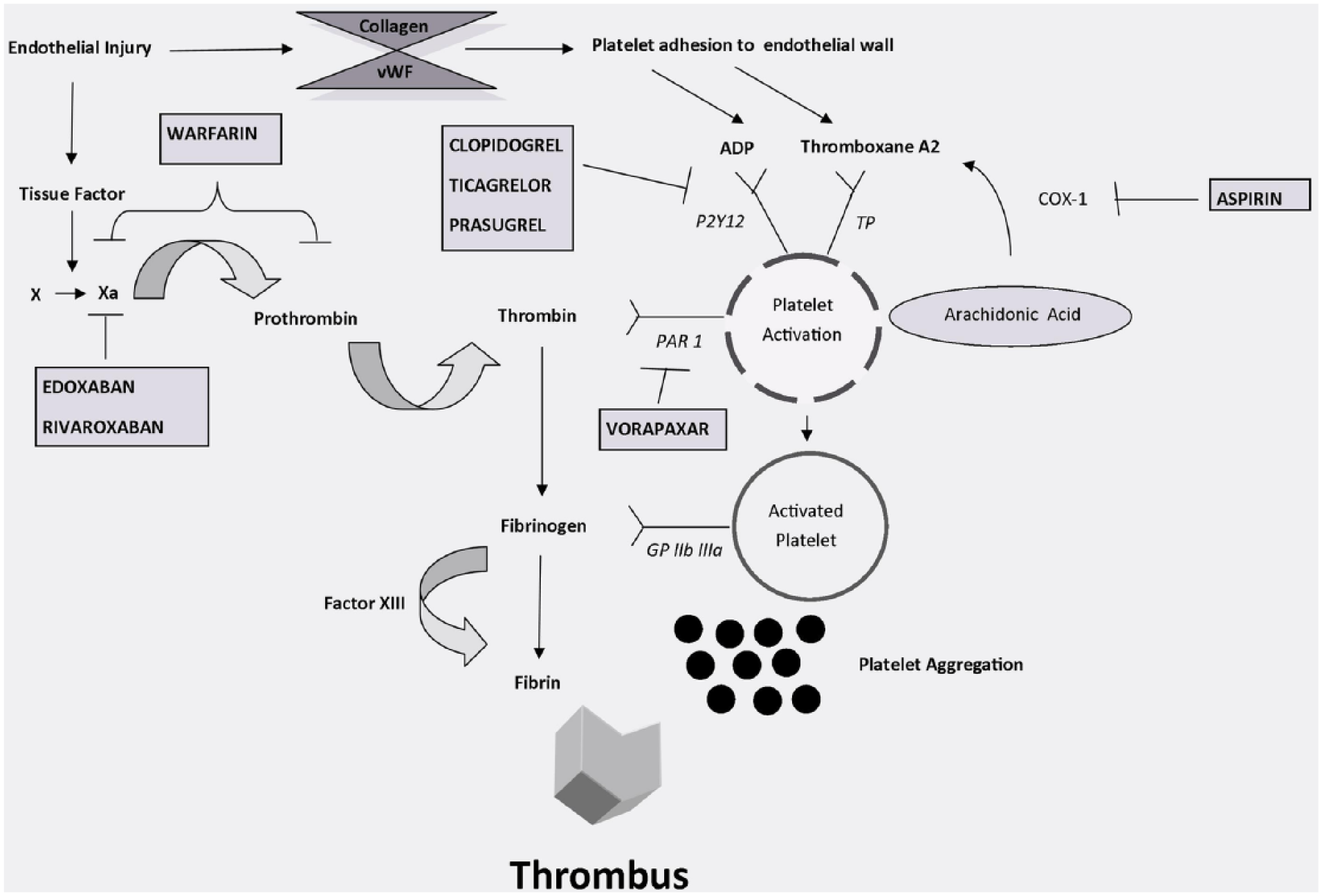
Mechanism of action of antithrombotic drugs for the treatment of peripheral artery disease. Currently available antithrombotic drugs for the treatment of peripheral artery disease target platelet activation and/or the coagulation cascade. Antiplatelet targets include the P2Y12 receptor (clopidogrel, ticagrelor, prasugrel), cyclooxygenase-1 (aspirin), and protease activated receptor 1 (vorapaxar). Anticoagulant targets include inhibition of vitamin K epoxide reductase, with a concomitant inhibition of Factors II, VII, IX, and X (warfarin), or more selective inhibition of Factor Xa (edoxaban, rivaroxaban). (vWF, von Willebrand factor; ADP, adenosine diphosphate; PAR 1, protease activated receptor 1; GP, glycoprotein; TP, thromboxane prostanoid receptor; COX, cyclooxygenase.)
These data support the concept that circulating platelets in patients with advanced PAD are characterized by increased reactivity, potentially lowering the threshold for thrombus formation in response to plaque rupture or erosion. More intensive or targeted antiplatelet therapy may therefore have particular benefit among patients with advanced PAD.
Antiplatelet therapy for the reduction of cardiovascular events
Patients with PAD have an increased risk of stroke and MI, and are six times more likely to experience a fatal cardiovascular event within 10 years compared to patients without PAD. 19 The use of antiplatelet therapy reduces secondary cardiovascular events in patients with cerebrovascular and cardiovascular disease, and is currently recommended in all patients with established PAD; however, the optimal antiplatelet strategy among patients with PAD is unclear. Numerous studies have explored the use of antiplatelet agents in both symptomatic and asymptomatic PAD. The findings of key trials and meta-analyses for each of the major antiplatelet agents are discussed below.
Antiplatelet monotherapy
Aspirin
Despite the advent of newer agents, aspirin remains the most widely used antiplatelet drug in the world. Aspirin reduces platelet aggregation by irreversibly inactivating cyclooxygenase-1 (COX-1), blocking the formation of thromboxane A2 from arachidonic acid. While the efficacy of aspirin in the primary prevention of cardiovascular events remains controversial, there is a robust body of evidence to support the use of aspirin for secondary prevention in patients with a prior MI or ischemic stroke.20–22
Current guidelines support the use of aspirin for the prevention of cardiovascular events in all patients with PAD, regardless of symptoms or prior ischemic events. 9 The rationale for this recommendation draws on the results of a meta-analysis published by the Antithrombotic Trialists Collaboration (ATC). 6 That meta-analysis included 9214 patients with PAD from 42 randomized trials and found a relative risk reduction of 23% in serious vascular events among patients prescribed antiplatelet therapy. However, nearly 60% of the data incorporated into that meta-analysis was derived from studies of antiplatelet agents other than aspirin, such as picotamide. An earlier meta-analysis performed by the same group included 4939 patients with PAD from 33 trials and found that aspirin use was not associated with a reduction in cardiovascular events in the absence of concomitant atherosclerotic disease in the coronary or cerebral vasculature. 23
Aspirin for asymptomatic PAD
Two major randomized trials have examined the role of aspirin in preventing initial cardiovascular events in patients with asymptomatic PAD. The Aspirin for Asymptomatic Atherosclerosis (AAA) trial compared once-daily aspirin 100 mg to placebo in 3350 asymptomatic patients with PAD, diagnosed by screening for an ankle–brachial index (ABI) < 0.95. 24 The primary endpoint was a composite of an initial fatal or non-fatal coronary event, stroke, or revascularization. After a mean follow up of 8.2 years, there was no significant difference in rates of the primary endpoint between the groups randomized to aspirin versus placebo (13.7 vs 13.3 events per 1000 person years, respectively; hazard ratio (HR) 1.03, 95% CI 0.84–1.27). No benefit was observed in the secondary endpoints of initial vascular events, a composite of a primary endpoint event, transient ischemic attack, angina, or new claudication (22.8 vs 22.9 events per 1000 person years for the aspirin vs placebo groups, respectively; HR 1.0, 95% CI 0.85–1.17) or all-cause mortality (12.8 vs 13.5 for the aspirin vs placebo groups, respectively; HR 0.95, 95% CI 0.77–1.16) between the two groups. Post hoc analyses of predefined subgroups with ABI cut points of 0.80, 0.85, 0.90 and 0.95 showed higher event rates with lower ABIs (24.3, 18.6, 15.7 and 13.7 events per 1000 person years, respectively) but no significant differences were identified between those receiving aspirin versus placebo. This lack of benefit observed with aspirin use was accompanied by an increased risk of bleeding.
Similarly, the prospective, randomized POPADAD trial compared aspirin (100 mg daily) and antioxidants to placebo in asymptomatic patients with PAD and diabetes. 25 A total of 1276 patients with an ABI less than 0.99 were enrolled in the study with a median follow-up period of 6.7 years. At baseline, patients had a mean age of 60 years and an ABI of 0.90. Again, despite the high-risk profile of the cohort, aspirin use was not associated with a benefit in terms of cardiovascular events or cardiovascular mortality. In the group randomized to aspirin, 116 primary fatal and non-fatal cardiovascular events occurred compared with 117 in the control group (HR 0.98, 95% CI 0.76–1.26). Bleeding rates did not differ significantly between groups.
Both the AAA and POPADAD trials were limited in their power to detect a small effect of aspirin. Given that these were primary prevention populations, albeit higher risk given the presence of subclinical atherosclerosis, a lack of statistical power may have contributed to the failure of either trial to show a significant reduction in mortality. Furthermore, the low absolute event rate (60% of original prediction) initially observed in the AAA trial led the authors to extend follow up from 5 to 9.5 years, potentially compromising adherence and contributing to crossover between study groups. 26 Additionally, it has been suggested that these trials may have shown a benefit with aspirin had they used lower ABI cut-off values to identify patients with more severe disease and, perhaps, a higher risk of atherothrombotic complications. Subgroup analyses of both the AAA and POPADAD trials, however, failed to show a benefit with aspirin therapy even when patients were stratified by ABI. Based on these data, aspirin may not have a significant benefit in the reduction of adverse cardiovascular events among patients with asymptomatic PAD.
Aspirin for symptomatic PAD
Patients with symptomatic PAD represent a higher risk cohort than those with subclinical disease identified through screening. Multiple mechanisms likely account for this increased risk, including more advanced atherosclerosis, increased prevalence of comorbidities, and increased activation of inflammatory cells, including platelets.27–29 Symptomatic patients have also been shown to have a greater pathologic burden of atherosclerotic disease characterized by ectopic neovascularization and active inflammation, thereby potentially increasing the risk of plaque rupture and associated atherothrombotic events such as MI and stroke. 30
The Critical Leg Ischaemia Prevention Study (CLIPS) investigated the efficacy of aspirin 100 mg daily in both symptomatic and asymptomatic patients with PAD, as defined by an ABI < 0.85 or toe index < 0.6. 31 Patients with advanced PAD, as reflected by a Fontaine Stage of III or IV, were excluded from the trial. The primary outcome was a composite of fatal and non-fatal vascular events (MI, stroke or pulmonary embolism) and incidence of critical limb ischemia (CLI). A total of 366 patients were enrolled in the trial and followed for up to 2 years. Among the 185 patients randomized to aspirin, only seven (3.8%) experienced a major vascular event, compared to 20 of 181 (10.5%) patients randomized to placebo, with a relative risk reduction of 64% (p = 0.016; HR = 0.35, 95% CI 0.15–0.82). The risk of fatal or non-fatal MI was also significantly reduced in the aspirin group (p = 0.03; HR = 0.18, 95% CI 0.04–0.82). However, the CLIPS trial failed to enroll its target sample size of 2000 patients and was terminated early due to difficulty recruiting patients due to non-trial use of aspirin. The benefit of aspirin observed in the CLIPS trial, while questionable given the study’s limitations and lack of sample size, stands in contrast to the findings of the AAA and POPADAD trials.
In 2009, Berger et al. published a meta-analysis of 18 prospective, randomized trials (including CLIPS and POPADAD) evaluating the effect of aspirin on cardiovascular event rates in patients with PAD. 32 The included trials enrolled both symptomatic and asymptomatic patients. A 34% relative risk reduction for non-fatal stroke was identified in the patients taking aspirin; however, there were no statistically significant reductions in overall cardiovascular events or mortality.
The findings of these studies illustrate the absence of compelling data supporting the use of aspirin for prevention of cardiovascular events in patients with PAD who have not suffered a prior MI or ischemic stroke; however, these studies are not representative of all clinical presentations of PAD. Patients with more severe disease, such as CLI, were not included in the AAA, POPADAD or CLIPS trials and were not well represented in the trials included in the meta-analysis. Such patients may represent a higher risk cohort more likely to benefit from aspirin therapy or more intensive antiplatelet therapy. Conversely, it is possible that the platelet activation seen in PAD may be less responsive to aspirin in general, thereby requiring treatment with alternative agents targeting other mechanisms of platelet activation.
P2Y12 inhibitors
Adenosine diphosphate activates platelet adhesion and aggregation by binding P2Y1 and P2Y12 receptors. Three ADP inhibitors (ticlopidine, clopidogrel, and ticagrelor) have been studied in PAD. Ticlopidine has been shown to reduce fatal vascular events and overall mortality in patients with claudication, but is no longer used clinically due to its adverse hematologic risk profile and limited patient tolerability.33,34 Currently, clopidogrel is the only P2Y12 inhibitor that is FDA-approved for use in patients with PAD.
Clopidogrel
The Clopidogrel versus Aspirin in Patients at Risk of Ischaemic Events (CAPRIE) trial was a randomized controlled trial comparing aspirin (50 mg to 325 mg daily) to clopidogrel 75 mg daily in 19,185 patients with atherosclerotic cardiovascular disease including recent ischemic stroke, MI, or symptomatic PAD. 7 The primary endpoint was a composite of ischemic stroke, MI or vascular death. Patients included in the PAD cohort had intermittent claudication of presumed atherosclerotic origin and an ankle-brachial index of < 0.85, or a history of intermittent claudication with previous leg amputation, reconstructive surgery or angioplasty. Patients were followed for up to 3 years (mean: 1.9 years), during which the primary endpoint occurred at an annual rate of 5.32% in the clopidogrel group versus 5.83% in the cohort receiving aspirin, for a small yet statistically significant relative risk reduction of 8.7% (95% CI 0.3–16.5; p = 0.043). Importantly, the subgroup of patients with PAD derived the most benefit from clopidogrel therapy, with an annual event rate of 3.7% compared to 4.9% in the aspirin group (relative risk reduction 23.8%, 95% CI 8.9–36.2; p = 0.0028). The results of the CAPRIE trial generally support the use of clopidogrel as antiplatelet monotherapy in patients with PAD, but the absolute benefit of clopidogrel over aspirin monotherapy is small.
Ticagrelor
Ticagrelor is an oral, reversible ADP receptor P2Y12 inhibitor that is currently FDA-approved for use in secondary prevention of thrombotic events in patients with unstable angina, NSTEMI (non-ST-segment elevation MI) or STEMI (ST-segment elevation MI). Unlike clopidogrel, ticagrelor is an allosteric antagonist of ADP, and is reversible. The Ticagrelor Versus Clopidogrel in Patients with Acute Coronary Syndromes (PLATO) trial compared ticagrelor to clopidogrel in acute coronary syndrome and found a reduction in the rate of cardiovascular death, MI or stroke (9.8% vs 11.7%, p< 0.001) associated with ticagrelor, with no significant difference in rates of major bleeding. 35
The Examining Use of Ticagrelor in PAD (EUCLID) trial is currently ongoing and aims to evaluate the use of ticagrelor in PAD. 36 This study, which is the first to directly compare two P2Y12 inhibitors in PAD, has randomized 13,500 patients with symptomatic PAD to ticagrelor (90 mg BID) or clopidogrel (75 mg QD). The primary objective of the EUCLID trial is to compare the effect of long-term treatment with ticagrelor versus clopidogrel on rates of cardiovascular death, MI and ischemic stroke. Patients will be followed for 18 to 36 months, with an anticipated median duration of 27 months. Enrolled patients must be symptomatic (claudication, ischemic rest pain, ulcers, gangrene) with objective evidence of PAD (ABI < 0.80 or prior lower-extremity revascularization for symptomatic and hemodynamically significant PAD). All patients enrolled in the study also have baseline genotyping performed to exclude those with poor metabolism of clopidogrel. The primary results of this trial are anticipated in 2017.
Thromboxane inhibitors
Picotamide antagonizes the thromboxane A2 receptor and inhibits thromboxane A2 synthase, thereby reducing platelet aggregation. In contrast to aspirin, picotamide does not affect cyclooxygenase. Its use in PAD has been studied in two major trials.
The Effect of Picotamide on the Clinical Progression of Peripheral Vascular Disease study (ADEP) enrolled 2304 patients with PAD, as evidenced by claudication and an ABI < 0.85 or claudication and prior amputation or surgical revascularization. 37 Patients were randomized to either placebo or picotamide 300 mg three times daily and followed for 18 months. A variety of major (cardiovascular death, MI, stroke, amputation) and minor (unstable angina, transient ischemic attack, renal failure, peripheral vascular disease (PVD) deterioration) vascular events were analyzed during the follow-up period. Patients receiving picotamide had an 18.9% relative risk reduction in a combined endpoint of major and minor events, but this did not reach statistical significance (p = 0.056). A subgroup analysis of 438 patients with diabetes identified a 45.2% relative risk reduction in the combined endpoint of major and minor cardiovascular events that was statistically significant (p = 0.022).
This post hoc analysis led to a subsequent study aimed at investigating the efficacy of picotamide versus aspirin in diabetic patients with PAD. The Drug Evaluation in Atherosclerotic Vascular Disease in Diabetics (DAVID) study enrolled 1209 patients with diabetes and PAD defined as the presence of intermittent claudication, loss of posterior tibial pulse, an ABI < 0.9 or > 1.3, or previous amputation, surgical revascularization or angioplasty. 38 Patients randomized to picotamide had a relative risk reduction of 45% in overall mortality at 2 years (HR 0.55, 95% CI 0.31–0.98), translating to a number needed to treat of 40 patients over 2 years to prevent one death. This finding was tempered by the fact that neither major ischemic cardiovascular events nor vascular mortality were significantly reduced by picotamide when compared with aspirin. Patients receiving picotamide demonstrated a trend toward a reduction in non-fatal cardiovascular events that did not reach statistical significance. The trials investigating antiplatelet monotherapy in asymptomatic or symptomatic PAD are summarized in Table 1.
Antiplatelet monotherapy.
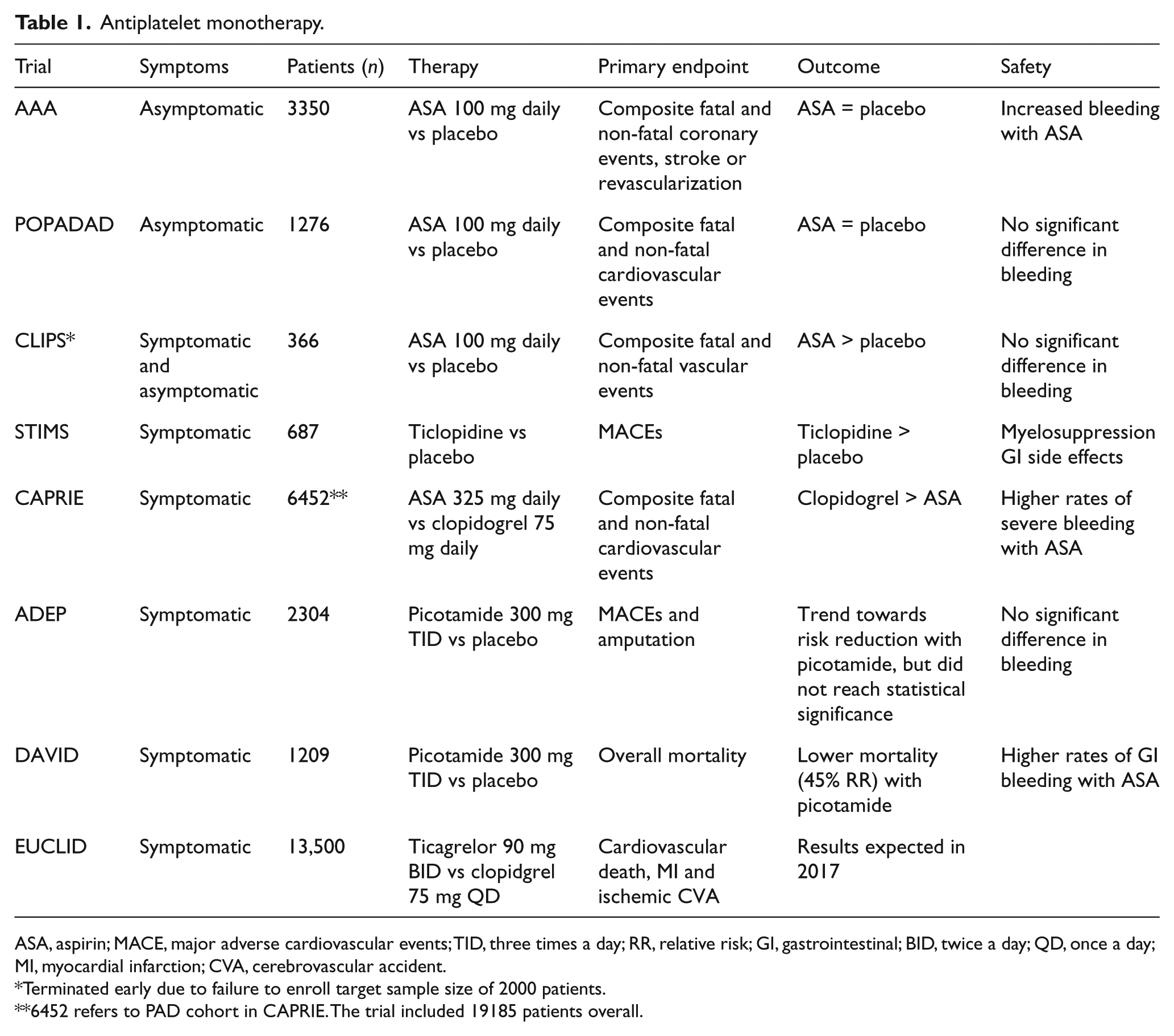
ASA, aspirin; MACE, major adverse cardiovascular events; TID, three times a day; RR, relative risk; GI, gastrointestinal; BID, twice a day; QD, once a day; MI, myocardial infarction; CVA, cerebrovascular accident.
Terminated early due to failure to enroll target sample size of 2000 patients.
6452 refers to PAD cohort in CAPRIE. The trial included 19185 patients overall.
In summary, the available data for antiplatelet monotherapy supports the use of aspirin or clopidogrel as a single agent in patients with PAD who have symptoms or who have experienced a previous MI or stroke. A subgroup analysis of the CAPRIE trial demonstrated a greater reduction in MACE in patients with PAD treated with clopidogrel compared to aspirin, but the overall benefit was small. In comparison, the data for aspirin use among patients with asymptomatic PAD may not support regular use of this agent.
Combination antiplatelet therapy
Clopidogrel and aspirin
The adhesion, activation and aggregation of platelets can be pharmacologically disrupted through a multitude of mechanisms. Targeting more than one pathway has the potential to amplify the net antiplatelet effect and mitigate the risk of future atherothrombotic events. Multiple trials have shown a benefit in both short and long-term outcomes of combination therapy with aspirin and clopidogrel in reducing recurrent atherothrombotic events in patients with prior MI,39–42 and current guidelines recommend dual antiplatelet therapy with aspirin and clopidogrel for 12 months following MI regardless of whether percutaneous coronary intervention was performed. The recently published Dual Antiplatelet (DAPT) study suggests that, in this patient population, the benefit of dual antiplatelet therapy may extend beyond 12 months due to a continued risk of MACE after PCI. 43 The PEGASUS study also suggested a possible additional benefit of DAPT with ticagrelor after prior MI (1–3 years prior). 44
The effect of dual antiplatelet therapy in patients with PAD was examined in the Clopidogrel and Aspirin versus Aspirin Alone for the Prevention of Atherothrombotic Events (CHARISMA) trial. 45 That study compared once-daily low-dose aspirin (75 to 162 mg) to clopidogrel (75 mg) plus aspirin in patients with documented cardiovascular disease (prior stroke, MI, PAD) or multiple risk factors. A total of 15,603 patients were enrolled with a median follow-up of 2.3 years. The primary endpoint was a composite of MI, stroke, or death from cardiovascular causes. In the overall cohort, there was no significant difference between treatment groups in the primary endpoint (6.8% in the DAPT group vs 7.3% in the aspirin monotherapy group; HR 0.93, 95% CI 0.83–1.05; p = 0.22). However, in a subgroup analysis of patients with symptomatic cardiovascular disease, there was a statistically significant reduction in the primary endpoint in patients receiving both clopidogrel and aspirin (6.9% in the DAPT group vs 7.9% in the aspirin monotherapy group; HR 0.88, 95% CI 0.77–0.998; p = 0.046). Importantly, the overall event rates in the CHARISMA trial were lower than those observed in CAPRIE, possibly owing to advances in medical therapy in the preceding decade.
Further analysis of the PAD cohort within CHARISMA identified 3096 patients that also failed to benefit from dual antiplatelet therapy compared to aspirin alone in reducing the composite endpoint of MI, stroke or death. 8 However, there was a significant reduction in the rate of MI among patients with PAD randomized to clopidogrel and aspirin versus those receiving aspirin alone (2.3% vs 3.7%, HR 0.63; p = 0.028).
More recently, an observational cohort analysis was conducted of 629 patients (mean age 67 years; 44% male) with claudication or CLI undergoing diagnostic or interventional lower-extremity angiography. In total, 348 patients were prescribed DAPT and 281 were prescribed aspirin monotherapy. 46 At baseline, patients in the DAPT group were more likely to have CAD (p = 0.007) or diabetes (p = 0.02) than those in the aspirin group. After 3 years of follow-up, during which antiplatelet prescriptions were verified on a monthly basis, DAPT use was associated with a reduction in MACE (HR 0.65, 95% CI 0.44–0.96) and overall mortality (HR 0.55, 95% CI 0.35–0.89). There were no differences in major amputation or major adverse limb events (MALE) between the two groups; bleeding rates could not be adequately assessed. The results of this study suggest that in patients with symptomatic PAD undergoing endovascular intervention, prolonged DAPT may be associated with a reduction in MACE and mortality.
Vorapaxar in combination with other antiplatelet agents
Vorapaxar, an antagonist of the primary thrombin receptor expressed on vascular endothelium and smooth muscle, was recently evaluated in 26,449 patients with stable vascular disease (MI, stroke or PAD) in the Trial to Assess the Effects of SCH 530348 in Preventing Heart Attack and Stroke in Patients with Atherosclerosis (TRA2P-TIMI 50) trial. 47 In this randomized, double-blind trial, patients were randomized to receive vorapaxar sulfate 2.5 mg daily or placebo. A total of 3787 patients were randomized into the PAD cohort, and median follow-up was 36 months. The majority of PAD patients (88%) were treated with concomitant aspirin, 37% were prescribed a thienopyridine at baseline, and 28% of the patients in the PAD cohort were prescribed dual antiplatelet therapy. The majority of patients (75%) had symptomatic PAD at enrollment (72% with claudication, 2% with rest pain, and 1% with ulceration, necrosis or gangrene).
The primary endpoint of the trial, a composite of cardiovascular death, MI or stroke, did not differ significantly among patients with PAD who were randomized to vorapaxar (11.3% vs 11.9%, HR 0.94; p = 0.53). However, rates of hospitalization for ALI (2.3% vs 3.9%, HR 0.58; p = 0.006) and peripheral artery revascularization (18.4% vs 22.2%, HR 0.84; p = 0.017) were significantly reduced in patients randomized to vorapaxar.
Importantly, the observed reduction in ischemic limb events was consistent regardless of baseline antiplatelet therapy with aspirin or thienopyridine. Furthermore, the risk of GUSTO moderate or severe bleeding with vorapaxar did not differ significantly between patients on aspirin monotherapy, clopidogrel monotherapy or dual antiplatelet therapy at baseline. However, patients receiving vorapaxar did have a higher risk of bleeding, including GUSTO moderate or severe bleeding (7.4% vs 4.5%, HR 1.62; p = 0.001).
The results of the TRA2P-TIMI 50 trial demonstrate an additive benefit of inhibition of the PAR-1 thrombin receptor, when superimposed on standard antiplatelet therapy, in reducing ischemic limb events in patients with PAD. Treatment with vorapaxar in this setting, however, is associated with a moderately increased risk of bleeding. Because patients with PAD were an overall small subgroup among all patients studied in the clinical trial, a dedicated follow-up trial may be necessary to further determine the benefit of vorapaxar in the reduction of MACE and adverse limb events among patients with PAD.
A recent post hoc subgroup analysis of the TRA2P-TIMI 50 explored rates of ALI in patients with symptomatic PAD taking vorapaxar. 48 There were 109 cases of ALI during the trial, the majority (54%) of which were due to surgical graft thrombosis. In patients with symptomatic PAD, the addition of vorapaxar to standard antiplatelet therapy (aspirin and/or clopidogrel) was associated with a 42% relative risk reduction in the incidence of ALI versus aspirin and/or clopidogrel alone (3-year event rate of 2.3% vs 3.9%, HR 0.58, 95% CI 0.39–0.86).
A second post hoc subgroup analysis of 5845 patients in the TRA2P-TIMI 50 trial with a history of PAD showed a reduction in rates of peripheral artery revascularization among patients taking vorapaxar in addition to background antiplatelet therapy with aspirin and/or clopidogrel. 49 A total of 934 peripheral revascularization procedures were performed over a median duration of 2.5 years; most interventions were performed for claudication (60%). Additional indications included CLI (22%) and ALI (13%). In patients taking vorapaxar, there was a significant reduction in the incidence of peripheral revascularization for claudication (HR 0.84, 95% CI 0.71–0.99; p = 0.04), and a trend towards fewer procedures for CLI and ALI. There was also a trend towards fewer amputations in those patients on vorapaxar, but this did not reach statistical significance. The trials investigating combination antiplatelet therapy in asymptomatic or symptomatic PAD are summarized in Table 2.
Combination antiplatelet therapy.
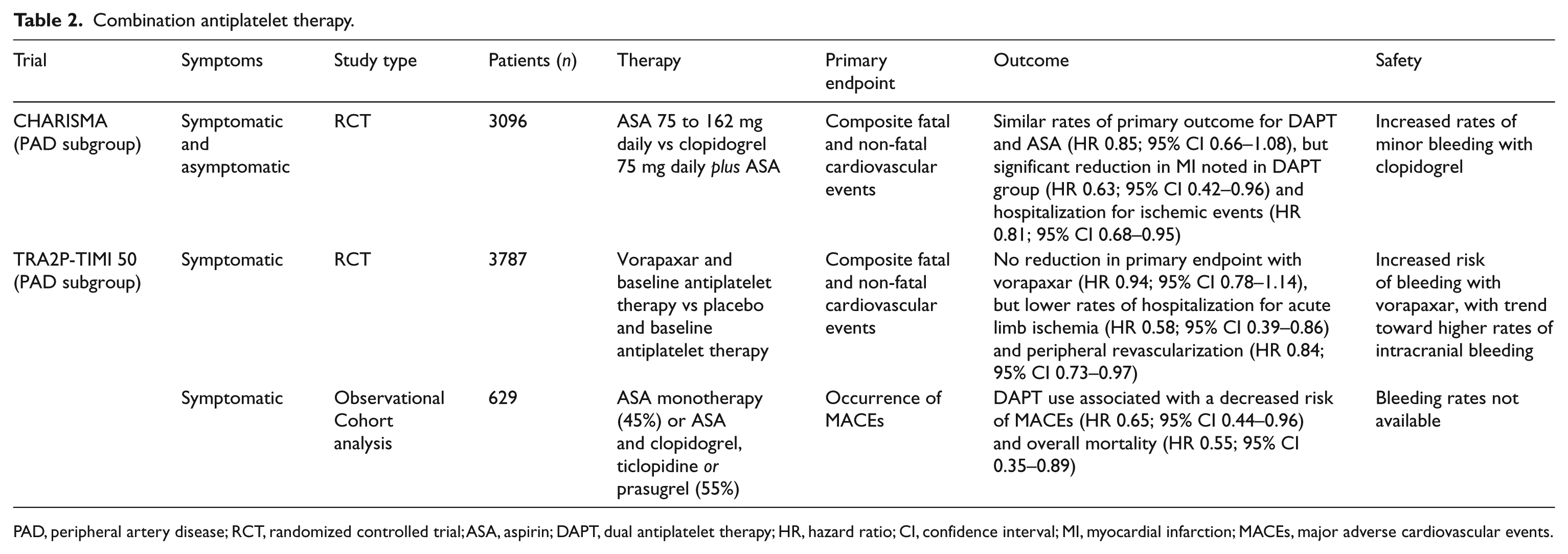
PAD, peripheral artery disease; RCT, randomized controlled trial; ASA, aspirin; DAPT, dual antiplatelet therapy; HR, hazard ratio; CI, confidence interval; MI, myocardial infarction; MACEs, major adverse cardiovascular events.
In summary, a subgroup analysis of patients with PAD enrolled in the CHARISMA trial demonstrated a reduction in MI in patients treated with DAPT, but no overall mortality benefit. Observational data suggests that DAPT may be associated with lower rates of MACE and mortality in patients with severe symptomatic PAD undergoing endovascular intervention. The use of vorapaxar in addition to aspirin and/or clopidogrel has been shown to reduce the risk of ALI and peripheral artery revascularization in patients with symptomatic PAD when compared to aspirin or clopidogrel alone. However, the use of vorapaxar in this setting is associated with an increased risk of moderate bleeding.
Antiplatelet therapy following surgical interventions for PAD
Approximately 10–15% of patients with PAD undergo lower-extremity revascularization with bypass grafting, most commonly for chronic CLI in an effort to avoid amputation. The annual rates of graft occlusion are approximately 15% for venous grafts and 20% for synthetic grafts; these rates rise to 45% and 75%, respectively, for bypass procedures below the knee.50–52
The Clopidogrel and Acetylsalicylic Acid in Bypass Surgery for Peripheral Arterial Disease (CASPAR) trial evaluated the effect of dual antiplatelet therapy with aspirin and clopidogrel versus aspirin alone in maintaining graft patency following below-knee bypass grafting. 50 The trial enrolled 851 patients who were randomized to clopidogrel (75 mg/day) plus aspirin (75 to 100 mg/day) or placebo plus aspirin (75 to 100 mg/day) for 6 to 24 months. The primary endpoint was a composite of graft occlusion or revascularization, above-ankle amputation of the affected limb, or death. No significant difference in the composite outcome was identified between patients receiving DAPT versus those receiving aspirin alone. However, a pre-specified subgroup analysis did demonstrate a statistically significant reduction in the primary endpoint among patients with prosthetic, rather than venous, grafts (HR 0.65, 95% CI 0.45–0.95; p = 0.025). Bleeding occurred more frequently in patients prescribed clopidogrel, but the rates of severe bleeding did not differ significantly between study groups.
In summary, for patients with PAD undergoing below-knee surgical revascularization with prosthetic grafts, DAPT is associated with improved graft patency and lower rates of amputation compared to treatment with aspirin alone, without an appreciable difference in rates of severe bleeding.
Antiplatelet therapy following endovascular interventions for PAD
Endovascular interventions such as angioplasty and stenting are frequently performed to treat claudication or limb ischemia in patients with PAD. Compared with surgical interventions, endovascular therapies are associated with lower morbidity and mortality in the short term, but potentially higher rates of restenosis in the long term. 53 These procedures have become increasingly effective over the past decade, but substantial rates of restenosis and subsequent re-intervention pose a disadvantage to this approach. Currently, there is large variability in duration of DAPT prescription after endovascular interventions, with most physicians prescribing 1–3 months of DAPT. However, these data are based primarily on extrapolation from coronary stenting data, rather than dedicated studies among patients with PAD.
The Management of Peripheral Arterial Interventions with Mono or Dual Antiplatelet Therapy (MIRROR) study was a randomized, double-blinded trial that enrolled 80 patients with PAD undergoing femoropopliteal interventions for treatment of either claudication (majority of patients) or CLI. 54 Participants initially underwent PTA followed by stenting with nitinol stents, if there was greater than 30% residual stenosis. Patients were then randomized to receive either aspirin (81 mg/day) or aspirin and clopidogrel (81 and 75 mg/day, respectively) for 6 months. At 6 months, there was a significant reduction in the need for target lesion revascularization in the DAPT group compared to the aspirin monotherapy group (5% vs 20%; p = 0.04). This translated into a significant reduction in the number of days spent in the hospital for patients receiving aspirin and clopidogrel. Interestingly, this reduction in the need for revascularization occurred despite there being no difference in the rate of binary restenosis (>50%) as detected by digital subtraction angiography or duplex ultrasound, suggesting that clopidogrel reduced symptoms even in patients with restenosis.
Of note, there were discrepancies in baseline characteristics between the two groups: patients in the DAPT group had significantly longer lesion lengths (p = 0.011), a factor known to adversely affect long-term patency following femoropopliteal interventions. Additionally, the clopidogrel group had more restenotic lesions at baseline (40% vs 12.5%; p = 0.010).
Given the clinical benefit observed at 6 months in the MIRROR study, a 12-month follow-up was conducted. All patients were taken off clopidogrel at 6 months and continued on aspirin monotherapy per the study protocol. By 12 months, the significant difference in target level revascularization (TLR) observed in the initial period was no longer apparent. This was equally true for patients who had received angioplasty alone and those who received stenting. At 12 months, similar TLR rates were observed in patients who received DAPT and aspirin monotherapy, suggesting a potential rebound effect in the DAPT group following cessation of clopidogrel.
Aspirin and clopidogrel resistance in patients undergoing endovascular intervention
The antiplatelet response to aspirin or clopidogrel varies among individual patients, and high on-treatment platelet reactivity (HTPR) to ADP is a risk factor for adverse clinical events. 55 Multiple studies have implicated resistance to clopidogrel as a potential contributing factor to thrombotic events following coronary interventions.56–58 The reported prevalence of clopidogrel resistance in patients with PAD ranges from 9.8% to 35.2%.54,59,60 The PRECLOP study included 100 patients undergoing infrainguinal angioplasty or stenting and receiving clopidogrel (75 mg/day) in the peri-procedural period. 61 Those patients who had residual platelet activity above the optimal cut-off value, identified by pre-procedure point of care testing, had a nearly 17-fold increase in adverse clinical events following endovascular intervention. Despite these promising results, no study has determined whether more intensive antiplatelet therapy among patients with baseline resistance to aspirin or clopidogrel is associated with improved clinical outcomes. The trials investigating antiplatelet therapy after surgical or endovascular revascularization for PAD are summarized in Table 3.
Antiplatelet therapy following endovascular or surgical revascularization.
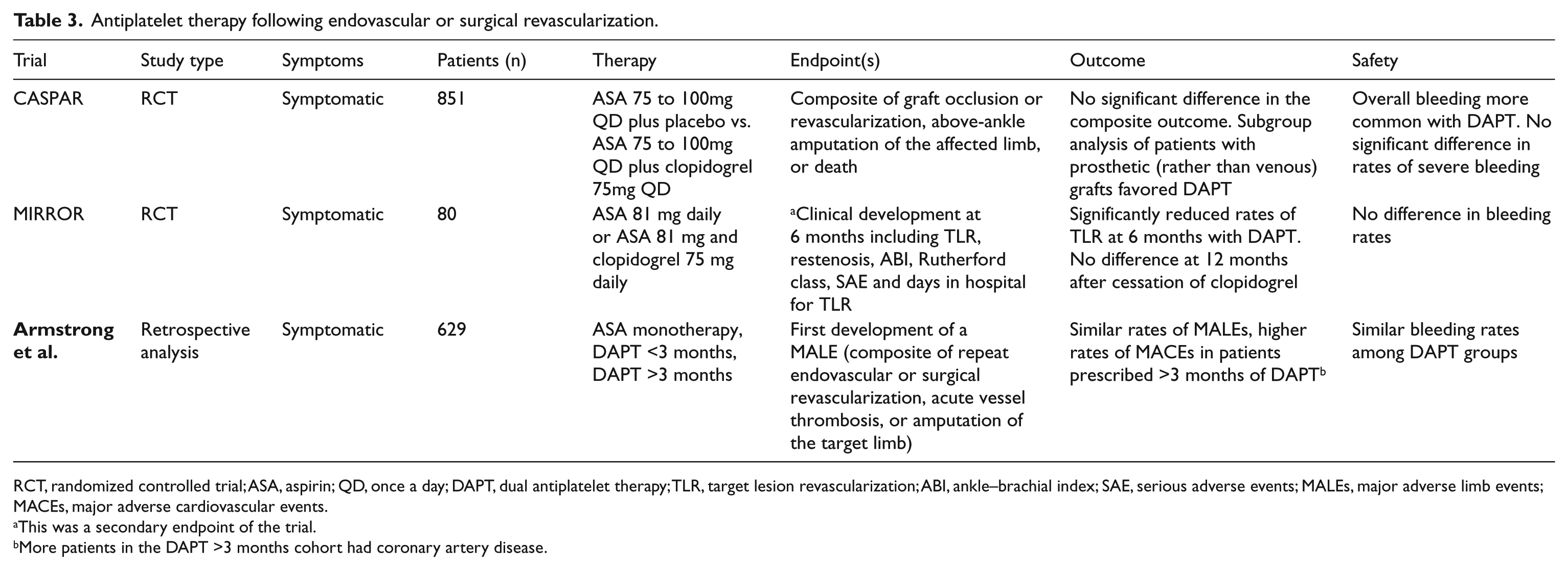
RCT, randomized controlled trial; ASA, aspirin; QD, once a day; DAPT, dual antiplatelet therapy; TLR, target lesion revascularization; ABI, ankle–brachial index; SAE, serious adverse events; MALEs, major adverse limb events; MACEs, major adverse cardiovascular events.
This was a secondary endpoint of the trial.
More patients in the DAPT >3 months cohort had coronary artery disease.
Anticoagulant therapy in PAD
In addition to having a higher proportion of activated platelets, patients with PAD have increased levels of circulating tissue factor, exaggerated thrombin production and diminished fibrinolytic potential when compared to healthy controls, consistent with an underlying prothrombotic state.62–64 Oral anticoagulants have long been incorporated into the treatment of patients with PAD despite conflicting data with regard to efficacy and safety.
The role of oral anticoagulant therapy in preventing adverse cardiovascular events in patients with PAD was evaluated in the Warfarin Antiplatelet Vascular Evaluation (WAVE) study. 65 This randomized controlled trial compared combination therapy with warfarin and an antiplatelet agent to an antiplatelet agent alone in patients with PAD. PAD was defined as symptomatic atherosclerosis of the arteries of the lower extremities, the carotid arteries or the subclavian arteries. Patients with lower extremity atherosclerosis were included if they had intermittent claudication, rest pain, non-healing gangrenous ulcers or previous amputation. A total of 2161 patients were enrolled with a mean duration of follow-up of 35 months. The primary endpoint was a composite of MI, stroke or death from cardiovascular causes. There was no significant difference between treatment groups in the occurrence of the primary endpoint (12.2% in the combination therapy group vs 13.3% in the antiplatelet monotherapy group; HR 0.92, 95% CI 0.73 to 1.16; p = 0.48). Furthermore, the secondary composite endpoint of MI, stroke, severe ischemia of the coronary or peripheral arteries or death from cardiovascular causes did not differ significantly between treatment groups (15.9% in the combination therapy group vs 17.4% in the antiplatelet monotherapy group; HR 0.91, 95% CI 0.74 to 1.12; p = 0.37). Despite the exclusion of patients with known risk factors for bleeding, rates of life-threatening bleeding (4.0% vs 1.2%; HR 3.41, 95% CI 1.84 to 6.36; p < 0.001) and moderate bleeding (2.9% vs 1.0%; HR 2.82, 95% CI 1.43 to 5.58; p = 0.002) were increased in the combination therapy group.
The findings of the WAVE study suggest that combination therapy with warfarin and an antiplatelet agent yields no significant benefit in patients with PAD when compared to antiplatelet therapy alone, and is associated with an increased risk of moderate and severe bleeding. The lack of benefit seen with warfarin therapy may be partially explained by the architecture of arterial thrombi. Unlike venous clots, which are composed primarily of erythrocytes and fibrin, arterial thrombi contain a higher concentration of platelets and few red blood cells.66–68 The trials investigating anticoagulation for patients with PAD are summarized in Table 4.
Anticoagulants in PAD.
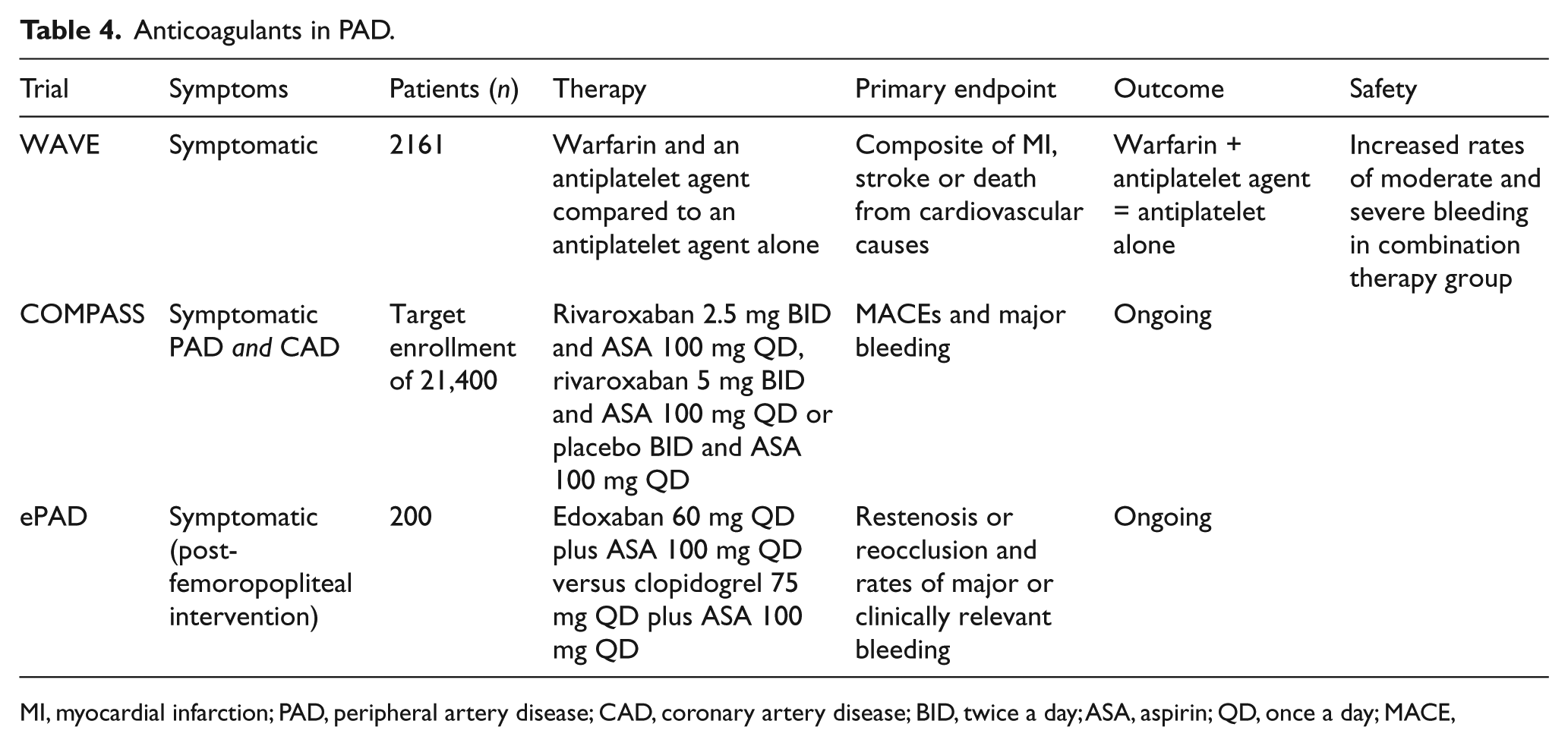
MI, myocardial infarction; PAD, peripheral artery disease; CAD, coronary artery disease; BID, twice a day; ASA, aspirin; QD, once a day; MACE,
Anticoagulant therapy following surgical interventions for PAD
A 2006 meta-analysis of nine randomized trials included a total of 4889 patients with PAD. Four of the trials compared oral anticoagulation with a control in patients with intermittent claudication or prior lower-limb revascularization. 69 Collectively, the mean patient age in these four trials was 62.6 years, and 23.8% were female; follow-up ranged from 3 to 10 years. Oral anticoagulation was associated with a significant reduction in graft occlusion (odds ratio (OR) = 0.63, 95% CI 0.44–0.89) and a trend toward reduced all-cause mortality (OR = 0.73, 95% CI 0.5–1.07). Rates of major bleeding were significantly higher in patients receiving oral anticoagulation (OR = 3.46, 95% CI 2.03–6.56).
This meta-analysis also included two trials comparing combination therapy with oral anticoagulation and aspirin to aspirin alone in patients with PAD undergoing infrainguinal bypass surgery. Among 887 primarily male (98%) patients with a mean age of 64.2 years and an average follow-up of 36 months, combination therapy with oral anticoagulation and aspirin was associated with a trend toward lower rates of graft occlusion (OR = 0.84, 95% CI 0.62–1.12; p = 0.2), at the expense of increased all-cause mortality (OR = 1.57, 95% CI 1.16–2.12; p = 0.004) and major bleeding (OR = 2.13, 95% CI 1.27–3.57; p =0.004).
The remaining two trials included 2781 patients undergoing infrainguinal bypass surgery, randomized to oral anticoagulation or aspirin. Collectively, the mean participant age was 69 years, 36% were female and the average follow-up was 21 months. No significant differences in graft occlusion (OR = 0.91, 95% CI 0.77–1.06) or mortality (OR = 1.04, 95% CI 0.85–1.29) were identified between the two groups. Oral anticoagulation was associated with increased rates of major bleeding (OR = 1.96, 95% CI 1.43–2.69; p = 0.0003).
The Dutch BOA Study was a multicenter, randomized controlled trial of 2650 patients undergoing infrainguinal bypass surgery for PAD. 70 Patients were randomized to warfarin with a target international normalized ratio (INR) of 3.0–4.5 or aspirin (100 mg/day) . The primary endpoint was bypass graft patency, which was assessed at 3 months and 6 months after surgery and every 6 months thereafter. After a mean follow-up of 21 months, no difference in the rate of graft occlusion was identified (308 events in the anticoagulation group vs 322 events in the aspirin monotherapy group; HR 0.95, 95% CI 0.82–1.11). A subgroup analysis showed similar rates of graft occlusion in patients with femoropopliteal (HR 0.97, 95% CI 0.81–1.16) and femorocrural bypass grafts (HR 0.95, 95% CI 0.7–1.30). Further stratification by graft material demonstrated a higher risk of occlusion of non-venous grafts in patients receiving anticoagulation rather than aspirin (HR 1.32, 95% CI 0.81–2.15). Rates of occlusion of autologous vein grafts were lower in patients receiving oral anticoagulation than those receiving aspirin (HR 0.71, 95% CI 0.5–1.02).
There was a significant reduction in rates of ischemic stroke in patients receiving oral anticoagulation (17 events in the anticoagulation group vs 34 events in the aspirin alone group; HR 0.50, 95% CI 0.28–0.89), but this was associated with a marked increase in rates of hemorrhagic stroke (14 events in the anticoagulation group vs four events in the aspirin group; HR 3.48, 95% CI 1.14–10.6). The study was underpowered to assess the risk of the composite outcome of vascular death, MI, stroke or amputation, but a trend toward lower rates with anticoagulation versus aspirin was found (HR 0.89, 95% CI 0.75–1.06). This was accompanied by a significant increase in rates of major bleeding (HR 1.96, 95% CI 1.42–2.71).
The Department of Veterans Affairs Cooperative Study was a prospective trial comparing the effect of warfarin plus aspirin therapy to aspirin therapy alone in 831 patients undergoing peripheral arterial bypass surgery with either prosthetic grafts (373 patients) or venous grafts (458 patients). 71 Following surgery, patients were randomized to warfarin with a target INR of 1.4–2.8 plus aspirin (325 mg/day) or aspirin (same dosage) alone. The primary endpoint was bypass patency. After an average follow-up of 36.6 months, 44 patients (23.5%) with prosthetic bypasses in the combination therapy cohort developed graft occlusion compared to 64 (34.4%) in the aspirin group (HR 0.62, 0.62, 95% CI 0.42–1.92; p = 0.017). Among patients receiving venous grafts, 57 patients (25.1%) receiving combination therapy and 57 patients (24.7%) receiving aspirin alone developed graft occlusion after an average follow-up period of 39.3 months. Combination therapy with warfarin and aspirin was associated with higher rates of major hemorrhagic complications than treatment with aspirin alone (OR = 2.05, 95% CI 1.02–4.15; p = 0.04). The trials investigating anticoagulants after surgical revascularization for PAD are summarized in Table 5.
Anticoagulants following surgical revascularization.
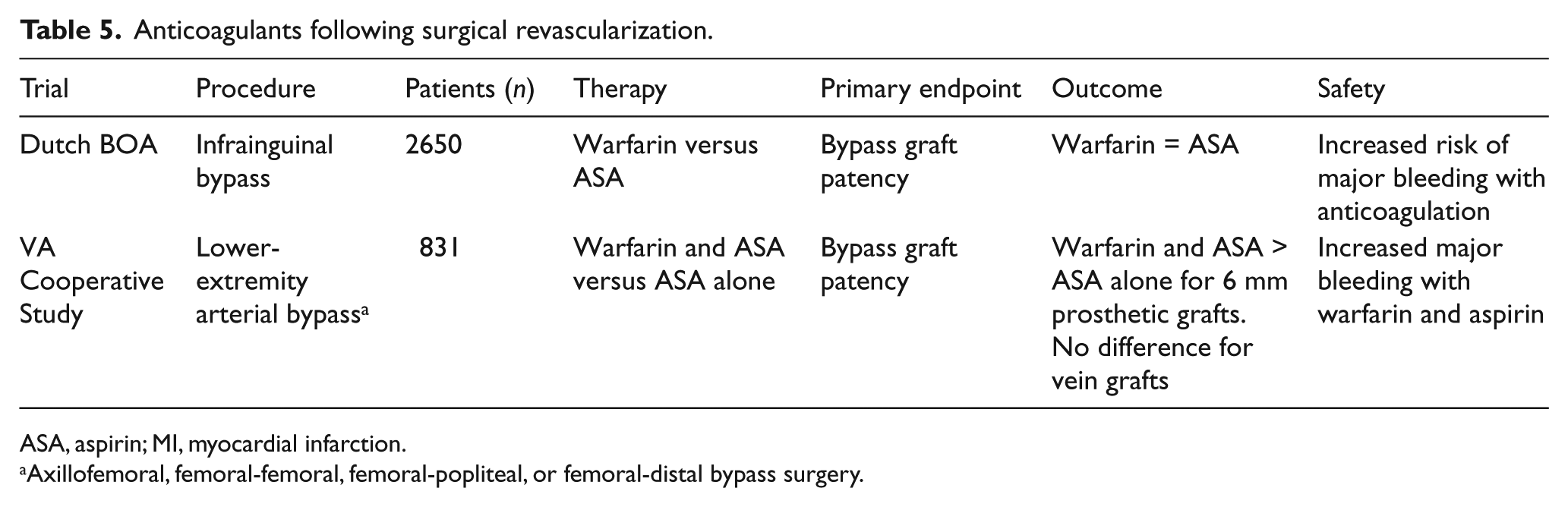
ASA, aspirin; MI, myocardial infarction.
Axillofemoral, femoral-femoral, femoral-popliteal, or femoral-distal bypass surgery.
In summary, the addition of warfarin to antiplatelet monotherapy in patients with symptomatic PAD is associated with an increased risk of life-threatening bleeding and no benefit in terms of adverse cardiovascular events. In patients who have undergone surgical revascularization for PAD, the use of warfarin increases the risk of major hemorrhagic complications without providing a clear benefit in terms of graft patency.
Direct oral anticoagulant agents in PAD
There is currently no prospective data available on the effects of direct oral anticoagulant (DOAC) agents in patients with PAD. The Rivaroxaban for the Prevention of Major Cardiovascular Events in Coronary or Peripheral Artery Disease (COMPASS) trial is an ongoing randomized controlled trial evaluating the role of rivaroxaban in patients with coronary or peripheral artery disease. 72 Patients are being randomized to one of three arms: (1) 2.5 mg of rivaroxaban twice daily and aspirin (100 mg once daily); (2) 5 mg of rivaroxaban twice daily and aspirin (same dosage); and (3) placebo twice daily and aspirin (same dosage). The primary outcome measures are time from randomization to first occurrence of MI, stroke or cardiovascular death, and time from randomization to the first occurrence of major bleeding. The study aims to enroll 21,400 patients and has an estimated completion date of February 2018.
The Edoxaban in Peripheral Artery Disease (ePAD) study is a recently completed multicenter randomized controlled trial evaluating the efficacy of edoxaban and aspirin versus clopidogrel and aspirin following femoropopliteal endovascular intervention. 73 The primary outcome measures are the proportion of patients with restenosis or reocclusion and rates of major or clinically relevant non-major bleeding at 6 months. Two hundred patients with Rutherford stage 2–5 PAD and > 50% femoropopliteal stenosis with at least one-vessel runoff to the affected foot were randomized to 60 mg of edoxaban once daily (two 30 mg tablets) plus aspirin (100 mg daily) for 3 months versus 75 mg/day of clopidogrel (after a loading dose of 300 mg once) plus 100 mg/day of aspirin for 3 months after endovascular intervention. Restenosis and reocclusion at the treated segment were assessed using duplex ultrasound at intervals of 1, 3 and 6 months following the procedure. The trial has completed enrollment, and the primary results are anticipated in 2016.
The use of rivaroxaban to prevent MACE and MALE in patients with PAD undergoing lower extremity revascularization is currently being investigated. The Efficacy and Safety of Rivaroxaban in Reducing the Risk of Major Thrombotic Vascular Events in Subjects With Peripheral Artery Disease Undergoing Peripheral Revascularization Procedures of the Lower Extremities (VOYAGER-PAD) is currently randomizing patients over 50 years of age with symptomatic lower extremity PAD who have undergone successful infrainguinal revascularization within the preceding 7 days to receive rivaroxaban (2.5 mg twice daily) or placebo in addition to standard background therapy. 74 The primary outcome measures include time from randomization to first occurrence of MI, stroke, cardiovascular death, ALI and major amputation, as well as time to first occurrence of major bleeding events. The study aims to enroll 6500 subjects, with an estimated study completion date of January 2019.
Conclusion
Patients with PAD are at significantly increased risk of atherothrombotic events such as MI, stroke and CLI. In patients with symptomatic PAD, this risk can be mitigated by the use of antiplatelet agents with an attendant increased risk of bleeding. Multiple pathways contribute to this elevated thrombotic risk in patients with PAD, providing targets for antithrombotic therapy. Ongoing trials investigating the use of antiplatelet and DOAC agents in this population will yield useful information for the management of patients with PAD.
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: EJA is a consultant/advisory board member to Abbott Vascular, Medtronic, Merck, and Spectranetics.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
