Abstract
Proton pump inhibitors (PPIs) are commonly used drugs for the treatment of gastric reflux. Recent retrospective cohorts and large database studies have raised concern that the use of PPIs is associated with increased cardiovascular (CV) risk. However, there is no prospective clinical study evaluating whether the use of PPIs directly causes CV harm. We conducted a controlled, open-label, cross-over pilot study among 21 adults aged 18 and older who are healthy (
Keywords
Introduction
The advent of proton pump inhibitors (PPIs) as antagonists of gastric acid secretion to treat a number of gastroesophageal disorders has radically transformed clinical practice in the management of several acid-related gastric disorders including gastroesophageal reflux disease (GERD), Barrett’s esophagus and
However, it is now evident that CV risk associated with PPIs is not restricted to those taking clopidogrel. For example, the Platelet Inhibition and Patient Outcomes (PLATO) study reported that PPIs promote risk amongst those taking ticagrelor, 16 a drug which does not require hepatic activation via the cytochrome p450 pathway. Intriguingly, recent meta-analyses have indicated that every member of the PPIs, including those that do not interact with CYP2C19, increase CV risk independent of clopidogrel use.14,17 One possible explanation for this increase in CV risk is an off-target effect of the PPIs that directly alters cardiovascular homeostasis. Recently, we published our finding that the PPIs directly inhibit the cardiovascular enzyme dimethylarginine dimethylaminohydrolase (DDAH), an enzyme that metabolically regulates the plasma concentration of asymmetric dimethylarginine (ADMA), an endogenous and competitive inhibitor of all the known nitric oxide (NO) synthase (NOS) enzymes. 18 Several clinical studies have indicated that elevated plasma ADMA is a CV risk factor.18–24 However, no clinical study has ever been conducted in humans to assess whether the use of PPIs directly alters plasma ADMA, and/or causes impaired vascular function.
To determine the effect of PPI use on vascular function and plasma levels of ADMA, we conducted a prospective 13-week, placebo-controlled, cross-over pilot study among healthy adults and patients with cardiovascular disease (CVD). This study also sought to correlate the impact of PPI use on routinely used measures of vascular function and plasma levels of the risk factor ADMA.
Methods
This clinical trial evaluating the effect of PPI use on vascular function was an open-label, placebo-controlled, non-randomized, cross-over study conducted among ambulatory subjects. Vascular function measurements were carried out at the Stanford University Medical Center. All study participants provided written informed consent, and the Stanford University Institutional Review Board (IRB) approved the protocol (IRB # 00000348).
Study subjects
The study subjects were community-dwelling adults aged 18 years and older who either had no pre-existing condition (
Baseline characteristics of study participants.
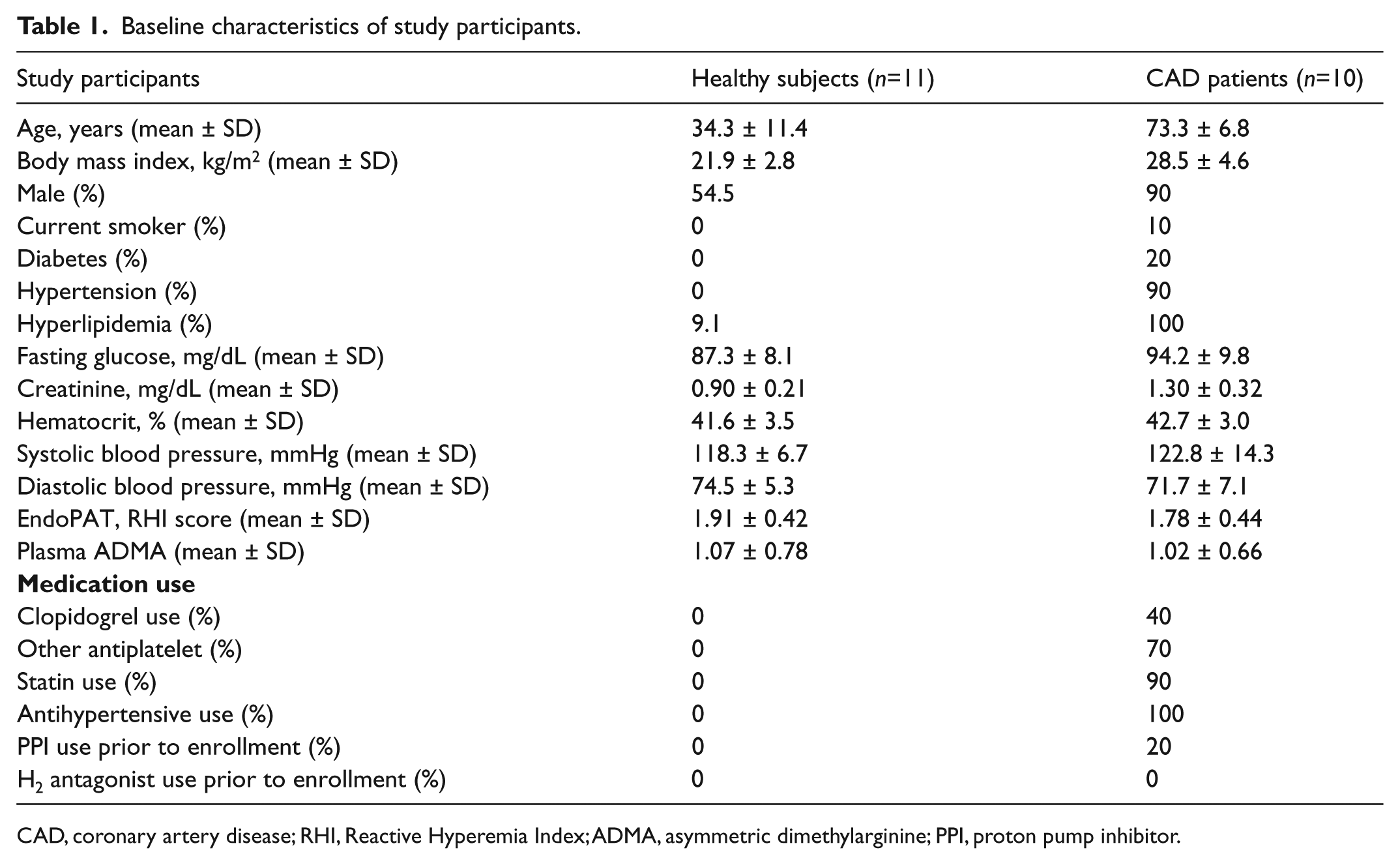
CAD, coronary artery disease; RHI, Reactive Hyperemia Index; ADMA, asymmetric dimethylarginine; PPI, proton pump inhibitor.
Treatment regimen
Following a 2-week run-in period, subjects were randomized to receive either PPI or placebo. The PPI lansoprazole (Prevacid; Takeda Pharmaceuticals, Inc.) was provided to participants in unit-dose containers, containing 14 counts of 15 mg tablets. Four containers (sufficient for a 4-week supply) were provided to each subject and the participants were instructed to self-administer two Prevacid tablets daily 30 minutes before a meal. The control arm of the study received 28 placebo tablets (each tablet containing 71% calcium carbonate, 25% microcrystalline cellulose, 2% stearic acid, 1% magnesium stearate and 1% starch) and participants were instructed to take a tablet once daily after a meal. As described below, all subjects crossed over to receive both treatments during the course of the study.
Study protocol
Volunteers were screened for eligibility (
We performed a run-in period of 2 weeks in order to acclimatize the study subjects to the study protocol, and also to evaluate the test-retest reliability of the EndoPAT measurements of vascular function. Baseline vascular function was measured and blood was collected prior to assigning the participants to PPI (
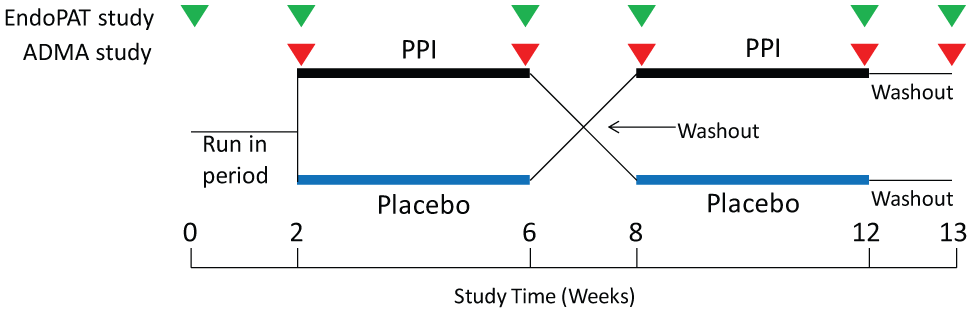
A pilot clinical study designed to evaluate the effect of a proton pump inhibitor (PPI) on vascular function.
At the end of the first 4 weeks, vascular function was assessed and a fasting blood sample was collected for measurement of plasma ADMA. Subsequently, a ‘washout’ period of 2 weeks preceded cross-over to the other treatment arm for the ensuing 4 weeks, after which period another fasting blood draw and vascular measurement was made. While our preclinical data suggested that the effect of the PPIs may no longer be evident 1 week after cessation of therapy (data not shown), we chose to include a 2-week washout before cross-over, to ensure that there would be no confounding carry-over effect to the second treatment window. Finally, a 1-week washout period, followed by collection of blood and vascular function measurements, completed the study (Figure 1).
Measurements
EndoPAT study
Vascular function was assessed by an operator unaware of the treatment groups using the EndoPAT technique: a non-invasive procedure commonly used in research settings to assess the effects of drugs and nutrition on vascular function. In brief, the EndoPAT device (Itamar Medical Inc., Franklin, MA, USA), equipped with standard blood pressure cuff and finger-probes, was set up in a dim, temperature-controlled room. Subsequently, one arm of each study participant (laid in a supine position) was occluded using the cuff while the contralateral arm was used as a control and the index fingers were attached to the sensor probes for assessment of pulse amplitude. Next, the EndoPAT 2000 software package associated with the device was launched to record flow-mediated vasodilation from the occluded and control arms (the ‘reactive hyperemia’ period). The Reactive Hyperemia Index (RHI), which represents the ratio of the post-to-pre-occlusion signal in the occluded side (normalized to the control side), was calculated in real time for assessment of vascular function in response to the study intervention. A normal RHI score is 1.67 and a finding below this value indicates vascular dysfunction: an independent predictor of cardiovascular morbidity and mortality.26–28 This vascular dysfunction may be related to an impairment of endothelium-dependent and/or endothelium-independent vasodilation.
Plasma ADMA measurement
Approximately 7 mL of blood was collected into EDTA-coated tubes (Fisher Scientific, Pittsburgh, PA, USA) following each study visit and was briefly kept on ice prior to centrifugation at 3300 rpm for 20 minutes to separate plasma. The plasma samples were transferred into sterile tubes and stored at −80° until assaying for ADMA: a CV risk factor recently implicated in PPI-mediated vascular dysfunction.25,29 The concentration of ADMA was measured in a blinded fashion using an ELISA assay (DLD Diagnostika, Hamburg, Germany) as previously described. 30 In brief, the samples were first acylated and then an antibody was used to capture the acylated ADMA in a microplate assay overnight. The next day, wash buffer was used to remove unbound analytes from the reaction plate. Subsequently, enzyme conjugate and TMB substrate were used to complete the reaction. Finally, stop buffer was added to terminate the reaction; absorbance (450 nm) was measured using a plate reader and the ADMA concentration in the plasma samples was estimated from a standard curve.
Statistical analysis
Baseline patient characteristics were compared using a
Results
Figure 1 illustrates the design of the study evaluating the effect of Prevacid on vascular function, including study visits for EndoPAT measurement and blood draw for ADMA level. Baseline characteristics of the subjects who participated in the study are shown in Table 1. No adverse events were reported in this study.
Reproducibility of the EndoPAT score during run-in period
As described above, vascular function was assessed using the EndoPAT technique. In order to compare the change in RHI score with each treatment during the course of the study and between the groups, each study participant underwent serial measurements at each of the pre-scheduled visits, including: two baseline measurements (at 2 weeks and 1 week prior to the start of the intervention), at the end of the first 4-week study block, at the end of the first washout period, at the end of the second 4-week block, and after the second washout period. Evaluation of test-retest reliability of the run-in period EndoPAT measurements (week 0 and week 2; Figure 2) yielded a correlation coefficient of 0.47 (
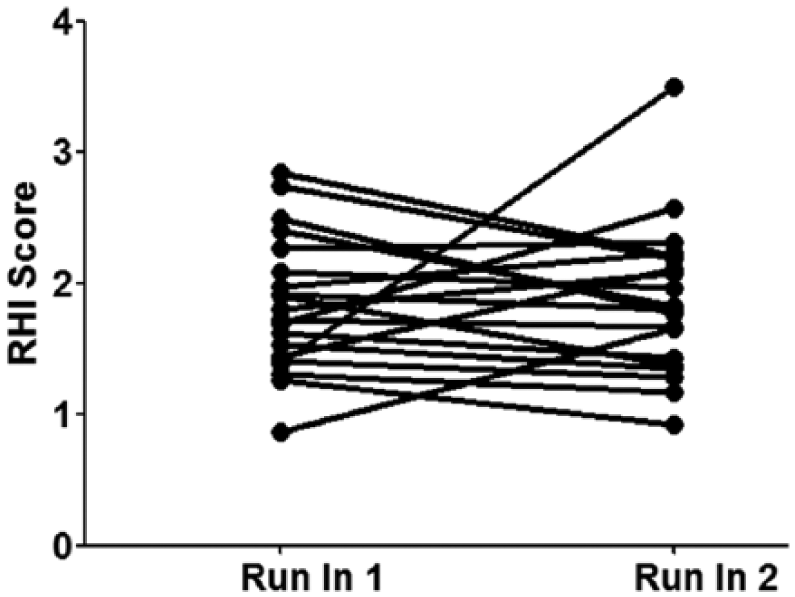
Evaluation of test-retest variability of the EndoPAT technique. Study participants were assessed for vascular function using the Reactive Hyperemia Index (RHI) score (
Effect of Prevacid on vascular function: EndoPAT score
Comparison of the change in EndoPAT score by treatment (Prevacid vs placebo) or by health profile of the study participants (healthy vs CVD patients) did not show a statistically significant difference in RHI score after treatment (Figure 3;
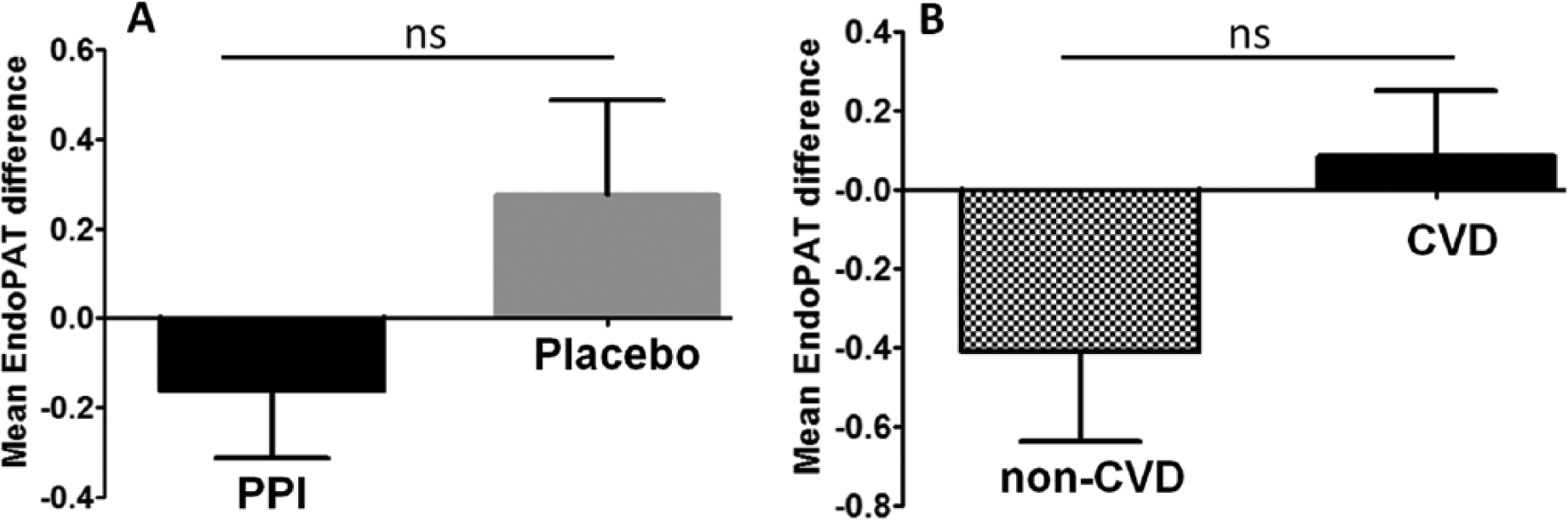
Comparison of the vascular effect of the proton pump inhibitor (PPI) Prevacid by EndoPAT. (A) Mean plus SEM values of the difference in the Reactive Hyperemia Index (RHI) score between the PPI and placebo group. (B) The effect of PPI treatment on vascular function is compared by study participants between healthy subjects (‘non-CVD’) and subjects with a history of cardiovascular disease (‘CVD’). (ns, not significant at
Effect of Prevacid on vascular function: Plasma ADMA concentration
Interestingly, our study evaluating the change in plasma ADMA concentration after treatment indicated that there was a non-significant trend towards a higher absolute change in ADMA when the participant was on PPI compared to when on placebo (0.12 µM vs 0.09 µM increase,
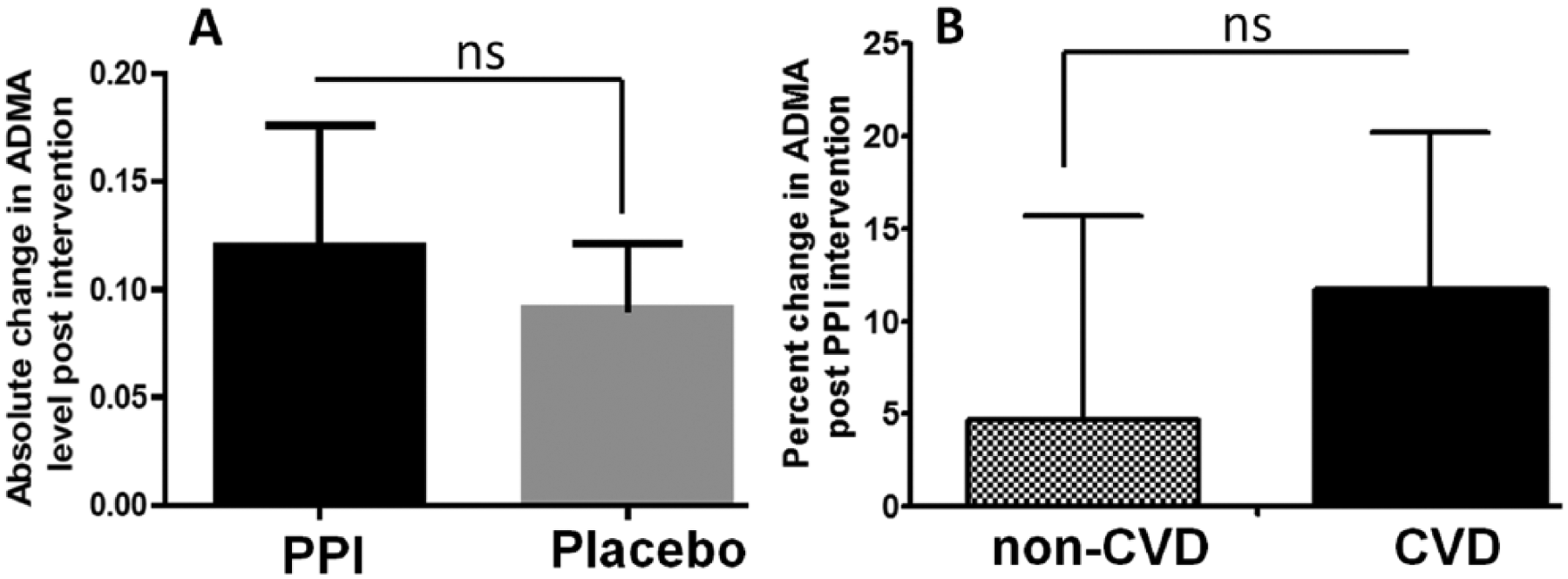
Effect of the proton pump inhibitor (PPI) Prevacid on plasma concentration of asymmetric dimethylarginine (ADMA). (A) Shows a trend towards an increase in absolute change in ADMA in plasma collected when a study participant is on PPI in comparison to placebo. (B) Shows subgroup analysis of plasma ADMA within the study groups comparing healthy participants (‘non-CVD’) with participants who had history of cardiovascular disease (‘CVD’). (ns, not significant at
Effect of Prevacid on vascular function: EndoPAT score and ADMA data correlation
Overall, our study demonstrated that there was a marginal inverse correlation (
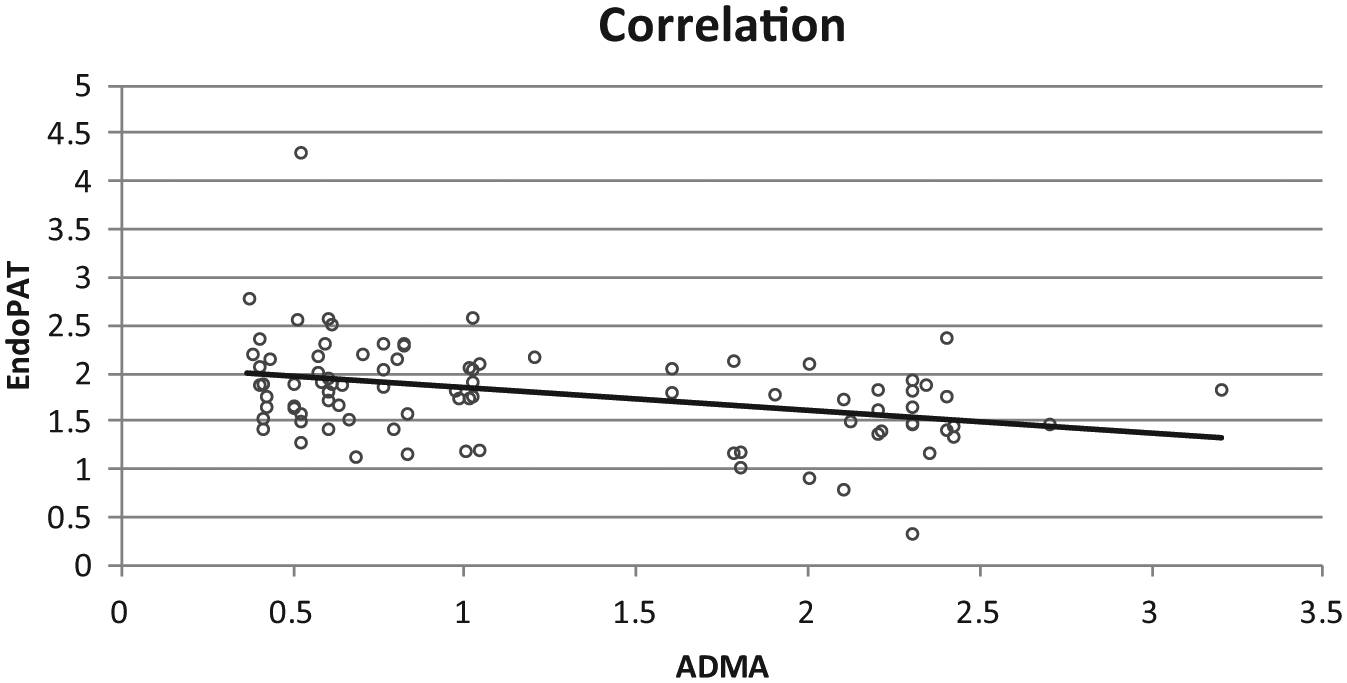
Correlation of EndoPAT score and plasma ADMA concentration. The
Discussion
PPIs have proven to be effective alternatives to the histamine (H2)-receptor antagonists in the treatment of several gastric acid-related disorders. In addition to their efficacy, the PPIs are well tolerated with minimal side effects when used intermittently. However, recent meta-analyses studies of acute coronary syndrome (ACS) patients have raised concern that the use of PPIs might be associated with more serious outcomes including the risk of myocardial infarction and rehospitalization.11–14,17 These and other studies 31 (and the references therein) raised the possibility that PPIs might reduce the cardiovascular benefit of concurrently used antiplatelet agents, such as clopidogrel, potentially due to their interference with a common metabolic pathway required for the activation of clopidogrel from its prodrug form, resulting in reduced bioavailability of the antiplatelet agent.
However, emerging evidence argues that the increased CV risk associated with PPI use is unlikely to be solely due to changes in gastric pH or interference with hepatic activation of clopidogrel. These conclusions are supported by the facts that: (1) a similar reduction in intragastric pH achieved with the use of H2-receptor antagonists does not increase CV risk 17 and (2) PPIs increase CV risk even when co-administered with antiplatelet agents other than clopidogrel, such as ticagrelor, that do not require hepatic activation. 16 These data suggest that PPIs might have an independent effect on the cardiovascular system, which is neither mediated via a change in drug absorption (due to change in gastric pH) nor a drug–drug interaction (due to interference with the cytochrome p450 pathway).
Recently, we reported our finding that the PPIs, as a class, directly inhibit the cardiovascular enzyme DDAH and elevate the CV risk factor ADMA in biochemical, cellular, ex vivo and preclinical studies. 25 ADMA poses CV risk due to its endogenous inhibition of endothelial nitric oxide synthase (eNOS) resulting in limited production of the vasodilator molecule nitric oxide (NO). Several clinical studies have demonstrated that CV risk factors such as atherosclerosis, hypertension, diabetes, insulin resistance and chronic kidney disease (CKD) are associated with impaired DDAH activity and elevated levels of plasma ADMA.32–36 However, whether the PPIs increase plasma ADMA in clinical settings has not been addressed. Meanwhile, our recent data-mining study of electronic health records, gathered from community-dwelling adults visiting tertiary health care institutions, indicates that the use of PPIs, as a class, is associated with an increased odds ratio for CV risk, independent of clopidogrel use, suggesting that PPIs pose CV risk in the general population. 37
In this prospective 13-week, open-label, controlled, cross-over pilot study of PPI use among healthy adults and subjects with cardiovascular disease, we found no significant effect of PPI use on vascular function, but did observe a trend towards a worsening in plasma ADMA levels.
It is possible that our study might have been confounded by several factors. First, the use of PPI, at clinically relevant concentrations, may not cause vascular dysfunction over the short 4-week time course studied in this pilot. It may be that a greater total exposure to PPIs is required to affect change. Indeed, our recent studies25,29 showed that PPIs can elevate levels of ADMA in cultured endothelial cells and in mice in vivo when given at a substantially higher concentration (20–100 µM) than is achieved in clinical settings (2–6 µM),15,38 as in this study. Alternatively, it is possible that the PPIs did impair the activity of endothelial NO synthase, and reduced endothelium-dependent NO generation, without affecting the hyperemic response. It is known that impairments of endothelial NOS activity can be balanced by activation of other vasodilator pathways. 39 Accordingly, vasodilator function can be maintained, at least in the short term.
Our current data suggesting that subjects with CVD may be more sensitive to PPI-induced changes in ADMA (Figure 4B) raise the possibility that vasculopathic patients may have an impaired DDAH reserve, rendering them more susceptible to the deleterious effects of PPIs. Thus, it is possible that these discordances in concentration, subject susceptibility and study duration might account for the observed differences between the preclinical and clinical settings.
Another limitation of our trial was the observed low test-retest reliability of the EndoPAT measurements within study participants, which likely contributed to a significant reduction in the precision of our analysis. Furthermore, the sample size utilized in this pilot study was based on effect sizes and their standard deviations observed in preclinical murine models, given that the effect of PPIs on vascular function had not previously been studied in humans. 25 However, post-hoc power calculations suggest we would have needed to study 88 subjects per group in order to detect a significant difference between treatment groups (with a power of 80% and an alpha of 0.05) given the observed means and standard deviations found in the current analysis.
In addition, we did not control for the amount of dietary nitrate (NO3) and nitrite (NO2) consumed by the study participants, which are both factors known to directly impact systemic NO production and vascular reactivity.40,41 Furthermore, this pilot study was unblinded due to the cost of obtaining placebo tablets of the same size and shape as the Prevacid pills. Given that vascular reactivity is known to be highly sensitive to emotional and cognitive stressors, it is possible that the knowledge of study drug assignment may have influenced the subject’s performance during the EndoPAT testing.
42
These potential confounders, together with the small sample size (
In conclusion, our pilot trial did not detect an adverse effect of PPI use on vascular function over a 4-week exposure. However, our study did suggest that PPI use may be associated with an increase in plasma ADMA, an emerging risk factor for major adverse cardiovascular events. The change in this biomarker appeared to be accentuated amongst subjects with CVD. Future prospective, randomized and blinded clinical trials conducted in a larger cohort over a longer period of follow up will be important to understand the mechanism by which this widely prescribed drug appears to promote CV risk.
Footnotes
Acknowledgements
The authors would like to thank all participants of the study.
Declaration of conflicting interest
YTG and JPC are inventors on patents owned by Stanford University that protect the use of agents that modulate the NOS/DDAH pathway therapeutically. They are also co-founders of Altitude Pharma Inc., a biotech developing products that regulate the NOS/DDAH pathway.
Funding
This pilot study was funded by grants from the Translational Research and Applied Medicine (TRAM) Program in the Department of Medicine at Stanford University [grant number 1144447-121-DHAXB]. This study was also supported in part by grants to JPC from the National Institutes of Health (NIH) [grant numbers 1R01 EY02060901, 1U01 HL100397] and internal funding from the Houston Methodist Research Institute (HMRI). YTG was a recipient of postdoctoral fellowships from the Stanford School of Medicine Dean’s Office [grant number 1049528-149-KAVFB] and the Tobacco-Related Disease Research Program (TRDRP) of the University of California [grant number 20FT-0090]. He is currently supported by the National Heart, Lung, and Blood Institute (NHLBI) [grant number 1K01 HL118683-01] and HMRI [grant number 25150001].
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
