Abstract
The cardiovascular risk factors that contribute to coronary calcification have been extensively studied while those related to tibial artery calcium are less well defined. We sought to determine the associations between cardiovascular risk factors and tibial artery calcification in a cohort of patients with and without significant peripheral atherosclerosis. A total of 222 patients without end-stage renal disease were identified in a prospectively maintained database containing tibial artery calcification (TAC) scores, and demographic, cardiovascular, and biochemical risk factor information. Patients with prevalent tibial artery calcification were more likely to be older, male, and have a history positive for hypertension, hyperlipidemia, diabetes, and tobacco use. Patients with an abnormal ankle–brachial index (ABI) or symptoms of peripheral artery disease (PAD) were also more likely to have higher calcium values. In analyses using multivariable logistic regression, age, gender, diabetes, and tobacco use maintained their association with prevalent tibial calcification while hypertension, hyperlipidemia and body mass index did not. These associations remained when PAD was added to the model. After adjusting for relevant cardiovascular risk factors, we found that only abnormal ABI, current PAD symptoms, and lower serum calcium values were associated with the presence of tibial artery calcification. In conclusion, in patients without end-stage renal disease, tibial artery calcification has risk factors that are similar but not identical to those for coronary artery calcification and peripheral atherosclerosis.
Keywords
Introduction
The risk factors for calcification in the coronary arteries have been previously defined. Age, gender, hypertension, hyperlipidemia, tobacco use, and diabetes are associated with increased calcium in intimal atherosclerotic plaque. 1 Medial calcification is more commonly associated with diabetes, renal failure, and markers of inflammation. 2 Risk factors for calcification in different vascular beds are generally similar but important differences exist. 3 Knowledge of such risk factors has greatly contributed to basic investigations on the pathologic mechanisms that contribute to arterial calcification and it can aid in developing potential treatments.4,5
Calcification in lower extremity arteries is associated with increased lower extremity events. 6 For example, linear calcifications in the femoral arteries of diabetic patients as seen on plain X-rays predict worse cardiovascular outcomes, including amputation. 7 Also, both patency and freedom from restenosis were worse in calcified femoral arteries after endovascular interventions. 8 Efforts to understand the mechanisms that contribute to lower extremity arterial disease are underway; however, the relationships between individual risk factors, biochemical markers, and tibial artery calcification have not been defined. Such knowledge may aid in our understanding of its pathologic mechanism while allowing us to predict which patients are at increased risk for lower extremity events, including amputation. We thus sought to identify the associations between cardiovascular risk factors, serum markers, and prevalent tibial artery calcification in a cohort of non-uremic patients with and without peripheral artery disease (PAD).
Materials and methods
Subjects
A total of 293 patients were recruited for a prospective study on tibial artery calcification from the vascular clinics of a single academic university hospital, an affiliated Veterans’ Administration Hospital, and the community. Vascular clinic patients (n=132) were from a referral population sent for evaluation of PAD symptoms. Community volunteers (n=161) responded to flyers in the hospital. All research investigations were conducted under a protocol approved by the Institutional Review Board (IRB) of Vanderbilt University Medical Center. From the initial cohort we excluded 71 patients due to either chronic kidney disease with an estimated glomerular filtration rate (eGFR) less than 30 (n=37) or incomplete biochemical or demographic evaluation (n=34), leaving 222 patients for the present analysis. Patients with an eGFR below 30 were excluded to avoid confounding by renal disease and associated mineral bone disorder.
Assessments
Patients were queried regarding symptoms of lower extremity arterial disease including claudication, rest pain and ischemic ulcers. Complete medical histories and cardiovascular risk factors were collected including the presence or absence of diabetes, tobacco use, hypertension, and hyperlipidemia. The presence of diabetes, hypertension, and hyperlipidemia were confirmed by review of the medical record and medication history. History of hypertension was confirmed by documentation in the medical record or current use of at least one anti-hypertensive medication. History of hyperlipidemia was based solely on self-reported history or current statin therapy. Tobacco use was self-reported. Lower extremity vascular exam and noninvasive arterial Doppler testing were performed in order to confirm the presence of PAD.
Biochemical measurements
Patients’ biochemical markers were evaluated within 30 days of computerized tomography (CT) evaluation using standard hospital-based laboratory procedures. Biochemical measurements included serum calcium, phosphate, total cholesterol, high-density lipoprotein (HDL), low-density lipoprotein (LDL), triglycerides, eGFR, and C-reactive protein (CRP).
Tibial artery calcium scoring
Standardized calcium scoring software (Heartbeat CS; Phillips Medical Systems, Amsterdam, The Netherlands) was used to score the tibial arteries. Investigators blinded to patient assessment performed the scoring. Individual tibial artery calcification (TAC) scores were derived for each extremity using the Agatston system. This was performed on non-contrast CT scans of the abdomen, pelvis, and bilateral lower extremities obtained during routine clinical assessment of PAD patients, or on CT scans that were obtained from the mid-thigh to the toes for community volunteers under an IRB-approved protocol. 9 Briefly, on cross-sectional images through the lower extremities, areas of calcification along the tibial arteries with a cross-sectional area greater than 1 mm2 and with a density of 130 Hounsfield units (HU) were identified automatically. Regions of interest along the distal popliteal, anterior tibial, posterior tibial and peroneal arteries were manually selected and labeled. To insure consistency, measurements were performed using boney landmarks starting at the bottom of the patella and ending at the widest portion of the tibial and fibular malleoli at the ankle. This usually included the bottom half of the popliteal artery and the entire length of the three tibial arteries down to the ankle. The average score was then computed by summing the two values (left and right) and dividing by 2. For statistical analyses, we used TAC > 0 to distinguish patients with calcification and log of (TAC +1) for correlations.
Statistical analysis
Descriptive statistics were calculated as median with interquartile range (median [IQR]) or mean with standard deviation (mean ± 1 SD) for continuous variables. Frequency expressed as percent was used for categorical variables. The normality of continuous variables was assessed using the Kolmogorov–Smirnov test. Values for arterial calcification were significantly skewed. Spearman’s rank correlation coefficients (rho) were calculated in order to evaluate the correlation between the TAC score and other continuous variables. Wilcoxon’s rank sum test was used to compare the TAC score for patients with and without hyperlipidemia, hypertension, diabetes, and tobacco use. Binary logistic regression was used to assess adjusted odds ratios and 95% confidence intervals (CIs) for the presence of tibial artery calcification related to demographic and cardiovascular risk factors. The Wald test was used to test for the real p-value of biochemical and inflammatory markers after adjusting for age, gender, tobacco use, and diabetes. Statistically significant results were concluded if the two-sided p-values were less than 0.05. Analyses were performed using R version 3.0.1 (www.r-project.org) and IBM SPSS version 22 (Armonk, NY, USA).
Results
The mean age for all patients was 57 years and those with tibial calcification were significantly older and more likely to be male. Patients with quantifiable arterial calcification on non-contrast CT scans were also more likely to have a history of hypertension, hyperlipidemia, diabetes, and tobacco use. Total cholesterol levels were lower in patients with positive TAC values but this may have been due to concurrent statin use. Calcium and the calcium × phosphate product were interestingly lower in patients with positive tibial calcium, but renal function, as measured by the eGFR, was decreased. There was no relation between tibial calcification and race, body mass index (BMI), triglycerides, or phosphate levels in our cohort. The percentage of patients with an abnormal ankle–brachial index (ABI) and PAD symptoms was significantly higher in patients with tibial artery calcium (Table 1).
Characteristics of study subjects by presence or absence of tibial artery calcification.
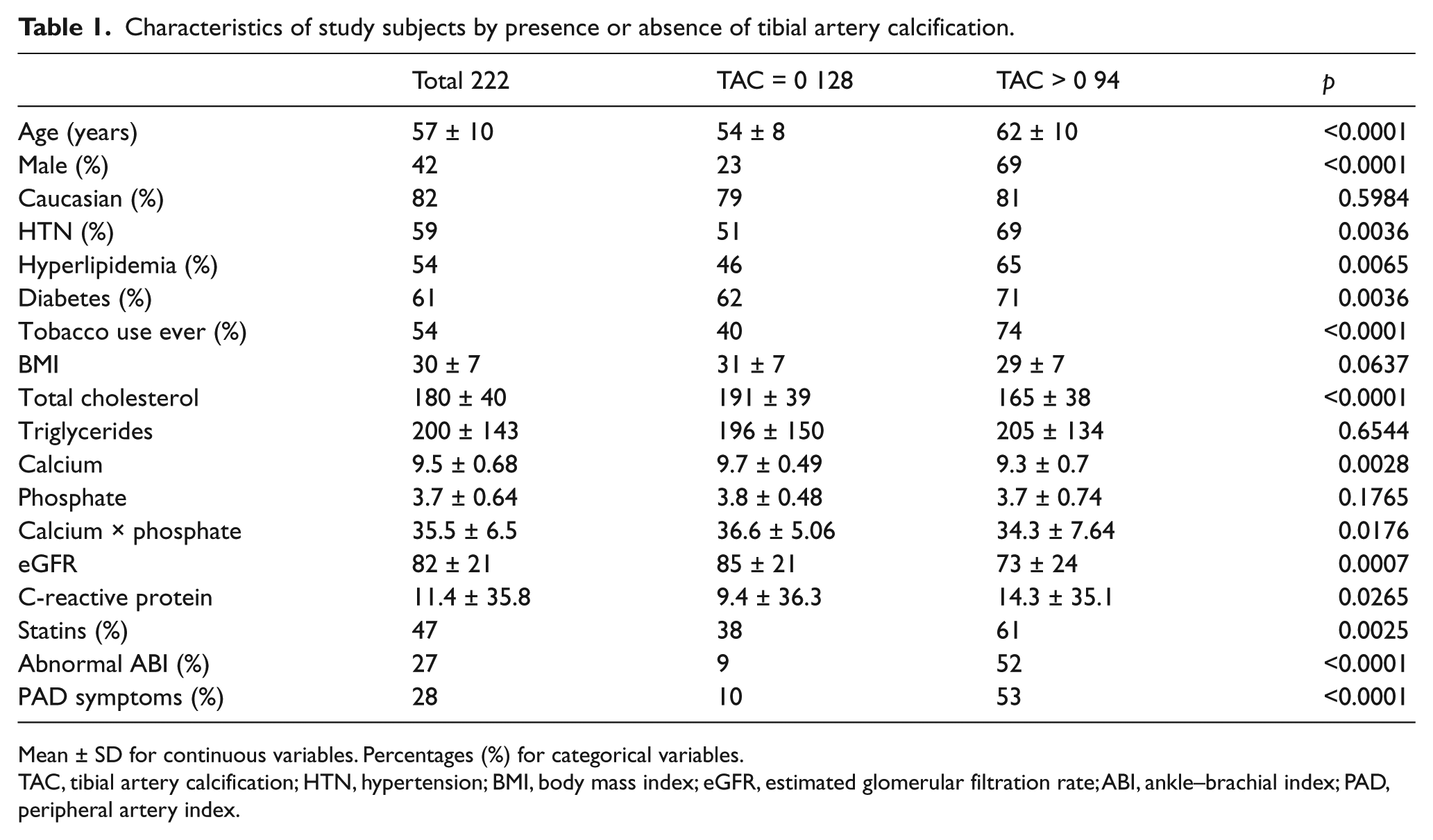
Mean ± SD for continuous variables. Percentages (%) for categorical variables.
TAC, tibial artery calcification; HTN, hypertension; BMI, body mass index; eGFR, estimated glomerular filtration rate; ABI, ankle–brachial index; PAD, peripheral artery index.
When we compared patients according to the presence or absence of PAD, we found that our PAD patients were older and more likely to be male. They had a higher incidence of hypertension, hyperlipidemia, and tobacco use. Diabetes was not more common in our PAD cohort, likely due to the high percentage in our non-PAD population. Again, total cholesterol and serum calcium levels were lower in patients with PAD (Table 2).
Characteristics of study subjects by presence or absence of peripheral artery disease (PAD).
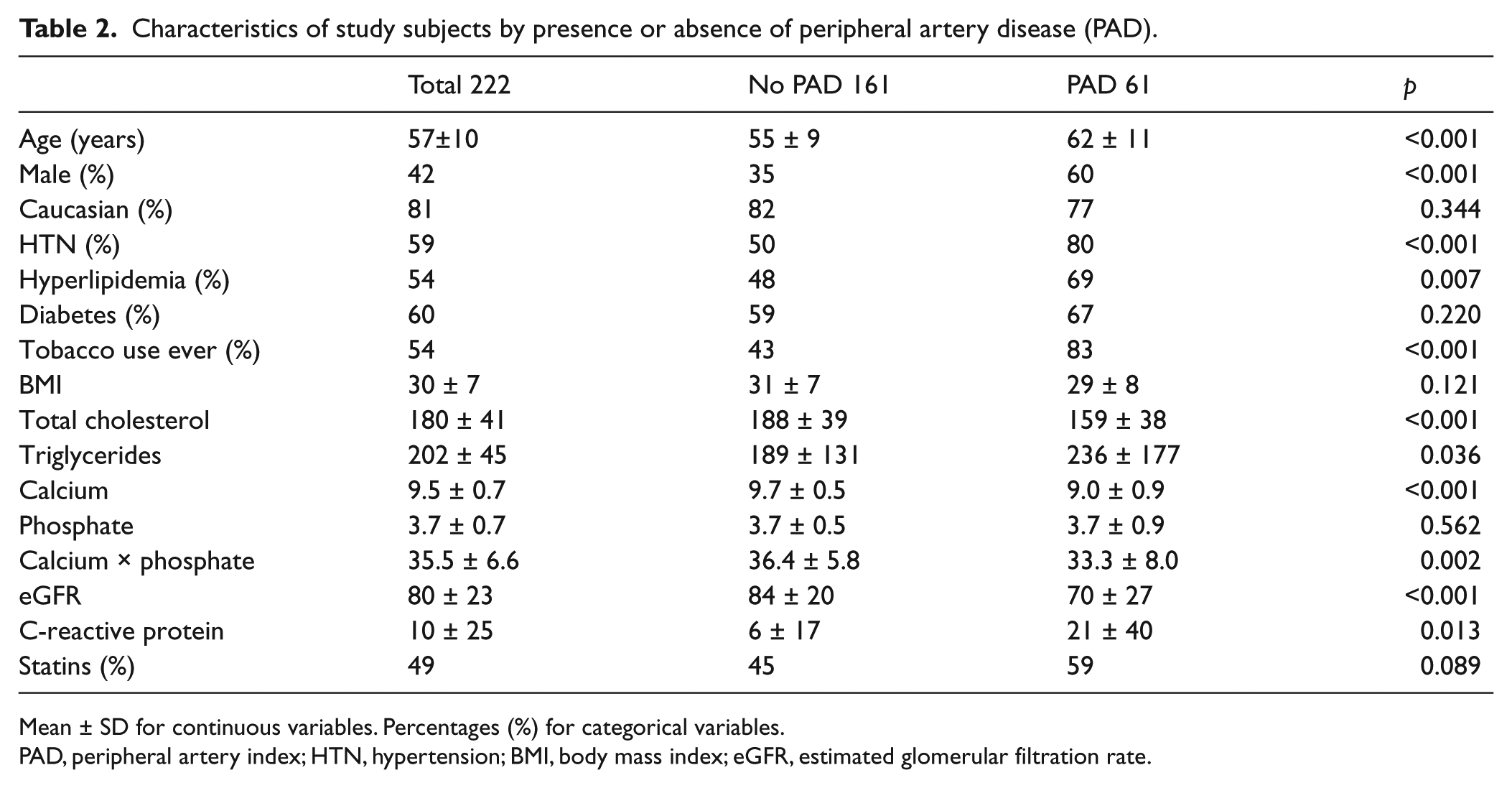
Mean ± SD for continuous variables. Percentages (%) for categorical variables.
PAD, peripheral artery index; HTN, hypertension; BMI, body mass index; eGFR, estimated glomerular filtration rate.
On univariate analysis, the TAC score was positively correlated with age, male sex, hypertension, hyperlipidemia, diabetes, and tobacco use, while it was negatively correlated with BMI (rho = −0.204, p = 0.003). Interestingly, in our patient population, the TAC score was inversely correlated with serum calcium levels (rho = −0.268, p <0.001), serum phosphate levels (rho = −0.163, p = 0.018), and the serum calcium × phosphate product (rho = −0.262, p <0.001). Patients with abnormal ABIs or symptoms of PAD also had significantly higher TAC scores (Table 3, Figure 1).
Univariate analysis: Tibial artery calcification (TAC) score.
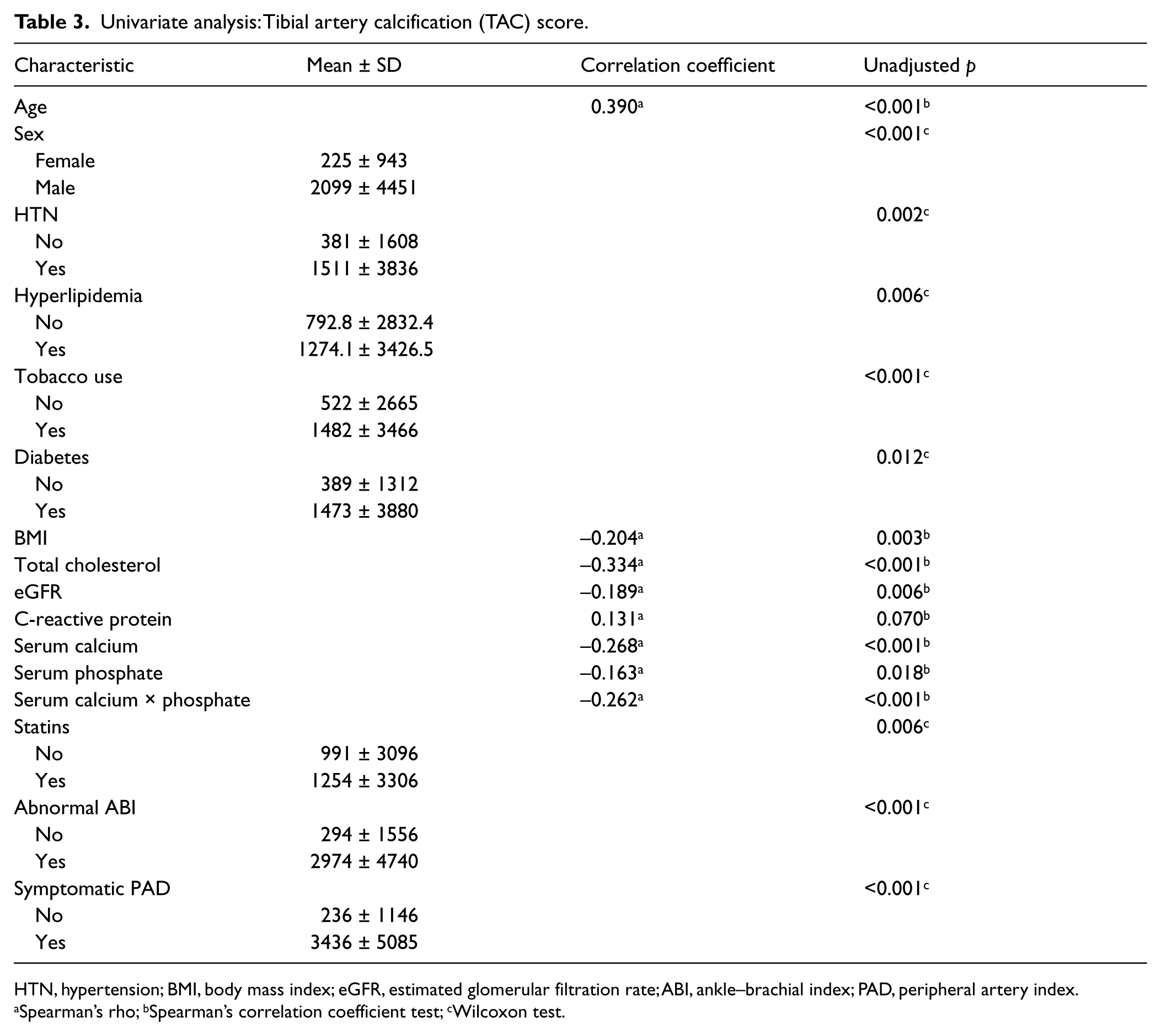
HTN, hypertension; BMI, body mass index; eGFR, estimated glomerular filtration rate; ABI, ankle–brachial index; PAD, peripheral artery index.
Spearman’s rho; bSpearman’s correlation coefficient test; cWilcoxon test.
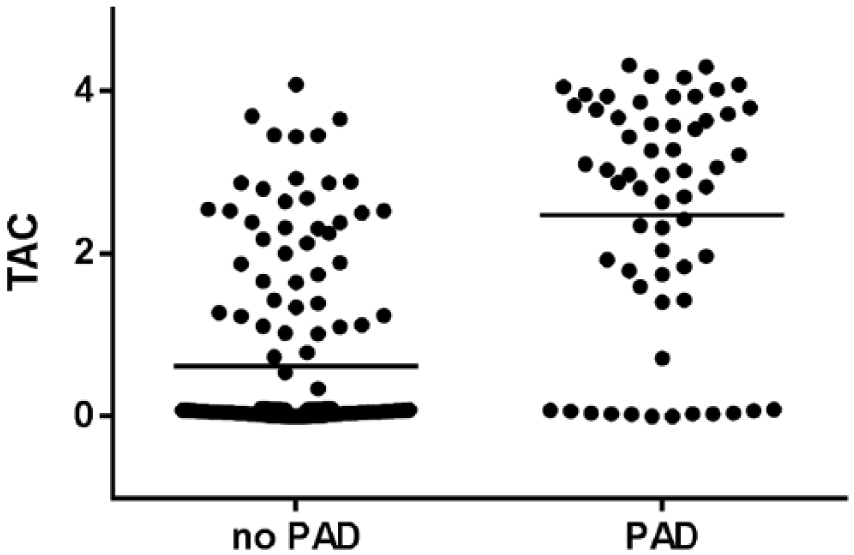
Scatter plots of individual tibial artery calcium (TAC) scores in patients with and without peripheral artery disease (PAD).
In multivariable analysis, age, male sex, diabetes, tobacco use and symptomatic PAD were independently associated with calcification while hypertension and hyperlipidemia were not (Table 4). When evaluated separately, our subgroup without PAD showed a persistent association between age, male sex, and diabetes with tibial calcification, while tobacco use failed to reach significance. For the group with PAD, age, male sex, and tobacco use maintained their significant association with tibial calcification while diabetes with an odds ratio of 3.2 failed to reach significance.
Multivariable analysis: Presence of tibial artery calcification based on the logistic regression model.
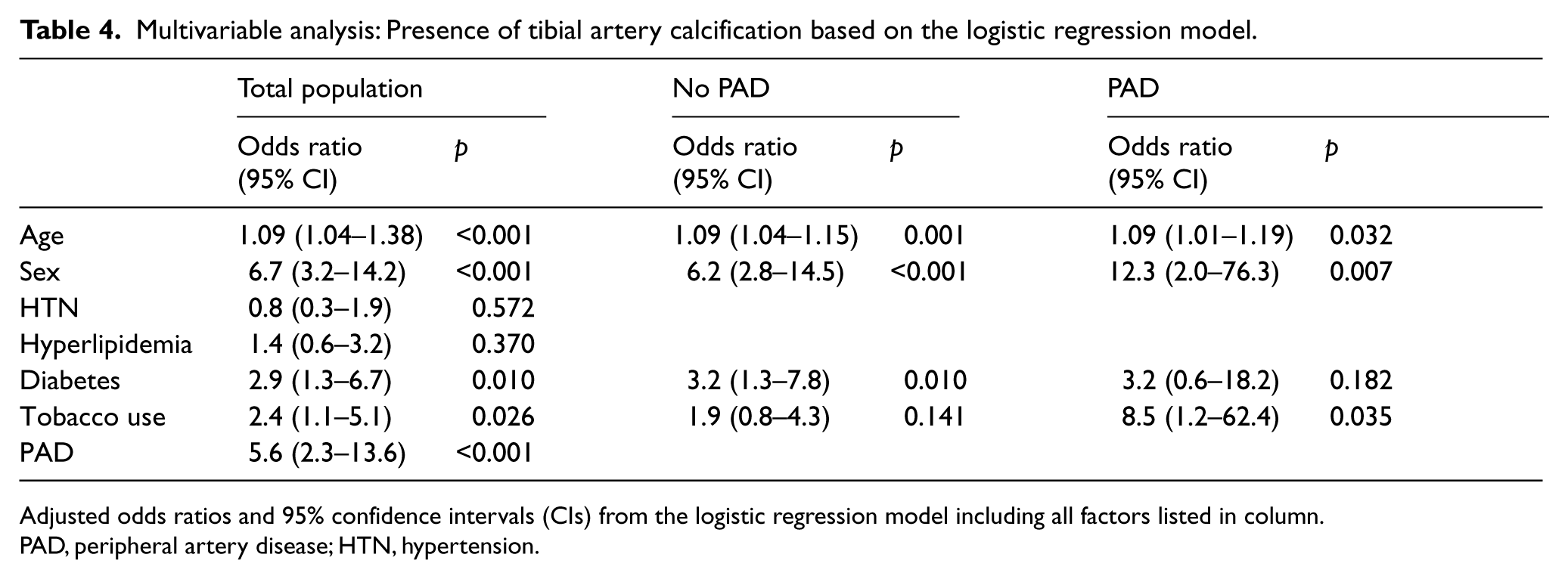
Adjusted odds ratios and 95% confidence intervals (CIs) from the logistic regression model including all factors listed in column.
PAD, peripheral artery disease; HTN, hypertension.
No association was found between the biochemical factors including total cholesterol, triglycerides, CRP, and eGFR and tibial artery calcium in logistic regression that adjusted for age, male sex, diabetes, and tobacco use. There was no correlation between the TAC score and serum phosphate levels or the calcium × phosphate product; however, interestingly, the inverse association between serum calcium and the TAC scores remained (OR [95% CI], p-value) (0.501 [0.27–0.93], p = 0.020) (Figure 2). Additionally, both the presence of an abnormal ABI (8.80 [3.12–24.9], p<0.001) or symptoms of PAD (7.36 [2.86–18.9], p<0.001) were strongly associated with prevalent tibial artery calcification.
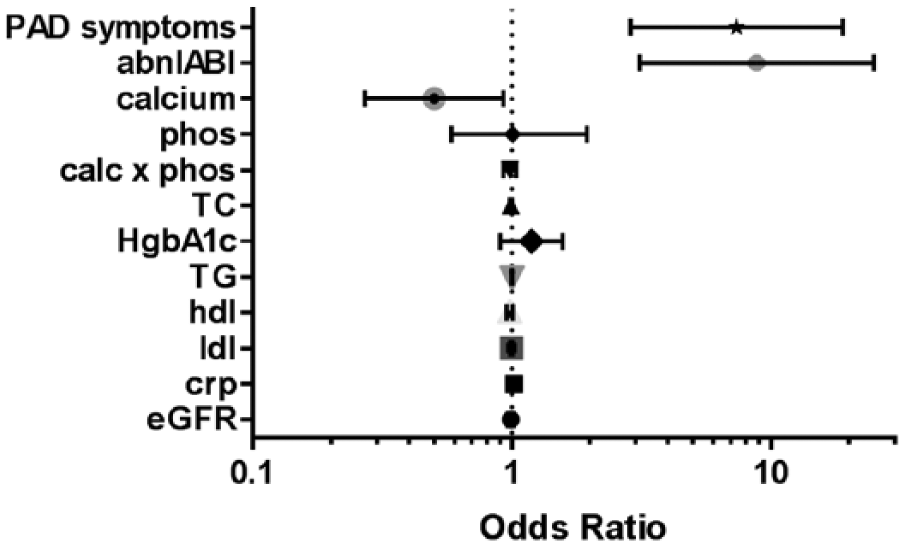
Individual adjusted odds ratios and 95% confidence intervals for arterial calcification after adjusting for age, sex, history of tobacco use, and diabetes in the logistic regression model. (PAD, peripheral artery disease; abnl ABI, abnormal ankle–brachial index; phos, phosphate; calc, calcium; TG, Total cholesterol; HgbA1c, glycated hemoglobin; TG, triglycerides; hdl, high-density lipoprotein; ldl, low-density lipoprotein; crp, C-reactive protein; eGFR, estimated glomerular filtration rate.)
Discussion
The main findings in this manuscript are that in patients without severe renal disease, tibial artery calcification is associated with age, male sex, diabetes, and tobacco use but not hypertension, hyperlipidemia, or BMI. These risk factors remain even after including the presence of PAD in our analysis. There is no association between tibial calcification and biochemical factors including serum cholesterol, triglycerides, HgbA1c, and CRP. There was a mild inverse correlation with serum calcium but not phosphate levels or the calcium × phosphate product after adjusting for cardiovascular risk factors. It is also strongly associated with abnormal ankle pressures and symptoms of PAD.
Recently, investigators have shown in patients with diabetes that below-knee calcification is associated with cardiovascular events and tobacco use; however, individual risk factors including hypertension, diabetes, and hyperlipidemia were not addressed.10,11 In the coronary arteries, where an understanding of calcification and its predictive value have developed over the past two decades, investigators have identified separate determinants of intimal versus medial calcium deposition. Age, diabetes, and renal failure have been most strongly associated with medial calcification, while the additional factors of male sex, dyslipidemia, hypertension, and tobacco use have all been associated with intimal calcification. 2 In a recent study on patients with metabolic syndrome, hyperglycemia, hypertension, hyperlipidemia, and BMI were all associated with an increased risk of coronary artery calcification (CAC). 12 In women, hormone replacement therapy is associated with decreased coronary calcium while LDL-cholesterol and HDL-cholesterol levels predicted increased coronary and aortic calcium scores. 13 In the population-based Rochester Heart Study of patients with a strong family history of coronary artery disease, the most important predictor of coronary calcium after increasing age was plasma triglyceride levels. 14 Thus, while there is clear overlap between CAC and tibial calcification risk factors, there are also some clear differences. Hypertension, BMI, lipid levels, and biochemical factors do not appear to be as strongly associated with tibial calcification in our cohort. Importantly, in our study, the use of CT scans to quantify calcification does not allow for the distinction between intimal and medial calcification. It is possible, if not likely, that much of the tibial artery calcification seen on CT scans is located in the media and the relevant risk factors may reflect this.
In patients with PAD, calcification of the tibial vessels predicts amputation better than cardiovascular risk factors and the ABI combined. 15 Also, tibial artery calcification is associated with diabetic foot ulcers in a manner that is independent of the amount of atherosclerosis. 16 Recent data suggest that extensive calcification hampers efforts to deliver therapeutic agents using drug-eluting balloons. 17 Interestingly, we found that age, male sex, tobacco use, and diabetes maintained their association with tibial calcium even after adjusting for presence of PAD. This may reflect the more medial localization of calcification in the tibial vessels or the relatively higher proportion of non-atheromatous lesions recently reported by O’Neill et al. 18 It also is possible that atherosclerosis and calcification share overlapping risk factors. This would support the concept that tibial calcification independently contributes to limb ischemia and poor outcomes; however, the mechanisms behind these associations have not been identified.
Our finding of lower serum calcium levels in patients with increased TAC scores is intriguing. The factors that control serum calcium levels are complicated and include intestine-, bone-, and renal-based mechanisms. In patients on dialysis, increased serum calcium levels are particularly important as they are associated with the presence and 1-year progression of CAC. 19 Serum factors, including calcium, phosphorous, osteoprotegerin, and increased inflammatory markers, have also been associated with coronary calcium presence and progression. 20 Similar associations also have been identified in patients with near normal renal function. 21 Serum calcium levels were also positively associated with aortic calcification in elderly women. 22 Our cohort involves patients both with and without diabetes and they were younger. We did not measure vitamin D, parathyroid hormone, FGF-23 levels or other markers of calcium homeostasis as this was not the original intent of our study. Future efforts will focus on these assessments. Serum calcium values were not adjusted for albumin levels and thus the inverse association with tibial calcification may reflect nutritional status. It is also possible that some patients with eGFR > 30 may have had significant renal disease with altered mineral metabolism.
The study has several limitations in that it is cross-sectional and therefore we are unable to perform any predictive analyses using either the TAC score, risk or biochemical factors. The study was performed in a mixed population of patients with some having extensive vascular disease and others being asymptomatic. Most patients with high calcification scores were recruited from a tertiary referral practice. Such patients with symptomatic PAD may have been under intensive risk factor modification regimens and this may have affected our results.
In summary, we have found that tibial artery calcification is associated with traditional cardiovascular risk factors including diabetes and tobacco use but not with hypertension, hyperlipidemia, or BMI. The associations are not significantly diminished by PAD. Biochemical factors including cholesterol, triglycerides, and CRP are not associated but serum calcium is lower in patients with tibial calcification. These findings may help to develop a better understanding of the pathologic mechanisms involved in calcification of tibial arteries and guide efforts aimed at treating patients with PAD.
Footnotes
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This research was funded by grants from the NIH R21DK067368 and NIH R01HL105641 to RG. AM was supported through NIH 2T32DK007061.
