Abstract
B-vitamin trials failed to demonstrate beneficial effects on cardiovascular outcomes, but hyperhomocysteinemia still stands out as an independent cardiovascular risk factor, particularly in elderly individuals. B-vitamins may influence early vascular dysfunction, such as endothelial dysfunction, or may have adverse effects, for example on inflammation. We investigated the effect of B-vitamins on endothelial function and inflammation within an interventional study. This study was conducted within the framework of the B-PROOF trial, which included 2919 hyperhomocysteinemic elderly individuals, who received daily vitamin B12 (500 μg) and folic acid (400 μg) or placebo for 2 years. Using an electrochemiluminescence platform, we measured intercellular adhesion molecule 1 (ICAM-1), vascular adhesion molecule 1 (VCAM-1), serum amyloid A (SAA), vascular endothelial growth factor (VEGF) and C-reactive protein (CRP) at baseline and follow-up in a subsample of 522 participants (271 intervention group; 251 placebo). Treatment effects were analyzed with ANCOVA. The participants had a mean age of 72 years, and 55% of them were male. At the 2-year follow-up, B-vitamins did not change the ICAM-1 (+36% change in the intervention group versus +32% change in the placebo group; p = 0.72), VCAM-1 (+27% vs +25%; p = 0.39), VEGF (–1% vs +4%; p = 0.40), SAA (+34% vs +38%; p = 0.85) or CRP levels (+26% vs +36%; p = 0.70) as compared to placebo. In conclusion, in elderly patients with hyperhomocysteinemia, vitamin B12 and folic acid are unlikely to influence either endothelial function or low-grade systemic inflammation.
Introduction
Hyperhomocysteinemia is an independent risk factor for cardiovascular disease, in particular among more elderly individuals. 1 Several homocysteine-lowering trials with B-vitamins have been performed; however, no beneficial effects on cardiovascular disease or venous thrombosis have been observed, 2 with the possible exception of stroke.3,4 To date, this overall lack of benefit of lowering homocysteine remains unexplained. One possible explanation is that the association between homocysteine and cardiovascular disease is not causal. Alternatively, it has been proposed that beneficial cardiovascular effects of homocysteine-lowering therapy could be masked by adverse effects of high-dose B-vitamins, such as an increase in proliferation or inflammation. 5
It is possible that B-vitamins only influence vascular dysfunction at an early stage, like endothelial dysfunction, rather than benefit the process of advanced atherosclerosis or arterial stiffness. Earlier, we demonstrated no effects of B-vitamins on arterial stiffness and intima-media thickness, a surrogate measure of atherosclerosis. 6 An increased homocysteine level has previously been related to endothelial dysfunction.7–10 If homocysteine-lowering therapy did have a favorable effect on endothelial function, the time required to see this translated into a clinical endpoint may be considerably longer than the 2- to 5-year follow-up of the current homocysteine-lowering intervention trials.
Trials investigating the effect of B-vitamins on endothelial function usually measure flow-mediated dilatation, for which the results are conflicting.11–16 There are also several small trials investigating the effects of B-vitamins on endothelial biomarkers, again with conflicting results.13,17–20 Most of these trials were performed in selected samples, such as patients with coronary artery disease or diabetes, and did not select patients with high baseline homocysteine. Also, these trials were of relatively short duration (up to 40 weeks), and included relatively few individuals (maximum of 90). Furthermore, only a small variety of biomarkers were used, and data regarding the more important and relevant markers of endothelial function (intercellular adhesion molecule, vascular adhesion molecule, and vascular endothelial growth factor; ICAM, VCAM, and VEGF, respectively21–25) were often lacking.
As an alternative explanation for the lack of clinical benefit, adverse effects of high doses of B-vitamins on the cardiovascular system have been postulated. Folate fuels one-carbon metabolism, and may stimulate many metabolic active cells, including proliferating or inflammatory cells. Since the atherosclerotic process is partly inflammatory, involving macrophages and T-cells, it is possible that folate enhances inflammation in atherosclerotic lesions, which makes them prone to rupture. 5 Direct assessment of inflammation in atherosclerotic lesions is difficult, but the response of inflammatory biomarkers to B-vitamins may represent a proof-of-concept for pro-inflammatory effects of B-vitamins.
Based on these considerations, we set out to study the effects of B-vitamins on biomarkers of endothelial function and inflammation in a large population of hyperhomocysteinemic elderly individuals.
Materials and methods
Study participants
The B-PROOF trial is a multicenter, randomized, placebo-controlled, double-blind, intervention study. B-PROOF is an acronym for ‘B-vitamins for the PRevention Of Osteoporotic Fractures’. A detailed description of this randomized controlled trial has been reported elsewhere. 26 In short, the B-PROOF trial included 2919 participants from three areas in The Netherlands. Inclusion criteria were age of 65 years and older and an elevated homocysteine level (12–50 μmol/l). Fifty-one percent of the screened sample could be included based on their homocysteine level. The main outcome was osteoporotic fractures. Main exclusion criteria were renal insufficiency (creatinine level > 150 μmol/l) and presence of a malignancy in the past 5 years. All participants gave written informed consent before the start of the study. The B-PROOF study has been registered with The Netherlands Trial Register (http://www.trialregister.nl) under identifier NTR 1333 since 1 June 2008, and with ClinicalTrials.gov under identifier NCT00696514 since 9 June 2008. The Wageningen Medical Ethics Committee approved the study protocol. At the baseline measurement, participants were invited for vascular measurements and dual X-ray absorptiometry. In total, 522 participants underwent both additional measurements. In those participants who underwent the additional measurements, we measured biomarkers for endothelial function and inflammation, which we used for this substudy.
Intervention
The intervention period was 2 years. Participants were randomly allocated in a 1:1 ratio to the intervention group or to the control group. We stratified the randomization for study center, sex, age (65–80 years, ⩾ 80 years), and homocysteine concentration (12–18 μmol/L, ⩾ 18 μmol/L). The intervention group received a daily tablet with 500 μg vitamin B12 and 400 μg folic acid, and the control group received a daily placebo. Both tablets contained 15 μg (600 IU) vitamin D3 to ensure a normal vitamin D status. 27 The intervention and placebo tablets, produced by MCO Health (Almere, The Netherlands), are indistinguishable in taste, smell and appearance. The random allocation sequence and randomization were generated and performed using SAS 9.2 by an independent research dietician. Recruitment took place from August 2008 until March 2011.
Baseline and follow-up evaluations
All data and measurements were obtained at baseline and at follow-up. Participants were scheduled for a follow-up visit at 2 years after the intervention period. Adherence was judged by counting tablets, which the participants were asked to send back every 6 months. In case of non-reply, the participants were periodically phoned. Compliance was defined as more than 80% adherence over the 2-year period, based on the total amounts of tablets provided during the study.
Clinical characteristics
All measurements were performed at the baseline visit and follow-up visit. Alcohol intake, smoking habits, cardiovascular history and the presence of diabetes were determined using a structured questionnaire, 26 which was requested at both baseline and at follow-up measurements. Cardio-vascular history was defined as myocardial infarction, (unstable) angina pectoris, heart failure or arrhythmia thrombosis, peripheral arterial disease and cerebrovascular accidents (including transient ischemic attacks).
Laboratory measurements
Homocysteine and creatinine
Plasma homocysteine, serum creatinine, vitamin B12, folate, methylmalomic acid (MMA) and holo-transcobalamin (holoTC) were measured. Details regarding laboratory techniques are described elsewhere.26,28 The estimated glomerular filtration rate (eGFR) was calculated with the Modification of Diet in Renal Disease (MDRD) formula in ml/min/1.73 m2. 29
ICAM-1, VCAM-1, SAA, VEGF, CRP
A multi-array electrochemiluminescence platform (MesoScale Discovery, Gaithersburg, MD, USA; www.mesoscale.com) was used to measure intercellular adhesion molecule 1 (ICAM-1), vascular adhesion molecule 1 (VCAM-1), serum amyloid A (SAA), and C-reactive protein (CRP) simultaneously in serum. For VEGF, a single-array platform was used. Each serum sample was frozen after being drawn at baseline or follow-up and both baseline and follow-up samples were measured simultaneously. The samples were measured singularly and all measurements were done within a period of 30 days.
Statistical analysis
We calculated this study had to include 408 subjects in order to have a more than 80% power for detection of an improvement of 1.5 standard deviations (~ 10% change), taking into account an α-level of 0.05.
For comparisons between treatment groups, Student’s t-test was used for continuous variables and the chi-squared test for categorical variables. The Mann–Whitney U-test was used for variables which were not normally distributed.
Both intention-to-treat and per-protocol analyses were performed. In the intention-to-treat analyses, all participants with a baseline and follow-up measurement were included. In the per-protocol analysis, all non-compliant participants were excluded.
Associations between homocysteine levels and endothelial markers at baseline were tested using multivariable linear regression analysis, with standard adjustment for age, sex, study center and eGFR.
Differences between treatment groups were analyzed with analyses of covariance (ANCOVA), using the follow-up measurement as the dependent factor, the baseline measurement as covariate and treatment allocation as the fixed between-subject factor. This analysis was done for all continuous outcome measures. Before performing the ANCOVA, the assumption of linearity and homogeneity of variance was checked. The ANCOVA was first done unadjusted. Secondly, covariates were included in the final model as potential confounders, if a variable was different between the two treatment groups (p < 0.2) and contributed to a more than 10% change in F. Age, sex and study center were all added standard as covariate to the model. To investigate the effect of treatment in specific groups, interaction terms with treatment were tested for age, sex, cardiovascular disease history, homocysteine level, vitamin B12 and folic acid level. If an interaction term was significant (p < 0.05), stratified ANCOVA was performed. For continuous variables, strata were created based on tertiles.
To account for biological variability of the biomarkers of interest and increase statistical power, aggregate scores of endothelial function and inflammation were created. First, we calculated the differences between baseline and follow-up of all biomarkers. From this, we calculated overall Z-scores by dividing the difference of each participants’ individual level minus the mean level, by the standard deviation of the total sample. From these Z-scores, means were calculated. The inflammation score included CRP, SAA and ICAM-1. The endothelial function score included ICAM-1, VCAM-1 and VEGF. ICAM-1 is expressed both in monocytes and the endothelium, and was therefore included in both scores. 30
Statistical analyses were done with use of the statistical software package SPSS version 20.0 (SPSS Inc., Chicago, IL, USA). Two-sided p-values of < 0.05 were considered statistically significant.
Results
The study sample consisted of 522 participants; a subsample of the total B-PROOF study in whom biomarker data were collected. The characteristics of this subsample are shown in Table 1. Both groups were well-balanced with regard to baseline characteristics and covariates (Table 1). The median homocysteine level was 14.2 [interquartile range 12.9–16.4] µmol/l. As compared to the total B-PROOF population, the included participants were significantly younger, more often male, had slightly lower homocysteine levels (1.4%), 0.9% lower MMA levels and 14.5% higher holoTC levels. There were no differences in their cardiovascular disease history or cardiovascular risk factors (Table 5, supplementary material).
Characteristics of the study sample at the start of the B-PROOF study.
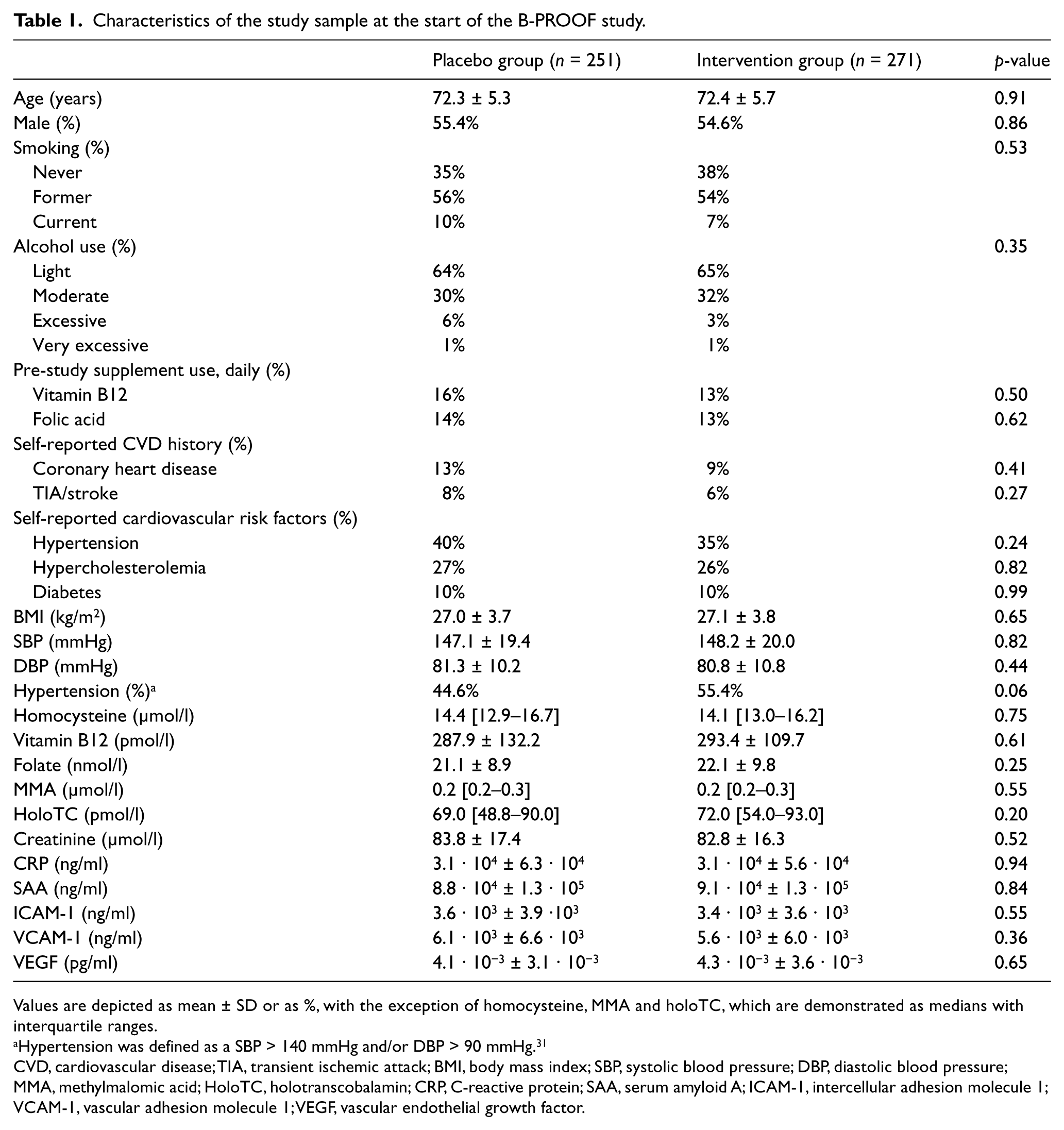
Values are depicted as mean ± SD or as %, with the exception of homocysteine, MMA and holoTC, which are demonstrated as medians with interquartile ranges.
Hypertension was defined as a SBP > 140 mmHg and/or DBP > 90 mmHg. 31
CVD, cardiovascular disease; TIA, transient ischemic attack; BMI, body mass index; SBP, systolic blood pressure; DBP, diastolic blood pressure; MMA, methylmalomic acid; HoloTC, holotranscobalamin; CRP, C-reactive protein; SAA, serum amyloid A; ICAM-1, intercellular adhesion molecule 1; VCAM-1, vascular adhesion molecule 1; VEGF, vascular endothelial growth factor.
At baseline, there was no association between homocysteine level and CRP, SAA, ICAM-1 or VCAM-1. There was a significant association between homocysteine level and VEGF level, adjusted for age, sex, study center and eGFR (beta 132.7 pg/ml [95% CI 55.6; 209.8]; p = 0.001) (Table 2, Figure 1).
Association between endothelial and inflammation markers and homocysteine level at baseline.
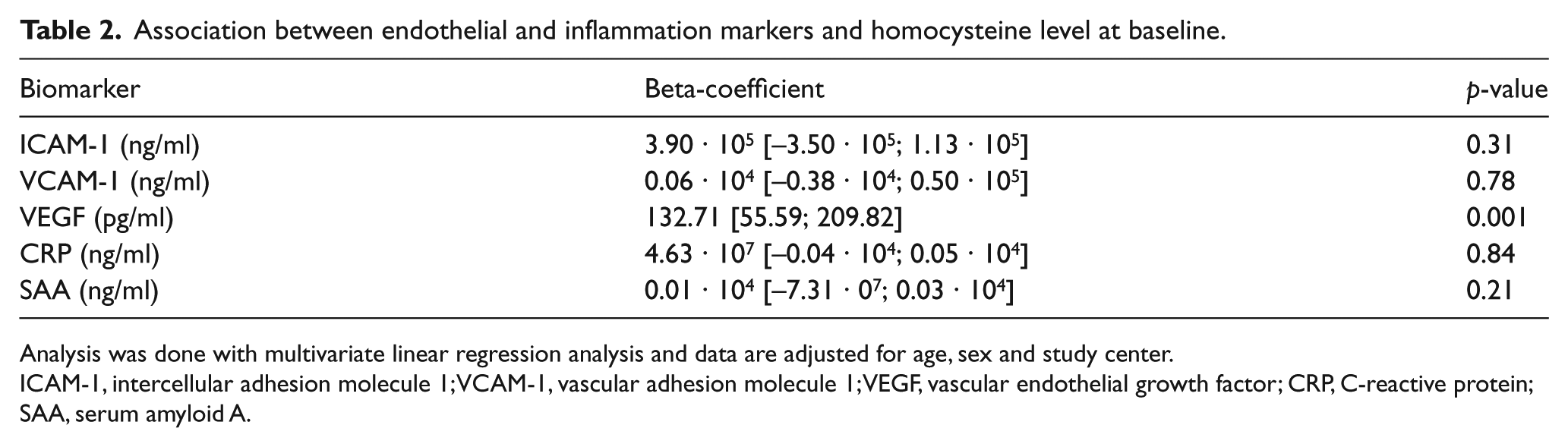
Analysis was done with multivariate linear regression analysis and data are adjusted for age, sex and study center.
ICAM-1, intercellular adhesion molecule 1; VCAM-1, vascular adhesion molecule 1; VEGF, vascular endothelial growth factor; CRP, C-reactive protein; SAA, serum amyloid A.
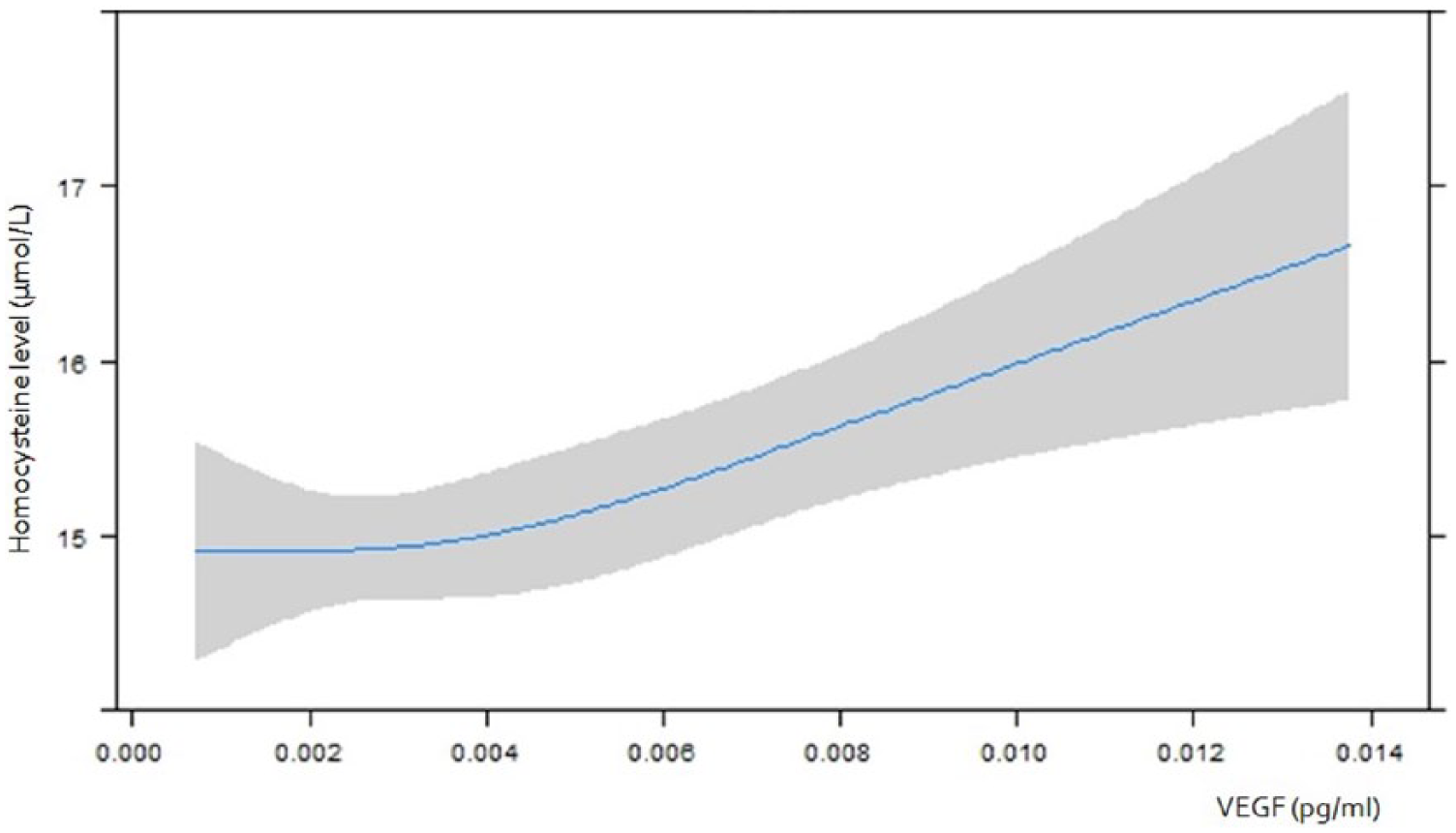
The association between homocysteine and vascular endothelial growth factor (VEGF) at baseline. This figure demonstrates the association between homocysteine level and VEGF cross-sectionally at the baseline measurement. The grey shaded area represents the 95% confidence interval.
After 2 years of follow-up, the homocysteine level decreased from 14.4 to 14.3 µmol/l (–0.1 μmol/l) in the placebo group, compared to a decrease from 14.3 to 10.3 µmol/l (–4.0 µmol/l) in the intervention group (p-value <0.05). B-vitamin treatment showed no effect on individual plasma biomarkers of endothelial function or on endothelial Z-scores (Table 3). For ICAM-1 levels, there was a 36% increase in the intervention group and a 32% increase in the placebo group between baseline and follow-up (p = 0.72). For VCAM-1 levels, the intervention group levels increased 27% versus 25% in the placebo group (p = 0.39). According to VEGF levels, there were no changes between baseline and follow-up in both groups (–1% versus +4%; p = 0.40). There was no effect of B-vitamin treatment on individual plasma inflammatory markers or on the inflammation Z-score (Table 4).
Changes in endothelial markers during the 2-year follow-up for the placebo and intervention groups.
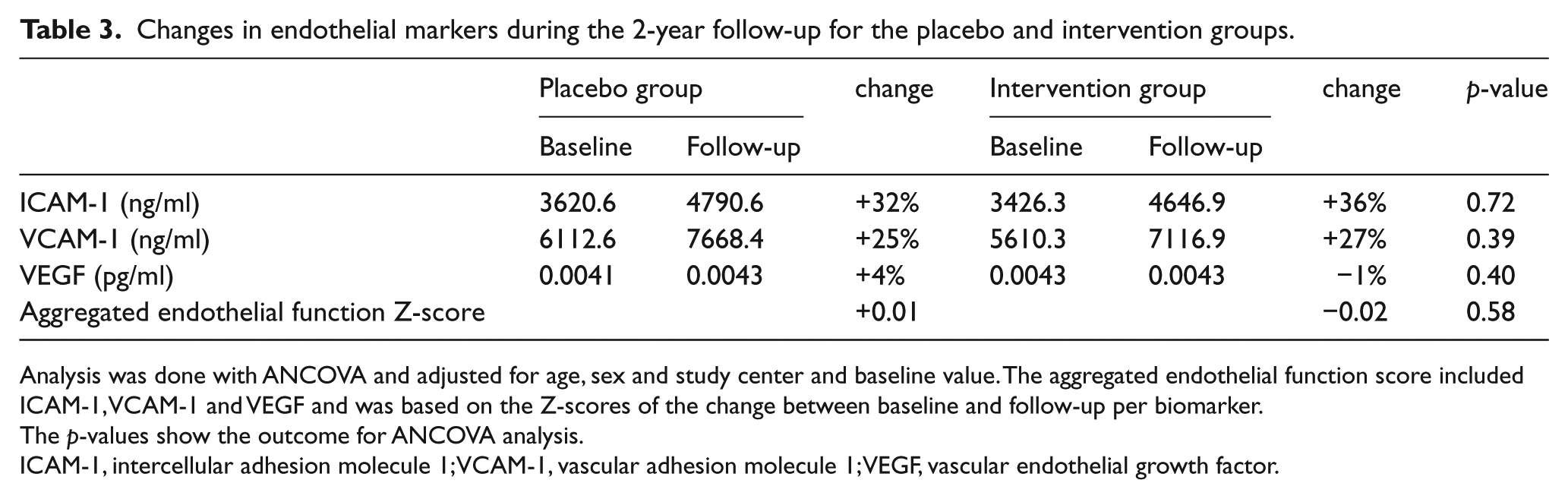
Analysis was done with ANCOVA and adjusted for age, sex and study center and baseline value. The aggregated endothelial function score included ICAM-1, VCAM-1 and VEGF and was based on the Z-scores of the change between baseline and follow-up per biomarker.
The p-values show the outcome for ANCOVA analysis.
ICAM-1, intercellular adhesion molecule 1; VCAM-1, vascular adhesion molecule 1; VEGF, vascular endothelial growth factor.
Changes in inflammation markers during the 2-year follow-up for the placebo and intervention groups.
Analysis was done with ANCOVA and adjusted for age, sex and study center and baseline value. The aggregated inflammation score included CRP, SAA and ICAM-1 and was based on the Z-scores of the change between baseline and follow-up per biomarker.
The p-values show the outcome for ANCOVA analysis.
CRP, C-reactive protein; SAA, serum amyloid A; ICAM-1, intercellular adhesion molecule 1.
Age, sex, homocysteine level, vitamin B12 and folic acid level did not modify the treatment effect on any of the studied biomarkers (all p-values > 0.05). The presence of cardiovascular disease appeared to be a significant effect-modifier for the treatment effect on SAA (p = 0.045), but not any of the other biomarkers (all p-values > 0.05). In individuals without cardiovascular disease, B-vitamins caused a 17% increase of SAA levels, whereas in persons with a cardiovascular history, SAA levels increased by 77% (Figure 2).
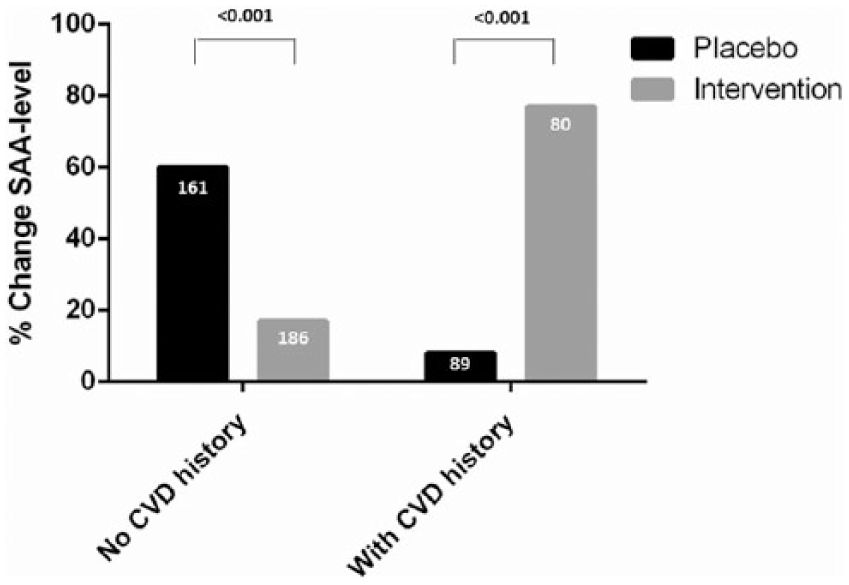
Stratified analysis participants with and without cardiovascular history based on percentage change in SAA level. Data represent the percentage of change of SAA levels in the placebo and intervention group stratified for participants without cardiovascular history (no CVD history) and with a positive cardiovascular disease history (with CVD history). The numbers in the bars represent the number of participants. The p-value of interaction was 0.045. Differences between placebo and intervention were not statistically significant.
In per-protocol analysis, 98% of participants remained in the intervention group and 95% remained in the placebo group. Also with this analysis, we observed no treatment effect of vitamin B12 and folic acid on endothelial function or inflammation.
Discussion
In our study, no effects of vitamin B12 and folic acid on 2-year changes in plasma biomarkers of endothelial function and inflammation were observed in hyperhomocysteinemic elderly subjects.
Other studies on the effect of B-vitamins on endothelial function showed conflicting results. Some suggested a beneficial effect, whereas others showed no effect at all.11–20,32–34 Most of these studies had a short follow-up time (maximum of 40 weeks) and were done in specific study groups. If a treatment effect of homocysteine-lowering therapy with B-vitamins is present, it would conceivably be observed most clearly in subjects with increased levels of homocysteine. We observed no treatment effect in such subjects after 2 years and no significant interactions. We believe this provides strong support for a lack of benefit of B-vitamins on endothelial dysfunction.
We selected ICAM-1, VCAM-1 and VEGF as biomarkers for endothelial function. ICAM-1 and VCAM-1 mediate the adhesion of endothelial cells to leukocytes. 35 Because ICAM-1 is also expressed on monocytes, this biomarker is also associated with inflammation. 30 High levels of ICAM-1 and VCAM-1 may be caused by damaged or activated endothelial cells. 36 VEGF induces the migration and proliferation of endothelial cells, and is also important for vascular permeability and thrombogeneticy. 37 Low-grade inflammation was assessed with CRP, SAA and ICAM-1 levels. CRP is a widely accepted biomarker for chronic low-grade inflammation and has been consistently associated with cardiovascular disease.38–41 SAA also predicts cardiovascular disease, but to a lesser extent than CRP. 40 ICAM-1 is expressed both in monocytes and the endothelium and was therefore applied in the inflammatory Z-score as well. 30
Our findings with respect to inflammation argue against the hypothesis that folate could increase systemic inflammation; however whether a lack of effect on circulating biomarkers excludes an effect on local inflammation in atherosclerotic plaques is unknown. Nevertheless, a small study in stroke patients did not observe a B-vitamin treatment effect on arterial wall inflammation, measured with positron emission tomography (PET) scans. 42 Further research is necessary to determine the intra-atherosclerotic plaque inflammatory process.
The possible difference in treatment effect on SAA levels between individuals with and without a history of cardiovascular disease implies we cannot completely rule out beneficial effects from B-vitamins in specific subgroups. In general, our study supports previous trials, 2 concluding that the cardiovascular system is generally unresponsive to B-vitamin treatment, as are cardiovascular disease outcomes, and arterial stiffness and endothelial function. This does not annihilate the strong association between homocysteine and cardiovascular disease, but further adds to the notion that we need to search for alternative explanations for this association.
Limitations
This study has its limitations. First, this study was performed in moderately hyperhomocysteinemic elderly individuals, therefore our results cannot be translated directly to the general population. The association between homocysteine levels and cardiovascular complications is gradual and fairly linear. 1 Nonetheless, a beneficial treatment effect of homocysteine lowering cannot be excluded in subjects with higher homocysteine levels than in our study population. Also, the biomarkers were only measured once, which does not ascertain a perfect quality of the measurements. Third, the use of an aggregated Z-score of endothelial function and inflammation ideally requires that each biomarker has an equal weight. Fourth, the outcome parameters of this report were not assigned as primary outcomes of the B-PROOF study at large. Further, circulating biomarkers do not cover the full spectrum of tests for assessing endothelial (dys)function and low-grade inflammation. Hence, we cannot exclude that, for example, tests of endothelial vasomotor function or of other circulating indicators of low-grade inflammation would show different results. However, the tests we used have proven relevance in their respective contexts and are widely used for study purposes similar to ours.
Conclusion
In conclusion, in elderly individuals with high–normal homocysteine levels, the use of vitamin B12 and folic acid did not influence endothelial function or inflammation.
Footnotes
Acknowledgements
The authors are grateful for all study participants and all dedicated co-workers who helped in the success of this trial. Further, we acknowledge the Department of Clinical Chemistry at Erasmus MC for their help with the electrochemiluminescence assays, in particular, Gerard Klein Heerenbrink.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B-PROOF is supported and funded by The Netherlands Organization for Health Research and Development (ZonMw, Grant 6130.0031), the Hague; unrestricted grant from NZO (Dutch Dairy Association), Zoetermeer; MCO Health, Almere; NCHA (Netherlands Consortium Healthy Ageing) Leiden/Rotterdam; Ministry of Economic Affairs, Agriculture and Innovation (project KB-15-004-003), the Hague; Nutricia Research Foundation, Singapore; Wageningen University, Wageningen; VU University Medical Center, Amsterdam; Erasmus Medical Center, Rotterdam. The sponsors do not have any role in the design or implementation of the study, data collection, data management, data analysis, data interpretation, or in the preparation, review, or approval of the manuscript.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
