Abstract
Acute systemic (anaphylaxis and anaphylactoid) reactions have been well described in patients with heparin-induced thrombocytopenia (HIT). Both necrotizing and non-necrotizing skin lesions at heparin injection sites have been reported and may occur in 10–20% of patients with HIT. We report herein a patient treated with subcutaneous enoxaparin sodium who developed non-necrotizing erythematous skin lesions at enoxaparin sodium injection sites. A subsequent intravenous bolus of unfractionated heparin produced a fatal anaphylactoid reaction. This suggests that caution should be exercised in the administration of intravenous heparin to patients with non-necrotizing erythematous skin lesions at prior heparin injection sites.
Introduction
Heparin-induced thrombocytopenia (HIT) is an adverse drug reaction that results from the formation of immunoglobulin G (IgG) antibodies to ultra-large heparin platelet factor 4 complexes. 1 It is associated with increased risk of arterial and venous thrombosis via platelet aggregation and micro-particle release.1,2 HIT occurs more commonly with unfractionated heparin (UFH) than with low-molecular weight heparin. 3 Skin lesions at heparin injection sites occur in 10–20% of patients with HIT and may precede thrombocytopenia. 4 We report herein a case of HIT associated with a fatal anaphylactoid reaction to UFH that was preceded by therapy with enoxaparin sodium and was heralded by a non-necrotizing erythematous rash at heparin injection sites.
Case history
A 64-year-old male with no history of cardiovascular disease or hematologic disorders underwent successful left total knee replacement at a community hospital. His pre-operative complete blood count, platelet count, international normalized ratio (INR) and activated partial thromboplastin time were normal. Enoxaparin sodium (Sanofi-Aventis), 30 mg subcutaneously every 12 hours, was initiated 24 hours after surgery with the intent of continuing it for 21 days. He experienced no adverse reaction to enoxaparin during his hospitalization and was discharged on post-operative day 5.
Four days later, the patient developed a non-necrotizing erythematous rash at enoxaparin sodium injection sites on right lower and left upper quadrants of the abdominal wall (Figure 1) and pitting edema in the right leg extending from the upper thigh to the ankle. He was evaluated in the emergency department of his community hospital. Vital signs were as follows: blood pressure, 125/52 mmHg; pulse rate, 87 beats per minute; breathing rate, 22 per minute; and temperature, 38.8°C. The cardiac and pulmonary examinations were normal. Fearing deep vein thrombosis his physician administered an intravenous bolus of 8000 units of UFH while awaiting duplex ultrasound evaluation of the lower extremity veins. Within 10 minutes the patient developed severe dyspnea, flushing and hypotension. Diphenhydramine (50 mg), methylprednisolone (125 mg) and epinephrine (1 mg of a 1:1000 solution) were administrated by intravenous bolus. Shortly thereafter, he developed monomorphic ventricular tachycardia followed by asystole. Basic and advanced cardiac life support were conducted for 40 minutes with eventual restoration of a pulse and blood pressure. His electrocardiogram showed new right bundle branch block. Serum creatine kinase (CK)-MB and troponin levels were normal.
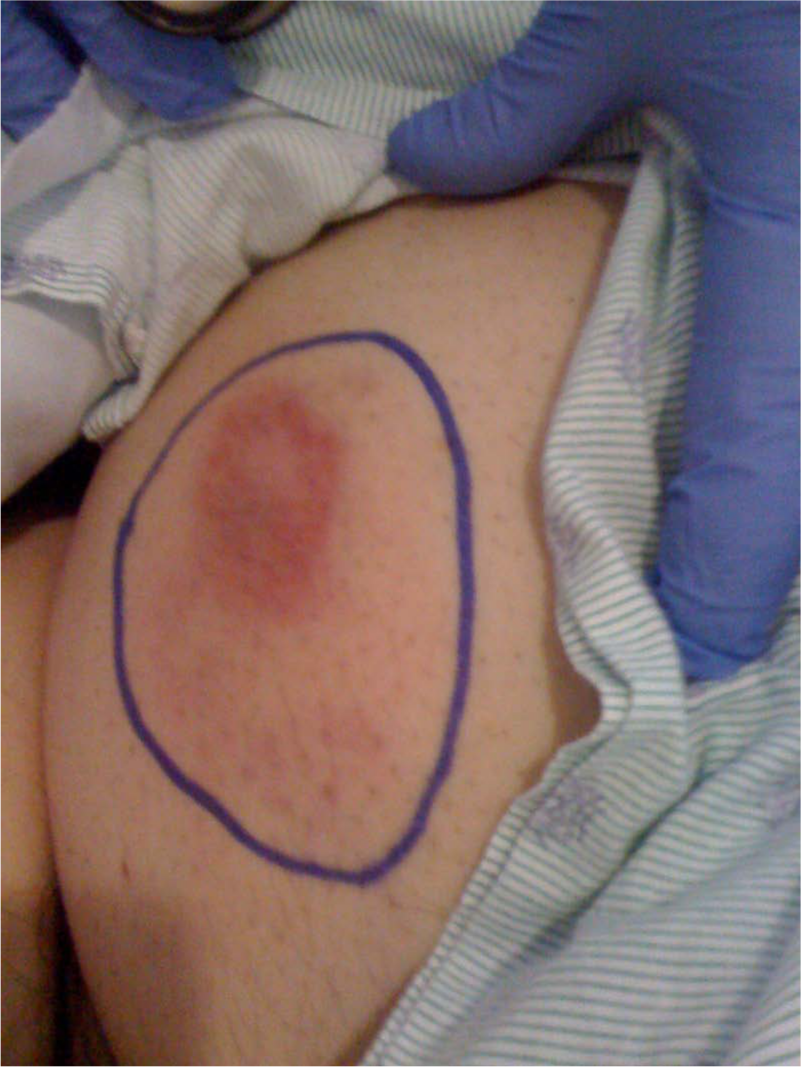
Non-necrotizing erythematous skin lesion at an enoxaparin sodium injection site in the right lower quadrant of the abdominal wall.
He was transferred to the Cleveland Clinic for further evaluation and management. On admission his blood pressure was 171/83 mmHg and his heart rate was 130 beats per minute (sinus rhythm). He was intubated and unresponsive, but afebrile. Neurological examination showed decerebrate posturing with absent gag and corneal reflexes. The erythematous rashes on the abdomen and pitting edema of the left leg were noted. Laboratory studies showed a leukocyte count of 12,800 cells/cm3, a blood hemoglobin of 8.4 g/dl, a platelet count of 45,000 cells/µl (Figure 2), a serum CK-MB of 5.4 ng/ml and a serum troponin of 1.13 ng/ml. Six hours later his platelet count was 90,000 cells/µl (Figure 1). Immune-mediated, HIT was suspected. Bivalirudin (Angiomax®; The Medicines Company, Parsippany, NJ, USA) 0.1 mg/kg/hour was administered intravenously. An ELISA assay for anti-platelet/heparin factor 4 antibodies was positive with an optical density of 2.148. Contrast-enhanced computed tomography showed no evidence of pulmonary emboli in the main, central, lobar or proximal segments of the pulmonary arteries. Duplex ultrasound examination of the lower extremity veins showed no deep vein thrombosis. A computed tomogram of the brain and a transthoracic echocardiogram were normal. The patient’s platelet count normalized within 24 hours (Figure 2). Ultimately, a serotonin release assay (Blood Center of Wisconsin, Milwaukee, WI, USA) confirmed the diagnosis of HIT. A non-contrast magnetic resonance imaging (MRI) scan of the brain suggested the presence of extensive hypoxic brain injury. At the family’s request, care was withdrawn and the patient died shortly thereafter. On autopsy, chronic total occlusion of the right coronary artery and subsegmental pulmonary emboli were identified.
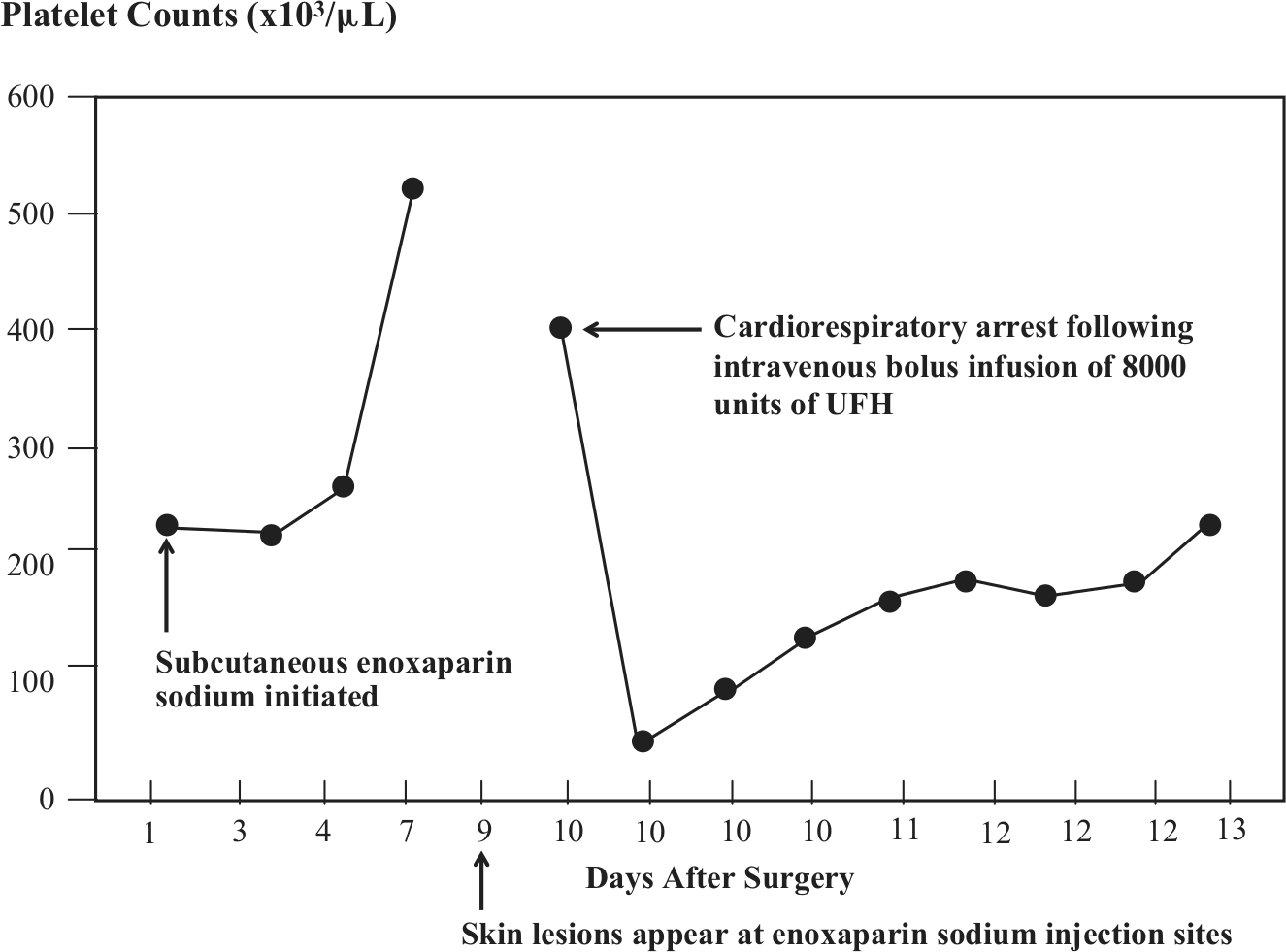
Platelet counts after initiation of enoxaparin sodium 30 mg subcutaneously b.i.d. and following cardiorespiratory arrest related to intravenous bolus administration of 8000 units of UFH (unfractionated heparin).
Discussion
Anaphylaxis is a rapid, progressive, life-threatening allergic reaction mediated by sudden mast cell and basophil degranulation. Immunologic (immunoglobulin E (IgE) binding to antigen) and non-immunologic mechanisms may precipitate anaphylaxis. Anaphylactoid reactions are those that resemble anaphylaxis, but are mediated by different mechanisms than anaphylaxis. Acute anaphylactoid reactions to bolus intravenous infusion of heparin have been described.5,6 Clinical features may include fever, chills, hypotension, tachycardia, dyspnea, chest pain, transient ischemic neurologic deficits, transient global amnesia and cardiorespiratory arrest within 5 to 30 minutes after a heparin bolus. 7 This sequence occurs most commonly in patients who have received recent subcutaneous heparin and are then exposed to an intravenous bolus infusion of heparin.6,7 Such anaphylactoid reactions have been linked to HIT. 8 Current guidelines recommend prompt measurement of the platelet count as thrombocytopenia may be abrupt, severe, and transient. 3 Prior reports indicate that IgG as well as IgE can mediate immediate hypersensitivity reactions to protamine, lepirudin and thiamine by activating the complement pathway leading to generation of anaphylatoxins (C3a, C4a and C5a).9–11 It is plausible that this could be the mechanism of heparin-induced anaphylactoid reactions.
In earlier reports, skin lesions at heparin injection sites were thought to represent a localized dermal expression of HIT characterized by microvascular thrombosis and associated with IgG anti-platelet factor 4 antibodies.1,4,12 More recently, non-necrotizing skin lesions associated with low-molecular weight heparin injections were described to be only weakly associated with HIT or anti-platelet/heparin factor 4 antibodies and were classified as a delayed-type hypersensitivity reaction. 12 However, in this case, these skin lesions clearly heralded the onset of HIT.
In summary, a patient ultimately diagnosed with HIT suffered a fatal anaphylactoid reaction to intravenous UFH following administration of subcutaneous enoxaparin sodium that was heralded by a non-necrotizing erythematous rash at injection sites. Accordingly, we urge caution in the administration of intravenous heparin to patients with such skin lesions at prior heparin injection sites.
Footnotes
Conflict of interest
The authors report no conflicts of interest.
Funding
This research received no specific grant from any funding agency in the public, commercial or not-for-profit sectors.
