Abstract
Chronic mesenteric ischemia (CMI) is a rare condition that is usually the result of atherosclerotic obstructive disease affecting the mesenteric arteries. The classic triad of post-prandial pain, food aversion and weight loss is not always present, often leading to low clinical suspicion for CMI and underdiagnosis. Non-invasive evaluation for CMI usually starts with mesenteric arterial duplex scanning, followed by computed tomography angiography, magnetic resonance angiography or conventional angiography, the latter being the gold standard for establishing its diagnosis. However, angiography alone has been demonstrated in coronary and other vascular beds to be inaccurate in predicting the physiologic and hemodynamic significance of a certain subset of atherosclerotic stenoses. We present the case of a patient with risk factors and symptoms suggestive of CMI who underwent angiography. However, angiography was equivocal and invasive physiologic testing was required to confirm the diagnosis and guide revascularization.
Introduction
It has been previously documented that both qualitative (visual) and quantitative estimations of percent stenosis severity can underestimate the functional significance of a vascular stenosis. The physiologic significance of a vascular stenosis can be better assessed by the demonstration of a threshold pressure gradient across the narrowing – the higher the gradient the more functionally significant the stenosis. The traditional technique of invasive assessment of a gradient in the non-coronary bed has been to use an end-hole catheter advanced across the stenosis. When applied to smaller caliber arteries this technique is not very accurate, and is potentially harmful as the measuring catheter may itself physically obstruct the flow and thereby lead to an overestimation of the gradient. 1 Instead, the technique of fractional flow reserve (FFR) assessment with a 0.014” pressure wire as used in the coronary circulation can be extrapolated to the visceral arteries in order to precisely assess the physiologic significance of a lesion, as highlighted in renal artery stenosis intervention. Additionally, the use of a 0.014” guidewire as opposed to a 4-French catheter helps to minimize the potential for catheter-induced vessel trauma, including dissection and atheroembolization.
Clinical vignette
A petite 62-year-old female initially presented with an acute myocardial infarction, requiring emergent coronary artery bypass grafting due to underlying left main and multi-vessel coronary artery disease. After recovery, she underwent endovascular intervention of the distal abdominal aorta and bilateral common iliac arteries for severe obstructive disease causing disabling intermittent claudication, which resolved after intervention. During this procedure her inferior mesenteric artery (IMA), which had a severe ostial stenosis, was covered with an aortic endograft resulting in its occlusion.
Four weeks after the aorto-iliac intervention, the patient developed complaints of post-prandial abdominal discomfort. She would develop epigastric pain radiating to the right upper quadrant, followed by nausea and vomiting after meals. These prompted a gastrointestinal evaluation which was fairly unremarkable with the exception of revealing gallstones without evidence of cholecystitis. Given the patient’s significant history of extensive peripheral artery disease, chronic mesenteric ischemia (CMI) was considered a likely culprit. Abdominal computed tomography angiography (CTA) was performed, which did not reveal a high-grade stenosis affecting the celiac axis (CA) and superior mesenteric artery (SMA) (Figure 1). It was noted that the IMA was covered at its origin, with no contrast opacification demonstrated on the CTA. Given the high index of clinical suspicion, the patient was referred for mesenteric angiography.
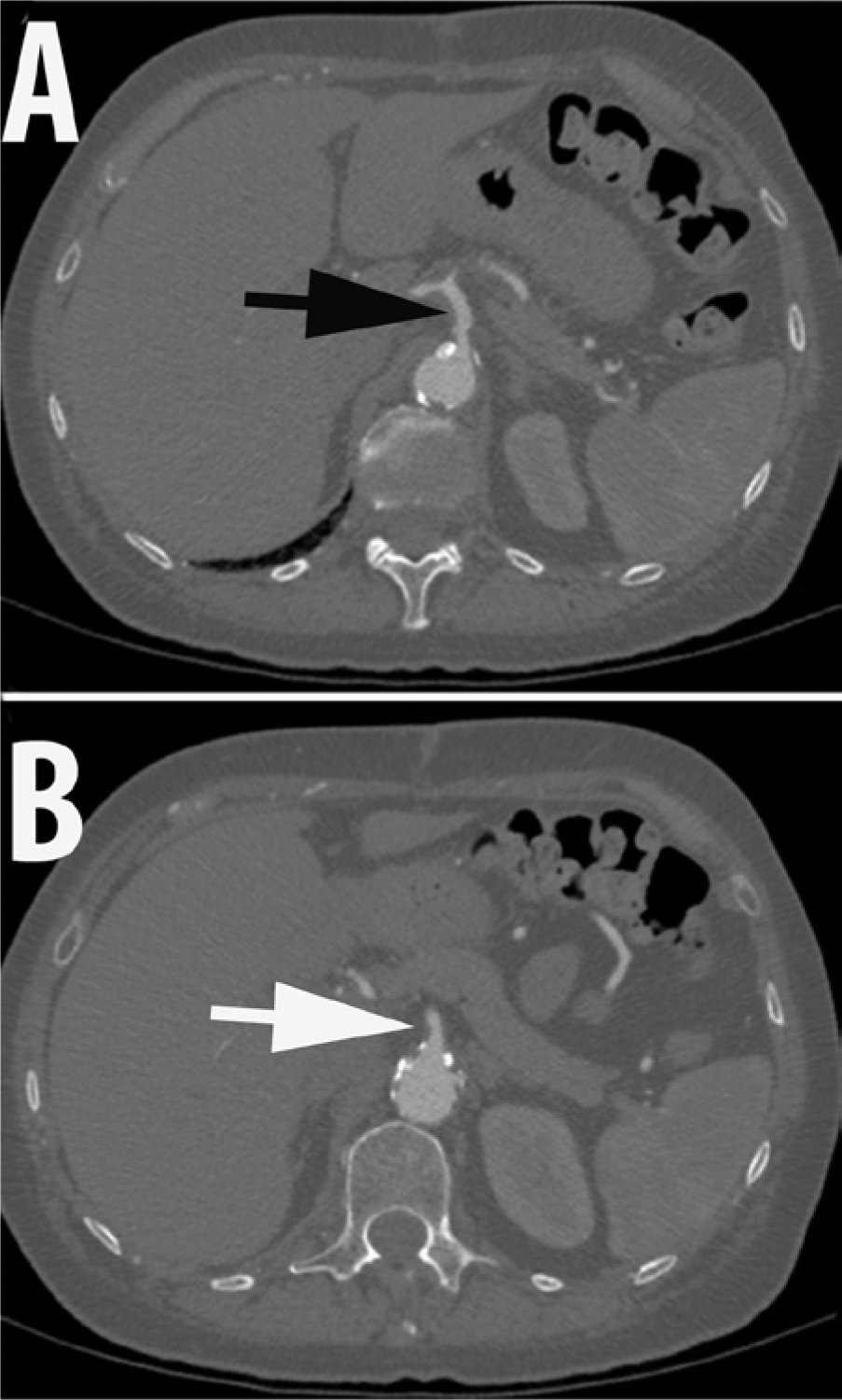
Computed tomography angiography of the abdomen ((A) Black arrow: celiac artery; (B) White arrow: superior mesenteric artery).
Abdominal aortography and selective angiography of the CA and SMA were performed in anterior-posterior and lateral projections. Angiography demonstrated mild to moderate disease in the proximal segment of the CA, and a focal severe stenosis in the proximal splenic artery (Figures 2 and 3). The SMA had moderate disease in the ostial and proximal segments, with brisk reflux of dye into the aorta. The SMA was noted to provide robust collaterals to the IMA via the arc of Riolan (also known as the ‘meandering’ artery) as well as the marginal artery of Drummond. The IMA had no antegrade flow.
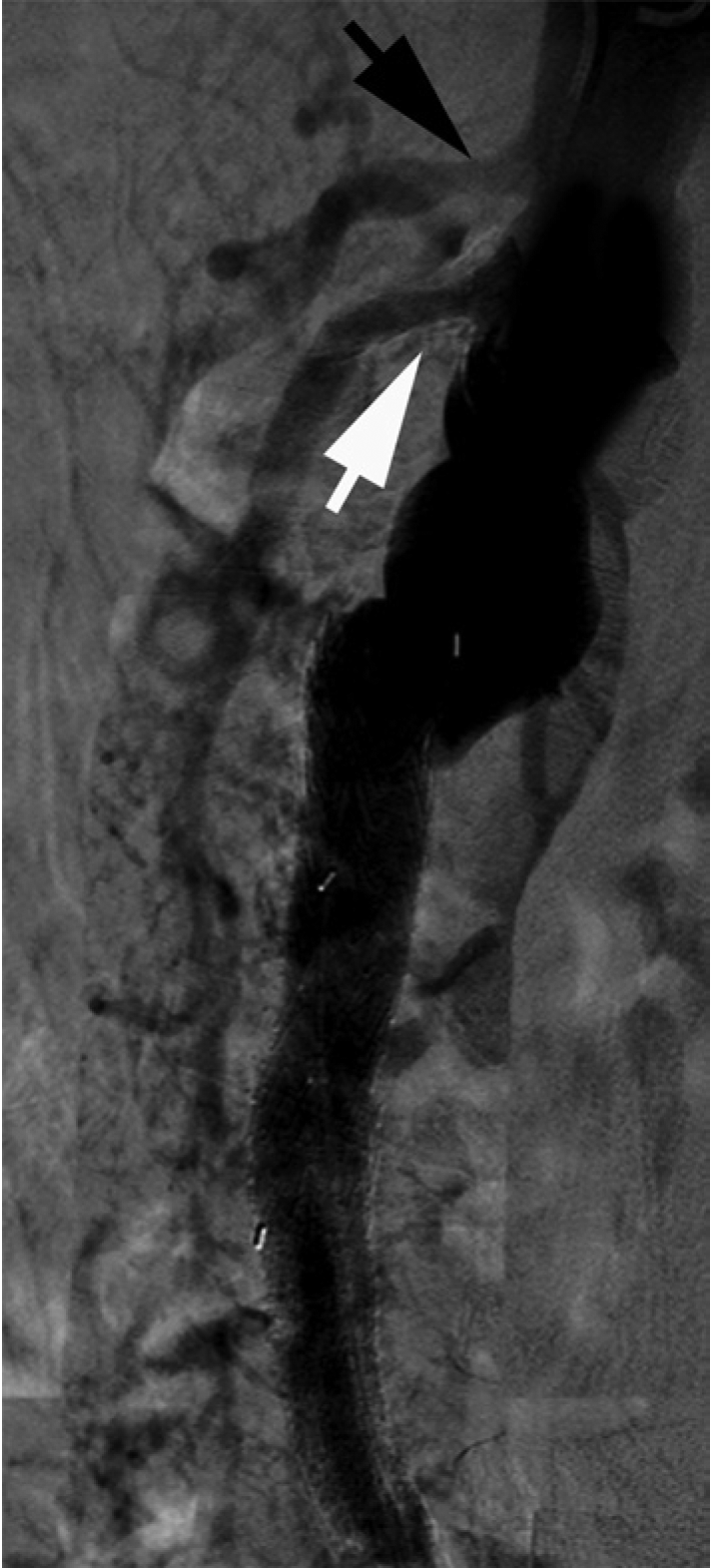
Abdominal aortography, lateral projection (black arrow: celiac artery; white arrow: superior mesenteric artery).
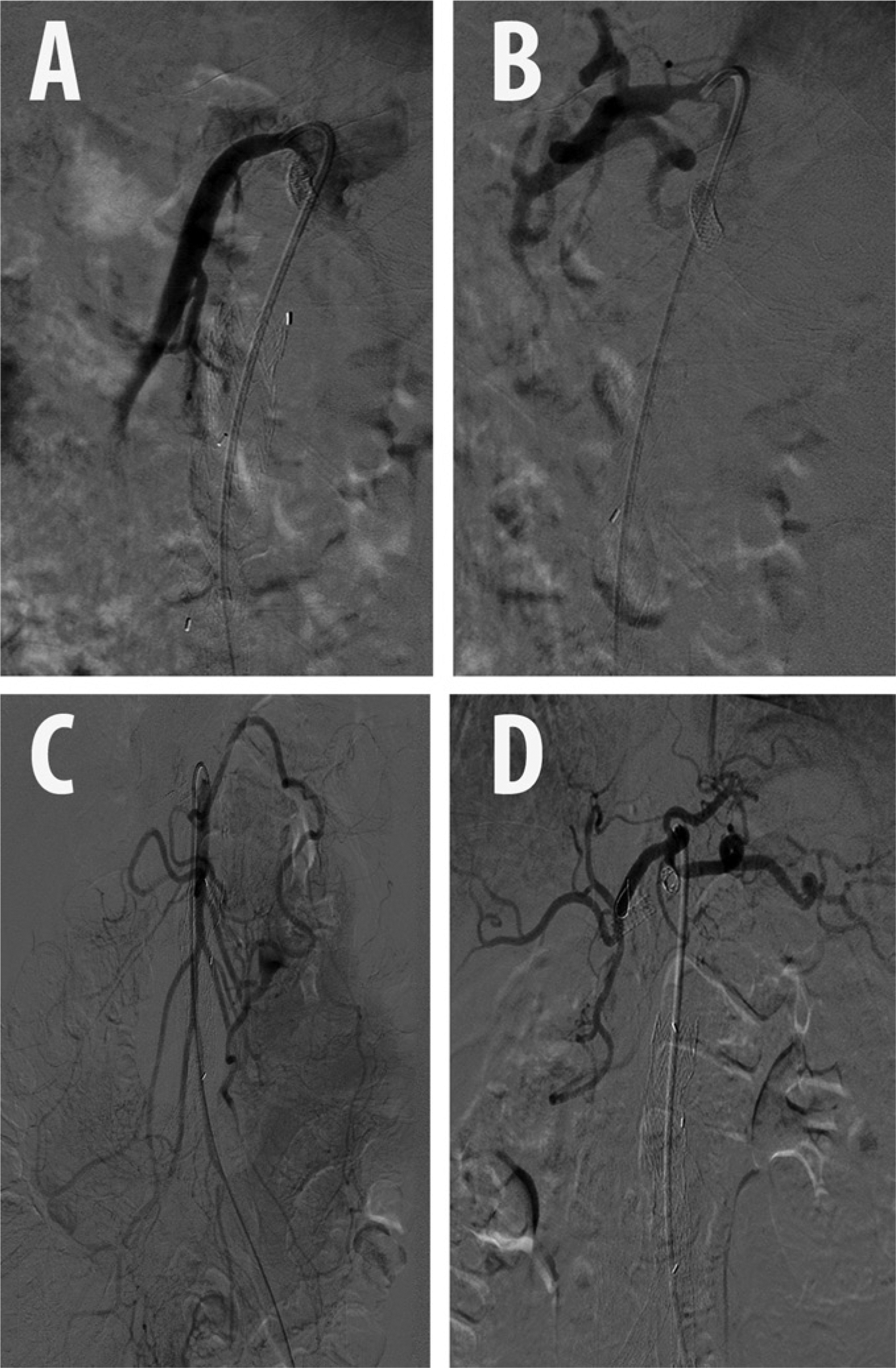
Pre-stenting angiography: (A) superior mesenteric artery, lateral; (B) celiac artery, lateral; (C) superior mesenteric artery, anterior-posterior; (D) celiac artery, anterior-posterior.
The hemodynamic significance of the stenoses affecting the SMA and CA was assessed sequentially using a 0.014” 175 cm pressure wire (PressureWireTM; St Jude Medical Inc., St Paul, MN, USA). A 6-French IM guiding catheter was used to engage the ostium of the SMA in the lateral projection. The pressure wire was advanced into the ostium of this vessel. While the transducer of the wire (3 cm proximal from the distal tip) was still in the aorta and outside the ostium of the SMA, the guiding catheter was withdrawn 1 cm and the pressures were equalized. The guiding catheter was re-engaged and the pressure wire was advanced into the proximal SMA. A baseline pressure gradient of 14 mmHg (FFR=0.87) was recorded (Figure 4A). After this, 300 µg of intra-arterial (IA) nitroglycerin (NTG) was injected into the SMA through the guiding catheter. A hyperemic pressure gradient of 37 mmHg (FFR=0.65) was recorded (Figure 4B and 4C). Since this finding was unexpected, an Eagle Eye® Platinum intravascular ultrasound (IVUS) catheter (Volcano Corp., San Diego, CA, USA) was used to evaluate the proximal SMA to identify the offending stenosis (Figure 5). The IVUS revealed moderate diffuse plaque in the proximal SMA with a focal, concentric, severe stenosis near the ostium of this vessel. Moderate calcification was noted as well. The minimal luminal diameter was noted to be 4 mm, while the reference vessel diameter was 9 mm. Given the FFR and IVUS data, the patient underwent stenting of the ostial/proximal segment of the SMA with a balloon-expandable 7.0 mm × 12 mm Palmaz® Blue® (Cordis Corporation, Bridgewater, NJ, USA) stent. Upon repeat assessment, the FFR was 0.95 without hyperemia, and 0.85 (Figure 6) with IA NTG, showing a significant improvement post-intervention.
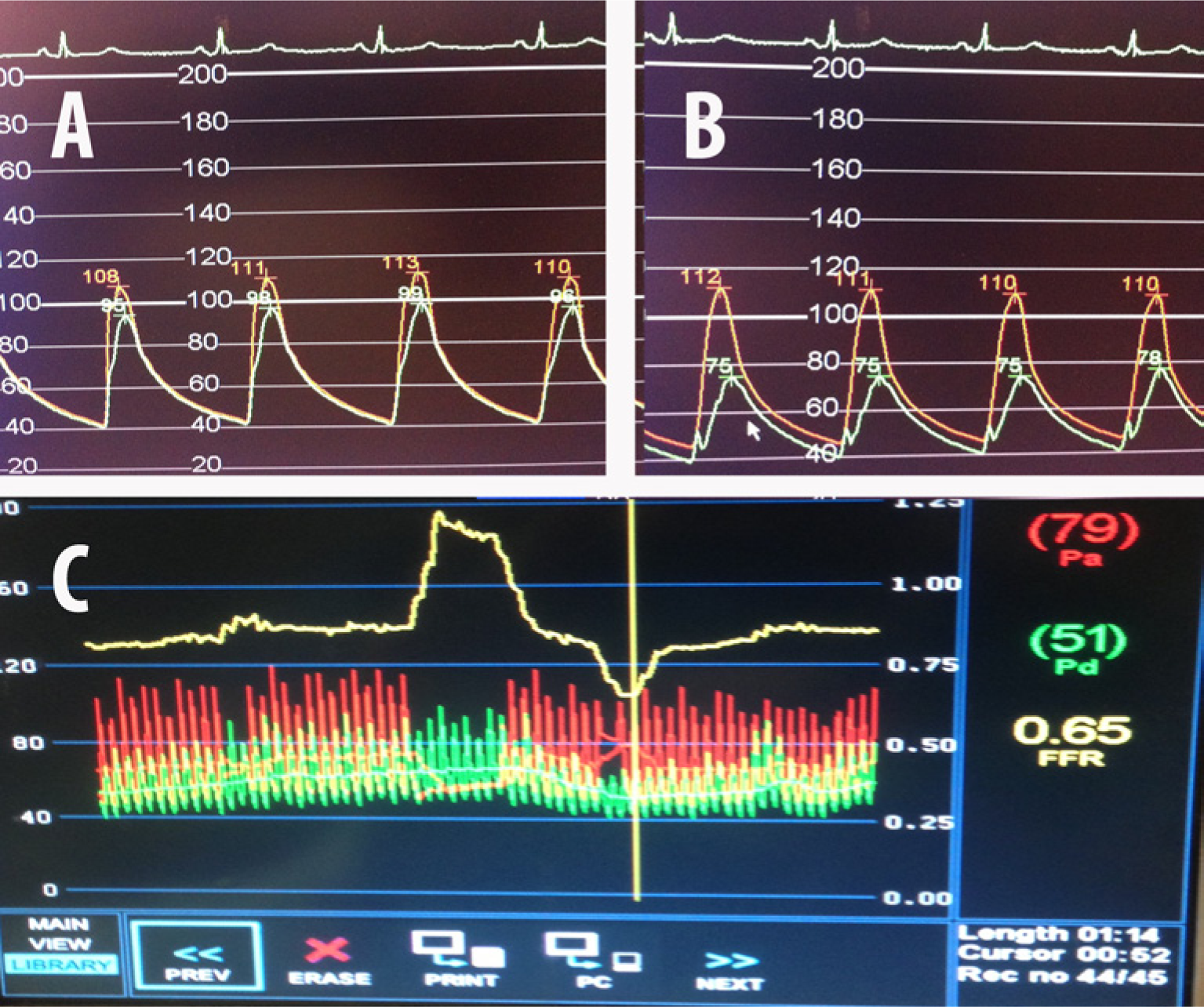
Superior mesenteric artery FFR prior to stenting: (A) without intra-arterial nitroglycerin, FFR=0.87; (B) after intra-arterial nitroglycerin; (C) composite FFR post nitroglycerin = 0.65.
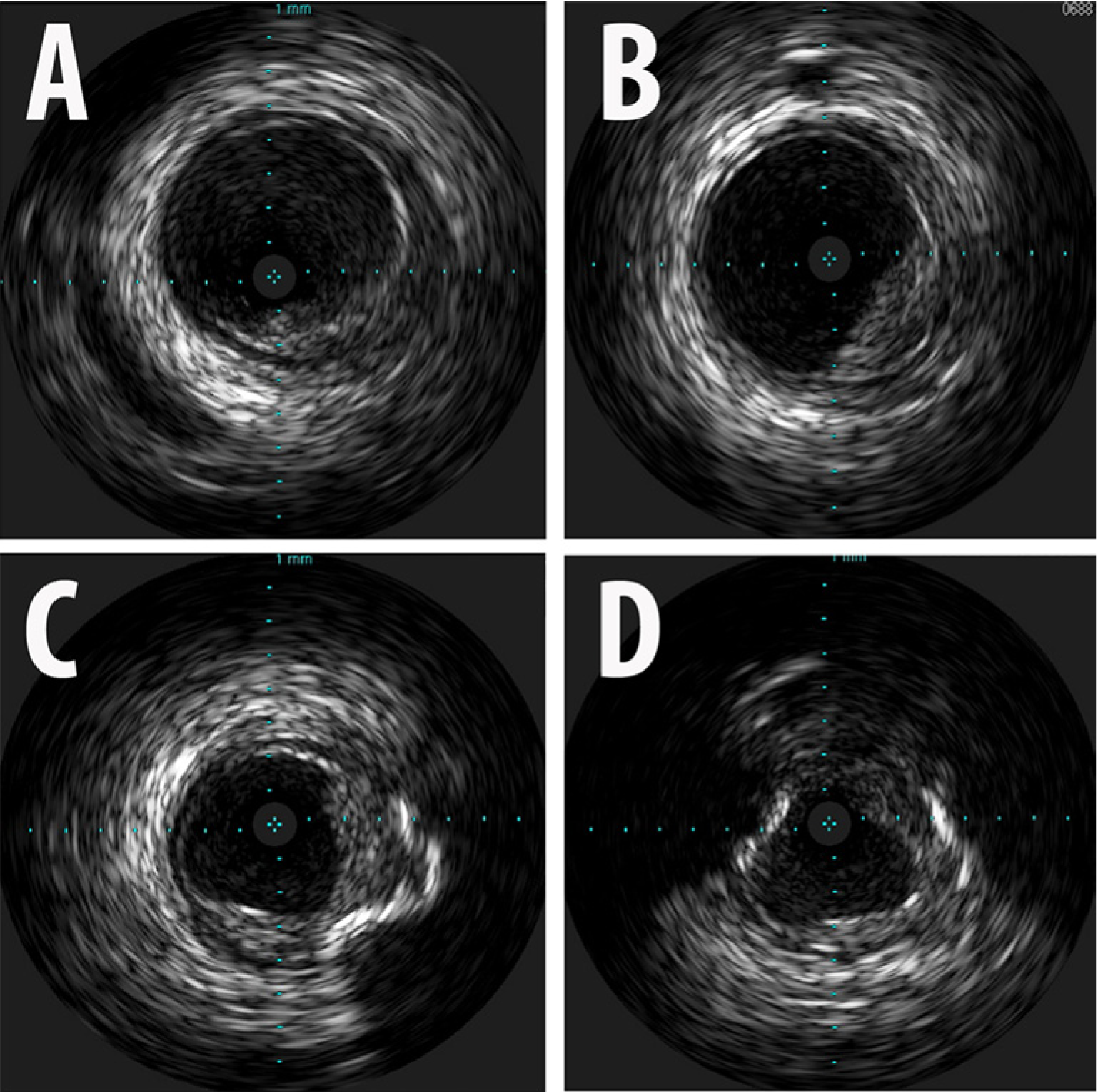
Intravascular ultrasound images from the superior mesenteric artery (SMA): (A) distal SMA; (B) mid SMA; (C) proximal SMA; (D) ostial SMA. Reference vessel diameter: 9 mm; minimal luminal diameter: 4 mm.
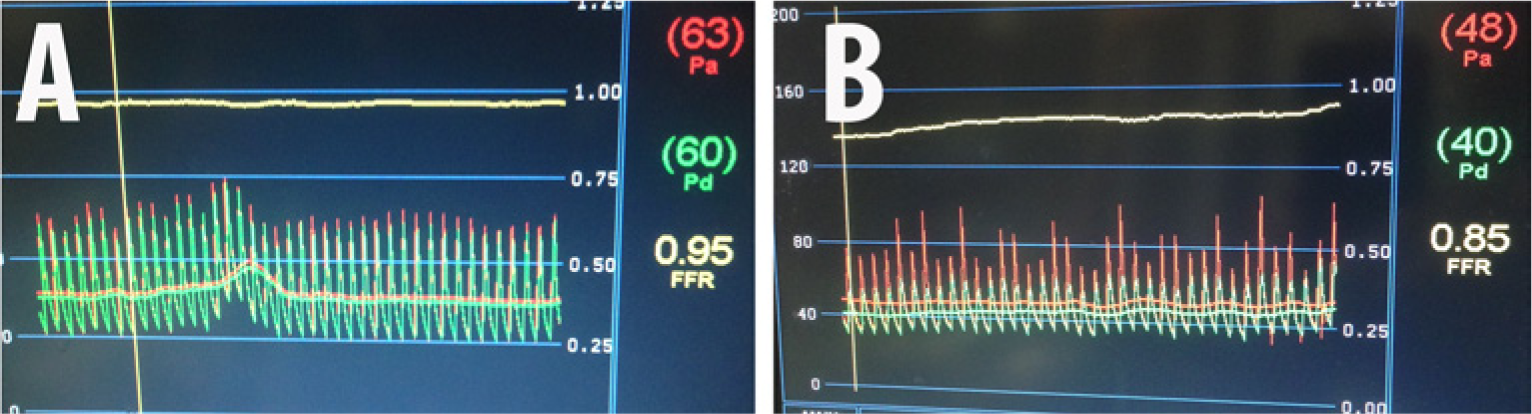
Superior mesenteric artery FFR after stent placement: (A) without intra-arterial nitroglycerin, FFR=0.95; (B) after intra-arterial nitroglycerin, FFR=0.85.
Employing the same techniques detailed for evaluating the SMA, gradients and FFR were measured across the proximal stenosis in the CA (Figure 7A). The resting gradient was 30 mmHg (FFR=0.82), which increased to 40 mmHg following hyperemia (FFR=0.75) with 300 µg of IA NTG (Figures 7B and 7C). As a result of these objective findings, a balloon-expandable 7.0 mm × 12 mm Palmaz Blue stent was successfully deployed and post-dilated in the standard fashion along the ostial/proximal CA. Repeat FFR pre- and post-IA NTG (0.97 and 0.82, respectively) confirmed significant hemodynamic improvement (Figure 8).
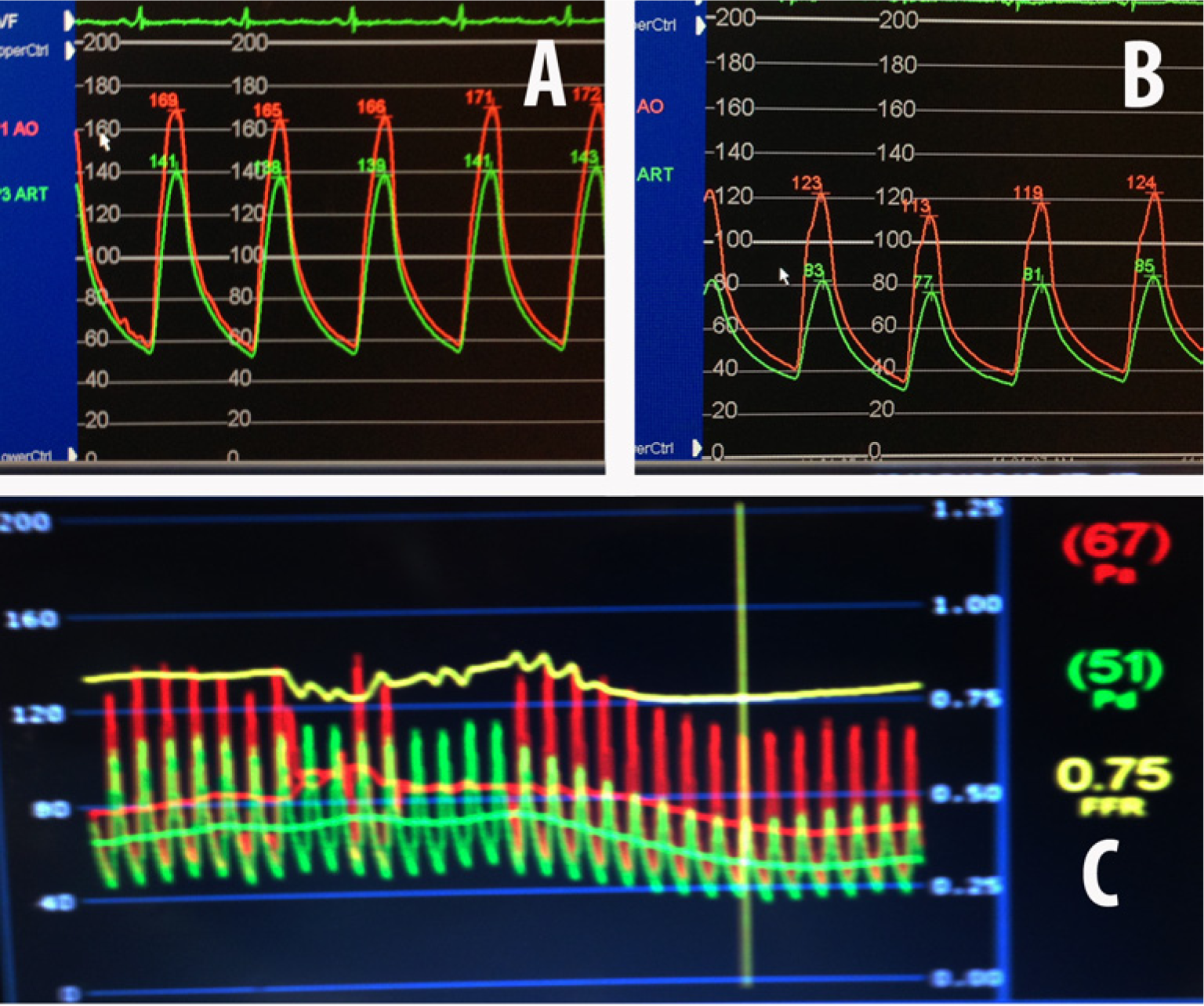
Celiac artery FFR before stenting: (A) without intra-arterial nitroglycerin, FFR=0.82; (B) after intra-arterial nitroglycerin, FFR=0.75; (C) composite FFR post intra-arterial nitroglycerin.
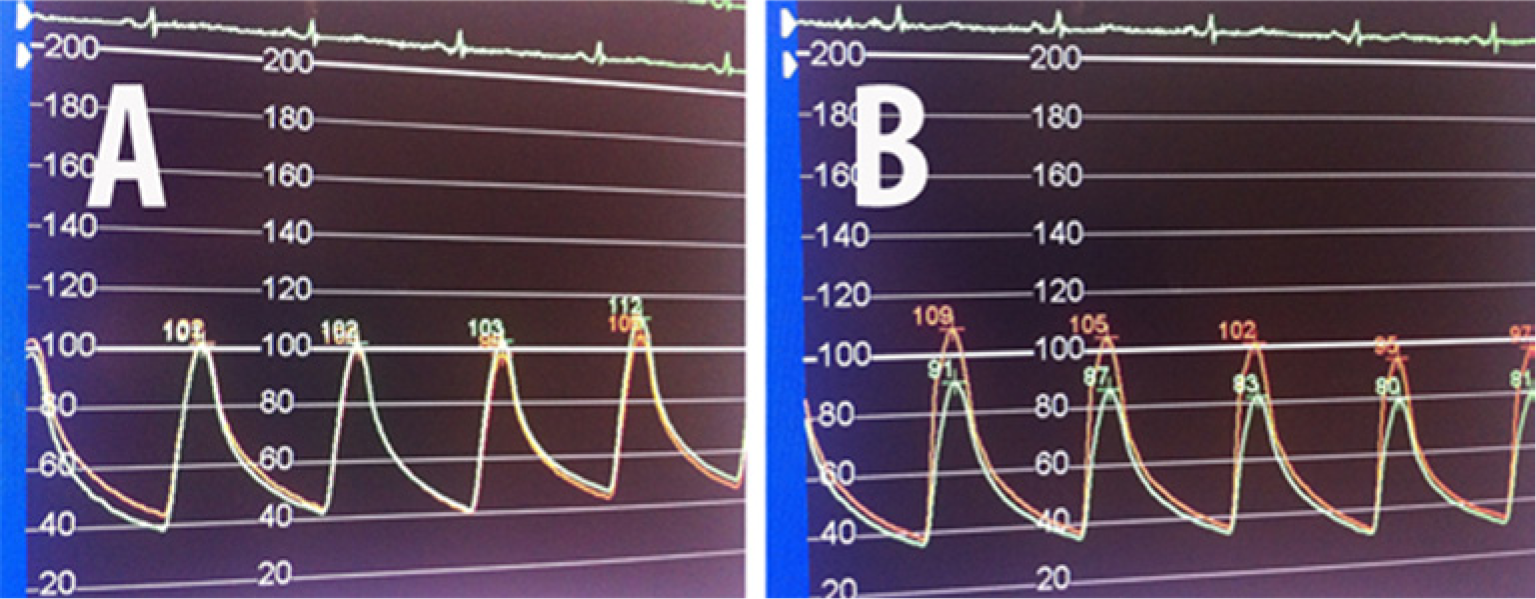
Celiac artery FFR after stenting: (A) without intra-arterial nitroglycerin, FFR= 0.97; (B) after intra-arterial nitroglycerin, FFR=0.82.
Patient outcome
Our patient had significant improvement in her abdominal discomfort within the first 4 weeks and she gained approximately 5 pounds (2.3 kg) of weight over the following 6-week period. While her pain and anorexia abated, she continued to have intermittent mild nausea.
Discussion
Patients with symptomatic CMI have limited underlying reserve against a subsequent ischemic insult. Therefore, it is critical to identify these patients before further deterioration in their clinical status. Traditional non-invasive evaluation consists of duplex ultrasonography to assess the peak velocities during systole and diastole of the mesenteric arteries as a measure of flow in these vascular beds. Despite its accuracy, this modality has several limitations that plague all ultrasound techniques, such as patient body habitus and vessel course, as well as technician skill and expertise. Although advanced non-invasive imaging such as CTA or MRA provide improved anatomic detail and minimize subjective influences on data acquisition, these modalities, like conventional angiography, neglect physiologic evaluation. Thus, when patients are referred for invasive mesenteric angiography and indeterminate disease is identified, additional hemodynamic assessment is recommended to conclusively rule out CMI. 2
This case illustrates the critical importance of assessing the functional significance of SMA and CA stenoses using FFR, and correlating these findings with IVUS. The angiographic appearance of the superior mesenteric and celiac arteries from orthogonal views did not suggest flow-limiting disease in either of these vessels, despite clinical suspicion suggesting otherwise. On the basis of objective hemodynamic assessment, our patient went on to have a definitive therapeutic intervention of the SMA and CA, with near-immediate relief of her previously debilitating symptoms.
FFR is an index of the physiological significance of a coronary stenosis, and is a well-validated tool used to determine the hemodynamic severity of an angiographically indeterminate coronary artery stenosis. It is defined as the ratio of maximal blood flow in a stenotic artery to normal maximal flow (pressure distal/pressure proximal or Pd/Pa). The technique involves measuring a baseline gradient across a stenosis. If this gradient is not considered significant, hyperemia is induced pharmacologically and the measurement repeated. While the principle of this hemodynamic assessment has been applied to non-coronary vascular beds such as the renal arteries, certain differences exist. Studies have indicated that an FFR measurement (Pd/Pa ratio) of 0.90 in the renal arteries may be considered the threshold value for hemodynamic significance as opposed to 0.80 in the coronaries.3,4 An elaborate study by Leesar and colleagues 5 demonstrated the predictive value of a hyperemic systolic gradient on the therapeutic success of renal artery stenting, while others have resorted to using a non-hyperemic assessment.3,4 This latter approach is intuitively appealing as renal artery flow is subject to minimal physiologic hyperemia, and any changes in renal vasomotor tone are brief and transient. In the mesenteric arterial bed, however, the flow is dynamic with routine post-prandial hyperemia. This underscores the importance of measuring a hyperemic gradient in the mesenteric arteries, particularly the SMA, as flow in this situation actually mimics post-prandial flow. While at present the threshold of a clinically significant gradient or FFR in the mesenteric bed has not been validated, one could postulate that using a stricter cut-off, as in the coronary arteries, may be feasible.
The importance of concurrent IMA disease cannot be overstated. 6 In our patient, the symptoms of CMI developed several weeks after the iatrogenic total occlusion of her previously severely narrowed IMA. It is almost certain that this, combined with the hemodynamically significant residual disease involving the CA and SMA, served as the tipping point in the onset of mesenteric ischemia in a patient that was previously in a state of clinical equipoise with regards to her intestinal perfusion. It is likely that in the presence of a narrowed but patent IMA, the superior mesenteric and celiac arteries perfusing the small intestine were subclinically burdened, providing collateral flow to the large intestine. However, after the occlusion of the IMA, a phenomenon of ‘intestinal steal’ occurred, with blood diverted for the IMA collateralization, causing small intestinal ischemia. It is indeed interesting to note that, judging by the scheme proposed by Lotun et al., 7 our patient’s symptoms reflected small-bowel ischemia more than large-bowel ischemia despite relative patency of the CA and SMA.
While our case clearly demonstrates the feasibility of adjunctive invasive diagnostic testing in the assessment of patients with CMI, further studies are required to define the precise role of FFR in the evaluation of indeterminate angiographic lesions of the mesenteric arteries, the specific flow reserve value pertinent to CMI, and its impact on clinical outcomes after intervention.
Footnotes
Declaration of conflicting interest
The authors declare no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
