Abstract
Polyunsaturated fatty acids have been associated with beneficial influences on cardiovascular health. However, the underlying mechanisms are not clear, and data on the relations of polyunsaturated fatty acids to subclinical disease measures such as vascular stiffness and cardiac function are sparse and inconclusive. In a large community-based cohort, we examined the relations of omega-3 and other fatty acids to a comprehensive panel of vascular function measures (assessing microvascular function and large artery stiffness), cardiac structure and left ventricular function. Red blood cell (RBC) membrane fatty acid composition, a measure of long-term fatty acid intake, was assessed in participants of the Framingham Offspring Study and Omni cohorts and related to tonometry-derived measures of vascular stiffness and to a panel of echocardiographic traits using partial correlations. Up to n=3055 individuals (56% women, mean age 66 years) were available for analyses. In age- and sex-adjusted models, higher RBC omega-3 content was moderately associated (p≤0.002) with several measures of vascular stiffness and function in a protective direction. However, after multivariable adjustment, only an association of higher RBC omega-3 content with lower carotid-femoral pulse wave velocity (a measure of aortic stiffness) remained significant (r = −0.06, p=0.002). In secondary analyses, higher linoleic acid, the major nutritional omega-6 fatty acid, was associated with smaller left atrial size, even after multivariable adjustment (r = −0.064, p<0.001). In conclusion, in our cross-sectional community-based study, we found several associations consistent with the notion of protective effects of omega-3 and linoleic acid. The clinical significance of these modest associations remains to be elucidated.
Keywords
Introduction
Fish consumption has been associated with a reduced risk of cardiovascular disease (CVD) outcomes, including heart failure, atrial fibrillation, stroke, fatal myocardial infarction and sudden cardiac death in several observational studies.1–5 It has been suggested that polyunsaturated omega-3 fatty acids underlie the cardioprotective associations of fish intake, in particular eicosapentaenoic acid (EPA, 20:5ω-3) and docosahexaenoic acid (DHA, 22:6ω-3). However, the literature is inconclusive in this regard. Of four recently published meta-analyses, two concluded that omega-3 polyunsaturated fatty acids (PUFAs) were cardioprotective6,7 whereas the other two did not.8,9
Different biological mechanisms may underlie the potential cardioprotective effects of omega-3 fatty acids, in particular lower systemic inflammation, lower blood pressure and a healthier lipid profile. 10 Beyond these potential biological effects on systemic biomarkers and cardiovascular risk factors, information on the association of omega-3 fatty acids with markers of cardiovascular target organ damage is sparse. Very limited data suggest that omega-3 fatty acids may have favorable effects on vascular stiffness, 11 a major determinant of age-related cardiovascular morbidity and mortality, including systolic hypertension. 12 However, large-scale studies investigating this premise are lacking. Omega-3 fatty acids also have been related to favorable effects on cardiac function and geometry in experimental models and smaller epidemiological studies,13,14 and therapeutic administration has demonstrated beneficial effects in the treatment of chronic congestive heart failure in some studies.15,16
Other PUFAs also have been associated with cardiovascular risk. In particular, the intake of omega-6 fatty acids (primarily linoleic acid (18:2ω-6)) has been associated with reduced cardiovascular risk and with a lower prevalence of future left ventricular (LV) hypertrophy in some studies,17,18 but the published literature is not conclusive.19,20 Linoleic acid can be converted into arachidonic acid (20:4ω-6), which is a critically important precursor for prostaglandins and leukotrienes. Higher circulating arachidonic acid concentrations appear to be associated with a lesser inflammatory state. 21 In contrast, trans fatty acids, mainly originating from partially hydrogenated plant oils, have been associated with an increased cardiovascular risk. 22
Accordingly, we investigated the relation of red blood cell (RBC) omega-3 fatty acid (EPA+DHA) content (a measure of long-term omega-3 intake), with a comprehensive panel of measures assessing microvascular function, large artery stiffness, cardiac structure and LV function in the Framingham Offspring Study. In secondary analyses, we also explored the relation of RBC linoleic acid, arachidonic acid, and trans fatty acid content with vascular stiffness, cardiac structure and LV function.
Subjects and methods
Study sample
We studied participants of the Framingham Offspring Study (FOS) and Omni cohorts.23,24 The Offspring cohort started in 1971 with the recruitment of 5124 individuals who were the children of the original Framingham cohort participants or the children’s spouses. Participants are evaluated approximately every 4 years. Each examination cycle includes standardized questionnaires, anthropometry, blood pressure and electrocardiograms, and phlebotomy for assessment of cardiovascular risk factors. Data for the current study were obtained at Offspring cohort examination cycle 8 (2005–2008). The Omni cohort contains non-white minorities and was recruited in 1994–1998. 24 Data for the current study were retrieved from Omni cohort examination cycle 3 (2005–2008). The study protocols were approved by the Boston University Medical Center Institutional Review Board and conform with the principles outlined in the Declaration of Helsinki. Written informed consent was obtained from all participants.
Of 3021 eligible Offspring and 298 Omni participants, 3196 had RBC fatty acid measurements available. Of these, 3170 had at least one echocardiography or tonometry trait available. After exclusion of 115 participants with missing covariates (see below), we obtained a final sample size of 3055. For analyses of echocardiographic traits, we further excluded 169 individuals with prevalent heart failure or history of myocardial infarction (possibly violating geometric assumptions inherent in some echocardiographic LV measurements). The study protocol was approved by the Boston University Medical Center Institutional Review Board; written informed consent was obtained from all participants.
Assessment of red blood cell fatty acid composition
RBC membrane fatty acid composition was measured using gas chromatography as previously described. 25 In short, RBC samples were heated to 100°C for 10 minutes with methanol containing 14% boron triflouride and hexane to generate fatty acid methyl esters from the membrane glycerophospholipids. After the addition of water, the methyl esters were recovered and analyzed in a GC2010 (Shimadzu Corporation, Columbia, MD, USA) gas chromatograph using a SP-3560, 100-m capillary column. Fatty acid composition was expressed as a percent of total identified fatty acids. Throughout, the term ‘omega-3 fatty acids’ will refer to EPA+DHA.
Arterial tonometry and peripheral arterial tonometry
Tonometry measures of large arteries were obtained as previously described in detail.26,27 Briefly, after about 5 minutes’ rest brachial systolic and diastolic blood pressures were obtained in the supine position. Arterial tonometry with simultaneous electrocardiogram (ECG) recording was performed on the brachial, radial, femoral and carotid arteries,26,27 with all measurements being performed on the right side of the body. Transit distances were estimated by body surface measurements from the suprasternal notch to the pulse recording site. Details of signal analyses and computational data processing have been published elsewhere.26,27
Peripheral arterial tonometry (PAT) measurements were acquired using a commercially available device (Endo-PAT2000; Itamar Medical, Caesarea, Israel) as previously described. 28 In short, the PAT device contains a pneumatic plethysmograph that applies uniform pressure to the surface of the distal finger, allowing measurement of pulse volume changes in the finger. Baseline pulse amplitude was measured from each fingertip for 2 minutes 20 seconds. Arterial flow was then interrupted for 5 minutes by a cuff placed on a proximal forearm. Pulse amplitude was recorded electronically in both fingers. Automatic signal analysis algorithms provided the average pulse amplitude for each 30-second interval after forearm cuff deflation up to 4 minutes. The PAT ratio was calculated as the natural logarithm of the ratio of post deflation to baseline pulse amplitude in the hyperemic finger divided by the same ratio in the contralateral finger.
We assessed the following traits derived from large artery tonometry and PAT: carotid-femoral pulse wave velocity (CFPWV) as the reference standard measure of aortic stiffness; central pulse pressure as a measure of central aortic pressure pulsatility; reflection factor as a measure of relative peripheral pressure wave reflection; PAT ratio as a measure of small artery vasodilator function; and the mean (log-transformed) baseline digital pressure pulsatility, a measure of small artery pulsatility. 28
Echocardiography
Echocardiography was performed using a Sonos 5500 (Philips) ultrasound machine. Two-dimensionally guided M-mode tracings were recorded with a minimum of three frames for measuring and averaging LV mass and left atrial dimensions. All echocardiograms were evaluated by an experienced sonographer or cardiologist using a standardized reading protocol. Left atrial (LA) end-systolic diameter, diastolic LV diameter and posterior and anteroseptal wall thickness were measured using a leading edge technique as recommended by the American Society of Echocardiography (ASE). 29 LV mass was calculated according to ASE guidelines, applying the formula proposed by Devereux et al. 29 as 0.8[1.04(LV internal dimensions+septal wall thickness+posterior wall thickness)3 – (LV internal dimensions)3]+0.6 g. The sum of the diastolic thicknesses of the septum and posterior wall was used to estimate LV wall thickness (LVWT). Diastolic LV function was assessed as the E/E’ ratio, with E being the maximum early diastolic mitral inflow velocity, and E’ being the maximum early diastolic mitral annulus velocity, measured by tissue Doppler at the lateral mitral annulus.
Statistical analyses
LV mass was natural-logarithmically transformed for statistical modelling to normalize its distribution and harmonize variances between men and women. Carotid-femoral PWV was inverse-transformed to normalize its distribution and reduce heteroscedasticity and was then multiplied by –1 to restore directionality.
We used Pearson correlations to investigate the relations of fatty acids with echocardiography and tonometry traits. Potential confounders in these relations were accounted for using multivariable-adjusted (partial) Pearson correlations.
Our primary exposure of interest was RBC omega-3 content; secondary exposure variables were linoleic acid, arachidonic acid and trans fatty acids. Correlation analyses were performed in a two-stage design: (1) adjusting only for age and sex; (2) additionally adjusting for potential clinical confounders based on our previous work on RBC fatty acids, 25 tonometry traits, 30 and echocardiographic traits. 31
Accordingly, for tonometry traits we assessed two models: (1) adjusting for age, age squared (to account for the non-linear age-slope of vascular stiffness), and sex; and (2) adjusting for cohort, age, age squared, sex, height, weight, heart rate, diabetes, current smoking, prevalent CVD, blood glucose, total to high-density lipoprotein cholesterol ratio, hormone replacement therapy, hypertension treatment, use of aspirin, and lipid-lowering medication. Since about 10% of our study sample reported intake of fish oil supplements, 25 which may potentially confound any observed association, we also included fish oil supplementation use as a binary covariate. Similarly, partial correlations between fatty acids and echocardiographic traits were estimated adjusting for (1) age, sex and height, and (2) adjusting for cohort, age, sex, height, weight, heart rate, systolic blood pressure, antihypertensive treatment, diabetes, and fish oil supplementation. Since we tested 12 echocardiography and tonometry traits, we a priori defined p<0.004 (0.05/12) as statistically significant using a Bonferroni correction. With this threshold of significance, we had 80% power to detect partial correlations of 0.0705 (n=2800) to 0.0745 (n=2500).
Results
Study sample
The characteristics of our study sample are listed in Table 1. The unadjusted correlations between fatty acids, vascular stiffness and echocardiographic measures are listed in Supplementary Table 1.
Characteristics of the study sample (n=3055 unless indicated otherwise).

Continuous variables are presented as mean ± standard deviation; binary traits as percent prevalence.
BMI, body mass index; BP, blood pressure; RBC, red blood cell; EPA, eicosapentaenoic acid; DHA, docosahexaenoic acid; LV, left ventricular; LA, left atrial; E/E’, ratio of maximum diastolic mitral inflow Doppler velocity to maximum diastolic mitral annulus tissue Doppler velocity; CFPWV, carotid-femoral pulse wave velocity; PAT, peripheral arterial tonometry.
Available n were as follows: LV fractional shortening n=2596, LV mass n=2597, LV diastolic diameter n=2600, LV wall thickness n=2745, LA diameter n=2849, E/E’ n=2777, CFPWV n=2727, central pulse pressure n=2780, mean arterial pressure n=2834, reflection factor n=2780, PAT ratio n=2487, PAT baseline pulsatility n=2487.
Omega-3 fatty acids and vascular stiffness
The age- and sex-adjusted correlation coefficients between RBC omega-3 fatty acids and vascular stiffness measures are given in Table 2 (upper part). After considering multiple testing, higher RBC omega-3 fatty acid content was modestly associated with lower CFPWV in age- and sex-adjusted (r = −0.098, p<0.001) and multivariable-adjusted analyses (r = −0.060, p=0.002). Omega-3 fatty acids were also associated with the reflection factor (r=0.059, p=0.002), PAT ratio (r=0.061, p=0.002) and PAT baseline pulsatility (r = −0.066, p=0.001) in age- and sex-adjusted analyses.
Correlation of RBC omega-3 content with vascular stiffness and echocardiographic traits.
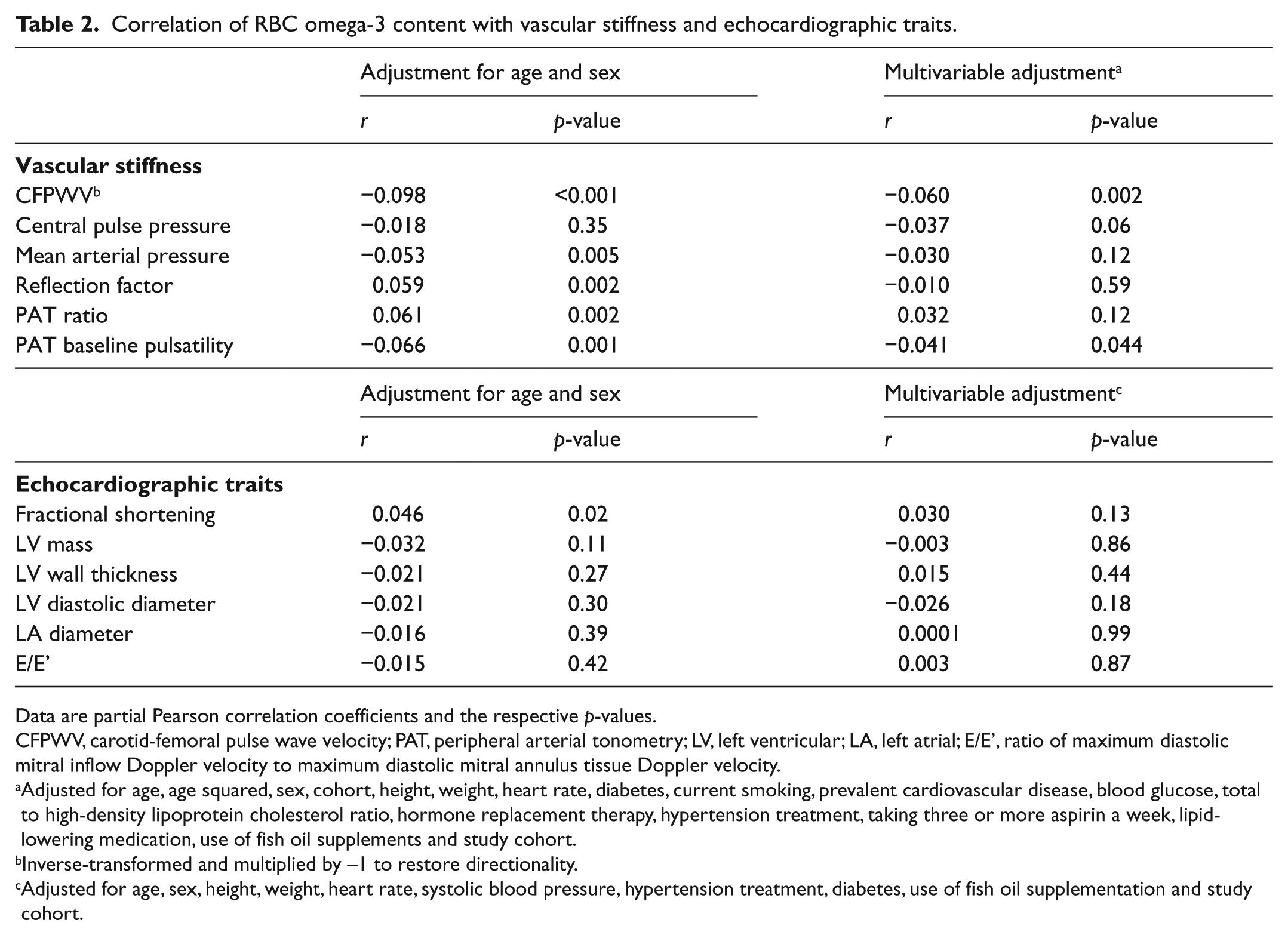
Data are partial Pearson correlation coefficients and the respective p-values.
CFPWV, carotid-femoral pulse wave velocity; PAT, peripheral arterial tonometry; LV, left ventricular; LA, left atrial; E/E’, ratio of maximum diastolic mitral inflow Doppler velocity to maximum diastolic mitral annulus tissue Doppler velocity.
Adjusted for age, age squared, sex, cohort, height, weight, heart rate, diabetes, current smoking, prevalent cardiovascular disease, blood glucose, total to high-density lipoprotein cholesterol ratio, hormone replacement therapy, hypertension treatment, taking three or more aspirin a week, lipid-lowering medication, use of fish oil supplements and study cohort.
Inverse-transformed and multiplied by –1 to restore directionality.
Adjusted for age, sex, height, weight, heart rate, systolic blood pressure, hypertension treatment, diabetes, use of fish oil supplementation and study cohort.
To further explore the relation of omega-3 fatty acids with CFPWV, we divided our study cohort into three groups based on tertiles of RBC omega-3 content, and calculated age- and sex-adjusted mean CFPWV for each group. These adjusted means of CFPWV for each group are depicted in Figure 1.
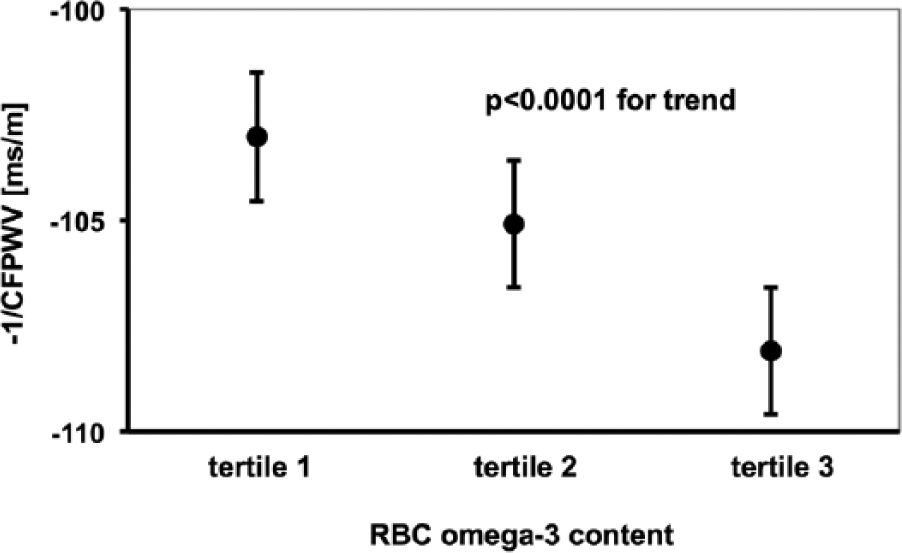
Age- and sex-adjusted carotid-femoral pulse wave velocity (CFPWV; inverse-transformed and multiplied by –1 to restore directionality) partitioned by tertiles of red blood cell (RBC) omega-3 fatty acid content. Error bars indicate standard errors.
Omega-3 fatty acids and cardiac structure and function
The correlations between RBC omega-3 fatty acids and echocardiographic variables are given in Table 2 (lower part). In neither age/sex-adjusted nor multivariable-adjusted models were omega-3 fatty acids associated with any echocardiographic trait (all p-values exceeded 0.004).
Secondary analyses: linoleic acid, arachidonic acid and trans fatty acids
Correlations between linoleic acid and measures of vascular stiffness and cardiac structure and function are given in Table 3. In age- and sex-adjusted models, higher RBC linoleic acid was related to lower LV mass, lower LVWT and lower LA diameter. After multivariable adjustment, only a moderate association with lower LA diameter remained (p<0.001). Arachidonic acid was only associated with greater LA diameter and only in an age- and sex-adjusted model (Table 4). Trans fatty acids were not associated with vascular stiffness or echocardiography traits in any model (Table 5).
Correlation of RBC linoleic acid content with vascular stiffness and echocardiographic traits.
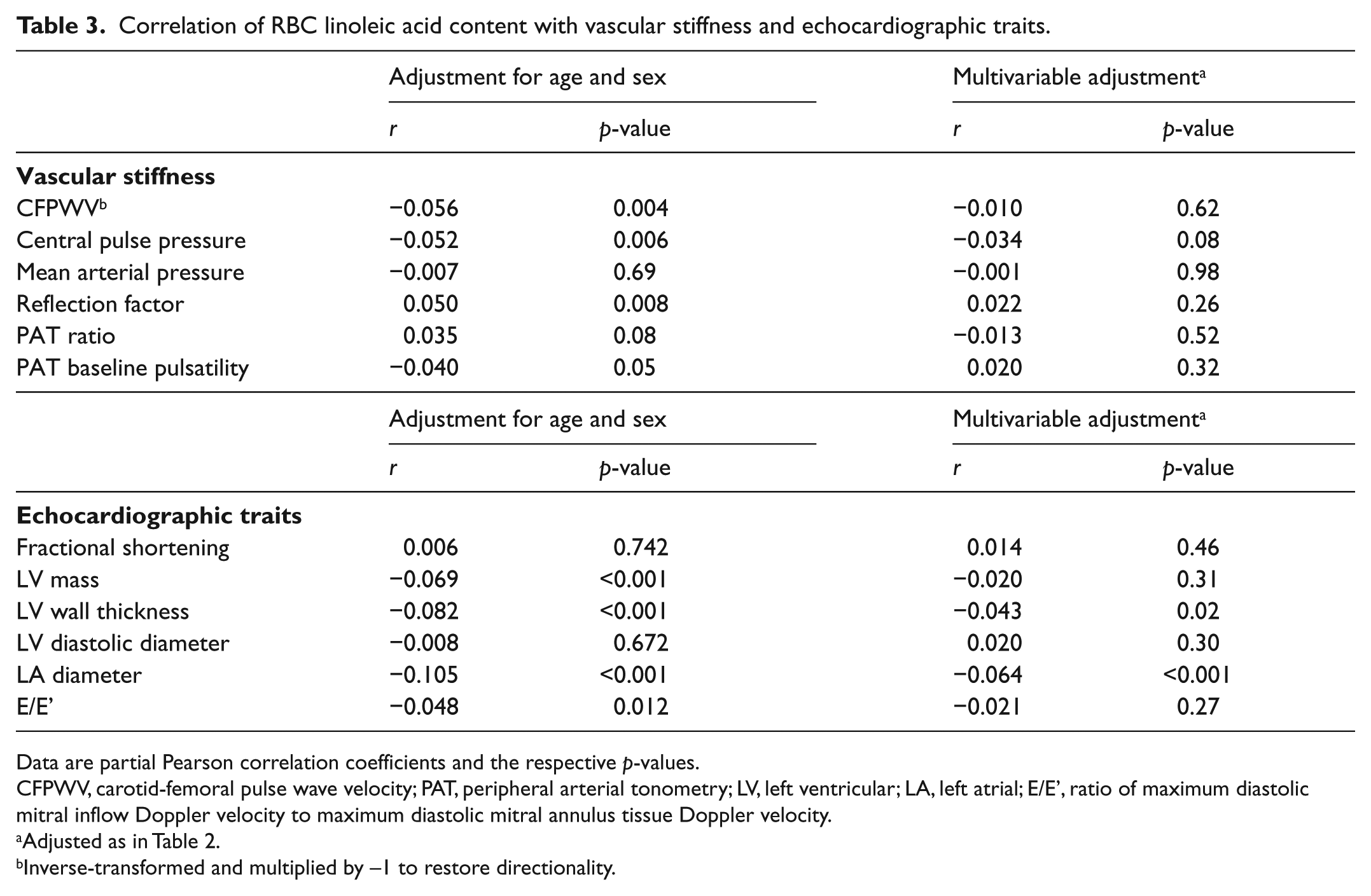
Data are partial Pearson correlation coefficients and the respective p-values.
CFPWV, carotid-femoral pulse wave velocity; PAT, peripheral arterial tonometry; LV, left ventricular; LA, left atrial; E/E’, ratio of maximum diastolic mitral inflow Doppler velocity to maximum diastolic mitral annulus tissue Doppler velocity.
Adjusted as in Table 2.
Inverse-transformed and multiplied by –1 to restore directionality.
Correlation of RBC arachidonic acid content with vascular stiffness and echocardiographic traits.
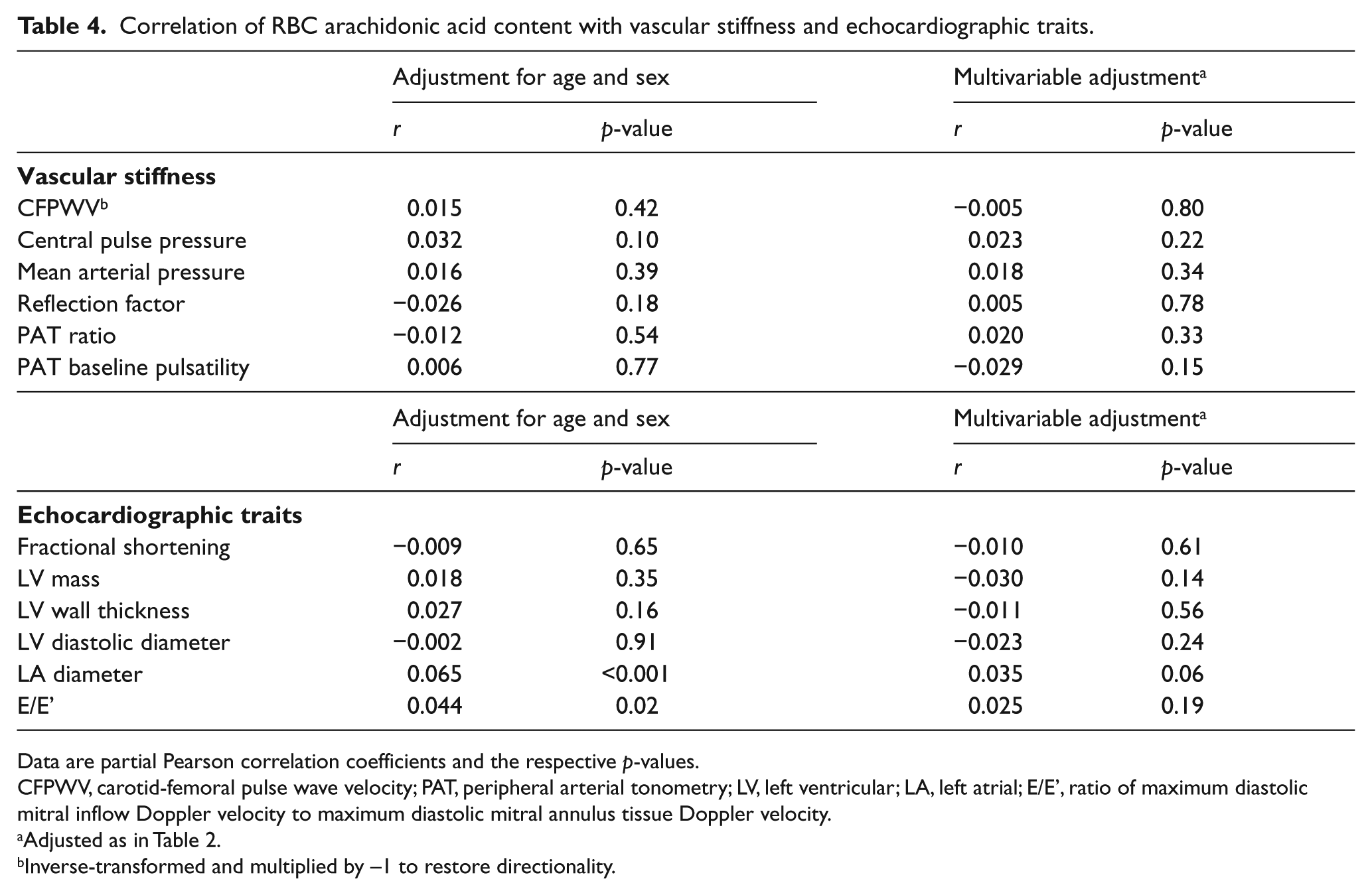
Data are partial Pearson correlation coefficients and the respective p-values.
CFPWV, carotid-femoral pulse wave velocity; PAT, peripheral arterial tonometry; LV, left ventricular; LA, left atrial; E/E’, ratio of maximum diastolic mitral inflow Doppler velocity to maximum diastolic mitral annulus tissue Doppler velocity.
Adjusted as in Table 2.
Inverse-transformed and multiplied by –1 to restore directionality.
Correlation of RBC trans fatty acid content with vascular stiffness and echocardiographic traits.
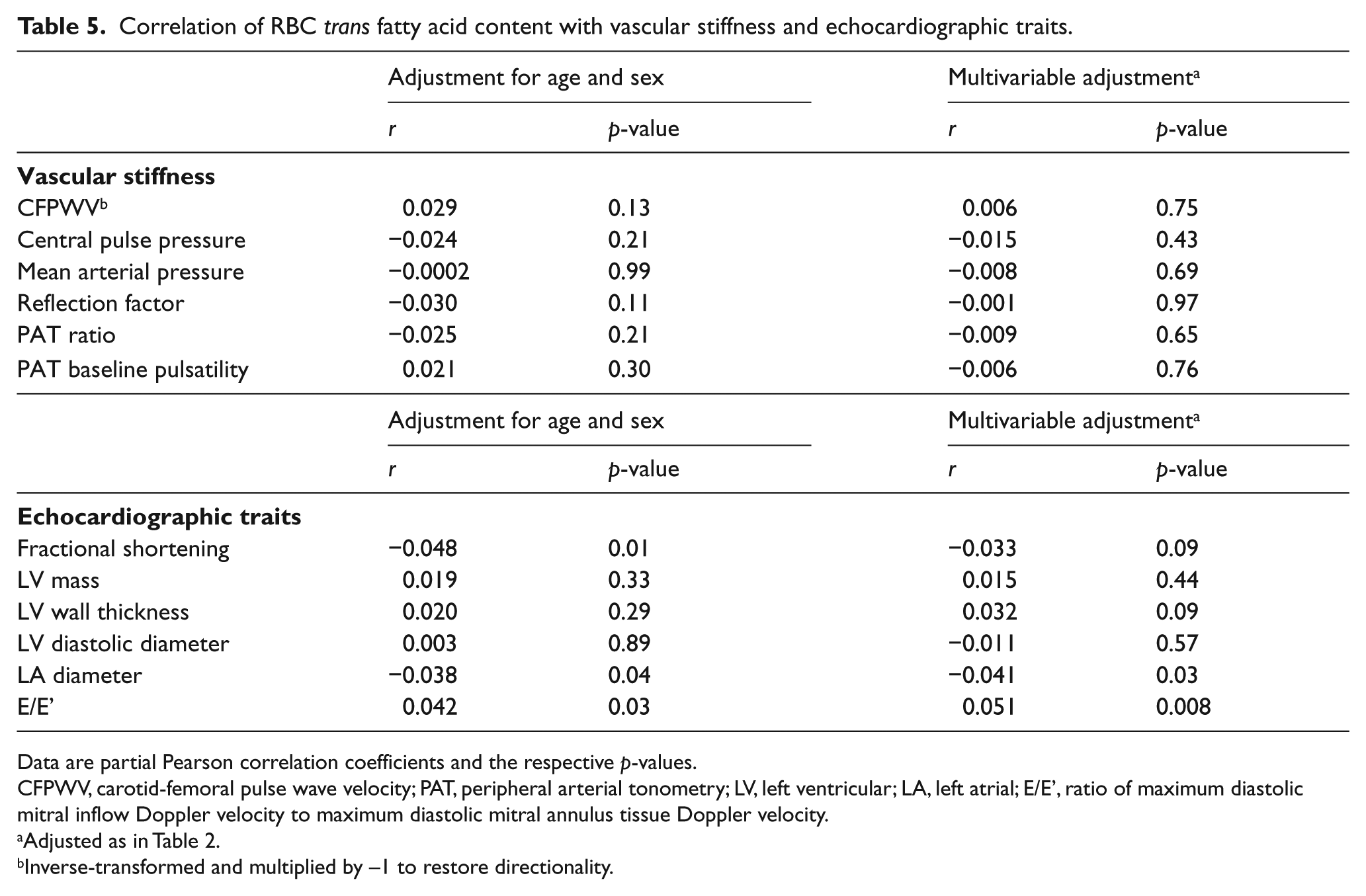
Data are partial Pearson correlation coefficients and the respective p-values.
CFPWV, carotid-femoral pulse wave velocity; PAT, peripheral arterial tonometry; LV, left ventricular; LA, left atrial; E/E’, ratio of maximum diastolic mitral inflow Doppler velocity to maximum diastolic mitral annulus tissue Doppler velocity.
Adjusted as in Table 2.
Inverse-transformed and multiplied by –1 to restore directionality.
Discussion
We examined the cross-sectional relations of RBC omega-3 content with measures of vascular stiffness and cardiac structure and function in the Framingham Offspring and Omni cohorts. In minimally (age- and sex-) adjusted models we observed several associations in a protective direction. However, after multivariable adjustment, only a modest association of higher RBC omega-3 content with lower aortic stiffness persisted. In secondary analyses, we observed a modest inverse relation between RBC linoleic acid content and left atrial size in a multivariable-adjusted model.
Omega-3 fatty acids and vascular stiffness
Whereas several epidemiological studies suggest a protective association of omega-3 PUFA on arterial blood pressure, 32 very few investigations have related them to directly assessed vascular stiffness (a major determinant of hypertension and strong predictor of cardiovascular events 33 ), and the published results have been inconsistent. Yamada et al. found no significant difference in CFPWV between 261 inhabitants of a Japanese fishing village (with high fish consumption) and 209 inhabitants of a farming village (with low fish consumption), 34 whereas in two other smaller studies arterial compliance was better in the fishing than in the farming communities.35,36 Schack-Nielsen et al. observed no association between eating habits and radial-femoral PWV in a sample of 87 10-year-old children. 37 On the other hand, plasma DHA and arachidonic acid levels were inversely correlated with aortic PWV (and 20-year mortality) in 174 subjects in another study. 38 Several interventional studies have assessed the impact of omega-3 supplementation on vascular stiffness. Some groups reported a beneficial effect of omega-3 supplementation on vascular stiffness,39–42 whereas others observed no effect.43,44 Generally, these investigations were of very limited size, and large studies are lacking.
To our knowledge, the present investigation is the first large-scale epidemiological study to investigate the relation between RBC fatty acids and vascular stiffness in a large middle-aged sample. RBC membrane omega-3 fatty acid composition reflects exposure over the life of the erythrocyte and is stable over several years in individuals with a consistent background fish intake. 45 Hence, RBC measurements provide more reliable estimates of long-term omega-3 intake than single occasion serum levels or food questionnaires. 46 In our large community-based sample, we observed a modest, but statistically significant, inverse association of cell membrane omega-3 fatty acid levels with aortic stiffness. The clinical relevance of this finding remains to be shown. Nevertheless, in light of the observation that aortic stiffness antedates and is an important risk factor for incident hypertension and cardiovascular disease events, 12 the role of increased omega-3 fatty acid intake, whether from food or supplements, as an approach for preventing hypertension merits further study.
Omega-3 fatty acids, cardiac structure and function
Several studies have suggested that marine omega-3 fatty acids correlate with cardiac structure or function, although the reported findings have been inconsistent.13,14,17,47 Mozaffarian et al. reported the association of EPA+DHA intake (estimated from diet questionnaires) with lower heart rate and greater echocardiographic LV stroke volume in the Cardiovascular Health Study. 48 Rupp et al. observed an inverse association of omega-3 plasma levels with LV diameter in a sample of 309 patients with dilated cardiomyopathy. 14 In animal models, fish-oil supplementation mitigated the development of LV hypertrophy induced by aortic banding.47,49 Conversely, Sundström et al. reported that higher serum EPA levels at age 50 years were associated with increased odds for LV hypertrophy 20 years later, possibly suggesting a detrimental effect of EPA. 17 Controlled trials in heart failure patients suggest that omega-3 supplementation may improve LV systolic function.16,50 These prior studies were limited by modest samples, referral bias, a select focus on people with prevalent disease or use of less robust measures of fish oil intake.
In the present investigation, we assessed RBC omega-3 content, which not only is a reliable measure of long-term circulating PUFA levels, it also very closely mirrors cardiac omega-3 status, and therefore may translate to a more direct relation with cardiac structure and function than other omega-3 measures. 51 However, our data showed no statistically significant association of RBC omega-3 content with echocardiographic traits, questioning a relevant impact of circulating omega-3 levels on cardiac function and structure in the general population.
Linoleic acid and cardiac structure and function
The cardiovascular effects of omega-6 fatty acids (with its main component being linoleic acid) are controversial. Theoretical considerations led to the hypothesis that linoleic acid may have detrimental effects because it inhibits endogenous EPA production (competing for the enzyme that converts plant alpha-linoleic acid to EPA). 18 Evidence from prospective cohorts, cross-sectional analyses, and experimental studies in humans and animals together supports a protective role of linoleic acid (as summarized elsewhere 18 ). The recent reanalysis of the Sydney Heart Study noted above questioned this notion, 19 but the dietary conditions in that study were quite extreme (15% energy from linoleic acid and very low alpha-linolenic acid) and may not reflect the physiological effects of the 5–7% of energy typically provided by linoleic acid in the USA. 52
Data regarding the relations of linoleic acid with cardiac structure and function are sparse. Sundström et al. reported that higher plasma linoleic acid levels were associated with lower prevalence of LV hypertrophy 20 years later. 17 In hypertensive heart failure rats, a linoleic acid-supplemented high-fat diet improved survival as compared to conventional high fat (lard) or a normal diet, but there were no echocardiographic differences between animals fed a high linoleic acid diet and those fed a conventional high-fat diet, questioning a specific effect of linoleic acid on cardiac structure and systolic function. 53 In our data, linoleic acid did not correlate with LV structure or function, but was moderately and inversely associated with LA dimension. Since LA size is a risk factor for atrial fibrillation, linoleic acid may protect against the development of atrial fibrillation, a premise that warrants further investigation and replication, given this finding in secondary analyses of our data.
Trans fatty acids
Despite the well-documented increase in cardiovascular risk associated with trans fatty acids, 22 we are not aware of any previous epidemiological study investigating their relation with vascular stiffness and function traits, or with cardiac structure and function. Trans fatty acids reduce endothelial nitric oxide production in vitro, 54 and thus may have detrimental effects on vascular function. However, our data do not support a statistically significant association of long-term trans fatty acid intake with vascular function (as assessed by PAT) in our sample.
Limitations
Some limitations of our study should be mentioned. Our investigation is observational and cross-sectional, and causal inferences cannot be made. RBC fatty acid composition was measured only once, which may introduce random error into our analyses, thereby biasing our results toward the null hypothesis of no association between fatty acid levels and tonometry and echocardiographic traits. Last, the approach of multivariable adjustment has to be discussed. It is possible that the impact of PUFA on vascular stiffness, cardiac geometry and LV function operates through pathways for which we adjusted, such as blood pressure, heart rate and lipid profile. Hence, our models may be deemed to be over-adjusted, thereby resulting in potential underestimation of the relations between PUFAs and the tonometry and echocardiography traits. We studied middle-age and older adults, who were predominantly of European origin. Our large sample size was well powered to detect even modest correlations of PUFAs with vascular and echocardiographic measures (see Methods for details). Yet, we likely lacked sufficient power to examine specific non-white ethnicities/race variation, and our findings may not generalize to younger individuals.
Conclusion
In our community-based sample, we observed an inverse association between RBC omega-3 content and aortic stiffness, consistent with a vasculoprotective effect of omega-3 fatty acids. In secondary analyses, we observed a protective association between RBC linoleic acid content and left atrial size, a finding that warrants replication. The clinical significance of these modest associations remains to be elucidated.
Footnotes
Declaration of conflicting interest
WSH is a scientific advisor to companies with interests in fatty acids including Monsanto, Aker BioMarine, Omthera Pharmaceuticals, Amarin and GlaxoSmithKline, and was a speaker for the latter. In addition, he is the owner of OmegaQuant Analytics, LLC and an employee of Health Diagnostics Laboratory, Inc., both of which offer blood fatty acid testing commercially. GFM is owner of Cardiovascular Engineering Inc. (a company that develops and manufactures devices to measure vascular stiffness) and serves as a consultant to Novartis and Merck. The other authors report no potential conflicts to disclose.
Funding
This work was supported by the National Heart, Lung, and Blood Institute (NHLBI; R01 HL089590) and by contract N01-HC-25195, the Framingham Heart Study (NHLBI) and Boston University School of Medicine, and by HL076784, G028321, HL070100, HL060040, HL080124, HL071039, HL077447, HL107385, and 2-K24-HL04334.
Research materials
Underlying research materials can be made available at the Framingham Heart Study upon request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
