Abstract
This study examined the effects of moderate exercise-induced heat stress (EIHS) on vascular function, central hemodynamic load and indices of coronary perfusion. Vascular-hemodynamic measures were collected in 12 healthy men (aged 22±3 years) pre and post 100 minutes of moderate, intermittent exercise in two randomized conditions: heat stress (HS; wearing firefighter personal protective equipment (PPE)), and no heat stress (NHS; wearing a cooling shirt and equivalent PPE weight). Aortic blood pressure, reflected wave pressure (Pb), systolic (SPTI) and diastolic pressure time-integral (DPTI), and aortic stiffness were assessed before and after each condition. SPTI was significantly greater, and DPTI and Pb were significantly lower for HS-post compared to NHS-post (p<0.05). Pulse wave velocity was not different between conditions. In conclusion, EIHS does not affect aortic stiffness, but increases indices of myocardial work and reduces indices of coronary perfusion which may be related to chronotropic responses to EIHS. The mismatch between oxygen demand and oxygen supply may increase cardiac vulnerability to ischemia during strenuous work in the heat.
Keywords
Introduction
Exercise-induced heat stress (EIHS) is routinely encountered in athletic and occupational settings. Heat stress results from a combination of metabolic heat production and environmental conditions and can be exacerbated by protective clothing that limits evaporative heat dissipation. 1 It has been well documented that EIHS limits exercise and work performance as evidenced by reductions in maximal oxygen consumption and maximal work capacity. 2 EIHS results in pronounced reductions in total peripheral resistance, necessitating an increase in cardiac output to prevent decreases in arterial blood pressure. 3 Cardiac output during high heat load scenarios is primarily achieved through alterations in heart rate (HR) and cardiac inotropic properties3,4 resulting in increased cardiovascular strain (e.g. increased myocardial work).
Understanding the effects of heat stress is particularly important for firefighters as the cardiovascular strain of firefighting may precipitate sudden cardiac death in vulnerable individuals.5–7 In fact, approximately 45% of firefighter line-of-duty deaths each year occur from sudden cardiac events,7,8 accounting for the highest proportionate cardiovascular mortality of any occupational group. 5 The exact mechanisms responsible for firefighters’ apparent increased risk of cardiovascular events remain largely unknown. The effect of EIHS on vascular function is not yet understood and may offer insight into increased cardiovascular strain related to heat stress.
Matching of oxygen demand and perfusion of the myocardium is largely mediated by blood pressure. Increases in systolic pressure increase afterload and myocardial work. Due to physical compression of coronary vessels during systole, the heart is primarily perfused during diastole (~80%) and depends on the duration of diastole and absolute diastolic pressure to establish a pressure gradient.9,10 Thus, reductions in aortic diastolic pressure reduce coronary perfusion. According to wave transmission-reflection theory, the blood pressure waveform is a combination of forward and backward traveling waves. Left ventricular ejection instigates the genesis of a forward traveling pressure wave, 11 the magnitude of which depends primarily on left ventricular contraction and the elastic properties of the aorta. 10 When the forward traveling pressure wave encounters bifurcations or smaller vessels in the peripheral circulation, part of the wave will be reflected back toward the heart. The speed and magnitude of this reflected wave is affected by several hemodynamic factors including changes in arterial stiffness, changes in downstream vasomotor tone, and distance to bifurcations. In healthy arteries, reflected waves typically arrive at the ascending aorta during diastole and act to increase diastolic blood pressure, thereby increasing coronary perfusion pressure. 10 A faster traveling reflected wave (as seen with stiffer arteries) arrives at the ascending aorta earlier, in mid-late systole, augmenting systolic pressure.12,13 This augmented pressure from wave reflections increases afterload, and thus cardiac systolic workload, while simultaneously reducing coronary perfusion pressure owing to a temporal shift in the confluence of pressure waves (i.e. more in systole means less in diastole). In this manner, changes in aortic stiffness and forward and/or backward traveling waves play an important role in determining systolic workload, and thereby oxygen demand, and also coronary perfusion and oxygen supply. 14 The purpose of this study was to explore the effect of moderate EIHS compared to normothermic exercise on aortic stiffness, pressure from wave reflections and noninvasive estimates of myocardial work/coronary perfusion. We hypothesized that moderate EIHS would: 1) increase myocardial work while decreasing myocardial coronary perfusion and 2) increase aortic stiffness and pressure from wave reflections.
Methods
Participants
Twelve healthy, physically-active men (22±3 years, maximal oxygen intake (VO2max): 60.3±4.4 mL∙kg−1∙min−1) in overall good health (BMI 24.6±2.8 kg∙m−2, body fat 15.0±3.7%) from the local college community were recruited to participate in this study. The study was approved by the Institutional Review Board of the college and all participants provided written informed consent and health history prior to testing. Additionally, all participants were non-smokers, received medical clearance to enroll in the study, and had no history of cardiovascular disease.
Study design
Using a repeated measures design, participants completed an alternating exercise/rest protocol under two different, randomized conditions: normothermic exercise (no heat stress (NHS)) and heat stress (HS). Experimental visits were conducted at the same time of day, separated by at least 48 hours, and completed within a two-week period. All study visits were conducted in a thermo-neutral laboratory (21.2±0.9°C; 28.8±12.1% relative humidity).
Conditions
EIHS was induced by having the participant wear structural firefighting personal protective equipment (PPE). The PPE consisted of turnout pants and coat, gloves, thermal hood, helmet (G-Xtreme; Globe Manufacturing Co, LLC, Pittsfield, NH), boots, and self-contained breathing apparatus (SCBA). The total weight of this equipment was 18.7±0.7 kg. For the NHS condition, the participant carried the SCBA and wore a weighted vest equated to the mass of the PPE, matching the metabolic work between conditions. Additionally, to mitigate the rise in core temperature during the NHS condition, participants wore a cooling shirt throughout the protocol (Cool Shirt, Shafer Enterprises, Stockbridge, GA). The Cool Shirt pumped cold water (≈5–6oC) from a cooler through tubing in the shirt, and was worn under the weighted vest. Participants were also cooled by a fan (1500CFM; Air King, West Chester, PA) during the exercise and rest bouts to maximize evaporative cooling. All participants began each study visit in a euhydrated state (urine specific gravity <1.020; Schueco Clinical Refractometer 5711-2020, Tokyo, Japan) and water was consumed during the exercise protocol to maintain this hydration status.
Experimental protocol
Participants completed familiarization sessions prior to experimental visits. During the familiarization sessions, participants walked on a treadmill while wearing PPE in a euhydrated state. Core temperature (Tco) and heart rate (HR) were recorded throughout the session (described below). These sessions were used to allow the participant to become accustomed to walking in PPE, to determine the workload for the experimental conditions, and to determine body water loss during the protocol. The workload was individually adjusted so that the individual was working at 75–80% of their maximum HR during the third exercise bout, which corresponded to approximately 40% of VO2max. Changes in body water were calculated based on nude body weight changes pre/post exercise and were used to determine the amount of total fluid lost during exercise. This volume was then provided during the exercise protocol in both conditions in order to prevent net fluid loss and dehydration.
Participants reported to the laboratory 24 hours prior to each experimental visit to determine hydration status. A urine specific gravity (USG) of ⩽1.020 was used to define euhydration. In the 24 hours preceding the experimental visit, participants obtained a nude body weight and were instructed to drink 30 mL·kg−1 of water (water provided). Participants were also given a telemetric gastrointestinal pill (HQ Inc., Palmetto, FL) used for the assessment of Tco, and were instructed to ingest the pill eight hours prior to testing.
Upon arrival at the laboratory on testing days, nude weight was obtained prior to a baseline rest period. Participants arrived following an overnight fast, and were provided with a standardized meal (protein bar; GNC, Pittsburgh, PA) consisting of 390 kcal (12 g fat, 49 g carbohydrate, 30 g protein) to ingest during the baseline rest period. Following a 20-minute supine rest period, baseline vascular measures were obtained. Participants then donned the condition-appropriate gear. A 100-minute alternating exercise/rest protocol consisted of three 20-minute bouts of treadmill walking with a 20-minute rest between each bout. This intermittent exercise/rest protocol was designed to mimic the routine practices employed by firefighters during emergency operations and has been previously used in our lab. 15 Work cycles of 20 minutes account for the approximate time it takes firefighters to use one cylinder of air. Rest cycles were analogous to the recommended rehabilitation periods for firefighters (20 minutes) when they are permitted to take off parts of the turnout gear. Participants walked at a 5% gradient at the individually-determined speed identified during the familiarization sessions. Each participant’s individual treadmill speed and gradient were kept constant for both testing visits. Upon completion of the exercise protocol, nude body weight was recorded, and vascular measures were again obtained in the supine position, approximately 15–30 minutes after cessation of exercise.
For the HS condition, during rest periods, participants removed the helmet and other components of PPE (SCBA, turnout jacket, thermal hood and gloves). If an individual began to feel cool they were free to put the turnout jacket back on. In instances where Tco dropped to pre-exercise levels, the researchers asked participants if they felt comfortable to wear the turnout jacket during the remainder of the recovery period. During the NHS condition, participants removed the weighted vest and SCBA and sat in front of a fan while still wearing the cooling garment. The total volume of fluid necessary to maintain hydration (determined during familiarization testing) was consumed in equal boluses during rest periods in both conditions.
Measurements
Descriptive characteristics
Height was determined using a stadiometer (Seca, Hanover, MD; accuracy ±0.01 m) and body mass was measured using a standard physician scale (599KL; Health O Meter, Alsip, IL; accuracy ±0.1 kg). After completion of all study visits, a total body dual energy x-ray absorptiometry scan (iDXA, GE Lunar, enCore 2010, Madison, WI) was used to estimate body composition. Maximal oxygen uptake (VO2max) was determined at the initial visit, prior to familiarization and experimental visits, using indirect calorimetry (TrueOne 2400 Metabolic Measurement system, ParvoMedics, Sandy, Utah) as the participant completed a graded exercise test on a treadmill (Bruce 1973) to volitional fatigue. VO2max was achieved if two of the following criterion were achieved: a) heart rate peak greater than 85% of the age-predicted maximum, b) rating of perceived exertion (RPE; Borg) greater than 17, c) a plateau in HR, or d) a plateau in oxygen consumption.
Physiological and perceptual measures
Heart rate (HR; Zephyr BioHarness BT2, Annapolis, MD), Tco (HQ Inc., Palmetto, FL), RPE and thermal sensations (TS) were measured at baseline (final minute of pre-exercise testing) and at the end of exercise.
Cardiovascular measures
Vascular testing was conducted in a quiet, dimly-lit laboratory with all measures performed on the right side of the body while in the supine position. Applanation tonometry (AtCor Medical, Sydney, Australia) was performed for measures of pulse wave velocity (PWV) and pulse wave analysis (PWA). Arterial tonometry was obtained from two anatomical sites (carotid, femoral) to assess aortic stiffness, which is also commonly referred to as central stiffness. The distances from the suprasternal notch to the femoral and carotid arteries were measured using a standard tape measure to the nearest millimeter. Consistent waveforms were collected over 10 s from the carotid and femoral arteries using a high-fidelity tonometer. PWV was calculated using the time delay between the carotid/femoral waveforms and the transit distance between the carotid and femoral arteries. The time delay was assessed as the time from peak R wave from simultaneous ECG gating to the foot of the corresponding pressure waveform. The transit distance was measured using a flexible tape measure and was corrected for parallel transmission at the aortic arch by taking the difference between the sites (i.e. femoral–carotid).
Radial waveforms were collected for PWA and the waveforms were algorithmically transformed to estimate an aortic pressure waveform using a generalized transfer function. The transfer function is well accepted,16,17 reproducible, and has been validated at rest and with various perturbations such as Valsalva maneuver and exercise.18–20 The aortic blood pressure waveform was then calibrated against brachial systolic and diastolic pressure, obtained manually just prior to applanation tonometry using a standard sphygmomanometer and stethoscope. Noninvasive estimates of myocardial work and perfusion were computed using pressure-time integrals calculated from aortic pressure waveforms. This is an accepted technique 14 based on preliminary invasive work by Buckberg et al. 9 The area under the systolic portion of the aortic pressure waveform (systolic pressure-time integral; SPTI) is directly related to myocardial oxygen requirements and, thus, has been used as a proxy measure of myocardial work. 21 SPTI was calculated as mean systolic pressure × ejection duration × HR. The area under the diastolic portion of the aortic pressure waveform (diastolic pressure-time integral; DPTI) is correlated with subendocardial perfusion 9 and was calculated as mean diastolic pressure × diastolic time × HR, where diastolic time was calculated as heart period – ejection duration. The ratio of DPTI to SPTI is defined as the subendocardial viability ratio (SEVR) and provides insight into myocardial work/perfusion matching. 9 Rate pressure product (RPP) was calculated as aortic systolic blood pressure × HR as an additional measure of cardiac work. HR was measured during tonometry by the SphygmoCor device over the 10-s epoch.
Post-hoc analyses were performed on aortic waveforms to separate the pressure waves into forward (Pf) and reflected (i.e. backwards traveling; Pb) constituents using wave separation analysis (WSA). WSA has been proposed as a robust measure of pressure attributable to wave reflection (i.e. wave reflection magnitude) that allows interrogation of the independent contributions of wave reflections to the blood pressure waveform and their possible effects on cardiac performance. 22 This technique is widely accepted14,23 and has been described in detail previously.22,24 A wave reflection index (RIx) was calculated by dividing Pb by Pf as a measure of reflection magnitude (i.e. wave reflection magnitude relative to forward wave magnitude).
Statistical analysis
Data were analyzed using the statistical software package SPSS (version 21.0, SPSS, Inc., Chicago, IL). Students’ paired t-tests were used to determine differences between baseline hydration status (USG, post-exercise nude body weight), Tco (baseline Tco, end-exercise Tco), and exercise responses (end-exercise HR, RPE, TS). A 2x2 repeated measures analysis of variance was performed to determine condition, time, or condition–time interaction effects for hemodynamic outcome variables. Post-hoc two-tailed t-tests were used to investigate significant interactions between pre and post measures and NHS and HS conditions. Data are presented as mean ±SE, unless otherwise noted, with significance set a priori at p<0.05.
Results
Experimental visits were completed within an average span of 10±5 days. There were no significant differences in hydration status, pre- or post-exercise body weight, or resting Tco between conditions (Table 1). The intermittent exercise protocol resulted in greater heat stress for HS compared to NHS, as evidenced by greater perceptual strain (p<0.001), and significantly greater Tco (p<0.001) and HR (p<0.001), which continued throughout post-testing.
Hydration, core temperature, and exercise responses between conditions (mean ±SE).
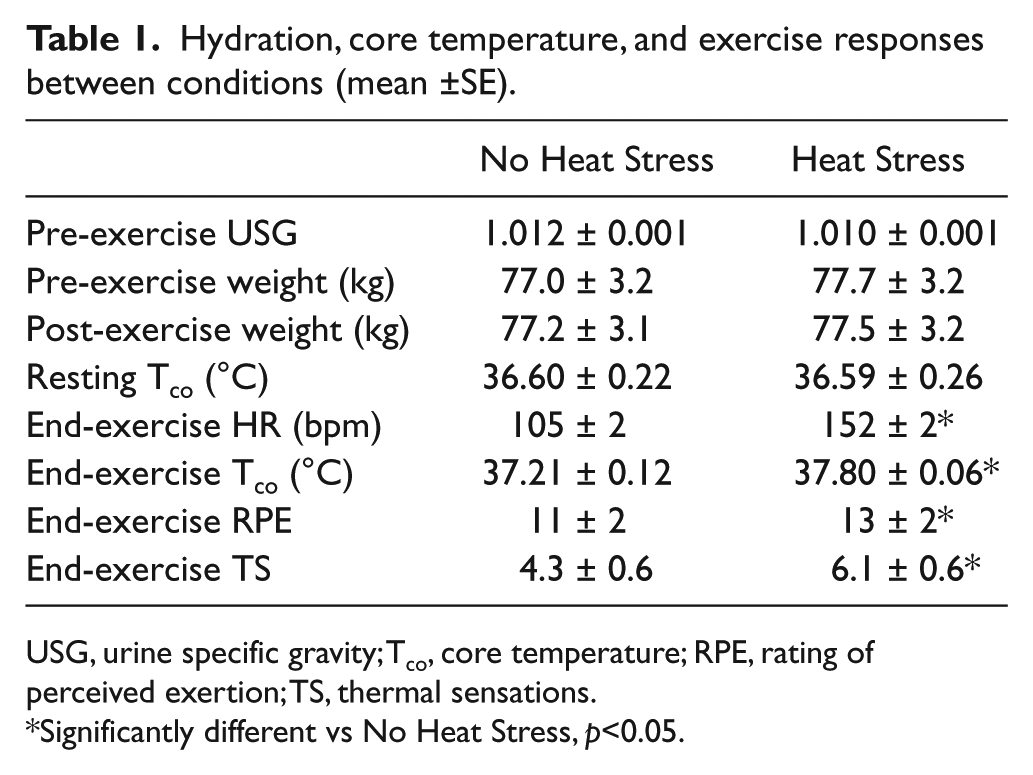
USG, urine specific gravity; Tco, core temperature; RPE, rating of perceived exertion; TS, thermal sensations.
Significantly different vs No Heat Stress, p<0.05.
Hemodynamic responses to EIHS
There were no significant differences in resting vascular-hemodynamic variables between conditions prior to the exercise protocol. Brachial blood pressure, aortic blood pressure, aortic stiffness, and ejection duration were not significantly different pre/post exercise or between conditions (Table 2). There was a significant condition-by-time interaction for heart rate which was significantly elevated post-exercise for HS (p=0.001). There was no significant difference in HR pre/post the exercise protocol in the NHS condition (p=0.17). Significant condition-by-time interactions were also detected for SPTI, DPTI, and SEVR. SPTI significantly increased (p<0.001), and DPTI (p=0.004) and SEVR (p<0.001) significantly decreased post-exercise for HS compared to NHS. A significant condition-by-time interaction was found for RPP (p<0.001). RPP was significantly elevated post-exercise for HS (p<0.001). There were no differences in RPP with the NHS condition (p=0.15). WSA analysis revealed a significant condition-by-time interaction for Pb (p=0.049), but not for Pf (p=0.90). Post-hoc testing indicated Pb decreased following the HS condition (p=0.012). Pb did not significantly change with the NHS condition (p=0.94). Changes in RIx mirrored changes in Pb, as RIx significantly decreased post-exercise for HS (p<0.001) compared to the NHS condition which was unchanged (p=0.71).
Hemodynamic variables before (pre) and after (post) intermittent exercise protocol (mean ±SE).
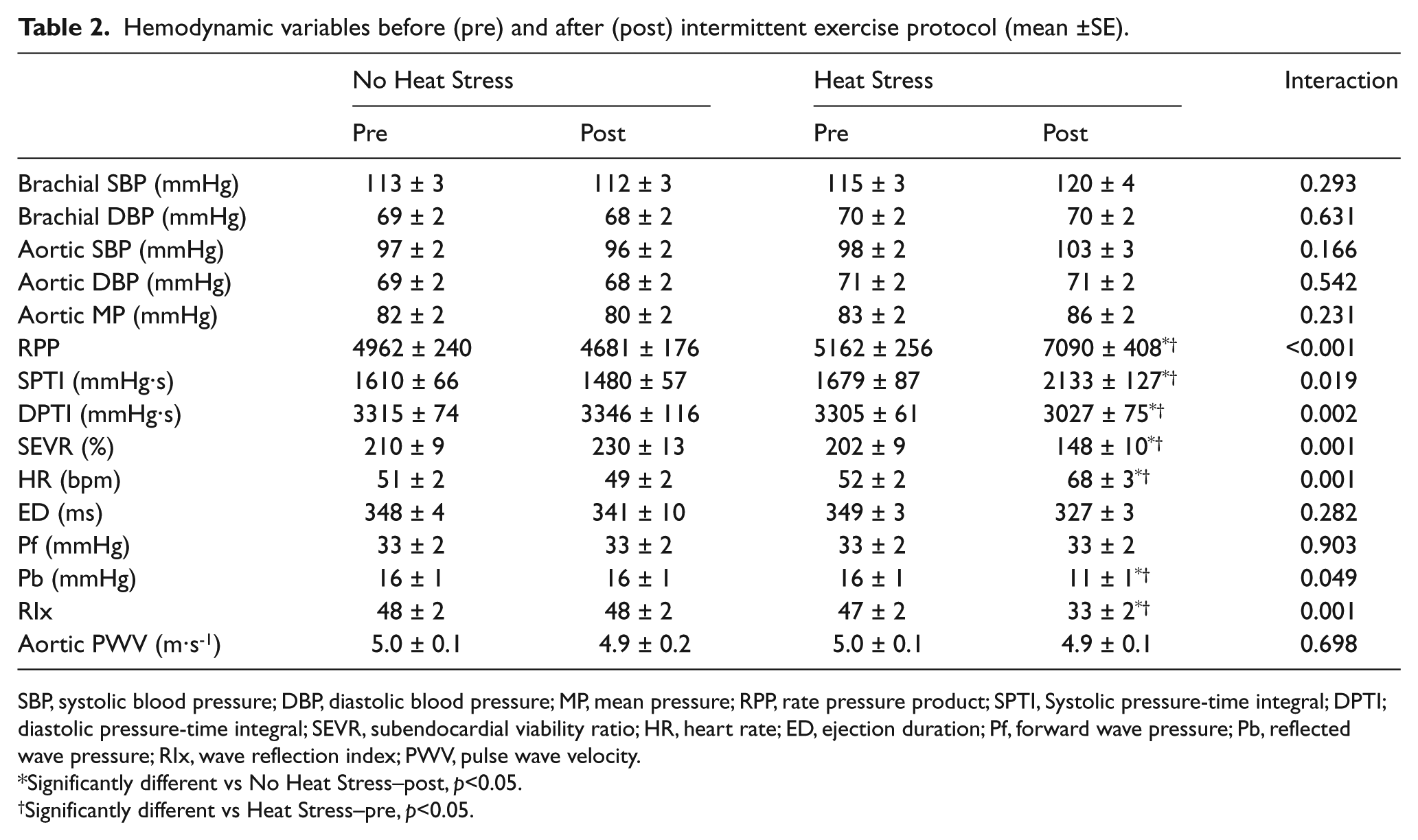
SBP, systolic blood pressure; DBP, diastolic blood pressure; MP, mean pressure; RPP, rate pressure product; SPTI, Systolic pressure-time integral; DPTI; diastolic pressure-time integral; SEVR, subendocardial viability ratio; HR, heart rate; ED, ejection duration; Pf, forward wave pressure; Pb, reflected wave pressure; RIx, wave reflection index; PWV, pulse wave velocity.
Significantly different vs No Heat Stress–post, p<0.05.
Significantly different vs Heat Stress–pre, p<0.05.
Discussion
Heat stress results in increased cardiovascular strain in an attempt to accommodate thermoregulatory balance. When coupled with physical exertion this may increase risk for a sudden cardiac event in at-risk individuals, particularly in such occupational settings as firefighting. Increases in aortic stiffness and pressure from wave reflections have been shown to have a negative effect on myocardial work 25 and coronary perfusion, and thus have been linked to sudden cardiac events. This study investigated the effect of EIHS on novel measures of vascular stiffness, central hemodynamic burden and noninvasive estimates of myocardial work and coronary perfusion. The main findings of this study are as follows: 1) EIHS increases measures of myocardial systolic work while concomitantly decreasing indices of coronary diastolic perfusion; 2) EIHS has no effect on aortic stiffness; 3) EIHS decreases reflected pressure wave magnitude (i.e. reduced Pb relative to Pf); 4) EIHS results in increased HR which may mediate changes in estimates of myocardial work and perfusion. Taken together, these findings suggest that moderate EIHS results in increased myocardial work and reduced coronary perfusion. This mismatch in myocardial demand and supply could lead to ischemic changes that make the myocardium more susceptible to arrhythmias and is a potential mechanism for the increased risk of cardiac events following fire suppression activities.
The combination of physical exertion and heat stress poses one of the greatest challenges to the cardiovascular system.3,26 This cardiovascular strain typically manifests as markedly elevated HR3,4,15,27–29 that results in increased RPP and myocardial work. Consistent with previous literature, our findings illustrate significant cardiovascular strain following EIHS compared to NHS. Following moderate EIHS, we documented significant increases in myocardial work (indicated by increased RPP and SPTI). Additionally, DPTI was significantly reduced following EIHS compared to the NHS condition, suggesting reduced myocardial perfusion. This contrasts to what is reported with steady state exercise which results in increased systolic workload, indicated by SPTI, without impacting coronary perfusion 30 minutes post-exercise. 30 Our data suggest that EIHS results in greater systolic work and reduced diastolic perfusion, creating an oxygen supply–demand mismatch.
Appropriate coupling between the left ventricle and the vasculature results in an optimal transfer of blood from the left ventricle to the periphery/systemic circulation. 31 Aortic stiffening, independent of changes in downstream peripheral stiffness, is capable of elevating systolic pressure, 32 increasing cardiac afterload 25 and thereby increasing myocardial work. When the heart ejects blood into a stiff, rather than compliant vessel, there is an increase in myocardial oxygen consumption although stroke volume is maintained. 33 Aortic stiffness has been reported to increase following live firefighting drills, 27 although it is unaffected by passive heat stress. 28 According to the current findings, EIHS did not affect aortic stiffness, suggesting that the increased myocardial work and decreased perfusion observed following EIHS in firefighter PPE may not be related to changes in the elastic properties of the aorta.
There were no changes in forward wave pressure magnitude (Pf) following EIHS. However, we observed a reduction in the magnitude of pressure from wave reflections (i.e. RIx and Pb). Changes in reflected wave magnitude, observed herein, appear to be independent of changes in pressure wave velocity, as there was no change in aortic stiffness, and may provide some insight into reduced coronary perfusion. Reductions in the magnitude of pressure from wave reflections arriving at the heart during diastole might decrease the area under the diastolic portion of the blood pressure waveform (DPTI). 13 Reductions in DPTI would impair diastolic coronary perfusion because of the coronary vessels’ dependence on diastolic pressure in establishing the perfusion gradient. 13 Thus, reductions in wave reflection magnitude may reduce DPTI and negatively affect the balance between myocardial oxygen supply and demand.
Heart rate was markedly elevated post-exercise in the HS condition compared to NHS, contributing to differences in cardiovascular strain. Elevated HR increases cardiac work secondary to more frequent ventricular contractions and also decreases time spent in diastole, reducing overall time available for coronary perfusion.13,34 In diseased coronary arteries, elevated HR may precipitate angina and myocardial ischemia 34 which may have particular implications for the firefighting community and their prevalence of high cardiovascular risk profiles. 35 Increases in HR also impact pressure from wave reflections. 36 HR is inversely associated with measures of global wave reflection owing to temporal shifts in the arrival of the reflected pressure wave. 36 In the current study HR was significantly inversely associated with wave reflection and diastolic perfusion (Pb, R2 = -0.17; DPTI, R2 = -0.21). In order to account for the effect of HR on the timing of wave reflections and diastolic perfusion we adjusted Pb by HR, and DPTI by diastolic time at each respective time point. This additional analysis did not change the findings of our study. However, it should be underscored that HR is not simply a statistical confound; rather, it is a physiological response that affects cardiovascular strain via multiple different mechanisms and should not be dismissed.
Implications
Our data suggest a myocardial supply and demand imbalance occurs in healthy males following moderate EIHS in a low-risk setting. The myocardial supply–demand imbalance seen herein may be more pronounced in the firefighting community due to the prevalence of obesity and high cardiovascular risk profile in the firefighting community. 35 The supply–demand imbalance may be further exacerbated when coupled with the severe physiological and psychological perturbations that accompany live-fire scenarios including intense physical work while in a hot, hostile environment wearing heavy protective gear (>25kg). 6 Indeed, these data are consistent with the theory that the combination of increased cardiac stress, physical work, and myocardial oxygen demand coupled with decreased cardiac perfusion and relaxation may be sufficient to precipitate exertional angina14,37 and may provide insight into the high occurrence of sudden cardiac events in the Fire Service.
Limitations
This study utilized healthy, fit, young men because of the strenuous and physically taxing nature of the HS trial. Although a sample of less fit individuals may have been more similar to fitness levels seen in the Fire Service, we chose to conduct our initial study in a homogeneous group of young healthy men to limit a number of variables that are known to affect vascular function. In light of this, our findings may not be generalizable to the population of firefighters. Our results would likely be more pronounced in unfit or obese individuals. Specifically, findings may differ in populations with stiffer arteries since passive heat stress has been reported to decrease aortic stiffness in participants with greater aortic stiffness at baseline. 28 Older firefighters, for example, may experience greater changes in stiffness following EIHS, which may exaggerate reductions in wave reflections, effectively reducing the beneficial contribution of wave reflections to diastolic perfusion of the coronary vessels. This may be of particular importance for firefighters since the high prevalence of obesity in the Fire Service has been linked to greater arterial stiffness 38 and reduced SEVR 35 at rest compared to less obese counterparts. Additionally, future research should investigate vascular responses to EIHS among females. Moderate heat stress was achieved in this study by performing moderate intensity exercise in firefighter PPE in a thermoneutral laboratory (peak Tco, 37.80±0.06oC), which may be a lower Tco than is experienced by athletes or those in other occupations. Although these environmental conditions differ from those encountered during fire suppression, the change in Tco documented in our study (∆Tco ≈ +1.21oC) is similar to previous reports from simulated firefighting (∆Tco from +0.73 to +1.54oC).39,40 The responses reported in this study may be exaggerated in scenarios with greater heat stress or if combined with dehydration, as often occurs with firefighting.
Vascular function in the current study was not assessed immediately following exercise cessation, and therefore the hemodynamic changes associated with peak Tco may not have been captured. However, the post-exercise time period is particularly relevant to firefighters as approximately 15% of cardiac incidents occur shortly after fire suppressions have ended (while returning from the emergency or at the station, 41 a time when Tco may not be severely elevated). In the NHS condition, we minimized the rise in Tco during exercise by using a cooling vest; thus, our NHS condition involved cooling which may have altered some vascular measures.
WSA was not performed with simultaneous measurement of aortic blood flow, but rather using an estimated flow waveform derived from the contour of the aortic blood pressure waveform via the triangulation method. This method assumes aortic flow is triangular in shape, with the start, peak and end set at the pressure wave upstroke, initial inflection point, and dicrotic notch, respectively. 22 Using the pressure and estimated flow wave, characteristic impedance can be calculated and used to determine the composition of forward and backward pressure waves. 22 Wave separation results based on the triangular flow estimation are well correlated with those obtained from direct flow measurements, suggesting the triangulation method provides an acceptable approximation of flow based on pressure alone.22,24 Future research utilizing measured flow and central pressure to derive Pf and Pb is needed to corroborate the present findings. The myocardial supply–demand balance was estimated noninvasively from the aortic pressure wave contour which correlates with myocardial oxygen consumption 42 and coronary perfusion. 9 Additional studies are needed to confirm the present findings using additional measures of coronary perfusion (e.g. coronary flow reserve from Doppler-ultrasound/echocardiography).
Conclusion
In conclusion, there were significant decreases in noninvasive estimates of diastolic coronary perfusion coupled with increases in systolic myocardial work following moderate EIHS compared to a normothermic-exercise NHS condition. Reductions in coronary perfusion may be partially related to decreases in pressure from wave reflections and moderated by changes in HR following EIHS, and not changes in aortic stiffness per se. Aortic stiffness was unaltered following EIHS and thus may not explain the changes in wave reflections observed in our sample of young, fit males. These data suggest a greater myocardial oxygen supply–demand imbalance with EIHS and may provide insight into potential mechanisms behind the prevalence of sudden cardiac events in the Fire Service.
Footnotes
Appendix
Term index
| Term | Definition |
| DPTI | Diastolic pressure time integral |
| EIHS | Exercise-induced heat stress |
| HR | Heart rate |
| HS | Heat stress condition |
| Pb | Reflected wave magnitude |
| Pf | Forward wave magnitude |
| PPE | Personal protective equipment |
| PWA | Pulse wave analysis |
| PWV | Pulse wave velocity |
| RIx | Wave reflection index |
| RPP | Rate pressure product |
| SCBA | Self-contained breathing apparatus |
| SEVR | Subendocardial viability ratio |
| SPTI | Systolic pressure time integral |
| Tco | Core temperature |
| USG | Urine specific gravity |
| VO2max | Maximal oxygen uptake |
| WSA | Wave separation analysis |

Acknowledgements
We would like to express our appreciation to the study participants for their time and effort, Patrick Segers for his work on developing the WSA program, Stephen Ives for his review of the manuscript, and to the staff members of the First Responder Health and Safety Laboratory for their assistance with data collection.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by a research grant from the Department of Homeland Security through a Federal Emergency Management Agency Assistance to Firefighters Grant (EMW-2010-FP-01992, Smith, PI).
