Abstract
Angiotensin converting enzyme inhibitors (ACEIs) or angiotensin receptor blockers (ARBs) are recommended for secondary prevention in peripheral artery disease, but their effectiveness in patients with critical limb ischemia (CLI) is uncertain. We reviewed 464 patients with CLI who underwent diagnostic angiography or endovascular intervention from 2006–2013 at a multidisciplinary vascular center. ACEI or ARB use was assessed at the time of angiography. Major adverse cardiovascular events (MACE), mortality, and major adverse limb events (MALE) were assessed during three-year follow-up. Propensity weighting was used to adjust for baseline differences between patients taking and not taking ACEIs or ARBs. ACEIs or ARBs were prescribed to 269 (58%) patients. Patients prescribed ACEIs or ARBs had more baseline comorbidities including diabetes and hypertension (p<0.05). Patients prescribed ACEIs or ARBs had lower three-year unadjusted rates of MACE (40% versus 47%) and mortality (33% versus 43%). After propensity weighting, ACEI or ARB use was associated with significantly lower rates of MACE (hazard ratio [HR] 0.76, 95% confidence interval [CI] 0.58–0.99, p=0.04) and overall mortality (HR 0.71, 95% CI 0.53–0.95, p=0.02). There was no significant association between ACEI or ARB use and MALE (HR 0.97, 95% CI 0.69–1.35, p=0.2) or major amputation (HR 0.74, 95% CI 0.47–1.18, p=0.1). ACEI/ARB use is associated with lower MACE and mortality in patients with CLI, but there was no effect on limb-related outcomes.
Keywords
Introduction
Peripheral artery disease (PAD) affects more than 202 million people worldwide and has increased in prevalence by an estimated 24% in the last decade.1,2 Critical limb ischemia (CLI), characterized by ischemic rest pain, ulcers, and gangrene due to arterial insufficiency, is the most severe stage of PAD.3–5 Among patients with CLI the mortality rate is 50% within five years after initial presentation and the major amputation rate is 40% within the first six months.3,4 Although CLI represents a small proportion of the overall population of patients with PAD, it accounts for considerable morbidity and mortality.
The beneficial effects of angiotensin-converting enzyme inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) have been well established in patients with coronary artery disease as well as those at high cardiovascular risk.6–11 The Heart Outcomes Prevention Evaluation (HOPE) Study found that treatment with the ACEI ramipril was associated with a reduced risk of vascular death, myocardial infarction (MI), and stroke in a broad range of patients who were at high risk for cardiovascular events, including those with PAD. 6 More recently, ACEIs have been shown to improve walking times in patients with intermittent claudication 12 and increasing evidence has supported the role of ACEIs in inhibiting the atherosclerotic process as well as improving vascular endothelial function.9,13–15
Practice guidelines for the management of patients with PAD indicate ACEIs or ARBs as a Class IIa recommendation.5,16 However, these guidelines are based primarily on data from patients with claudication or PAD defined by ankle-brachial index (ABI) <0.9. No prior study has specifically examined the association of ACEIs or ARBs with clinical outcomes among patients with CLI. These patients are at highest cardiovascular risk and may derive significant benefit from more intensive secondary prevention. The goal of this study was to analyze the effects of ACEI or ARB therapy on cardiovascular and limb outcomes in patients with CLI who underwent peripheral angiography. We hypothesized that patients with CLI who were treated with ACEIs or ARBs would have reduced rates of major adverse cardiovascular events (MACE) and major adverse limb events (MALE) during long-term follow-up.
Methods
This was a retrospective study utilizing data from the PAD-University of California, Davis Registry, the details of which have previously been described. 17 The study protocol was approved by the Institutional Review Board at the University of California, Davis Medical Center. All patients in the registry with CLI (N=464) were analyzed. All patients underwent lower extremity angiography or endovascular intervention as part of the cohort inclusion criteria.
Demographic, clinical, laboratory, and procedural data were obtained through pre-procedure clinical notes, admission history, in-patient documentation, and angiographic review. Comorbidities that could affect the decision to prescribe ACEIs or ARBs, including patient history of myocardial infarction, stroke, and coronary artery disease, were also recorded. Medical prescribing patterns were verified by pharmacy prescriptions both pre procedure and during follow-up. The medication prescriptions were obtained from both pharmacy orders and a standardized pre-procedure evaluation that included current medications. All records were reviewed by trained chart abstractors and verified by a board-certified cardiologist.
Patient outcomes were determined based on review of post-procedural clinical visits, as well as Electronic Medical Record (EMR) documentation of subsequent hospitalizations and discharge summaries. MACE was defined as MI, stroke, or death. MI was defined as chest pressure and troponin elevation with infarct as identified by stress imaging or catheterization. Stroke was defined as focal neurologic deficit with imaging evidence of cerebral ischemic or hemorrhagic infarct. Mortality was confirmed with direct chart documentation or the Social Security Death Index. MALE were defined as major lower extremity limb amputation, thrombolysis, or surgical bypass. CLI was classified as Rutherford category 4–6 disease (ischemic rest pain, minor tissue loss, or major tissue loss, respectively).
Outcomes
The primary outcome was the occurrence of MACE during three years of follow-up. Secondary outcomes included MALE, the components of MACE and MALE, and death or major amputation.
Statistical analysis
Means with standard deviations were used to describe continuous variables and frequencies and percentages to describe categorical variables. Continuous variables were compared using the Wilcoxon rank sum test and categorical values using chi-squared or Fisher’s exact tests. Propensity scores were developed to adjust for covariates that may influence the decision to prescribe ACEIs or ARBs.18,19 Covariates in the model included age, gender, race/ethnicity (Caucasian, Hispanic, African American, Asian), history of diabetes, congestive heart failure, coronary artery disease, prior myocardial infarction, hypertension, stroke, carotid artery stenosis, chronic obstructive pulmonary disease, prior major amputation, glomerular filtration rate, left ventricular ejection fraction (in 5% increments from ⩽10% to ⩾65%), smoking (never smokers, prior smokers, and current smokers at the time of the procedure), prescription of concomitant medications (including beta blockers, statins, or clopidogrel), and year of procedure. Diagnostic tests to determine balance of the covariates after inverse probability of treatment weighting (IPTW) included the standardized difference before and after weighting and visual inspection of a kernel density plot to verify propensity score overlap between groups. Unadjusted survival curves were estimated using the Kaplan–Meier method, and adjusted survival curves were estimated using IPTW weighting. 20 All analyses were performed using STATA Version 13.1 (STATA Corporation, College Station, Texas). Hazard ratios are given with 95% confidence intervals. For all tests, a p-value <0.05 was considered significant.
Results
During the study period, 464 patients with CLI underwent lower extremity angiography. A total of 269 (58%) patients were prescribed an ACEI or ARB. The baseline characteristics of the two groups are summarized in Table 1. Patients prescribed an ACEI/ARB were of similar age and gender to those not prescribed an ACEI/ARB, but were more likely to have a history of diabetes (69% versus 55%, p=0.002), hypertension (95% versus 72%, p<0.001), or coronary artery disease (55% versus 46%, p=0.05). The baseline glomerular filtration rates were similar between groups (61 versus 62 ml/min, p=0.9). Patients prescribed an ACEI/ARB had similar rates of prescription of aspirin or clopidogrel to those not prescribed an ACEI/ARB, but were more likely to be prescribed statins (73% versus 52%, p<0.001) or beta blockers (58% versus 48%, p=0.03). The baseline ABIs were similar between the two groups (0.5±0.3 versus 0.5±0.2), and the majority of patients in both groups presented with Rutherford category 5 symptoms.
Baseline characteristics of patients with critical limb ischemia.
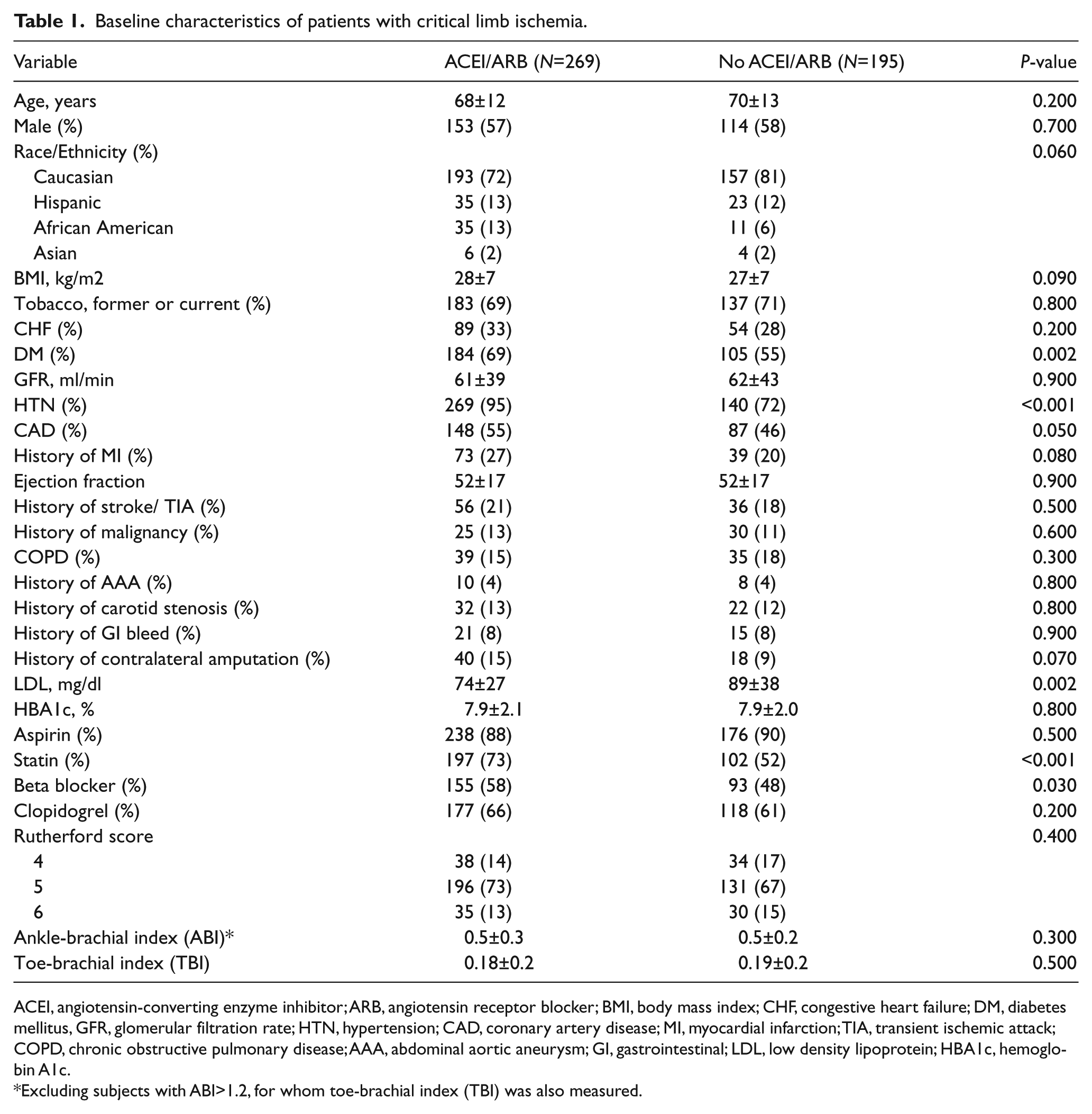
ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; BMI, body mass index; CHF, congestive heart failure; DM, diabetes mellitus, GFR, glomerular filtration rate; HTN, hypertension; CAD, coronary artery disease; MI, myocardial infarction; TIA, transient ischemic attack; COPD, chronic obstructive pulmonary disease; AAA, abdominal aortic aneurysm; GI, gastrointestinal; LDL, low density lipoprotein; HBA1c, hemoglobin A1c.
Excluding subjects with ABI>1.2, for whom toe-brachial index (TBI) was also measured.
The median follow up was two and a half years, with 79% of patients having complete three-year follow-up. At three years post procedure, the unadjusted rates of MACE (40% versus 47%) and overall mortality (33% versus 43%) were lower among patients prescribed an ACEI/ARB, despite a greater prevalence of cardiovascular comorbidities at baseline (Table 2). The rates of MALE and major amputation also trended lower, but were not statistically significant in unadjusted analyses.
Three-year outcome rates and unadjusted and adjusted hazard ratios.
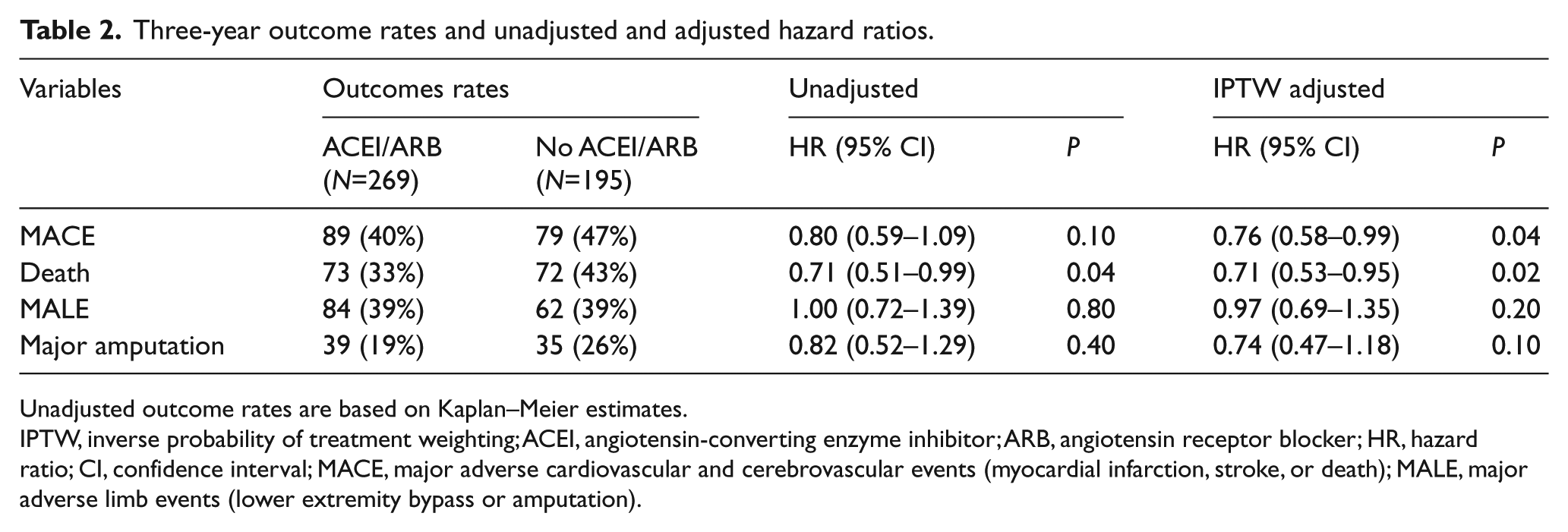
Unadjusted outcome rates are based on Kaplan–Meier estimates.
IPTW, inverse probability of treatment weighting; ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; HR, hazard ratio; CI, confidence interval; MACE, major adverse cardiovascular and cerebrovascular events (myocardial infarction, stroke, or death); MALE, major adverse limb events (lower extremity bypass or amputation).
Propensity scores were developed to account for baseline differences in patients that may influence the decision to prescribe an ACEI/ARB. Visual inspection of the propensity scores for each group demonstrated good overlap between groups prior to weighting (Figure 1). After inverse probability of treatment weighting, ACEI/ARB use was associated with significantly decreased MACE (adjusted hazard ratio [HR] 0.76, 95% confidence interval [CI] 0.58–0.99) and overall mortality (adjusted HR 0.71, 95% CI 0.53–0.95, Figure 2). In comparison, ACEI/ARB use was not associated with significantly decreased rates of MALE (adjusted HR 0.97, 95% CI 0.69–1.35), major amputation (adjusted HR 0.74, 95% CI 0.47–1.18), or amputation-free survival (adjusted HR 0.79, 95% CI 0.59–1.06). Similar point estimates for all outcomes were obtained when the analysis was limited only to ACEI use among patients with CLI.
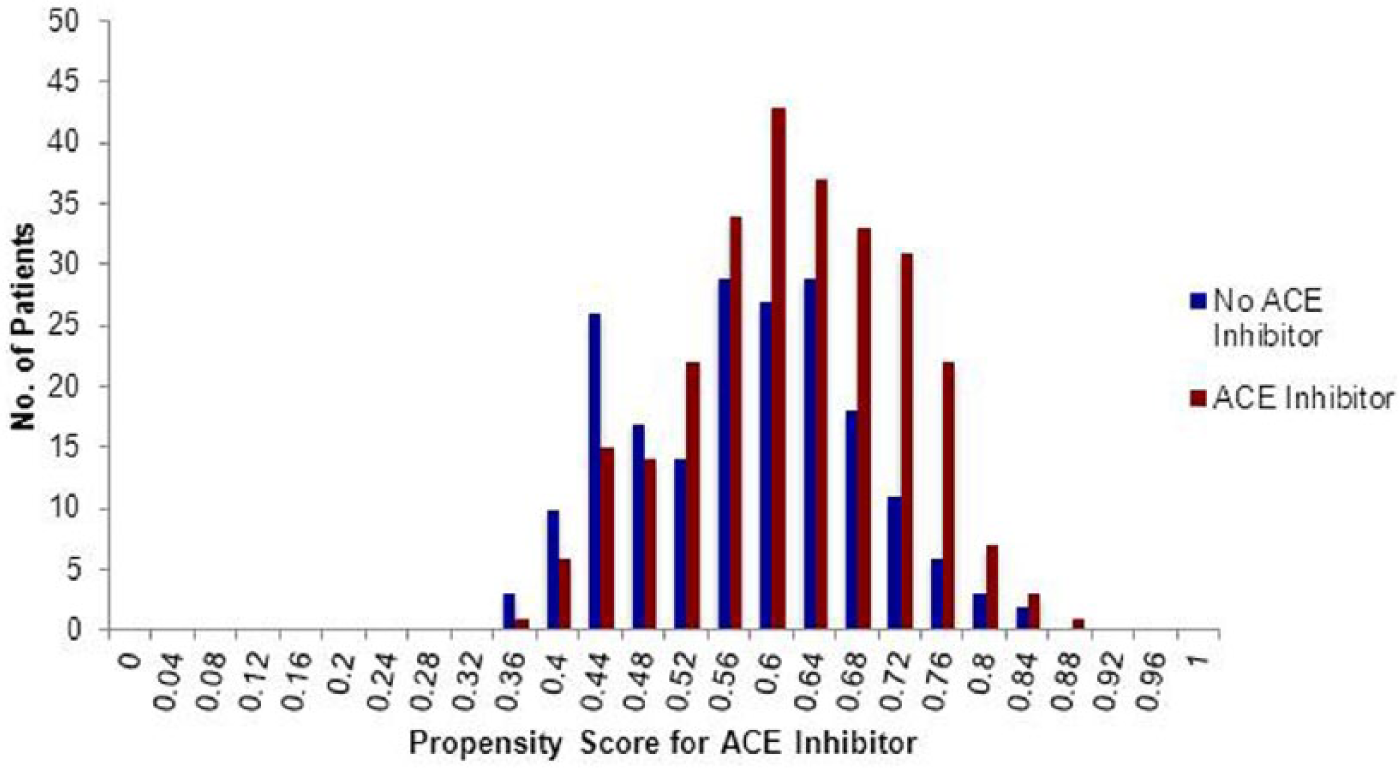
Propensity scores for ACEI/ARB therapy. The propensity score for ACEI/ARB therapy is the probability given baseline covariates that any patient in either group would be prescribed ACEI/ARB therapy.
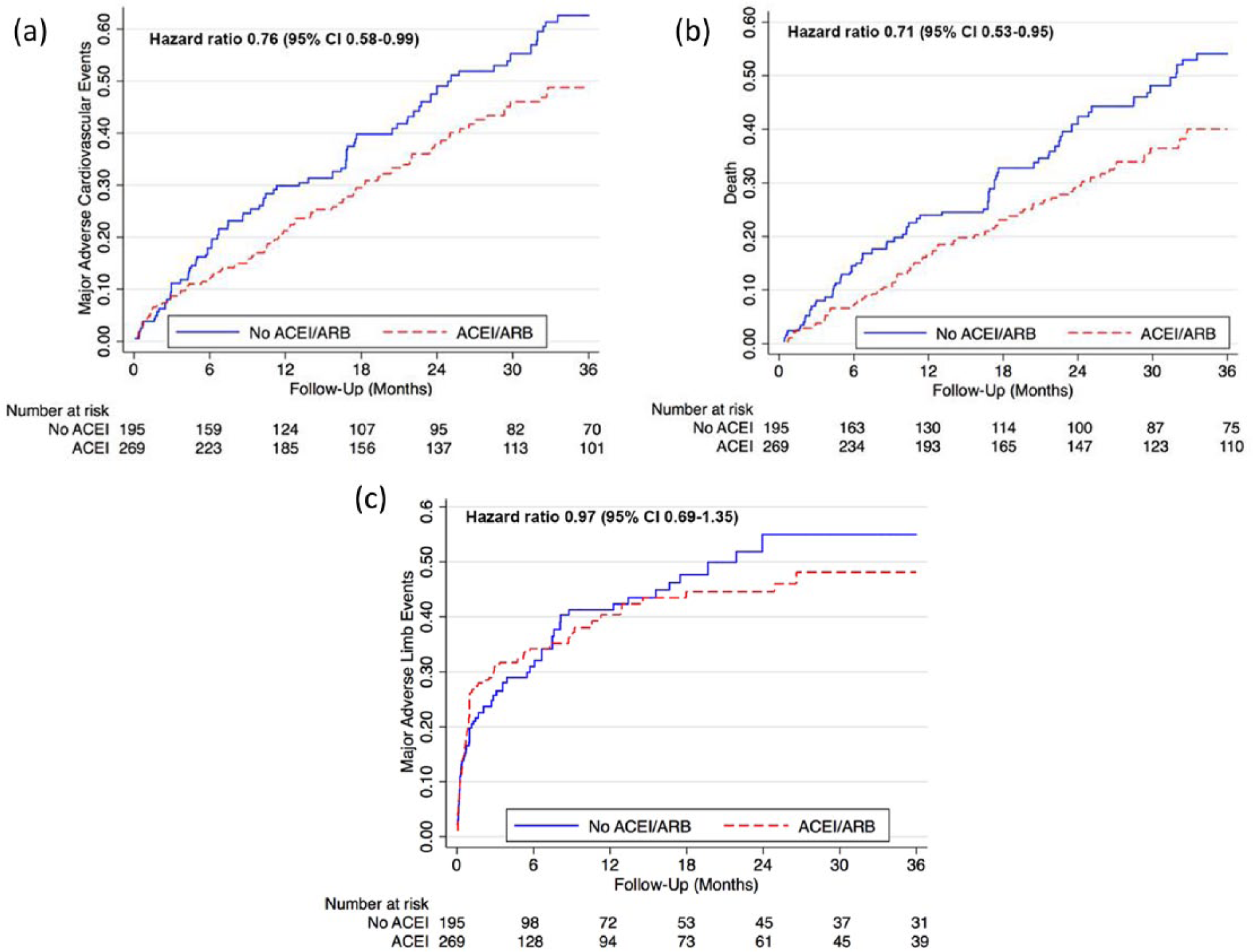
Major adverse cardiovascular events and limb outcomes among patients prescribed ACEIs/ARBs. Cumulative hazard curves to three years post procedure showing the event rates of (a) MACE (myocardial infarction, stroke, or death), (b) death, and (c) MALE (lower extremity bypass or major amputation). All curves are after propensity weighting.
Discussion
Our findings show that patients with CLI who were prescribed ACEIs or ARBs had significantly lower risk of major adverse cardiovascular and cerebrovascular events up to three years after index lower extremity angiogram. ACEI/ARB use was associated with a 24% reduction in MACE and a 29% reduction in overall mortality. Interestingly, there was no significant benefit of ACEI/ARB use on adverse limb events, including major amputation. We also found that patients who were treated with an ACEI/ARB had significantly more baseline comorbidities, including diabetes mellitus and hypertension (HTN), than patients not prescribed an ACEI/ARB. These findings suggest that use of an ACEI/ARB could significantly improve long-term health outcomes for patients with CLI.
Patients with PAD have a 10 to 15 times greater risk of cardiovascular-related death compared to those without PAD. 21 Multiple studies have also shown that PAD is associated with higher cardiovascular event rates than patients with coronary artery disease (CAD), and that patients with CLI have the highest rates of adverse cardiovascular outcomes.3,22–26 Given the known cardiovascular benefits of ACEIs/ARBs, these medications have been studied in multiple cohorts of patients with PAD. With a median follow-up of eight years, Feringa et al. 8 found that ACEI use was associated with a 20% reduction in risk of mortality among patients with PAD defined by ABI <0.9. As a follow-up investigation to the HOPE study, Östergren and colleagues 27 studied the impact of ramipril on specific PAD populations with varying ABIs. They reported that ramipril reduced risk of MACE in all groups of patients with symptomatic as well as asymptomatic PAD regardless of ABI. Among patients with symptomatic PAD, the MACE rate of those treated with ramipril versus a placebo was 20% versus 26% during a mean follow-up of four and a half years. 27 The ONTARGET study randomized patients at high vascular risk (including approximately 13% patients with PAD) to telmisartan, ramipril, or both, and found similar outcomes with each therapeutic strategy. 28 Our results contribute to the existing literature by studying outcomes specifically in patients with CLI, often considered the ‘end stage’ of PAD. We report three-year rates of MACE in the ACEI/ARB and placebo groups to be 40% versus 46%, respectively, which is approximately double the adverse event rates found by Östergren et al. 27 This finding further emphasizes the point that patients with CLI have more advanced disease as well as a higher risk for adverse cardiovascular events than the general PAD population. While the event rates in CLI are high, our report of a 24% reduction in MACE and a 29% reduction in mortality is similar to the risk reduction values reported by these aforementioned studies. This highlights the value of ACEI/ARB therapy in patients with various clinical manifestations and severities of lower extremity PAD.
In comparison to the significant reduction in MACE and overall mortality observed with ACEI/ARB use, we did not find an association between ACEI/ARB use and limb-related outcomes of MALE or major amputation. This suggests that the primary benefit of ACEI/ARB use on cardiovascular risk and mortality in CLI patients may stem from mechanisms promoting perfusion of other vascular beds. Multiple molecular mechanisms, including angiogenesis,29–31 vasodilation,32,33 and atherosclerotic plaque stabilization,13,15,34 have been suggested as contributors to the hemodynamic improvements observed in PAD patients taking an ACEI. For example, ACEI treatment has been associated with the development of coronary collateral circulation in patients with CAD 35 as well as increased levels of angiogenesis factors in PAD patients. In a recent study analyzing the potential vascular mechanisms of ramipril, Ahimastos et al. 36 found that among patients with intermittent claudication, ramipril was associated with a 64% increase in plasma levels of fibroblast growth factor-2 (FGF-2) and 38% increase in vascular endothelial growth factor-A (VEGF-A), both integral factors of angiogenesis. However, for late-stage CLI patients in our cohort with severe lower-limb tissue loss and ischemia, it is possible that ACEIs/ARBs are unable to significantly alter an already advanced stage of disease.
While ACEIs have not been shown to definitively affect limb-related outcomes in patients with CLI, inhibition of the renin-angiotensin system (RAS) has been shown to benefit walking ability in patients with intermittent claudication.12,37–41 A randomized controlled trial found that, relative to placebo, ramipril was associated with a 75-second (77%) increase in pain-free walking time and a 255-second (123%) increase in maximum walking time. 12 Proposed mechanisms include ACEI-mediated adaptations in skeletal muscles and improved capillary flow.12,36 Given the increasing evidence of functional capacity improvements in claudicants taking an ACEI, future studies should explore whether ACEI/ARB therapy in the general PAD population could play a role in inhibiting rates of disease progression to CLI.
Our data on the rates of ACEI/ARB use in CLI extend previous findings on this subject. Chung et al. 42 found that among patients with CLI the rate of ACEI use was 56%, which is similar to the 58% rate we found in our cohort. Among patients with claudication or CLI undergoing lower extremity revascularization, Slovut et al. recently reported a 48% rate of ACEI use at both admission and discharge. 43 In a study analyzing data from a general PAD population, Pande et al. 44 reported trends of secondary preventive therapies using the National Health and Nutrition Examination Survey and found a 21% rate of ACEI use. This substantial difference may be due in part to our inclusion of a higher-risk group of CLI patients who often present with more severe PAD symptoms and comorbidities, such as diabetes and HTN. Increased prevalence of baseline comorbidities along with evidence of increased use of statin medications (p<0.001) and beta blockers (p=0.03) among patients who were prescribed an ACEI in our study may imply that use of guideline-recommended therapies is likely driven by other cardiovascular comorbidities rather than by a diagnosis of PAD. While most other studies on ACEI use in PAD populations did not study ARBs, we included patients who were treated with an ACEI or ARB to obtain comprehensive data on all patients who received therapy that targeted the renin-angiotensin aldosterone system. In our cohort, 269 of 464 patients with CLI were prescribed an ACEI or ARB. Of the 269 patients, ACEIs were prescribed for 206 and ARBs for 65 patients (two patients were prescribed both ACEIs and ARBs). A meta-analysis of randomized comparative trials between ACEIs and ARBs found no difference between the two medications in terms of risk reduction of MI, cardiovascular mortality, or total mortality. 45
Several previous studies in the PAD population have documented undertreatment44,46,47 with ACEIs, statins, and antiplatelet agents, all medications that have demonstrated significant benefits in PAD patients.6,48–50 While attention to the effective use of all forms of secondary prevention is needed, ACEIs/ARBs tend to be relatively underutilized compared to other guideline-recommended therapies.42,51 Results from an analysis of data from the Reduction of Atherothrombosis for Continued Health (REACH) Registry indicate that patients with PAD only did not receive ACEI therapy as frequently as individuals with PAD and CAD/cerebrovascular disease (33% versus 49.4%, p<0.001) despite having comparable cardiovascular risk. 52 Notably, use of claudication medications was significantly higher in the PAD group compared to the polyvascular PAD group (32% versus 27%, p<0.001), indicating that symptom relief rather than risk factor modification was a probable focus of PAD therapy. Increased awareness of and education on the benefits of secondary prevention and ACEI/ARB therapy among patients with PAD are necessary. Particularly for patients with CLI, aggressive risk factor modification can provide meaningful cardiovascular improvements and decrease overall mortality.
Limitations
This study has several potential limitations. First, this was an observational cohort study and is therefore subject to possible residual confounding. We used propensity score weighting to minimize confounding based on measured covariates, although this cannot account for unmeasured confounding variables. Second, this was a retrospective analysis from a single center; therefore patterns of care and disease may differ at other clinical sites. Third, the dose of ACEI/ARB was not assessed in this study. Fourth, although we assessed the presence of ACEI/ARB use by conducting thorough reviews of prescription records and clinical chart notes, we could not fully assess the reasons for not prescribing an ACEI/ARB, which could include a prior diagnosis of renal artery stenosis or hyperkalemia. Last, our study did not include patients undergoing primary amputation or bypass without having undergone prior angiography.
Conclusions
Among patients with CLI, prescription of ACEIs or ARBs is associated with reduced rates of MACE and overall mortality. There was no association between ACEI/ARB use and limb-related outcomes. Increased attention to secondary prevention measures in patients with CLI could significantly improve long-term outcomes.
Footnotes
Declaration of conflicting interest
John Laird reports being a consultant for Boston Scientific, Covidien, Abbott, Bard, and Medtronic. He receives research support from WL Gore. Ehrin Armstrong reports being a consultant for Abbott Vascular and Spectranetics. All other authors report no conflicts of interest related to this study.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
