Abstract
Stenotic and occlusive diseases of the subclavian and brachiocephalic arteries can cause a significant morbidity as it can lead to symptomatic ischemia affecting the upper extremities, brain and, in some cases, the heart. An endovascular approach with primary stenting or provisional stenting has become the primary modality of revascularization of subclavian artery stenosis. In-stent restenosis can be treated with percutaneous transluminal angioplasty or repeat stenting and although stents offer superior long-term patency over balloon angioplasty alone for de novo lesions, there are no data regarding primary versus provisional stenting in subclavian in-stent restenosis. Here we describe a case of subclavian in-stent restenosis treated with just balloon angioplasty and demonstrate that provisional stenting with angioplasty alone when the percutaneous transluminal angioplasty results are excellent is a reasonable alternative to primary stent placement for subclavian in-stent restenosis.
Background
Stenotic and occlusive diseases of the subclavian and brachiocephalic arteries can cause significant morbidity as it can lead to symptomatic ischemia affecting the upper extremities, brain and, in some cases, the heart. 1 Primary stenting or provisional stenting (i.e. stenting after failed or suboptimal balloon angioplasty) has become the primary modality of revascularization for subclavian and brachiocephalic arteries because of a high clinical success rate and a lower rate of procedural complications as compared to open surgery.2,3 Endovascular revascularization of atherosclerotic subclavian stenosis with stenting is associated with a technical success rate of 92% to 100% with 2–5-year patency rates ranging from 77% to 89%,4,5 comparable to surgery. Stenting is preferred over percutaneous transluminal angioplasty (PTA) alone because of the uncertain long-term patency and high failure rate of PTA when treating occlusions. In-stent restenosis can be treated with PTA or repeat stenting but there is a paucity of comparative data regarding how best to treat subclavian artery in-stent restenosis. Paclitaxel-coated balloon angioplasty, 6 excimer laser debulking, 7 stent grafting, 8 drug-coated stents 9 and brachytherapy 10 have been used in the treatment of femoropopliteal in-stent restenosis with variable results, but the role of each of these technologies in the treatment of subclavian artery in-stent restenosis is unknown.
Case report
The patient is a 59-year-old woman who presented to our clinic with symptoms of left arm claudication with minimal arm activity. She was treated with bilateral subclavian artery stents about 10 years previously. She has a history of hypertension, hyperlipidemia and coronary artery disease with coronary artery bypass surgery using a left internal mammary arterial conduit to the left anterior descending artery. She currently denies angina pectoris.
She continues to smoke and has a history of peripheral arterial disease with common iliac artery stenosis treated with bilateral stents. She has a past history of ischemic stroke with extensive aortic arch atheroma identified on transesophageal echocardiography. On physical examination, systolic blood pressure was 50 mmHg greater in the right arm compared to the left arm. Magnetic resonance angiography (MRA), performed to investigate symptoms of stroke, demonstrated a possible near occlusion of the left subclavian artery ostium or an artifact (Figure 1). The utility of the MRA may be limited due to the void artifact caused by the metal stent.
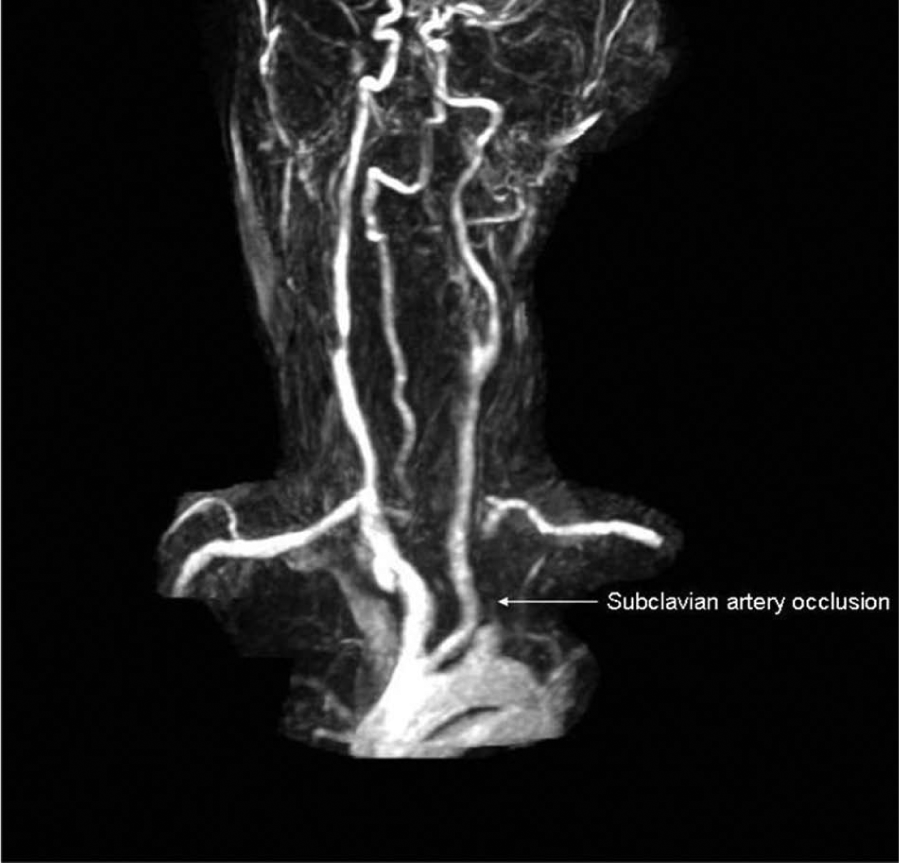
Magnetic resonance angiography showing a possible subclavian artery flow void or an artifact (arrow).
The MRA report was unreliable due to the metal artifact and thus the patient was taken to the catheterization laboratory for left subclavian angiography prior to possible endovascular intervention for her symptoms of left arm claudication. In the catheterization laboratory, the left radial artery access site was prepped and draped; it was chosen for the site of arterial access to avoid catheter contact with the extensive aortic arch atheroma that would occur with femoral artery access. We also felt that there was a possibility of an occluded left subclavian stent, which would be easier to cross retrograde from radial artery access, although a potential disadvantage of the radial technique is that it may be more difficult to see the proximal edge of the stenosis by retrograde angiography.
After local anesthesia, a 5 French sheath (Terumo Medical Corporation, Somerset, NJ, USA) was inserted into the left radial artery. Intravenous unfractionated heparin was employed as an anticoagulant during the procedure maintaining an activated clotting time of 250–300 seconds. A 5 French Judkins right diagnostic catheter was advanced over a 0.035 inch × 180 cm J-wire to the left subclavian artery and selective left subclavian angiography was performed demonstrating a moderately severe, 70% diameter narrowing within the previously placed subclavian stent (Figure 2). Using a 5 French Judkins right diagnostic catheter and a 0.035 inch × 180 cm stiff, angled Glidewire (Terumo Medical Corporation), the lesion was crossed and the stiff, angled Glidewire was positioned into the descending aorta. The 5 French Judkins right diagnostic catheter was then advanced over the stiff, angled Glidewire to the descending aorta. The stiff, angled Glidewire was removed and a pressure gradient of 30 mmHg across the proximal segment of the left subclavian artery stent was recorded. A 20 mmHg peak systolic or a 10 mmHg mean translesional pressure gradient is generally acceptable for intervention by expert consensus, not by evidence-based guidelines. The pressure gradient obtained during the catheterization was not simultaneous with the arm-to-arm blood pressure discrepancy. Also, the pressure gradient during the catheterization was translesional between the proximal segment of subclavian artery and descending aorta. Next, a 0.035 inch × 260 cm Amplatz Extra-Stiff Wire Guide (Cook Medical, Bloomington, IN, USA) was inserted into the Judkins right diagnostic catheter and positioned in the descending aorta. The Judkins right diagnostic catheter and 5 French sheath were then removed and exchanged for a 55 cm long 6 French Raabe sheath (Cook Medical). The in-stent restenosis was predilated with a 6.0 mm × 40 mm balloon (Mustang; Boston Scientific Corporation, Natick, MA, USA) followed by an 8.0 mm × 40 mm balloon (Figure 3), resulting in a residual stenosis of about 10% (Figure 4). The translesional pressure gradient was zero following balloon dilation.
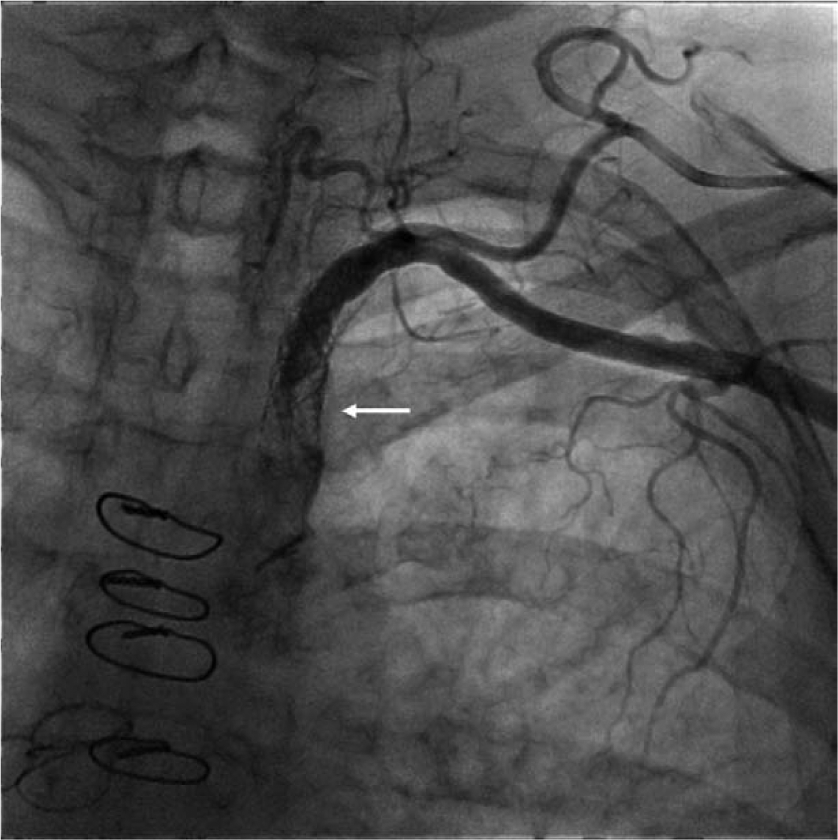
Retrograde subclavian angiography showing in-stent restenosis (arrow).
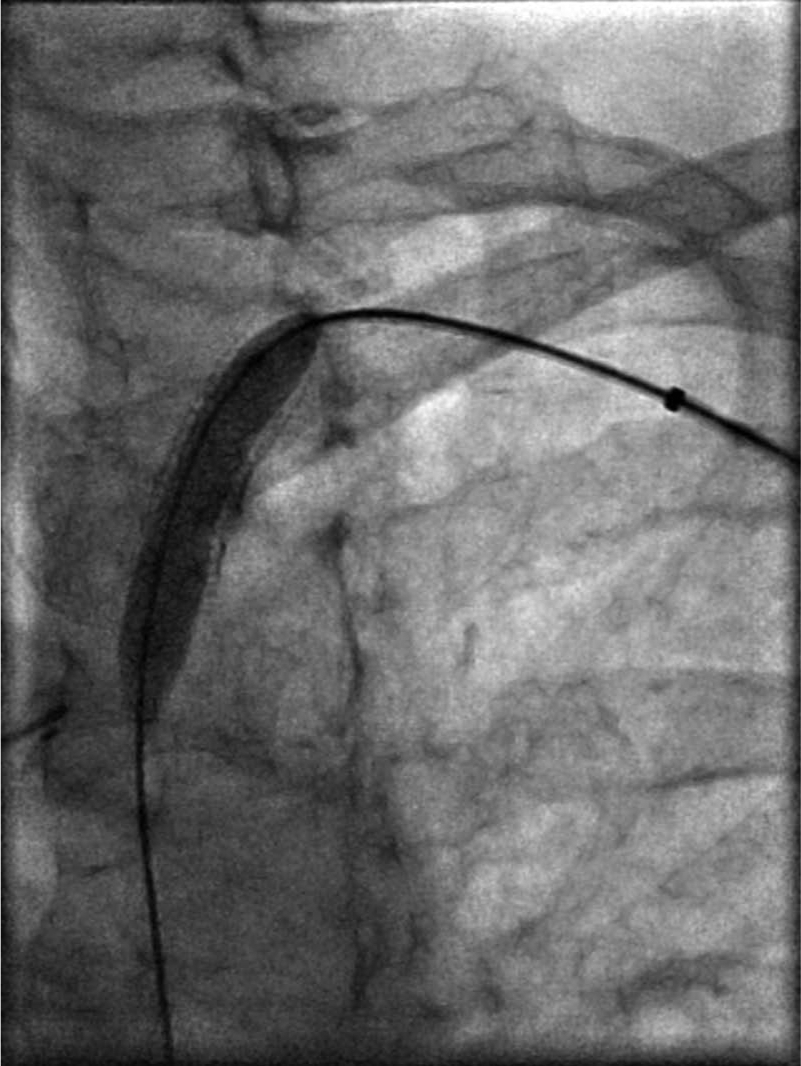
Percutaneous transluminal angioplasty with balloon inflation.
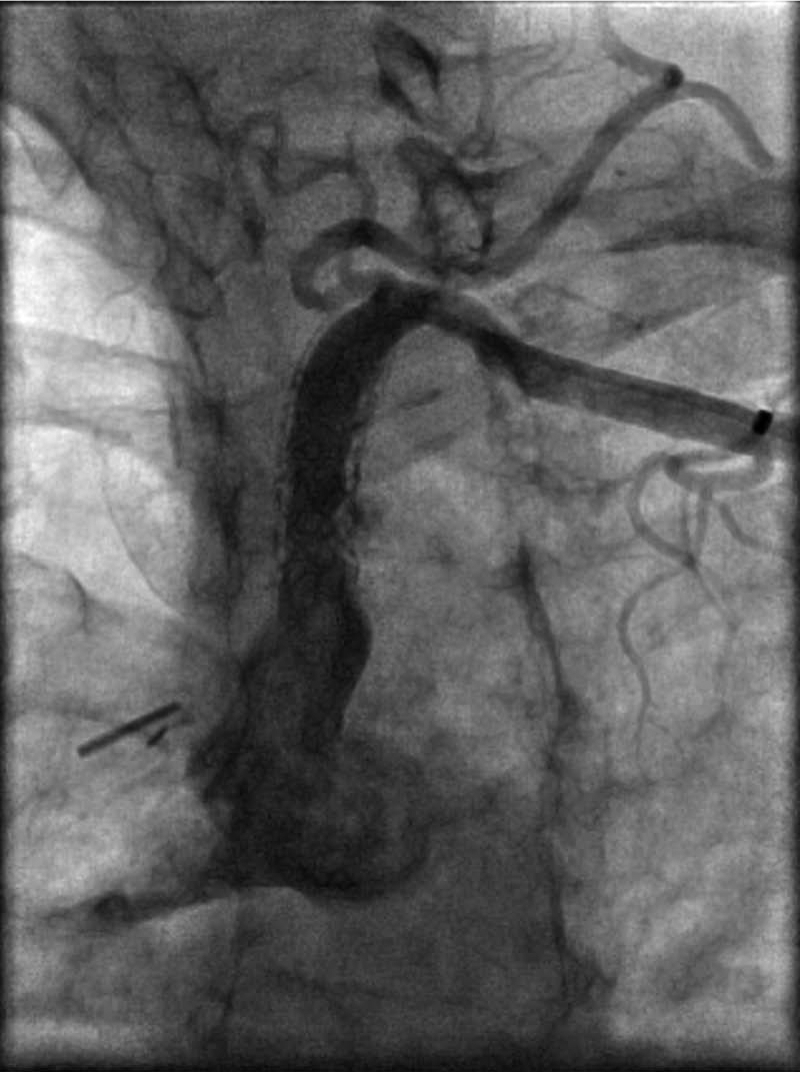
Angiography following balloon angioplasty demonstrating < 30% residual stenosis.
With an excellent angiographic result (< 30% residual stenosis without a flow-limiting dissection) and resolution of the translesional gradient, the decision was made to not place a new stent within the original stent. The catheters were removed and local hemostasis was obtained. Following angioplasty the patient did well and was discharged home. Her claudication resolved and she has been doing well 6 months subsequent to her procedure.
Discussion
Obstruction of the brachiocephalic or subclavian artery accounts for approximately 17% of symptomatic extracranial cerebrovascular disease 1 and frequently is responsible for ipsilateral arm claudication or subclavian steal syndrome. In patients with internal mammary artery (IMA) coronary bypass grafts, left subclavian stenosis may cause symptoms of coronary ischemia. 11 Brachiocephalic or subclavian artery revascularization is usually indicated for symptomatic patients and to protect inflow into internal mammary coronary graft, axillofemoral, axilloaxillary or subclavian-carotid graft. Restenosis, or reduction in lumen diameter after catheter-based percutaneous therapy of the subclavian artery, is the result of arterial damage with subsequent neointimal tissue hyperplasia. Binary angiographic restenosis is defined as ≥ 50% luminal narrowing at follow-up angiography, although standardized classifications, as described in the coronary beds, 12 are lacking in the brachiocephalic and subclavian arteries.
Balloon angioplasty of the subclavian and brachiocephalic arteries was first described in 1980 as an alternative to surgical revascularization. 2 Percutaneous catheter-based treatment offers a less invasive approach and carries a lower rate of procedural complications compared to open surgery, including lower peri-procedural morbidity, fewer deaths, and a lower stroke rate accompanied by a shorter hospital stay.2,3
The choice of vascular access (i.e. radial, brachial, or femoral) is critical in determining equipment choices such as sheaths, guide catheters, guide wires, balloons, or stents. Radial artery access offers significant advantages such as lower access site bleeding complications, early ambulation, and same day discharge. Radial access is also preferred when extensive aortoilliac disease is present, as in our case. A larger-sized sheath can potentially lead to thrombosis and occlusion of the radial artery. This can be avoided by using smaller-sized sheaths, using guiding catheters without sheaths, anticoagulation during the procedure and non-occlusive hemostasis post procedure. For the purpose of intervention, either 6 French or 7 French sheaths (Flexor Shuttle, Flexor Ansel or Flexor Raabe; Cook Medical) or 8 French or 9 French guide catheters are used. From the radial or brachial approach, both sheath and guide may be equally beneficial. We planned for balloon only, but were prepared to place another stent if there was recoil or an edge dissection. Even though the risk of radial artery occlusion is increased with a larger sheath size, we thought it was prudent to plan ahead for contingencies and easier to begin with an appropriate-sized sheath than to have to change in the middle of the procedure.
For subclavian ostial lesions, a guide catheter offers more control and steerability compared to a long sheath (80–100 cm) from the femoral access. From the brachial artery access, a 55 cm sheath may be used. A brachial or radial approach may limit visualization of the proximal edge of the stenosis by retrograde angiography. Using a diagnostic catheter such as a 4 French Multipurpose or a Judkins right catheter, the lesion may be crossed with a steerable 0.035 inch guide wire (Wholey Guidewire; Covidien, Mansfield, MA, USA) or it may be necessary to use a hydrophilic guide wire such as a 0.035 inch Glidewire (Terumo Medical Corporation). This steerable wire can then be exchanged for a more supportive and stiffer 0.035 inch guide wire such as the Amplatz Extra-Stiff Wire Guide (Cook Medical) intended to facilitate the placement and exchange of interventional devices such as the PTA balloon and stent during an interventional procedure. It is important to avoid disrupting the origin of the internal mammary artery. This goal is best achieved by delineating the origin of the vessel by performing the angiogram in the contralateral oblique projection. A 0.014 inch guide wire may also be placed in the internal mammary artery to protect the access to the vessel.
While the reported rates of restenosis at 3 years are as high as 21% with PTA alone, 13 primary stenting for symptomatic subclavian and brachiocephalic artery lesions, including occlusions, has been described to be successfully accomplished in 98% of patients with 83% primary and 96% secondary patency rates at an average of more than 5 years of follow up. 5 For in-stent restenosis, balloon angioplasty using an appropriately sized balloon may be adequate to treat the obstruction. Although stents offer superior long-term patency over PTA for de novo lesions, there are no data regarding primary versus provisional stenting in subclavian in-stent restenosis. Alternative treatment options for in-stent restenosis would include: (1) restenting; (2) covered stents; and (3) brachytherapy. There is no evidence to support an advantage for any of these options, but the placement of a covered stent is technically challenging and more difficult that the most simple solution, PTA. We prefer to escalate risk, starting with the simplest solution and progressing to more difficult if there is a second in-stent restenosis. The method of failure for covered stents is acute thrombosis rather than gradual restenosis, which is less attractive. Brachytherapy is not commercially available. When drug-coated balloons become available in the US market, they will be an attractive option. Again, using embolic protection complicates this procedure and must be weighed against the theoretical risk that this very rare complication might occur. There is no evidence that a cutting balloon has any utility for in-stent restenosis, but it has only been systematically studied in coronaries. The cutting balloon does cost significantly more than a conventional balloon, so it is hard to use without some supportive evidence.
With an excellent angiographic result and the gradient abolished, we treated in-stent restenosis with just balloon angioplasty alone. In conclusion, provisional stenting with angioplasty alone when the PTA results are excellent is a reasonable alternative to primary stent placement for subclavian in-stent restenosis.
Footnotes
Declaration of conflicting interest
The authors declare no conflicts.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
