Abstract
Treatment of an aberrant right subclavian artery (arteria lusoria) aneurysm is traditionally performed surgically or via a hybrid approach. To our knowledge, a fully percutaneous approach has not yet been reported. We describe the fully endovascular exclusion of an aberrant right subclavian artery and thoracic aortic aneurysm. This approach has the potential advantage of avoiding complications of an open surgical repair, particularly in patients of advanced age and/or with multiple comorbidities.
Keywords
Introduction
Bayford first described a patient with abnormal course of the right subclavian artery in 1794. 1 Autopsy studies have shown a 0.5–1.5% prevalence of an aberrant right subclavian artery. 2 The embryonic origin of the aberrant right subclavian artery is thought to be due to the involution or absence of the embryonic fourth right aortic arch which normally develops into the right subclavian artery. The proximal seventh intersegmental artery also known as the distal right aorta (normally involutes) remains attached to the descending aorta and evolves into the right subclavian artery. Proximally, it has a wide, broad base and often becomes Kommerell’s diverticulum. 3 Distally this crosses between the esophagus and vertebral column in 80% of cases, between the esophagus and trachea in 15% of cases, and anterior to trachea and esophagus in 5% of cases. 4
Clinically, most individuals with this anomaly are asymptomatic, but can present with dysphagia due to mechanical obstruction, coughing, or Horner’s syndrome. 4 Importantly, the aorta is often abnormal with a higher propensity for aneurysm formation, dissection, or rupture. 5 The physical examination is invariably normal. The diagnosis can be made by a barium esophagram or upper endoscopy, showing a pulsating compression of the posterior wall,6,7 or by imaging of the aortic arch with computed tomography (CT)-angiography or magnetic resonance (MR)-angiography. 8
Surgical therapy involves right subclavian-carotid bypass transposition, right subclavian-carotid bypass/transposition with subsequent closure of the aberrant right subclavian artery aneurysm or replacement of the descending aorta.9,10 Endovascular hybrid approaches include: (1) endovascular right subclavian artery aneurysm occlusion and right carotid-subclavian bypass and (2) thoracic endoluminal graft in the distal aortic arch and descending aorta and the option of unilateral right or bilateral carotid-subclavian hybrid bypass. 11 The challenge of a complete endovascular repair lies in the anatomy of the aneurysm. In the overwhelming majority of cases it includes the ostium and thereby renders straightforward endovascular stent graft repair impossible. To our knowledge, complete endovascular repair has not been reported.
Case report
The patient is an 85-year-old male with an aneurysm of a right aberrant subclavian artery. His past medical history is significant for a previously repaired abdominal aortic aneurysm, a 2.2 cm aneurysm of the right common iliac artery distal to the left iliac graft limb and immediately prior to the iliac bifurcation, and prior stenting for coronary artery disease. There were no symptoms related to the aneurysm. Specifically, there was no history of cerebral or peripheral embolization, and dysphagia was denied. The physical examination was unremarkable.
A CT scan demonstrated a 3.9 cm aneurysm of an aberrant right subclavian artery also known as Kommerell’s diverticulum (Figure 1). Significant aneurysmatic dilatation was apparent at the ostium of this artery and extended alongside the artery for a distance of 6.6 cm, sparing the right vertebral and internal mammary arteries. Anterior displacement of the esophagus by the aneurysm was noted. There were atherosclerotic changes throughout the thoracic aorta including mild adherent thrombus in the descending thoracic aorta. The aneurysm included the aorta at the take-off of the aberrant subclavian artery, where it measured 3.3 cm. With the exception of the aberrant right subclavian artery (feeding the right internal mammary and right vertebral artery) there were no anatomical variations of the remaining arch vessels. The anatomy is illustrated in Figure 2.
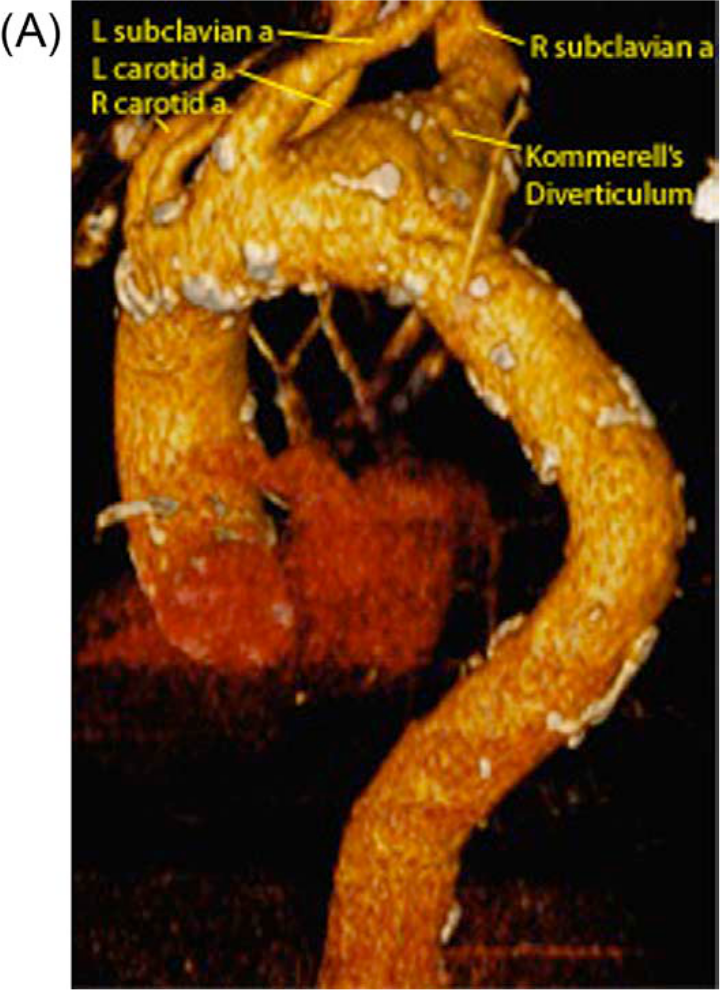
Computerized cardiac tomography showing the anteroposterior view of the thoracic aorta with aberrant right subclavian artery aneurysm.
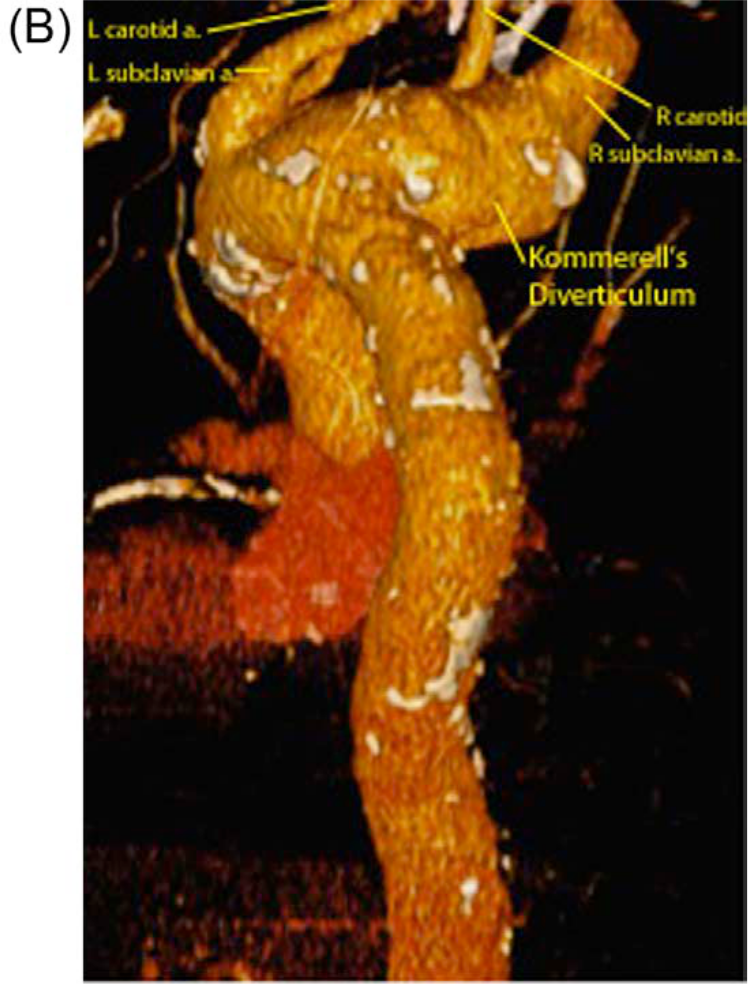
Computerized cardiac tomography showing the lateral view of the thoracic aorta with aberrant right subclavian artery aneurysm.
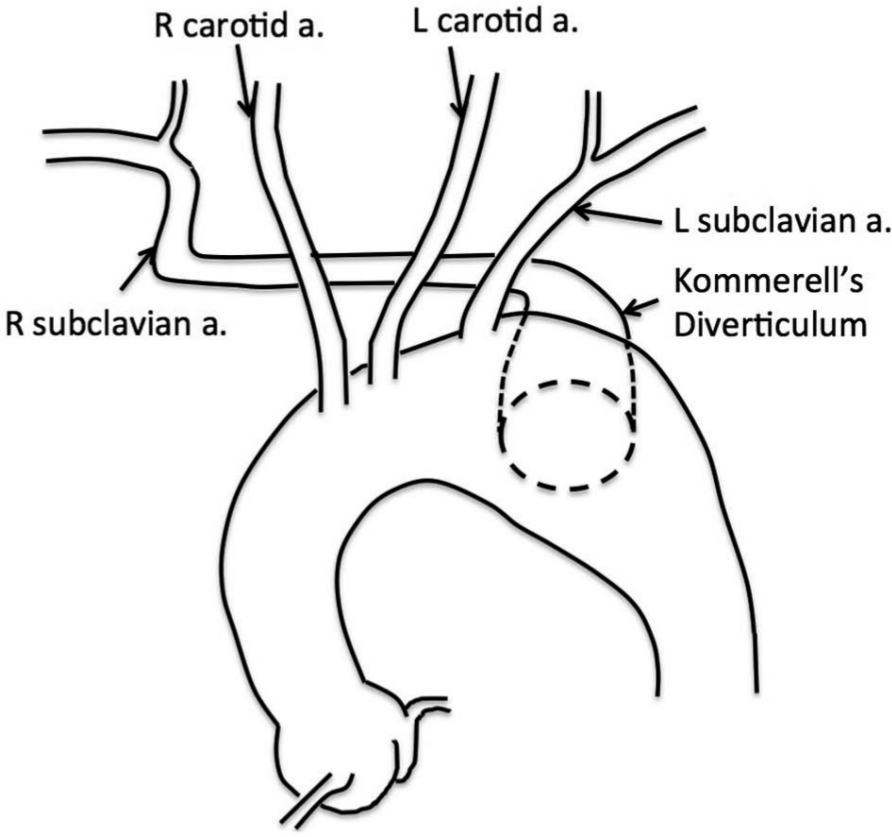
An illustration of the relationship between Kommerell’s diverticulum and branches of the thoracic aorta.
Despite the absence of symptoms, the concerning aneurysm size drove the decision to proceed with aneurysm exclusion. This was done after a full discussion of the risks, benefits, and alternatives with the patient. The deployment of a thoracic endovascular graft distal to the origin of the right common carotid artery while preserving the ostium of the left common carotid, left subclavian and aberrant right subclavian arteries was planned with deployment of a stent graft into the aberrant right subclavian artery via the orifice of the thoracic aortic endograft.
Custom thoracic aortic endograft
Based on computer modeling of the CT-images, a custom-made endovascular graft (Cook Medical Inc., Bloomington, IN, USA) was manufactured (Figure 3). This included the following - 1) a reinforced scallop proximally with a width of 20 mm and height of 20 mm, placed for the ostium of the left common carotid artery, followed by 2) a reinforced large fenestration with preloaded catheter and guidewire for the left subclavian artery and 3) an upward-facing fenestration at 4:00 on the clock face, allowing placement of a stent graft extending from the fenestration into the aberrant right subclavian artery excluding the aneurysm (Figure 3). Of note, the preformed prosthesis side branch shown in Figure 3 was removed leaving an orifice for delivery of the aberrant right subclavian artery stent graft.
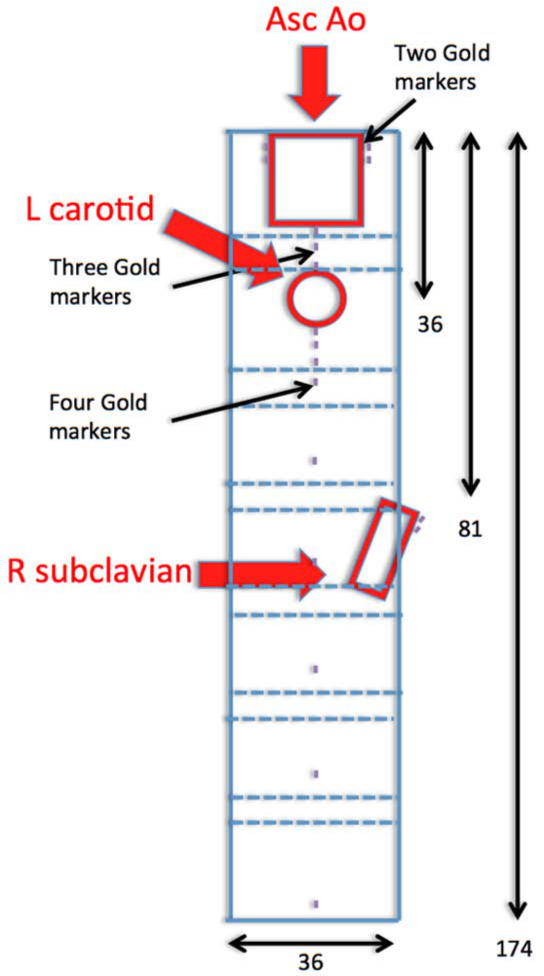
Technical illustration of the custom-made endoprosthesis.
Access
The procedure was performed under local anesthesia. The following access was obtained: 8 Fr right common femoral access (closed with an 8 Fr Angioseal (St Jude Medical, Minnetonka, MN, USA) at the termination of the procedure), 24 Fr left common femoral access (closed with a 10 Fr Prostar® (XL, Abbot Vascular, Abbott Park, IL, USA) suture closure at the termination of the procedure) and 6 Fr and 5 Fr left and right brachial access, respectively (closed with manual compression at the termination of the procedure). Next, a 5 Fr pigtail catheter was advanced into the ascending thoracic aorta and cine-angiography performed of the aortic arch and aberrant right subclavian artery.
Thoracic aortic stent graft deployment
A 5 Fr JR-4 catheter was advanced into the left subclavian artery via left brachial access. The endovascular stent graft was advanced via left common femoral artery access to the level of the ascending aorta while maintaining a 5 Fr pigtail catheter in the ascending aorta for angiographic views to facilitate stent graft positioning (Figure 4). Subsequently, a pre-positioned wire inside the undeployed stent graft was advanced into the left subclavian artery and snared and exteriorized via the left brachial access. This wire was then used as an anchor to mark the position of the left subclavian artery. The endograft was optimally positioned and deployed with the proximal edge just distal to the origin of the right common carotid artery (Figure 5).
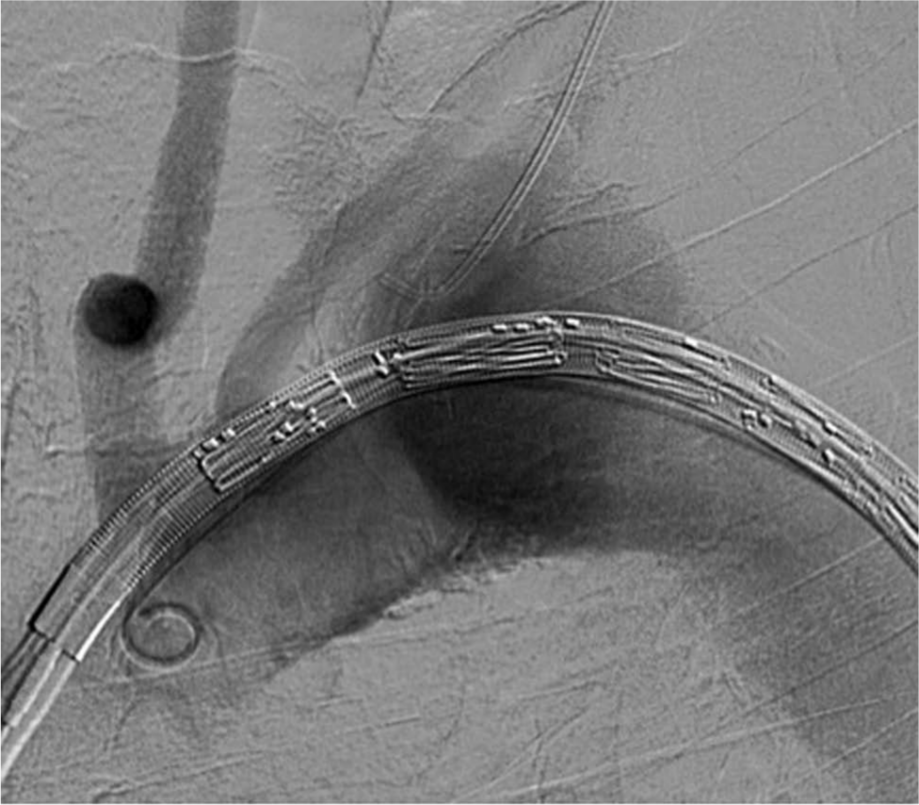
Aortic arch angiography with the thoracic aortic endograft positioned in the ascending thoracic aorta prior to deployment and a JR-4 catheter in the left subclavian artery.
The deployed thoracic aortic endograft with the wire loop into the left subclavian artery.
Left subclavian artery stent implantation
It was not possible to straighten the pre-established left subclavian wire loop (Figure 5). Therefore, it was removed and the left subclavian artery was cannulated via right femoral access through the deployed thoracic endograft. Two endografts were deployed at the left subclavian ostium (10 mm × 38 mm and 10 mm × 59 mm iCAST™ stents (Atrium, Hudson, NH, USA)), both of which dislocated distally during implantation missing the ostium/fenestration and requiring implantation of a third endograft (iCAST™ 10 mm × 36 mm) successfully covering the fenestration/ostium (Figure 6). This was postdilated with a 14 mm balloon.
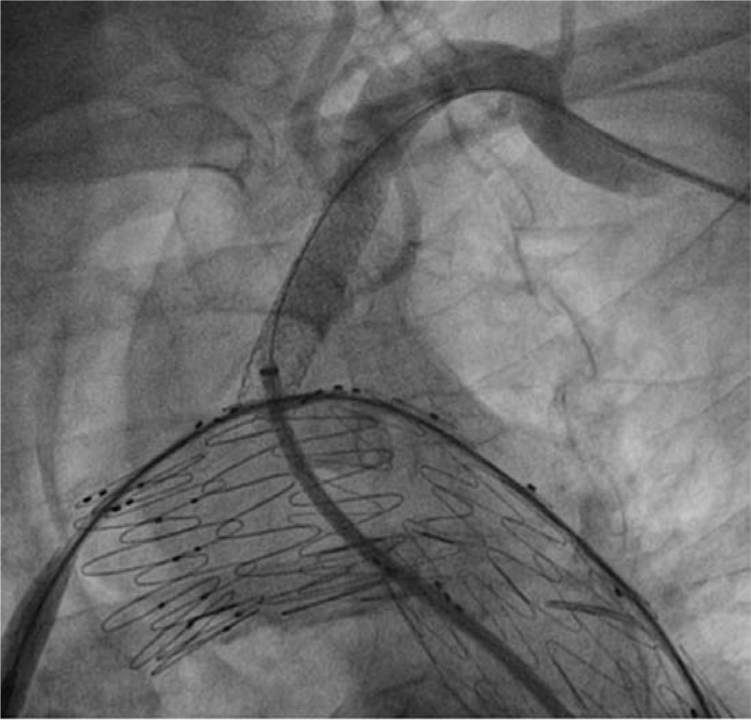
Left subclavian endografts have been implanted successfully.
Aberrant right subclavian artery stent implantation
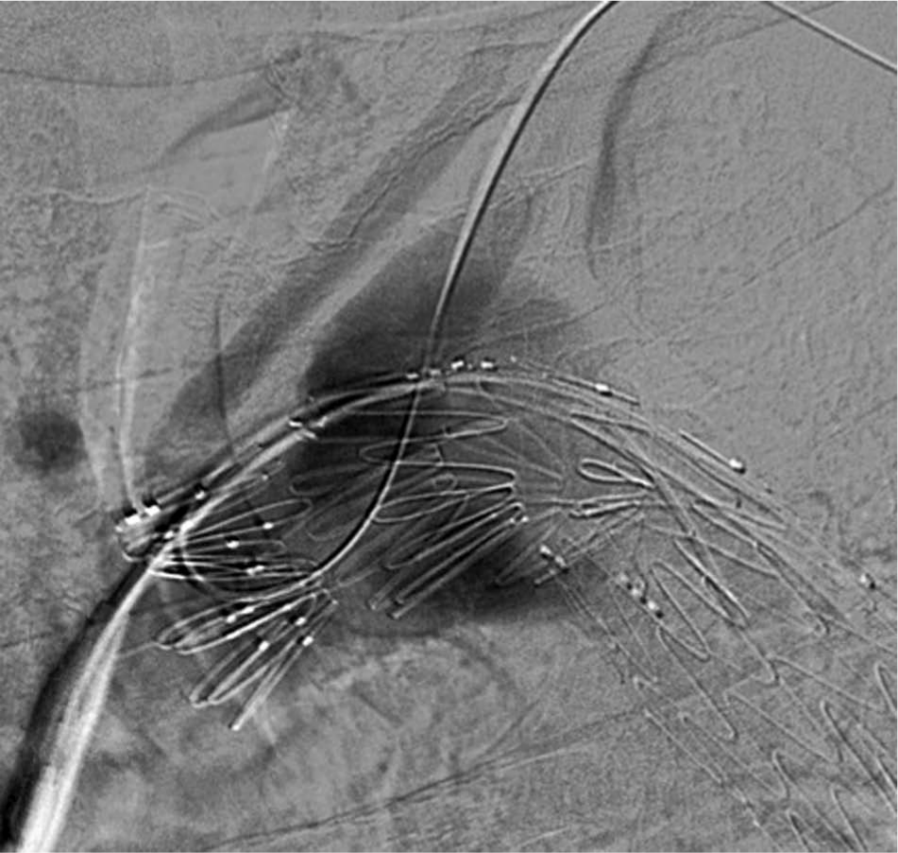
The aberrant right subclavian artery was cannulated via the preformed fenestration. This was done with exquisite care to avoid rupture of the right subclavian aneurysm. Next, the wire was advanced into the artery, snared and exteriorized (Figure 7), and a 10 mm × 15 cm Gore® Viabahn® (WL Gore and Associates, Inc., Flagstaff, AZ, USA) stent implanted with approximately 1 cm of the proximal edge extending into the aortic stent graft (Figure 8). Subsequently, a 13 mm × 10 cm Gore® Viabahn® stent was implanted distal to the previous stent, successfully excluding the right aberrant subclavian artery aneurysm (Figure 9). The landing zone was chosen based on the healthy vessel segment distal to the aneurysm. Final angiographic images demonstrate patent aortic arch vessels and patent right aberrant subclavian and left subclavian stents with successful aneurysm exclusion. Follow-up CT imaging at 6 months did not demonstrate any endoleaks.
The aberrant right subclavian artery has been cannulated with formation of a wire loop.
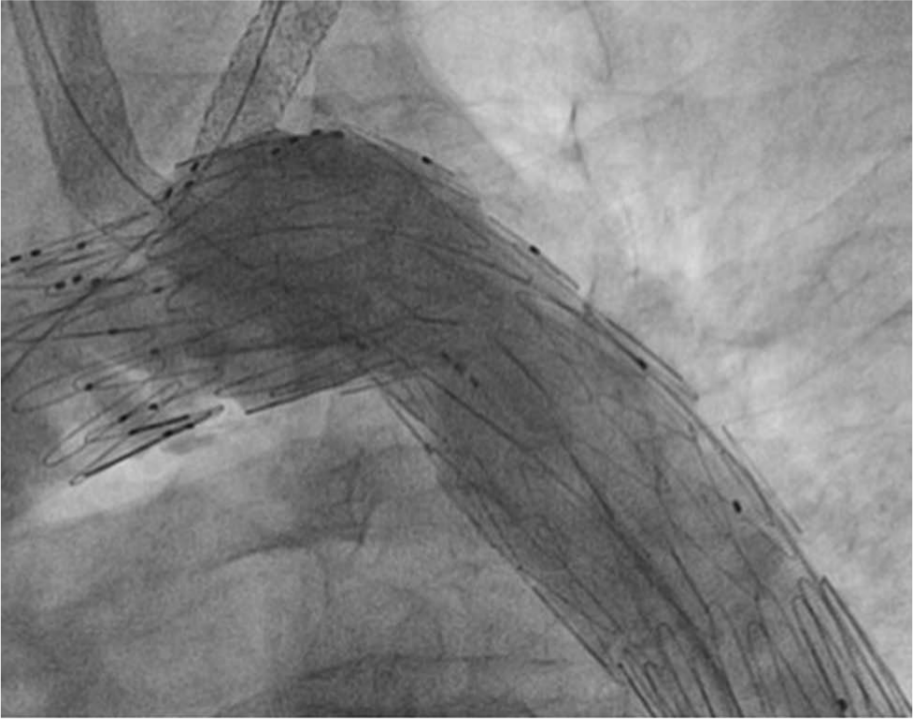
Successful deployment of the aberrant right subclavian artery endograft.
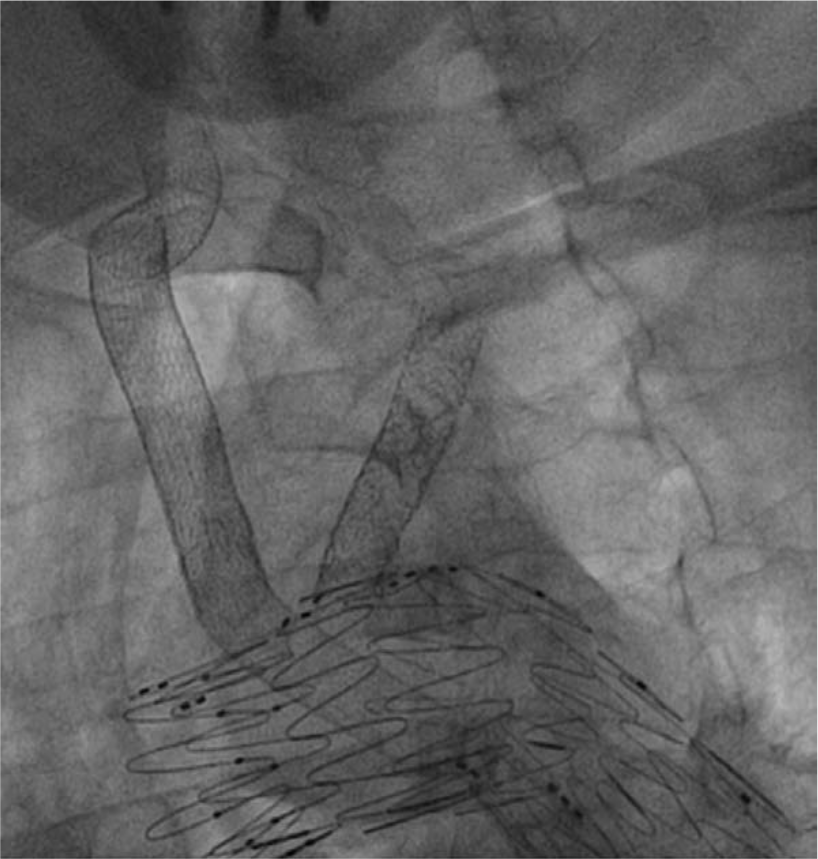
Final cine-image demonstrating the position of all endografts.
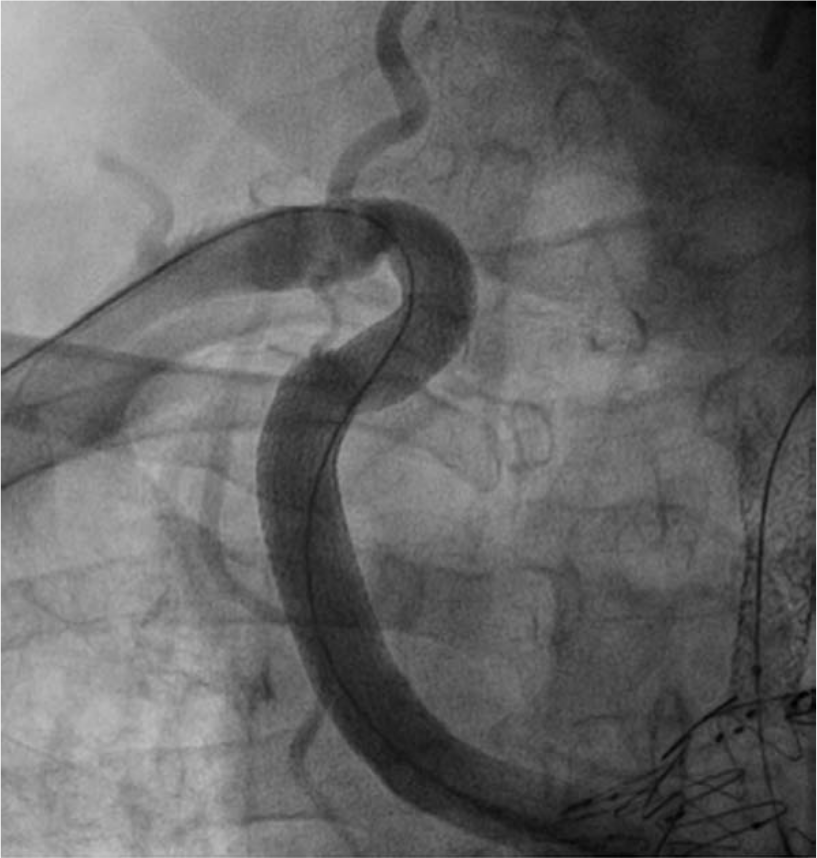
One-year follow-up angiography was performed. This showed excellent structural integrity and normal flow patterns (Figures 10 and 11).
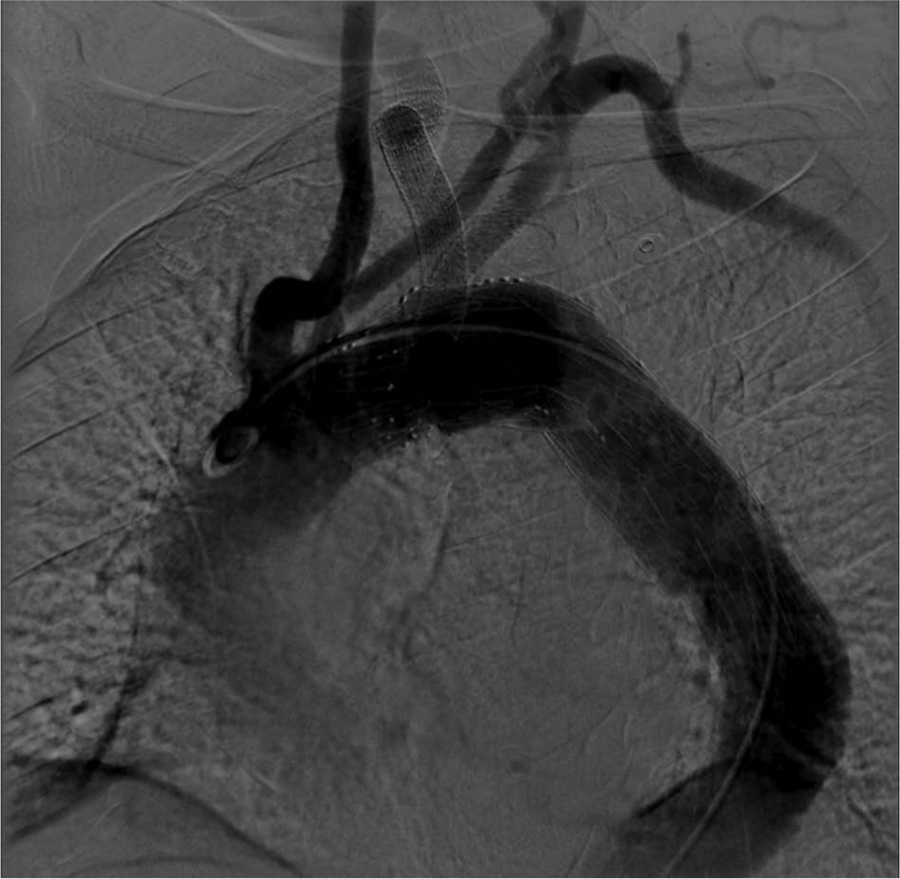
One-year follow-up angiography showing excellent flow pattern in the stent graft.
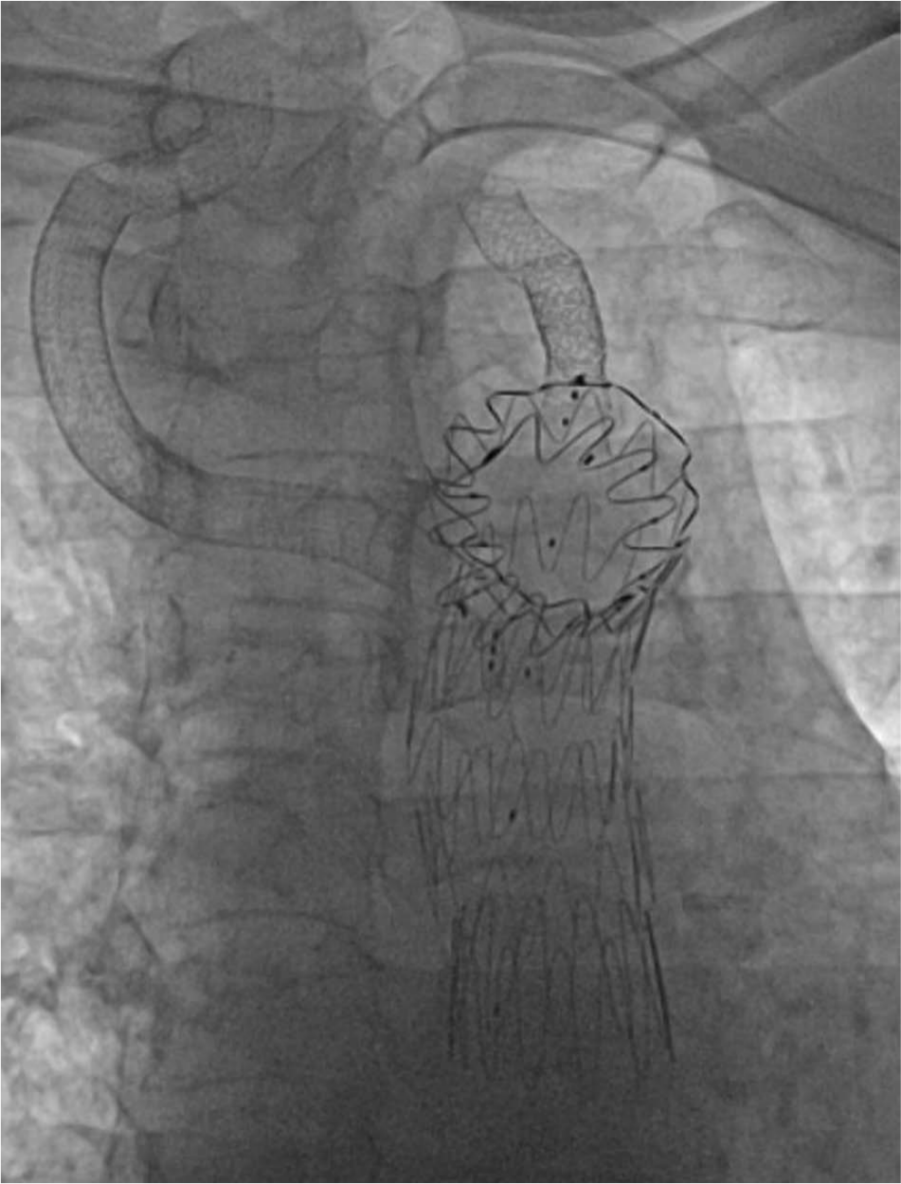
One-year follow-up evaluation showing excellent structural integrity (lateral view).
Limitations
Little is known about the natural history and rupture risk of an aberrant right subclavian artery aneurysm and even less regarding the prevention of risks (rupture/dissection/thrombus formation) associated with the aneurysm by conventional or hybrid repair. However, ruptures have been reported. 12 In addition, the rupture risk may be underestimated given a likely rapidly fatal outcome of rupture.
Discussion
To our knowledge, fully percutaneous treatment using customized stent grafts for an aneurysm of an anomalous aberrant right subclavian artery has not been described. This technique is the first in the literature and may provide options to patients and physicians wishing to avoid complications or technical issues associated with traditional surgical or hybrid surgical-endovascular procedures. In addition, it avoids placement of endovascular plugs and risk of erosion, plug failure, or plug migration.
Other approaches would involve a traditional open approach or a hybrid approach of open and percutaneous closure. A hybrid approach may involve a surgical right carotid-subclavian artery bypass and a percutaneous exclusion of the diverticulum and subclavian artery aneurysm with a Zenith iliac plug, as described by Kedora et al. 13 Amplatzer septal occluders have also been used for this purpose. 14 Left carotid-subclavian artery bypasses along with a TAG endoprosthesis have also been reported. 15 For our case, we wished to avoid open surgery given the patient’s advanced age and comorbidities and felt that a percutaneous option with a fenestrated graft in conjunction with covered stents for side branch preservation would allow successful aneurysm exclusion.
Given the added risks of general anesthesia and open surgery, it is our opinion that percutaneous approaches should be the initial choice in patients with suitable anatomy. Our case illustrates the feasibility of using thoracic aortic and subclavian stent grafts while preserving the take-off of all aortic arch vessels, effectively excluding the target vessel aneurysm.
Footnotes
Conflict of interest
Dr Sievert’s institution has ownership interest in or has received consulting fees, travel expenses or study honoraries from the following companies: Abbott, Access Closure, AGA, Angiomed, Arstasis, Atritech, Atrium, Avinger, Bard, Boston Scientific, Bridgepoint, Cardiac Dimensions, CardioKinetix, CardioMEMS, Coherex, Contego, CSI, EndoCross, EndoTex, Epitek, Evalve, ev3, FlowCardia, Gore, Guidant, Guided Delivery Systems, Inc., InSeal Medical, Lumen Biomedical, HLT, Kensey Nash, Kyoto Medical, Lifetech, Lutonix, Medinol, Medtronic, NDC, NMT, OAS, Occlutech, Osprey, Ovalis, Pathway, PendraCare, Percardia, pfm Medical, Rox Medical, Sadra, SJM, Sorin, Spectranetics, SquareOne, Trireme, Trivascular, Velocimed, Veryan.
All other authors have no disclosures.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
