Abstract
Mixed arterial venous disease is estimated to affect up to 26% of patients with lower extremity ulcerations. However, its clinical significance and pathophysiology are incompletely understood. Furthermore, there is no consensus on the optimal treatment modality, whether conservative or operative. In this review paper, we describe the current understanding of the pathophysiology of mixed arterial venous lower extremity ulcers. Guidelines for diagnostic tests for patients with mixed arterial venous diseases are discussed. We review some of the newer biological skin substitutes for conservative wound care. Finally, we propose a treatment algorithm based on current available data.
Introduction
Chronic lower extremity ulceration occurs in up to 5% of the population over 65 years of age. 1 The direct costs of treating lower extremity ulcerations, most secondary to venous disease, are approximately $3 billion per year in the United States. 2 It is estimated that 45–90% of all leg ulceration are of venous origin.1,3,4 The second most common etiology of lower extremity ulceration is peripheral artery disease (PAD), secondary to the high prevalence of cardiovascular risk factors, such as smoking, diabetes mellitus, and hyperlipidemia. 3 Less common etiologies of lower extremity ulcerations include vasculitis, exogenous factors, pyoderma gangrenosum, infection, neoplasia, calciphylaxis, and drug-induced. 5 The coexistence of both venous and arterial diseases is postulated to be present in up to 26% of patients with lower extremity ulceration (Figure 1). 6 However, the clinical significance and pathophysiology of this unique condition are not completely understood. In this paper, we discuss the pathophysiology and diagnosis of lower extremity ulcerations secondary to arterial venous disease. Finally, we review various treatment modalities, including arterial and venous interventions, and non-operative wound management directed at ulcer healing and, ultimately, limb salvage.
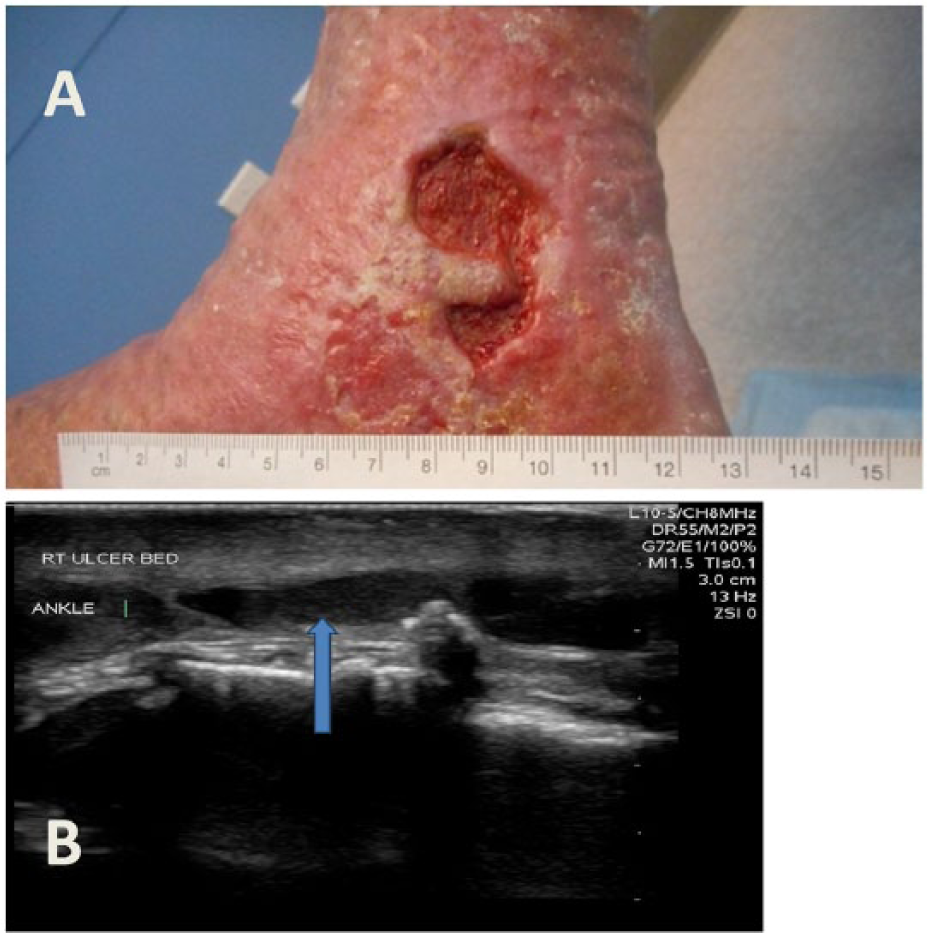
(A) Right medial malleolus ulcer with lipodermatosclerosis in a patient with long-standing venous and arterial disease. Patient had a right femoral to posterior tibial artery bypass graft in 2006 which has been maintained by endovascular interventions. Recent toe brachial index improved from 0.36 to 0.89 following inflow right superficial femoral artery angioplasty. (B) Duplex ultrasound revealing large vein (blue arrow) under ulcer bed; reflux > 2.5 seconds noted in greater saphenous and short saphenous veins.
Pathophysiology
Ulcerations in the lower extremities have long been attributed to chronic venous disease, secondary to venous reflux and/or obstruction. Arterial insufficiency from inadequate perfusion and oxygenation of the dermis can also lead to skin ulceration in the foot or leg. Treiman et al. defined combined arterial venous insufficiency as those patients with absent pedal pulses, an inflow arterial stenosis of >50%, and/or an ankle–brachial index (ABI) <0.90, along with superficial and deep venous insufficiency or reflux, or deep venous thrombosis (DVT) on duplex ultrasonography. 7
Venous reflux and/or obstruction can lead to increased ambulatory venous pressures, which, along with the inflammatory process and increasing hydrostatic pressure, leads to the clinical manifestations of chronic venous disease. Failure in the deep or superficial venous system or both correlates with the severity of venous disease and the development of symptoms that often lead to ulceration. 8 Both volume and pressure overload lead to progressive valve failure. 9 In some patients, symptoms of varicose veins and deep venous reflux can be reversed with treatment of the superficial venous reflux by ablation therapy or disruption of the superficial venous system.9,10
Genetic and environmental factors in venous disease include family history, female gender, pregnancy, estrogen levels, obesity, and prolonged sitting and/or standing. Genetic conditions such as Klippel–Trénaunay syndrome, cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy (CADASIL), FOXC2 gene mutations, desmulin dysregulation, and Ehlers–Danlos syndrome may present with venous disease, lower extremity edema, and varicose veins. 11 Secondary venous disease may involve thrombosis, inflammation, valvular damage, and recanalization resulting in venous wall damage and subsequent dilation. 11 In patients who develop post-thrombotic syndrome (PTS) following an episode of lower extremity DVT, there is a high likelihood of developing chronic lower extremity edema, skin changes, and ulceration.
Management
Lower extremity ulcers secondary to mixed arterial venous disease can present a particular challenge for clinicians. Although the optimal treatment algorithm for mixed arterial venous lower extremity ulcers remains unclear, the primary goal of therapy is still wound healing and limb salvage. The aims of management are to correct the underlying cause of the ulceration and achieve wound healing using the most current guidelines and available wound care therapies. A thorough clinical history, focusing on the duration and size of the ulcer with any associated lower extremity symptoms is the first step in patient management. A full evaluation of the patient’s comorbidities and overall health is important, as patients with arterial disease are likely to be older with significant cardiovascular comorbidities. A detailed history of DVT, family history of thromboembolism and/or venous disease, symptoms of arterial insufficiency, and history of prior treatments including arterial or venous interventions and compression use can provide information on the etiology of the ulcer and assist in treatment decision-making. Any contributing factors to poor wound healing (e.g. diabetes mellitus, smoking, vitamin deficiencies, vasculitides, or other systemic conditions) should also be identified. A thorough physical examination that also documents lower extremity pulses, presence of telangiectasia, varicose veins, edema; the location, size, and depth of the ulcers (healed or active); and any associated skin changes (e.g. hyperpigmentation, dermatofibrosclerosis, atrophie blanche, eczema, and/or discoloration) are critical for the patient’s evaluation. Finally, auscultation for bruits and reflux in the lower extremity are important to complete this evaluation.
Clinically, a venous ulcer differs from an arterial ulcer in several aspects. Venous ulcers are typically shallow/superficial and irregularly shaped. 12 Granulation tissue is often present along with signs of venous insufficiency, such as varicose veins or telangiectasia, associated venous disease skin changes, and edema. These wounds traditionally present in the lower third of the leg, especially around the gaiter region. Arterial ulcers can have a punched out appearance with distinct borders, and may involve the foot and/or the toes. 13 Gangrene may be present with arterial ulceration. Furthermore, patients with arterial ulcers may complain of more leg pain compared to patients with venous ulcers alone. In patients suffering from an ulcer of mixed arterial and venous origin, features of both ulcer types may be present (Figure 2). In these scenarios, clinical suspicion is important, especially in those with risk factors for both arterial and venous disease. On the other hand, neurotrophic wounds are distinct from arterial or venous ulcerations. They are typically found on the bony prominences of the foot such as the base of metatarsal heads and the lateral foot. These ulcers may also have a punched out appearance with hyperkeratotic borders. In contrast to those with phenotypic arterial or venous disease, a neurotrophic ulcer should be considered in those with impaired sensation of the foot. In patients with diabetes mellitus, neurotrophic ulcers form due to the presence of peripheral neuropathy, biomechanical pressure, and insufficient arterial perfusion.
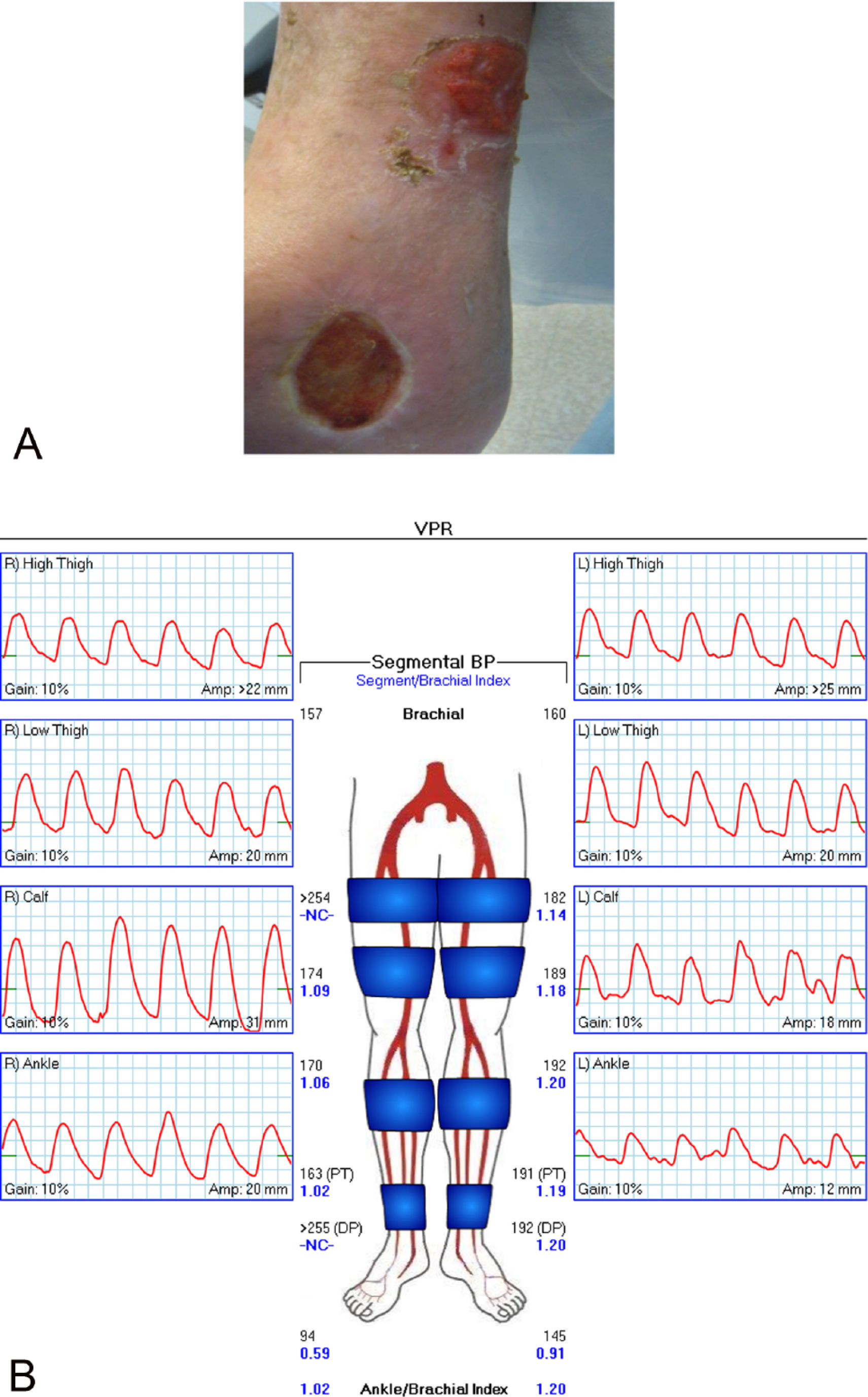
(A) Right leg and foot ulcers with induration in a patient with arterial and venous disease. (B) Pulse volume recording with mild to moderate dampening of waveforms, with a poorly defined dicrotic notch in the right lower extremity. The right toe-brachial index is abnormal, indicating mild inframalleolar occlusive disease.
Diagnostic studies
Documentation of arterial and venous disease is best achieved by non-invasive vascular laboratory studies in addition to clinical evaluation. Since the importance of adequate arterial perfusion in the healing of wounds cannot be overemphasized, ruling out PAD in patients with lower extremity ulceration is critical. Diagnostic tests include bilateral lower extremity pulse volume recording, ABI, and toe pressure measurements. An ABI <0.90 indicates PAD; however, near normal or elevated ABIs can be seen in patients with severe infragenicular disease in the presence of calcified and non-compressible tibial arteries, especially in those with diabetes mellitus and/or renal insufficiency or failure. 14 Conversely, an ABI <0.5 typically indicates severe arterial insufficiency. Toe pressures or toe–brachial index measurements can be useful in patients with non-compressible tibial arteries where ABIs may be falsely elevated. Pressure measurements are essential components in lower extremity arterial testing; however, the diagnostic accuracy of segmental pressure measurements decrease with multi-level disease, diabetic mellitus, vessel wall calcification, limb edema, and obesity. 15 A lower extremity arterial duplex ultrasound can be useful in identifying the level of arterial stenosis and/or occlusion to aid in treatment planning. In spite of this, duplex scanning of the infragenicular arteries can be limited due to artifacts from calcified vessels. 15
Over the last decade, several other modalities have been introduced, such as transcutaneous oxygen pressure measurement or laser Doppler measurement, which have also been used to determine the degree of arterial insufficiency based on lower extremity angiosomes but may not always be necessary. 16 The angiosome concept based on the pioneering work of Taylor and Palmer focuses on three-dimensional anatomic units of tissue supplied by specific source arteries. 17 For instance, in the foot, the peroneal, anterior tibial, and posterior tibial arteries supply six angiosomes. In recent years, investigators have evaluated the success of endovascular treatment for critical limb ischemia (CLI) based on angiosome-targeted arterial interventions. Limb salvage, skin perfusion, and ulcer healing rates are improved in the ‘direct’ revascularization groups where the specific angiosomes feeding the ulcers are targeted for treatment of ischemic or diabetic ulcerations compared to ‘indirect’ revascularization, where an unrelated angiosome is treated.18–20
Venous duplex ultrasonography with a combination of color flow venous duplex ultrasound along with Doppler and B-mode gray-scale imaging is typically performed to evaluate for venous thrombosis, reflux, and obstruction. It includes the following components: direct visualization of the deep, superficial, and perforator venous anatomy; assessment of compressibility; phasic venous flow with and without augmentation; and documentation of reflux by measuring valve closure times. 21 Valvular reflux has been traditionally defined by a closure time ⩾ 0.5 seconds. It is estimated that 74–93% of all patients with venous lower extremity ulceration will have reflux of the superficial venous system.22,23
Treatment
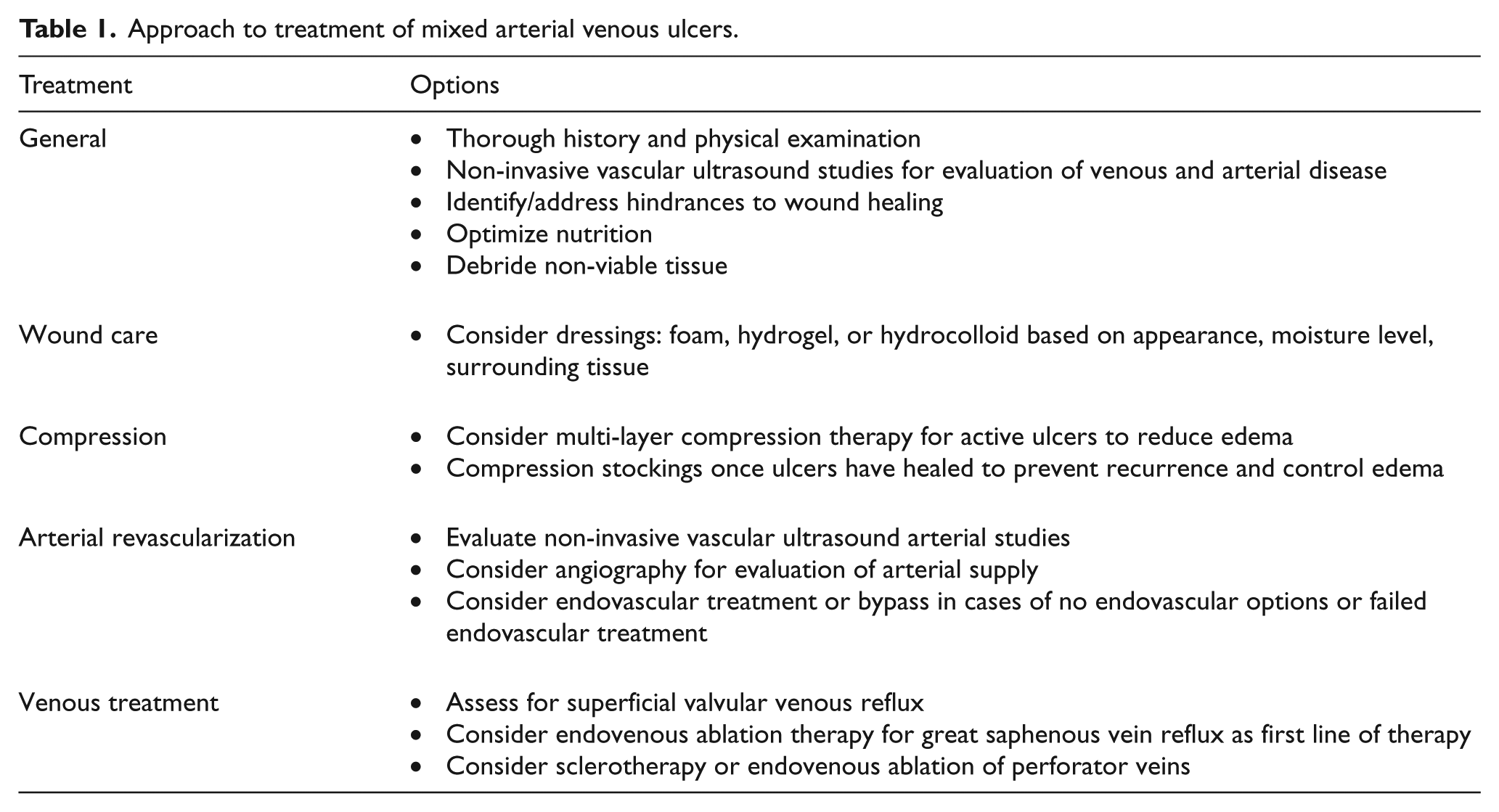
A multi-disciplinary and multi-level approach to treatment and management of mixed arterial venous lower extremity ulcers is critical in ensuring a successful outcome (Table 1). To ensure healing, wounds must be vascularized, free of necrotic and devitalized tissue, clear of infection, and kept moist. Ideal wound dressings absorb excess fluid while maintaining a moist environment, control bacterial invasion and/or proliferation, eliminate dead space, debride necrotic tissue, avoid maceration of healthy surrounding tissue, and minimize pain with dressing changes.
Approach to treatment of mixed arterial venous ulcers.
The many categories of wound dressings, designed for different stages of healing and different wound beds, should be adapted with the transformation of the different stages of wound healing. Chronic wound management can be simplified into the following categories: hydrogels for the debridement stage, foams for the granulation stage, and hydrocolloids for the epithelialization stage. 24 Foam dressings can absorb a large volume of exudate. Similarly, alginates are natural complex polysaccharides that may be used for moderately to heavily exudative wounds while keeping the wound base moist. Alginates can also be used to augment hemostasis and for packing deep spaces. Special dressings are suited to control the moisture level in and around the wound bed. Film dressings, such as Tegaderm™ (3M, St. Paul, Minnesota), may be applied to wounds for local protection; however, these dressings have limited absorptive capacity and may not be appropriate for heavily draining wounds. Hydrocolloids are dressings made of gel or foam on a self-adhesive film or polyurethane. Given their water-absorptive components (such as gelatin and pectin), these dressings can trap exudate and maintain a moist environment. Cadexomer iodine is a type of hydrocolloid, where the iodine is dispersed slowly when in contact with wound fluid. Research has found that cadexomer iodine enhances venous ulcer healing rates compared with standard of care. 25 Hydrogels consist of a matrix of various types of synthetic polymers with >95% water formed into sheets, gels, or foams, which are most useful for dry wounds. Some newer dressings contain active antimicrobial ingredients, such as silver, with concentrations ideal for antibacterial activity while preserving cell viability in healing wounds.
Wounds must be continually monitored, and if the healing progress of a wound is suspended, a different type of dressing should be considered. In the setting of the mixed arterial venous etiology, patients should be re-evaluated to ensure optimized arterial perfusion. Also, any additional factors that may affect wound healing should be addressed. These can include poorly controlled diabetes mellitus, smoking, malnutrition, increased bioburden, excessive pressure, and concomitant conditions/medications that impair wound healing.
For decades skin grafts using the patient’s own skin have been used to improve lower extremity ulcer healing with mixed results, with most trials showing no firm benefits. 26 In recent years, biologic cell-based dressings are being used increasingly for the treatment of chronic wounds. A recent Cochrane Review on ‘Skin grafting for venous leg ulcers’ found that bilayer artificial skin used in conjunction with compression dressings increased wound healing compared to traditional dressings with compression. 26 Cell-based dressings are composed of a live-cell construct that contains at least one layer of live allogenic cells. These materials mimic native skin consisting of extracellular matix materials, isolated (decellular) or in combination with various cell types such as fibroblasts or keratinocytes. 27 Skin substitutes function as sterile tissue grafts applied to wound beds and integrate with native tissue to stimulate cell migration, angiogenesis, and epithelialization.
Biologic dressings can be used when traditional dressings have failed or are deemed inappropriate. 28 Sheehan and colleagues demonstrated that when biological dressings are used for chronic wounds that have failed to heal at an appropriate rate of closure, a 55% reduction in wound area can be achieved within 4 weeks of treatment. 29 These types of dressings may utilize epidermal or dermal elements. Examples of these biologic dressings include Apligraf® and Dermagraft® (Organogenesis, Canton, Massachusetts). Acellular matrices serve as a scaffold, which may assist in wound healing and regeneration. Examples of these matrices include AlloDerm® (LifeCell, Bridgewater, New Jersey) and INTEGRA® (Integra:Integra LifeSciences, Plainsboro, New Jersey ).
The mainstay treatment for chronic venous insufficiency remains compression therapy with dressings (elastic or inelastic) or graded compression stockings. In the past, modified reduced compression was recommended for patients with ABIs of 0.5–0.7. 30 However, it has been demonstrated that in patients with mixed arterial venous ulceration of the lower extremities, with an ABI >0.5 and an absolute ankle pressure >60 mmHg, inelastic compression up to 40 mmHg does not impede arterial perfusion. 16 Supervised modified compression therapy has been shown to achieve a 68% and 53% ulcer healing rate in patients with moderate and severe arterial disease, respectively, at 36 weeks. 31 Compression therapy improves the healing of ulcers and reduces ulcer recurrence rates.30,32 Most medical compression stockings are double-layer and provide compression ranging from 23 to 40 mmHg. Compression therapy is believed to improve venous hypertension by improving the venous pump and lymphatic drainage.12,33 Local edema is relieved since the local hydrostatic pressure is increased and the superficial venous pressure is decreased. This in turn reduces the leakage of fluid and macromolecules. Furthermore, compression therapy leads to an increase in transcutaneous oxygen pressure secondary to increased cutaneous blood flow.33,34 During the initial treatment phase of mixed arterial venous ulcers, reducing lower extremity edema is critical. Inelastic (i.e. rigid bandages) or elastic and multi-layered bandages can be applied in combination with wound care. Elastic bandages can accommodate changes in volume of the leg associated with the edema. Compression should provide at least an 18–24-mmHg pressure for prevention of recurrence, allowing ulcer healing. 13
Intermittent pneumatic compression (IPC) has been evaluated for the treatment of venous ulcers with or without conventional compression therapy. Analysis of several randomized trials did not demonstrate consistent findings in regards to its efficacy for wound healing. Although intermittent compression assist devices for PAD have been evaluated, 35 little to no data exist for the use of IPC in mixed arterial venous ulcers. Although not routinely used, interface pressure measurement can be a useful tool in patients with suspected mixed arterial venous ulceration. Valuable information such as interface pressure may not only be important in treating venous ulcers but can also be important for the treatment of mixed arterial venous ulcers. Interface pressure measurement may provide objective information to better differentiate the therapeutic compression pressure range in those with PAD.
Endovascular obliteration of the great saphenous vein can be achieved in >90% of the cases using radiofrequency ablation or laser therapy with 4-year reflux-free rates of 86%.36,37 In our practice, we frequently begin the treatment algorithm with obliteration of superficial venous reflux via endovenous or surgical approaches. Venous Clinical Severity Scores (VCSS) can significantly improve with treatment of saphenous vein reflux. 38 The ESCHAR study demonstrated that in patients with active or healed ulceration, treating the great saphenous vein with compression therapy can reduce the ulcer recurrence rate by 25% at 4 years compared to compression alone. 39 In almost a third of the patients, deep venous reflux is obliterated when the superficial venous reflux is treated. 40 Following treatment of great and small saphenous vein reflux, correction of refluxing superficial and perforating veins is the next step in depressurization of the venous system. Reversal of venous reflux may convert a symptomatic patient to a healing patient and prevent progression to a more advanced stage of venous disease and ulceration. Practical considerations as to which vein to treat first and the techniques to employ may be challenging. New techniques also allow non-surgical options for the treatment of incompetent perforator veins, which combine the precision of a surgical approach with a minimally invasive option.
In the 1970s, DePalma and Edwards independently introduced a minimally invasive approach to perforator vein treatment addressing the problem of wound complications after sub-fascial ligation of incompetent perforators. 41 Popularization of endoscopy in surgery inspired the development of subfascial endoscopic perforator surgery (SEPS).42,43 Wound complications decreased with subfascial endoscopic ligation of perforator veins in SEPS; however, the occurrence of other complications, such as DVT (less than 1%), superficial thrombophlebitis (3%), and saphenous neuralgia (7%), as well as the technical complexity of the SEPS procedure and its high cost, stimulated interest in alternative techniques.44,45 In 1979, Fegan reported that successful injection sclerotherapy depends on several factors: (1) In the majority of patients with varicose veins and in almost all those patients with symptomatic venous disease, incompetent perforating veins are present; and (2) if these incompetent perforating veins are permanently occluded, the superficial veins are no longer subjected to an abnormal flow and are capable of regaining their normal tone and diameter, and their valves can regain competence. The aim of occluding these perforating veins is to prevent abnormal pressures and retrograde flow from the deep to superficial venous system. 41 Link et al. have described the treatment of a dilated popliteal fossa vein, by both ablation and sclerotherapy, which led to reversal of the deep venous reflux in the femoral and popliteal veins. 9
Strong associations of perforator incompetence with skin changes and ulceration have been well established. Incompetent perforators of 4–7 mm are best for treatment. Caution must be exercised in patients with severe arterial occlusive disease or active vasculitis as intra-arterial injection potentially leads to limb loss. Incompetence of a perforating vein can be determined by registering reversed flow (into the superficial veins) longer than 0.4 seconds, or by the size of the vein at the fascial opening exceeding 3.5 mm, or by the presence of both criteria. 41
The decision to treat arterial occlusive disease is based on several factors including the severity of arterial disease, clinical deterioration of the ulcer in the setting of conservative wound therapy and treatment of superficial venous reflux, as well as the patient’s overall health condition. Humphrey et al. reported the outcomes of 15 patients with mixed arterial venous ulcers with severe arterial insufficiency (ABI <0.5) who had a 13% 30-day mortality and 53% mortality at 36 weeks post-arterial revascularization. 31 These findings reiterate that patients with PAD are a higher risk population with significant cardiovascular comorbidities, making patient selection an important component of the treatment algorithm. Lantis et al. reported on a cohort of 27 patients with an ABI <0.7 who underwent percutaneous arterial revascularization of the lower extremities with a 100% technical success rate. 46 There was no 30-day perioperative mortality and the mean ABI improved by 0.4 ± 0.22. The majority of interventions were iliac artery or superficial femoral artery (SFA) angioplasty and/or stenting. The average time to ulcer closure was approximately 10 weeks. Four ulcer recurrences occurred due to failure of SFA interventions during a mean follow-up of 18 months. The success of arterial intervention depends on the patency of treated vessels as well as prior history of DVT. Treiman et al. demonstrated that despite patent bypass grafts performed for mixed arterial venous ulcers, a prior history of DVT was the risk factor most commonly associated with failure of lower extremity ulcer healing. 7 Arterial duplex surveillance following arterial revascularization to ensure patency of treated vessels is important to allow for timely reintervention if needed. 7 In general, we proceed with lower extremity angiography and revascularization for patients with mixed ulcers who have an ABI <0.7 if they have not had clinical improvements following treatment of superficial venous reflux (Figure 3).
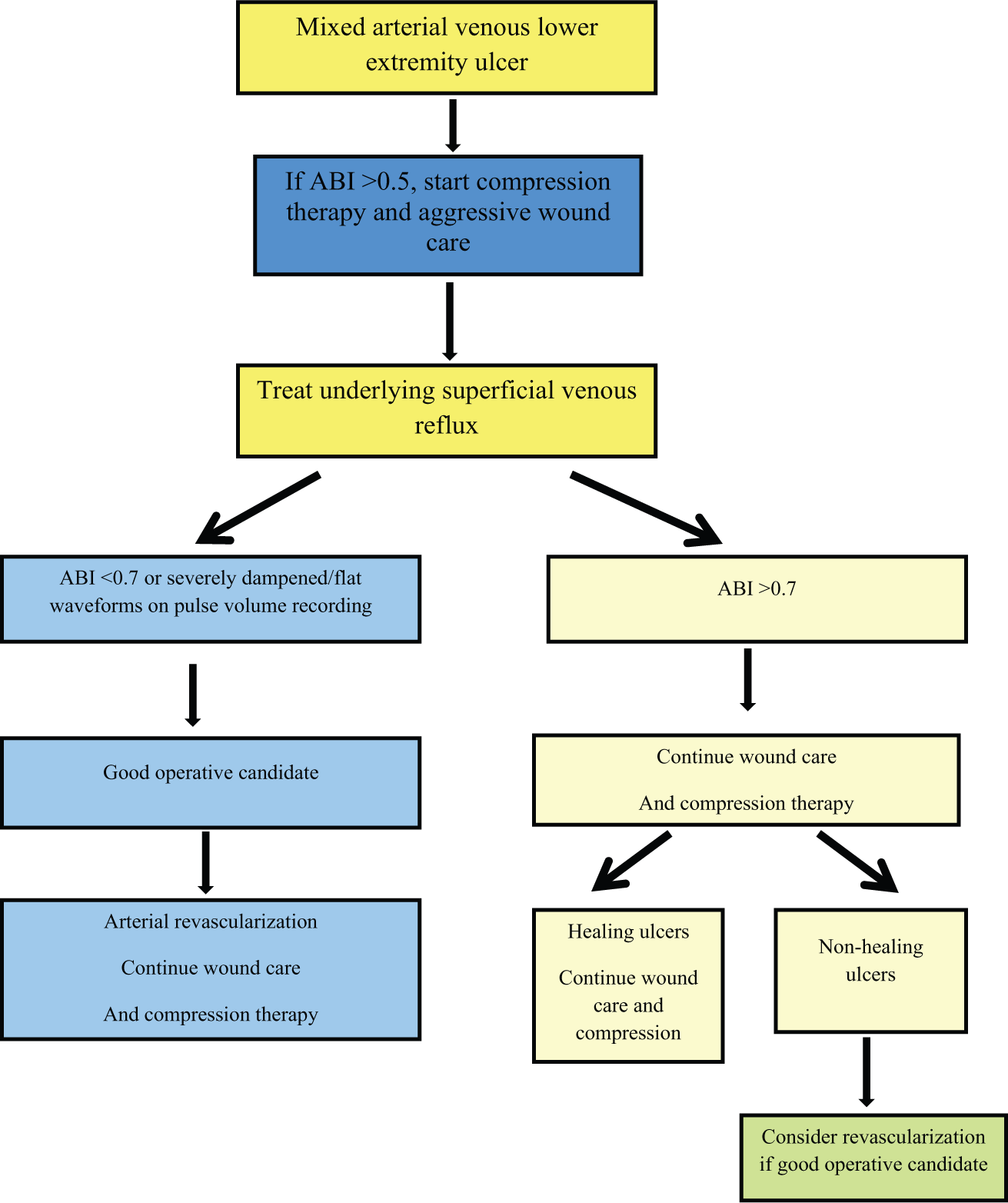
Treatment algorithm for patients with mixed arterial venous lower extremity ulceration.
For chronic lower extremity ulcers that have been unresponsive to multi-modality therapies and appear suspicious due to translucent granulation tissue or raised margins around the ulcer border, it is imperative to rule out neoplastic lesions. It has been documented that nearly 1–4% of all chronic leg ulcers will be positive for basal cell or squamous cell carcinoma.47,48
Conclusion
In summary, graded compression therapy and aggressive wound care using traditional methods or novel biologic dressings can be performed in the majority of patients with mixed arterial venous ulcerations. A full arterial and venous duplex evaluation is essential in treating patients with mixed arterial venous ulcer disease as these can affect management. Treatment of refluxing superficial lower extremity veins have been shown to be effective in achieving ulcer healing. In patients with mixed arterial venous ulceration who have severe PAD, revascularization is strongly advocated.7,46 Patients with an ABI <0.7 should be considered for revascularization via an endovascular or open bypass procedure to enhance ulcer healing (Figure 3); however, it is important to emphasize the importance of clinical judgment. Patients with an ABI <0.5 may need earlier arterial intervention to allow for aggressive compression therapy. Finally, following arterial intervention, standard wound care and compression therapy remain crucial to wound healing and the prevention of ulcer recurrence.
Future directions
As the optimal treatment of mixed arterial venous ulcerations remains a challenge for clinicians and patients, it is critical to explore other therapeutic options. Autologous and non-autologous cell therapies are being actively evaluated for the treatment of venous ulceration, as they have been for patients with severe PAD not amenable to revascularization and patients with diabetic foot ulcers. Another exciting research area is identifying biomarkers, such as bioactive cytokines and chemokines, 49 for prognostic test development to guide clinical decision making. This may help idenitfy those patients who may require earlier revascularization versus those who may respond well with conservative wound care alone.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
