Abstract
Cerebral angiography is an invasive procedure utilized without supporting guidelines in preoperative evaluations of infective endocarditis (IE). It is used to identify mycotic intracranial aneurysm, which is suspected to increase the risk of intracranial bleeding during cardiac surgery. Our objectives were to: (1) assess the utility of cerebral angiography by determining which subset of IE patients benefit from its performance; and (2) identify clinical and noninvasive screening tests that can preclude the need for invasive cerebral angiography. Retrospective analysis was performed of all patients treated surgically for IE from 7/2007 to 1/2012 and discharged with medical treatment for IE from 7/2007 to 7/2009 presenting to a large academic center. Of the 151 patients who underwent cerebral angiography, mycotic aneurysm was identified in seven (prevalence=4.6%; 95% CI 2.3–9.3%). Five had viridans group streptococci as the causative IE microorganism (p=0.0017). Noninvasive imaging and particularly absence of intracranial bleed on magnetic resonance imaging conveys a negative predictive value (NPV) of 0.977 (95% CI 0.879–0.996). Absence of a focal neurologic deficit or altered mental status convey a NPV of 0.990 (95% CI 0.945–0.998) and 0.944 (95% CI 0.883–0.974), respectively. Clinical suspicion for mycotic aneurysm and thus utilization of cerebral angiography is likely necessary only in the setting of acute neurologic deficits and when noninvasive imaging demonstrates acute intracranial bleed. A novel association between viridans group streptococci and intracranial mycotic aneurysm is demonstrated.
Introduction
Guidelines supporting the use of cerebral angiography to identify mycotic intracranial aneurysm in the preoperative evaluation of infective endocarditis (IE) are intentionally vague. The 2011 Society of Thoracic Surgeons clinical practice guideline states only that it is ‘reasonable’ to reserve catheter-based angiography for IE patients with evidence of intracranial bleeding or a suggestion of mycotic aneurysm on noninvasive vascular imaging (Class IIa, Level of evidence C). 1 The pooled American Heart Association-Infectious Diseases Society of America 2005 guidelines on endocarditis delineate the weaknesses of noninvasive imaging techniques and state only that cerebral angiography is indicated only when noninvasive studies are negative and ‘suspicion remains’ (no indication Class or Level of evidence given). 2 The European Society of Cardiology 2009 guidelines state ‘conventional angiography remains the gold standard and should be performed when non-invasive techniques are negative and suspicion remains. No randomized trials exist to guide management, and therapy must be tailored to the individual patient’. 3 But it remains unclear what should raise suspicion and how we define negative noninvasive techniques. Equally unclear is how therapy should be tailored. Cerebral angiography is an invasive procedure with definite associated risks and costs. 4 Mycotic aneurysms, if undiagnosed, are also associated with the risk of bleeding and stroke given the full anticoagulation required during cardiothoracic surgery.
The potential neurologic devastation of intracranial bleeding during cardiac surgery drives the utilization of cerebral angiography. The goal of cerebral angiography is to identify infectious mycotic aneurysm, a pathology originally described by Sir William Osler who coined the term ‘mycotic aneurysm’ meant to describe the mushroom shape of the infectious aneurysms he initially noted in the aorta. 5 There is no conclusive data supporting the belief that mycotic aneurysm increases the risk of bleeding when IE patients are subjected to full anticoagulation during cardiac surgery. Nonetheless, these aneurysms represent defects in the vascular wall and may theoretically increase bleeding risk in the perioperative period. The neurologic sequelae of IE, including development of mycotic aneurysms, are well documented, with prevalence of intracranial mycotic cerebral aneurysm ranging in registry data from 2% to 5% (depending on how cerebral angiography and intracranial imaging were utilized in the given populations).6–9 Which IE patients are at highest risk for mycotic aneurysm and in which subset cerebral angiography has the highest yield is unknown. Furthermore, the utility of noninvasive clinical or imaging tests in predicting the presence of mycotic aneurysm in preoperative IE patients is not established.
Methods
Study design
The Cleveland Clinic Infectious Endocarditis Registry was created by screening all patients who underwent cardiac surgery with a diagnosis of IE between 1 July 2007 and 31 December 2011 at the Cleveland Clinic, a single large academic referral center, and all patients who were discharged from the Cleveland Clinic on intravenous antibiotic therapy for the treatment of IE between 1 July 2007 and 30 June 2009. All patients were screened for elements of Duke criteria and only patients who met criteria for definite endocarditis were included. A total of 554 patients were included in the registry, of which 484 underwent cardiac surgery.
Three methods were used to identify all patients who underwent cerebral angiography. First, CPT (Current Procedural Terminology) codes for cerebral angiography were identified; then a meticulous medical record review was performed on all registry patients to determine if cerebral angiography was performed during the index hospitalization (either at the Cleveland Clinic or at a referring hospital) as part of IE management. ICD-9 codes for mycotic aneurysm were then searched to confirm that all patients with identified mycotic aneurysm were included in the cohort. Review of cerebral angiography reports was performed by a physician blinded from other clinical knowledge about the patient. There were 151 patients who underwent cerebral angiography. The frequency of performing cerebral angiography increased dramatically, particularly in the population that eventually underwent cardiothoracic surgery for IE: from 15% in the first half of 2007 to almost 40% in the second half of 2011 (Figures 1 and 2). At the Cleveland Clinic, every patient presenting with IE is evaluated by a combined clinical team including expert consultation in infectious disease, cardiovascular medicine, and cardiothoracic surgery. Neurology consultation was performed when any clinical or noninvasive evidence of neurologic pathology was present. The decision to proceed with cerebral angiography was typically made by consensus expert opinion among this infective endocarditis team. The retrospective nature of this work led to some clinician-dependent variability in interpretation of the available guidelines and thus practice patterns.
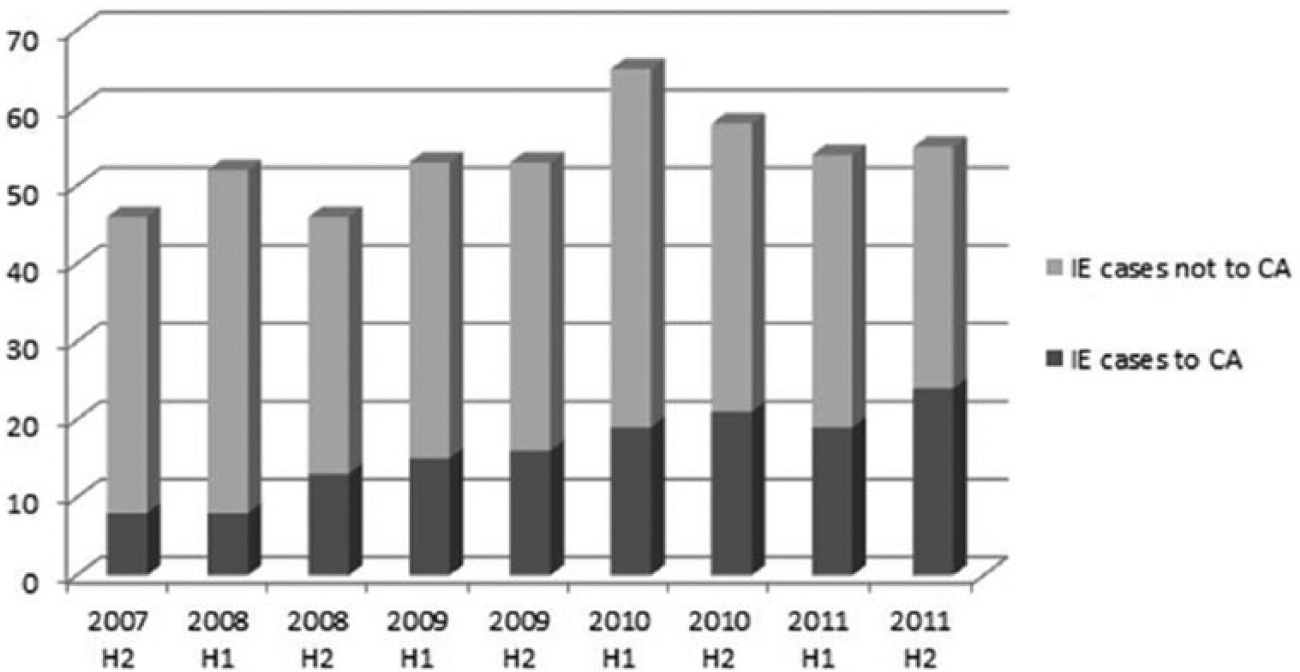
Graphical representation of the volume of surgically treated cases of infective endocarditis (IE) and identification of those evaluated with cerebral angiography (CA) between 1 July 2007 and 31 December 2011 as collected within the Cleveland Clinic Infective Endocarditis Registry. (H1, first half; H2, second half.)
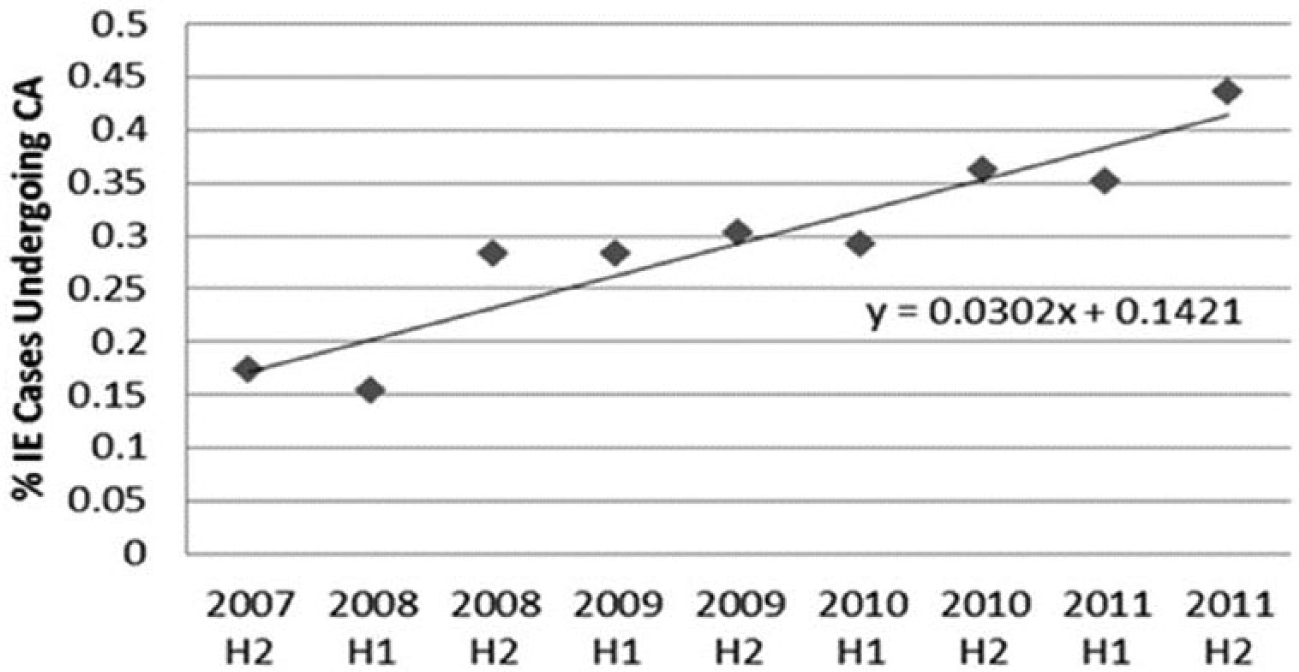
As a result of expert consensus opinion between neurology, infectious disease, cardiovascular medicine and cardiothoracic surgery at the Cleveland Clinic, the frequency of use of cerebral angiography increased significantly during the assessed time interval. (CA, cerebral angiography; H1, first half; H2, second half; IE, infective endocarditis.)
Imaging data
The electronic medical record was utilized to perform meticulous chart review of each record to assess for performance and results of noninvasive imaging studies, particularly computed tomography (CT), computed tomography angiography (CTA), magnetic resonance imaging (MRI), and magnetic resonance angiography (MRA). In patients who underwent surgery during their hospitalization, only imaging studies performed prior to surgery were evaluated. Review of noninvasive imaging reports was performed by a physician blinded from knowledge of the performance or results of cerebral angiography. In patients who underwent cerebral angiography, only imaging studies performed and interpreted prior to the date of cerebral angiography were utilized to avoid inclusion of noninvasive results biased by cerebral angiography data. Reports of studies performed at the Cleveland Clinic were directly evaluated and results of studies performed at a transferring institution were determined from chart review of admission and hospitalization documentation, including neurology team consultation. Of the 554 patients, 271 had at least CT, 108 had at least MRI, and 322 had either CT or MRI performed. Any studies documenting ‘acute’ or ‘subacute’ embolic pathology were adjudicated as positive for embolic stroke. Any studies documenting intracranial bleed were adjudicated as positive for bleed. Chronic vascular pathology was not adjudicated as a positive neurologic finding.
Neurologic data
The electronic medical record was utilized to obtain evidence of neurologic pathology, including altered mental status and focal neurologic deficit. Review of admission and hospitalization documentation including neurologic and infectious disease consultation was performed. Adjudication for altered mental status was deemed positive if clinical documentation of altered consciousness was present in the absence of medical sedation/iatrogenic causes (i.e. in intubated patients mental status was evaluated during ‘sedation holidays’). Adjudication for discrete neurologic symptoms was deemed positive if clinical documentation of discrete neurologic pathology was present on record review (most frequently with confirmation by expert neurologic consultation). Review of clinical documentation of neurologic pathology was blinded from knowledge of performance or results of cerebral angiography.
Statistical methods
Statistical comparisons were performed using basic chi-squared and t-tests. Fisher’s exact test was utilized in comparison of mycotic aneurysm positive versus mycotic aneurysm negative groups in the setting of the low number of positive outcomes demonstrated. Negative predictive values (NPVs) and likelihood ratios were also calculated.
Results
The baseline clinical characteristics were clinically and statistically similar between the group that underwent cerebral angiography and those that did not undergo cerebral angiography (Table 1). This includes similarities between the percentage of patients represented in each group with prosthetic valve endocarditis and the percentage of patients with underlying immunocompromised state.
The baseline clinical characteristics of all registry patients that did and did not undergo cerebral angiography.
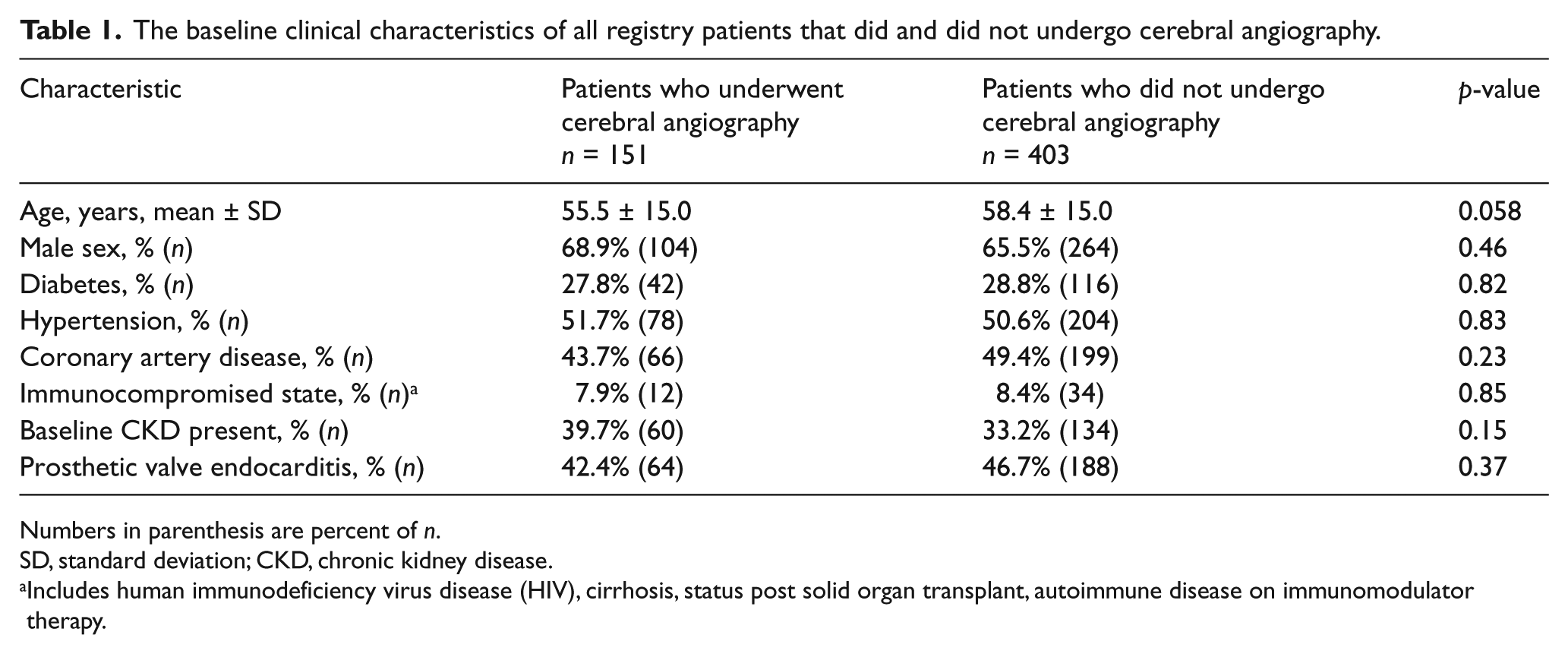
Numbers in parenthesis are percent of n.
SD, standard deviation; CKD, chronic kidney disease.
Includes human immunodeficiency virus disease (HIV), cirrhosis, status post solid organ transplant, autoimmune disease on immunomodulator therapy.
Those who underwent cerebral angiography were more likely to have undergone noninvasive neurologic imaging and to have pathologic findings on noninvasive imaging (Table 2). Significant differences also existed between the percentage of patients with focal neurologic symptoms and altered mental status among those who underwent cerebral angiography compared to those who did not.
Comparison of neurologic symptoms and utilization of noninvasive imaging between registry patients who did and did not undergo cerebral angiography.
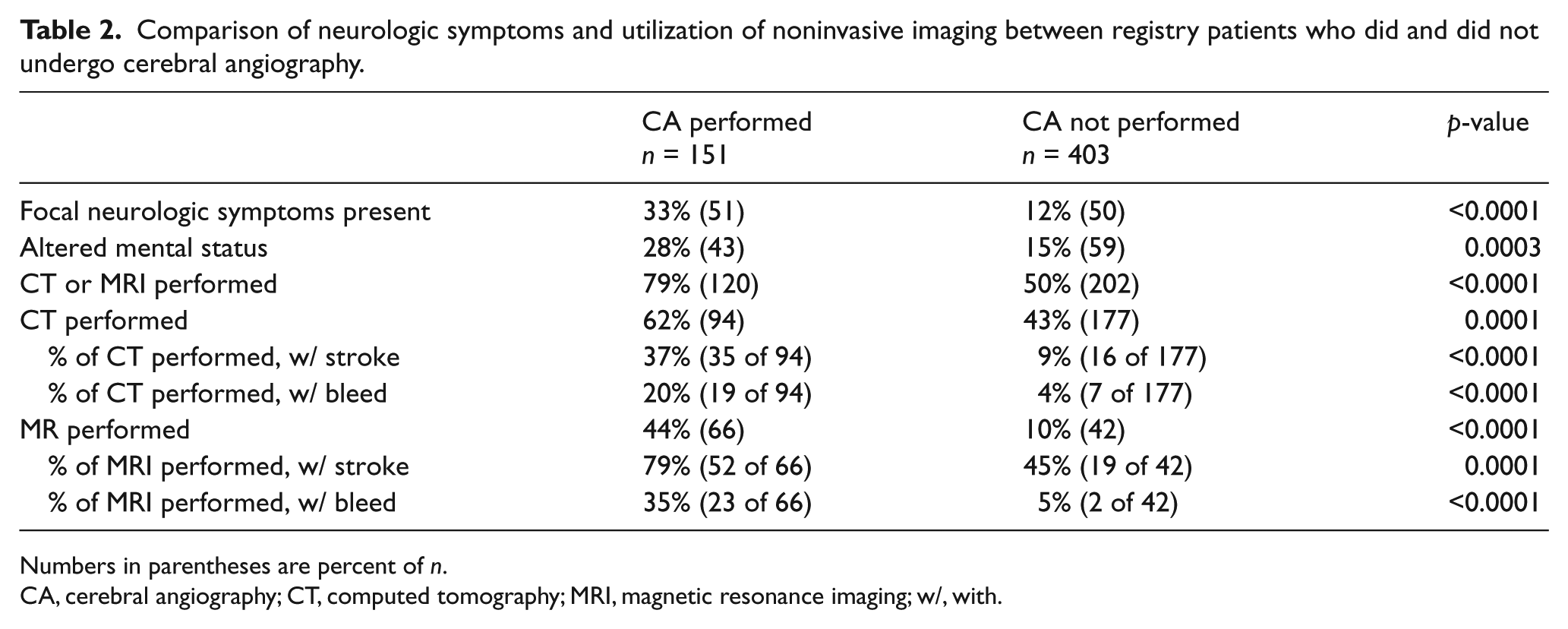
Numbers in parentheses are percent of n.
CA, cerebral angiography; CT, computed tomography; MRI, magnetic resonance imaging; w/, with.
Of the 151 patients assessed with cerebral angiography, seven (4.6%, 95% CI 2.3–9.3%) had mycotic aneurysm. Thus, 4.6% does not represent the prevalence of mycotic aneurysm in all patients presenting with IE. The overall prevalence of mycotic aneurym in all IE patients remains unknown simply because all IE patients were not assessed in this study with the gold standard of cerebral angiography. Rather, 4.6% represents the prevalence of mycotic aneurysm in the patients deemed high enough risk to be recommended for cerebral angiography by the multi-specialty IE team based on the limited currently available data and guidelines. Most baseline clinical characteristics were similar between those patients with and without mycotic aneurysm (Table 3). However, of the seven patients, five had viridans group streptococci as the causative organism of IE (Fisher’s exact test, p=0.0017); the other two patients had slow-growing microorganisms (HACEK and mold) (Table 3).
Comparison of clinical characteristics within the group that underwent cerebral angiography between those positive and negative for mycotic aneurysm.
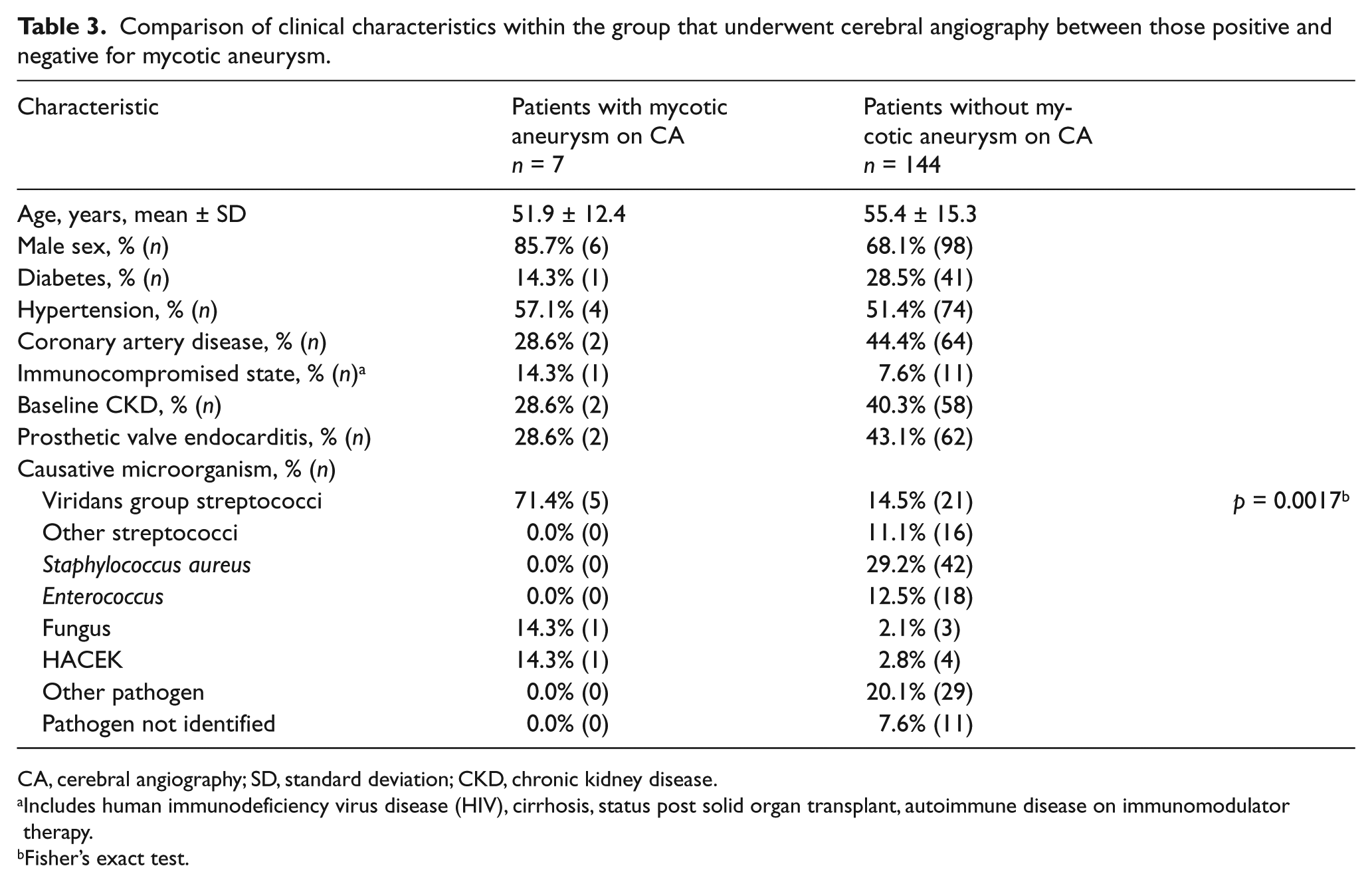
CA, cerebral angiography; SD, standard deviation; CKD, chronic kidney disease.
Includes human immunodeficiency virus disease (HIV), cirrhosis, status post solid organ transplant, autoimmune disease on immunomodulator therapy.
Fisher’s exact test.
Of the 151 patients assessed with cerebral angiography, 120 patients had CT or MRI or both performed. Only one had mycotic aneurysm directly documented by noninvasive imaging. The sensitivity of noninvasive imaging (CT or MRI) for direct documentation of mycotic aneurysm was thus very low at 0.17 (95% CI 0.01–0.64). The one patient with mycotic aneurysm demonstrated on noninvasive imaging had a particularly large mycotic aneurysm (3.4 × 3.4 mm) by comparison to the other aneurysms demonstrated in the group (n=6). All other mycotic aneurysms identified on cerebral angiography were smaller than 3 mm.
Despite low sensitivity to directly detect small mycotic aneurysms, nearly all patients with identified mycotic aneurysm on cerebral angiography did have pathologic findings of stroke or bleed on noninvasive CT or MRI, resulting in a good NPV for noninvasive imaging (Figure 3). This was particularly true for evidence of bleed on MRI, the absence of which had a NPV of 0.977 (95% CI 0.879–0.996). Of the 151 patients who underwent cerebral angiography, 51 patients had a focal neurologic symptom and 43 patients had altered mental status present prior to angiography. The NPV for the presence of mycotic aneurysm in the absence of a focal neurologic deficit in this population was 0.990 (95% CI 0.945–0.998) and that of absence of altered mental status was 0.944 (95% CI 0.883–0.974).
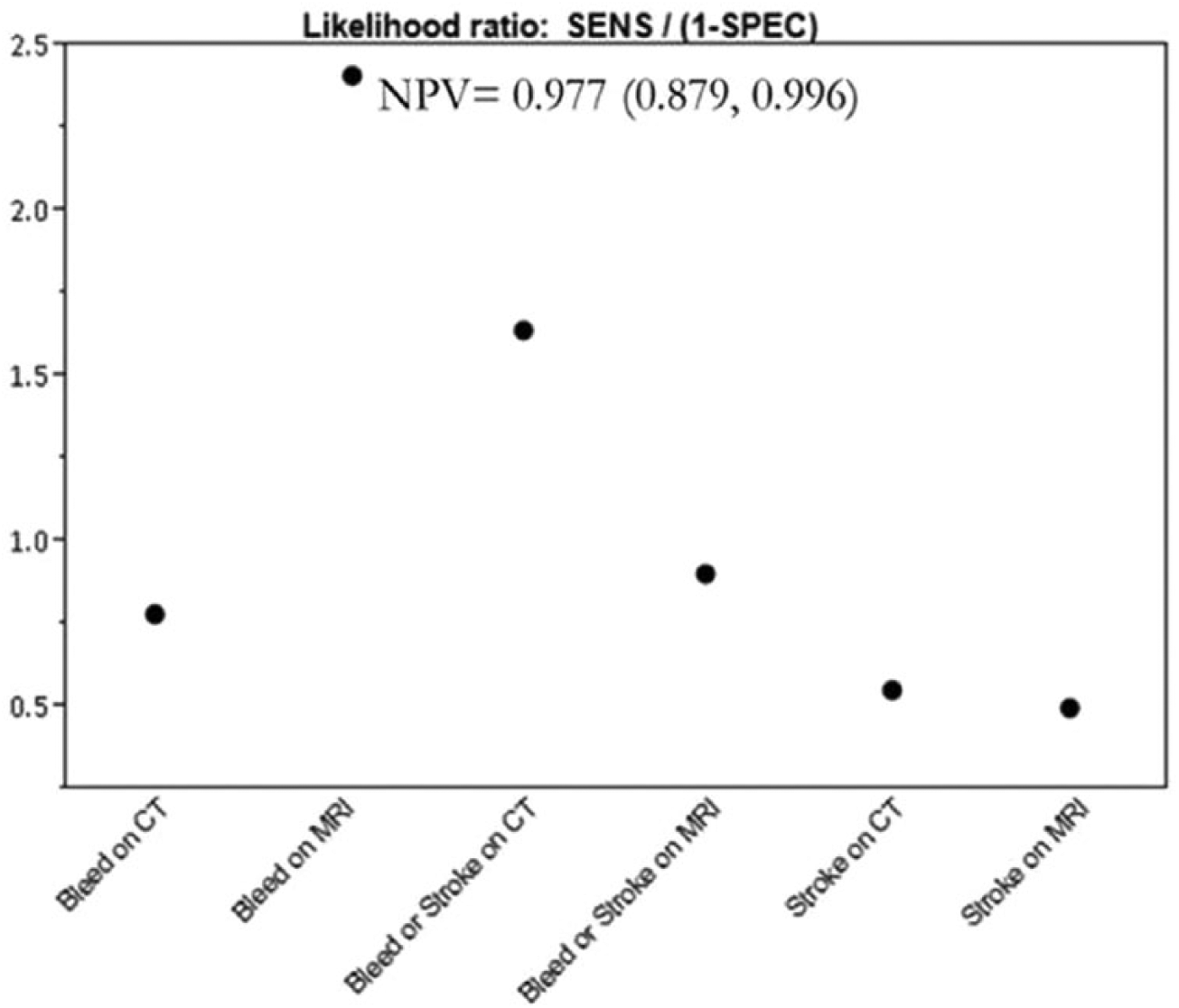
Likelihood ratios for stroke or bleed on CT or MRI and presence of mycotic aneurysm. Also noted is the NPV of bleed on MRI. (SENS, sensitivity; SPEC, specificity; CT, computed tomography; MRI, magnetic resonance imaging; NPV, negative predictive value.)
Of the seven patients with mycotic aneurysm, four underwent therapeutic embolization and one underwent repeat angiography to demonstrate aneurysm stability prior to open heart surgery (OHS). Six of the seven underwent cardiac surgery without any neurologic complications and one was refused surgery secondary to multiple coexistant co-morbidities. Of the six cardiac surgery patients, five were discharged from the hospital alive and there was one in-hospital death secondary to postoperative bowel ischemia and sepsis.
Discussion
In this, the largest-to-date assessment of cerebral angiography for mycotic aneurysm in the setting of IE, we found that cerebral angiography is most effectively utilized in patients with evidence of intracranial pathology on noninvasive imaging studies or in patients with evidence of neurologic pathology on clinical exam at presentation.
We demonstrate a low sensitivity for noninvasive imaging for directly detecting the presence of mycotic aneurysm. The reported overall sensitivity of single-slice CTA in the detection of intracranial aneurysms in general (not necessarily mycotic) has been reported to be between 77% and 100%.10–12 This diagnostic sensitivity for noninvasive imaging decreases dramatically for small intracranial aneurysms of less than 3 mm. 13 In our cohort, only one patient with a particularly large mycotic aneurysm had the aneurysm directly identified by noninvasive imaging. We demonstrate a very high NPV of normal noninvasive intracranial imaging as well as of absence of neurologic symptoms (focal symptom or altered mental status) for presence of mycotic aneurysm. The utility of NPVs must always be viewed cautiously for a pathology with a low prevalence. For mycotic aneurysm with a prevalence of 4.6% (95% CI 2.3–9.3%), these NPVs appear valid. NPVs were calculated using only the patients who underwent both cerebral angiography and noninvasive imaging tests (CT or MRI). Of the patients who did not undergo cerebral angiography, 23 had evidence of stroke or bleed on CT and 21 had evidence of stroke or bleed on MRI. Gold standard cerebral angiography data are by definition unavailable for these patients and thus they could not be included in NPV calculations. However, the IE patients who did undergo cerebral angiography were those deemed by the multidisciplinary heart team to be at elevated risk of mycotic aneurysm by clinical consensus. Therefore, the derived NPVs would likely prove even higher for noninvasive imaging and clinical tests used to screen all IE patients (a population that would be suspected to have a lower total prevalence of mycotic intracranial aneurysm).
In 1987, Salgado et al. compared the clinical course of 68 patients with IE with mycotic aneurysm and 147 with IE but no mycotic aneurysm. 14 Subarachnoid hemorrhage (SAH) or other intracranial hemorrhage (ICH) was noted in 57.4% of the patients with mycotic aneurysm in this study (as noted on cerebral angiography, at the time of surgery or at autopsy) and in 0% of patients without known mycotic aneurysm. This work was limited in that the patients who developed neurologic pathology were evaluated more thoroughly for mycotic aneurysm. Also, all patients in the study were not evaluated with the gold standard of cerebral angiography. This could have led to the development of selection bias (those developing SAH/ICH likely had more rigorous evaluation for mycotic aneurysm than those who never developed hemorrhage). It is also unclear how many of these patients underwent surgery for IE. In the setting of these limitations, the authors in this work drew the conclusion that cerebral angiography is favored for patients with a focal neurologic deficit with good recovery during the acute phase of illness. Salgado’s work displays the clear bleeding risk associated with intracranial mycotic aneurysm, a risk that only increases with addition of anticoagulation when cardiothoracic surgery is required. Our work here with cerebral angiography seeks to improve utilization of cerebral angiography, the gold standard for identifying patients with mycotic aneurysm facing the potential bleeding sequelae Selgado presented.
In 1992, van der Meulen et al. conducted a decision analysis in which they derived that cerebral angiography did not impact mortality in patients with IE and neurologic symptoms. 15 They noted that mortality was actually increased (from 0.25% to 0.35%) by the performance of cerebral angiography. This work too had limitations, comparing statistical results from various distinct types of studies and drawing conclusions from these comparisons.
Neither of these works tackled the questions of surgical morbidity and mortality necessary to take into account for a critical care population in consideration for open heart surgery. Our work acknowledges the risks involved with the invasive procedure of cerebral angiography that van der Meulen demonstrates and works to improve upon the utilization of cerebral angiography to guide its use.
Preexisting data are thus conflicting and there remain no guidelines supporting or opposing the performance of cerebral angiography in this population. Our work demonstrates that utilization of cerebral angiography is likely necessary only in the setting of acute neurologic deficits at presentation and when noninvasive imaging (including CT or MRI) demonstrate acute intracranial bleed. Optimization of the use of cerebral angiography is important because the procedure is invasive and there are definite associated risks and cost. 4 Kaufmann et al. retrospectively assessed neurologic and bleeding complications of 19,826 patients undergoing cerebral angiography for multiple indications including atherosclerotic cerebrovascular disease, tumor, and seizure. 4 They noted neurologic complications (stroke, transient ischemic attack and neurologic death) in 2.63% of these examinations, stroke with permanent disability in 0.14%, death in 0.06% and hematoma in 4.2%. Though this work was performed in a distinct patient population from that studied here, it does demonstrate the very tangible risks associated with the invasive procedure of cerebral angiography. Any of these complications could result in either delay of surgical treatment for IE or complications of cardiothoracic surgery (progression of hematoma, hemorrhagic conversion of thrombotic stroke, etc).
Sonneville et al., in 2011, in their analysis of all neurologic complications of IE, describe mycotic aneurysm in 10 patients among the 198 critically ill patients presenting with definitive left-sided IE to 23 university-affiliated and 10 general French hospitals. 6 The gold standard of cerebral angiography was used in very few of these patients and thus likely limits conclusions being drawn regarding mycotic aneurysm prevalence and clinical characteristics based on their findings. This prevalence was nonetheless quite similar to the 4.6% prevalence we demonstrated in our patient population undergoing cerebral angiography and thus helps to support the conclusions we draw about this population.
The more rigorous use of cerebral angiography in one arm of our work allows us to improve our knowledge of mycotic aneurysm prevalence and also to identify the clinical characteristics of the mycotic aneurysm patients. This includes a novel association between viridans group streptococci and mycotic aneurysm. Though based on a small cohort of patients, the finding that five of seven mycotic aneurysm patients had viridans group streptococci as the causative IE organism allows utilization of Fisher’s exact test to demonstrate this association. Staphylococcus and other more virulent organisms in general are more common causes of IE. Such organisms would likely present with acute severe valvular damage in need of treatment and repair. Conversely, less aggressive microorganisms such as viridans group streptococci would allow for a longer incubation period and thus for seeding of the vasculature and mycotic aneurysm formation. The other documented causative mycotic aneurysm organisms in our work are also slow-growing organisms: a HACEK variety, which is by definition slow growing, and a mold species. These findings are of course also subject to local microbiologic patterns. Little specific data exist regarding the microbiology of IE-associated mycotic aneurysm. A recent Spanish study also documented and discussed a similar preponderance of low-medium virulence microorganisms, including viridans group streptococci. 16
We begin to assess here as well the clinical impact of mycotic aneurysm, particularly as it relates to the performance of cardiac surgery. There is appropriate concern that the presence of mycotic aneurysm could affect the surgical risks of bleeding with anticoagulation during surgery, and thus affect both post-surgical neurologic outcomes and mortality. In the young population that often presents with endocarditis (in our work the average age of those with mycotic aneurysm is 51 ± 12.4 years), the morbidity associated with intracranial hemorrhage and resultant neurologic debility could be devastating. We demonstrate that six of the seven mycotic aneurysm patients in our series underwent OHS with full heparinization and without any neurologic bleeding. Four of the six underwent angiographically guided aneurysm embolization prior to cardiac surgery, one underwent reevaluation with repeat angiography at 2 weeks to demonstrate stability of the aneurysm, and one underwent no further treatment prior to OHS.
There are obvious limitations to this work. It is a retrospective, single-center study. The limited number of outcomes is prohibitive of the performance of propensity analysis or of the development of formal prediction models to further improve our ability to predict which patients are more likely to develop mycotic aneurysm in the setting of IE and thus improve the yield of invasive procedures such as cerebral angiography. Furthermore, this retrospective study did not represent the performance of cerebral angiography on all patients presenting with IE. Rather, only patients deemed at highest risk and thus clinically suitable for cerebral angiography by the multi-specialty IE team underwent the gold standard evaluation for mycotic aneurysm. Nonetheless, we can draw important conclusions from this large clinical series. Our study revealed an association between slow-growing organisms and mycotic aneurysm. Most importantly, it demonstrates that a normal neurologic examination as well as absence of intracranial bleed on noninvasive imaging tests can serve as good screening tests for mycotic aneurysm and thus safely eliminate the need for the performance of cerebral angiography in the majority of preoperative IE patients.
Footnotes
Declaration of conflicting interest
The authors have no conflict of interest relevant to this work. Dr Shishehbor is an educator and consultant to Abbott Vascular, Medtronic, Cordis, Covidien, Spectranetics, CSI, Bayer, and Cook, but has waived all personal compensation for this work in the past 4 years.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
