Abstract
Since the long-term patency of axillofemoral (AXF) grafts is inferior to aorto-bifemoral (ABF) grafts, limb salvage procedures are crucial in this group of patients. Emerging endovascular devices have helped in the successful restoration of flow for acute limb ischemia in both native arteries as well as bypass grafts. One such device, the Trellis™ thrombectomy system is being used more frequently in this setting. The device has previously been used in veins, native arteries, and rarely in aortofemoral grafts. We present its first successful use for the treatment of occluded AXF bypass graft. The use of this device helped to isolate the treatment zone in the occluded graft, which allowed the use of a lower dose of thrombolytics, less systemic release of thrombolytics, and less distal embolization. Resolution of extensive clot burden was achieved and, with subsequent stenting of the graft at the distal anastomotic site, arterial flow to the leg through the AXF graft was restored and a revision surgery was avoided.
Introduction
Aortoiliac disease is a frequent cause of lower extremity ischemia in patients with traditional cardiovascular risk factors. Aorto-bifemoral (ABF) bypass surgery has remained a cornerstone of severe aortoiliac disease owing to higher patency rates.1,2 However, elderly and debilitated patients can have adverse outcomes with direct anastomotic aortic reconstruction associated with ABF bypass strategy, and hence this modality of treatment may not be applicable to all patients. 3 Endovascular management of such conditions also has certain limitations. The TransAtlantic Inter-Society Consensus (TASC II) recommendations regarding endovascular repair, state that for type D lesions endovascular methods do not yield good enough results to justify them as primary treatment. For type C lesions, open revascularization produces superior long-term results; endovascular treatment should only be used when there is high risk associated with open repair. 4 Timaran et al. showed poor outcomes of iliac stenting compared to open surgical reconstruction in patients with type B and type C lesions who had poor run-off and external iliac disease. 5 Hence, for patients with diffuse aortoilliac disease, who remain unfit for the traditional ABF bypass surgery, axillofemoral (AXF) bypass grafting is occasionally considered to relieve claudication symptoms. The long-term patency of AXF grafts have traditionally been inferior to ABF grafts and hence limb salvage procedures are usually indispensable in this subgroup.6 –8 Surgical salvage options are limited considering the high risks associated with a second operation. Modern endovascular interventions are thus performed to preclude the risks associated with a second surgery. These interventions are being performed more efficiently and with seemingly good success with the advent of novel endovascular devices. One such device is the Trellis™ thrombectomy system (Covidien, Dublin, Ireland), which is being used more commonly for the treatment of acute limb ischemia. Though this device has previously been used in veins, native arteries, and, rarely, in aortofemoral grafts, we report its first successful use for the treatment of occluded AXF bypass graft.
Case report
A 56-year-old male with coronary artery disease, peripheral arterial disease (with a history of multiple lower extremity interventions), and hyperlipidemia, who was a current smoker, presented to our institution with Rutherford class IIb acute limb ischemia. The patient described complete numbness of the left lower extremity with eventual loss of power. He had a history of total occlusion of his left iliac and left common femoral artery about 3 years prior to this presentation. At that time, surgery was offered to the patient but he chose to be treated endovascularly. However, despite multiple endovascular techniques, recanalization of the occlusion was unsuccessful, and he eventually had a left AXF bypass graft placement. Of note, he also had a previous history of failed AXF graft on the right, which eventually led to right leg amputation. On examination, the patient was found to have absent left lower extremity pulses by palpation and Doppler. The ankle–brachial index (ABI) was 0.3. Since we had good evidence for acute limb ischemia, the patient was taken as a matter of urgency to the catheterization laboratory for evaluation.
Using a micropuncture technique and ultrasound guidance, a 5-Fr introducer was introduced retrograde into the left brachial artery. Left brachial, axillary, subclavian and left AXF bypass graft angiograms were performed with a Bernstein catheter (Infiniti Medical, Menlo Park, CA, USA). A total occlusion of the graft was noted (Figure 1). The lesion was crossed by an Asahi Astato guidewire (Asahi Intecc, Aichi, Japan) and further angiograms distal to the lesion revealed patent superficial femoral artery, diseased profunda femoris artery, and patent trifurcation vessels proximally with occluded posterior tibial artery.
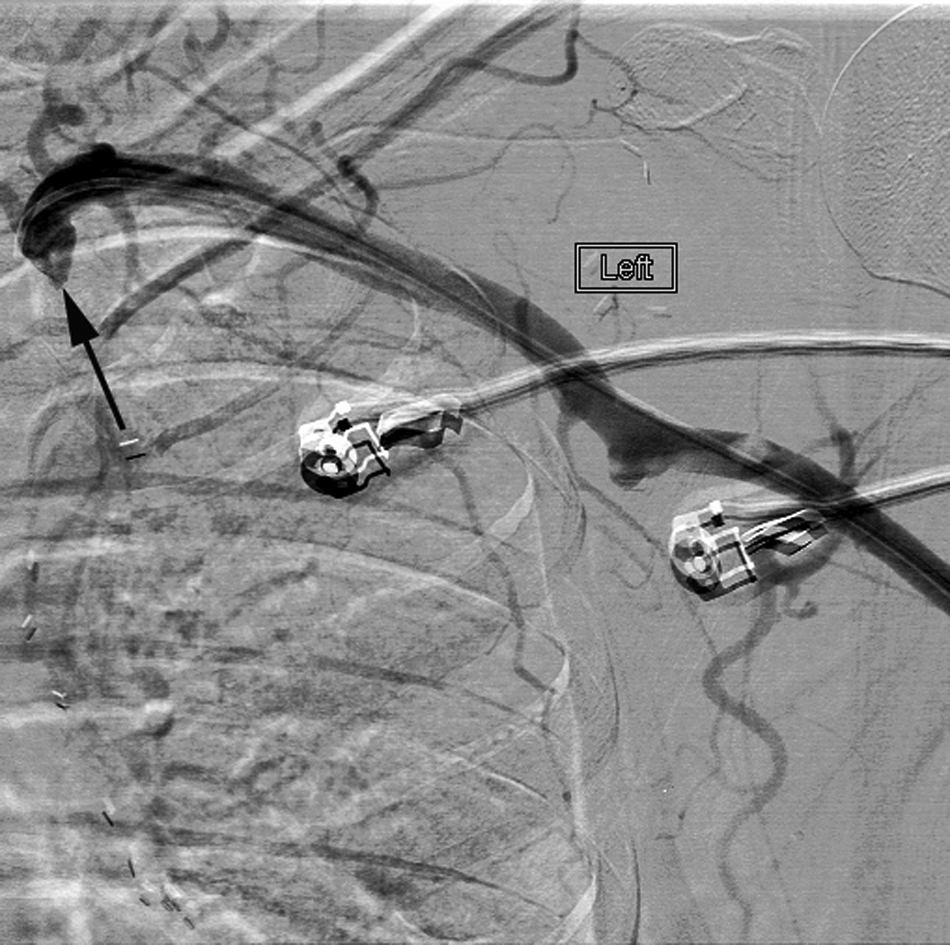
The initial angiogram reveals complete occlusion at the proximal part (arrow) of the axillofemoral graft.
After upsizing to a 6-Fr system, a 45 cm destination sheath was placed with its tip at the origin of the AXF bypass graft. Systemic heparin was started and 5 mg of recombinant tissue plasminogen activator (rtPA) was given to treat the distal thrombus. We then placed a 6-Fr 30 cm Trellis thrombectomy system (Covidien) with the distal balloon in the common femoral artery and the proximal balloon in mid-graft and administered 10 mg of rtPA over 12 minutes (Figure 2). A large amount of clot was removed. After this we placed the Trellis in the proximal part of the graft going from axillary to the mid-graft. Again, we gave 10 mg of rtPA for another 12 minutes and saw the entire clot burden relieved from the AXF bypass graft, subsequently restoring blood flow (Figures 3 and 4). There was a short stenosis noted at the distal anastomosis site, and hence we placed a 9 × 30 mm Sentinol® self-expanding nitinol stent (Boston Scientific, Natick, MA, USA) with about 1 cm extending into the common femoral artery. We then worked a Wholey™ (Covidien, Mansfield, MA, USA) wire up and down the tibio-peroneal vessels, restoring flow to the foot. Four milligrams of tPA was given through the distal catheter. The patient’s left foot showed remarkable improvement. It was pink with good capillary refill, and tone and power recovered immediately. The patient was transferred to the intensive care unit on aspirin, clopidogrel and heparin. The patient was discharged after attaining therapeutic anticoagulation on warfarin. A surveillance duplex done a month later demonstrated a patent graft, and a 6-month follow-up revealed the absence of any claudication symptoms since the procedure.
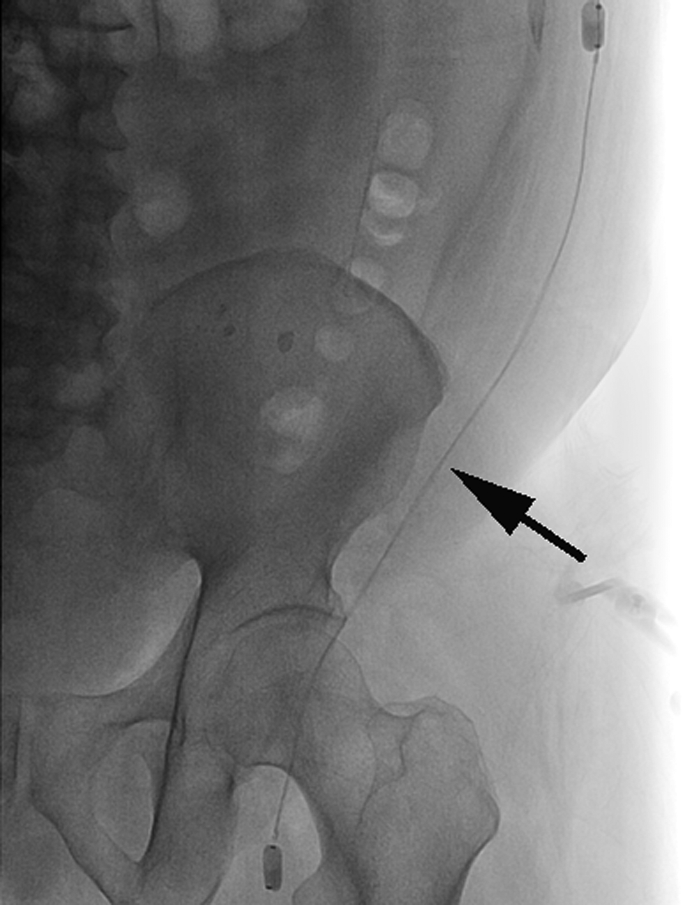
The arrow points to the Trellis™ device while it was being deployed in the axillofemoral graft.
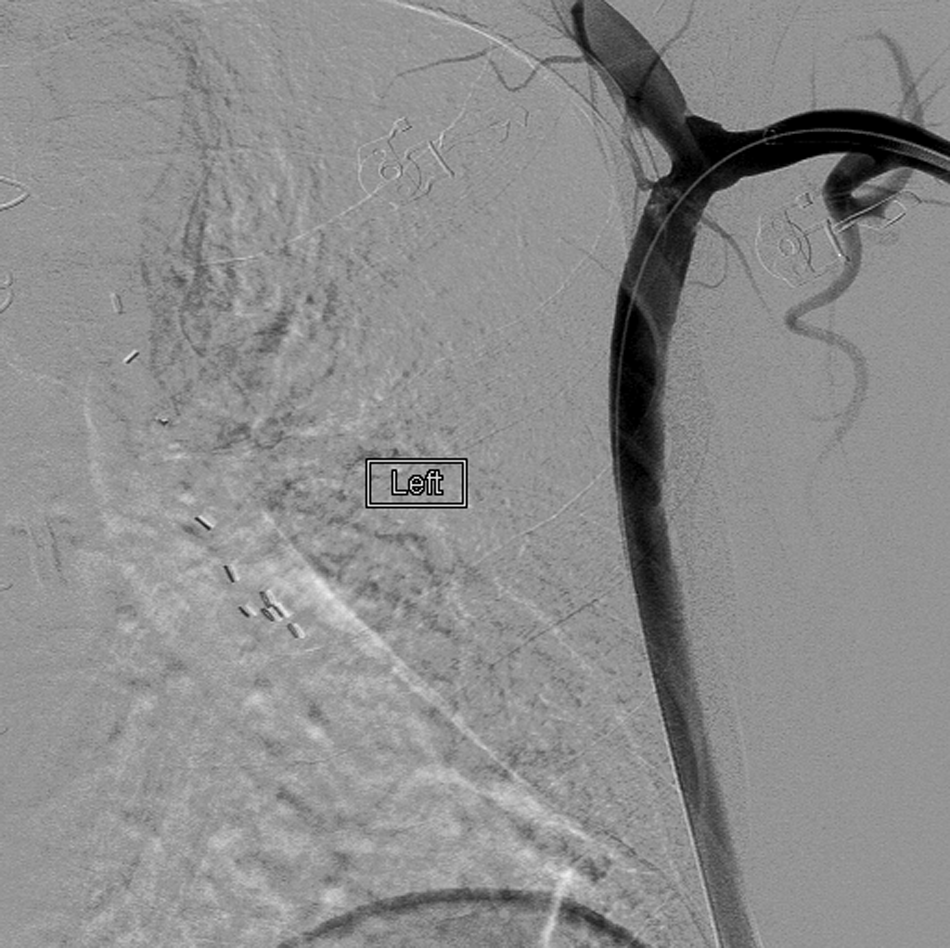
Restoration of flow in the proximal AXF graft.
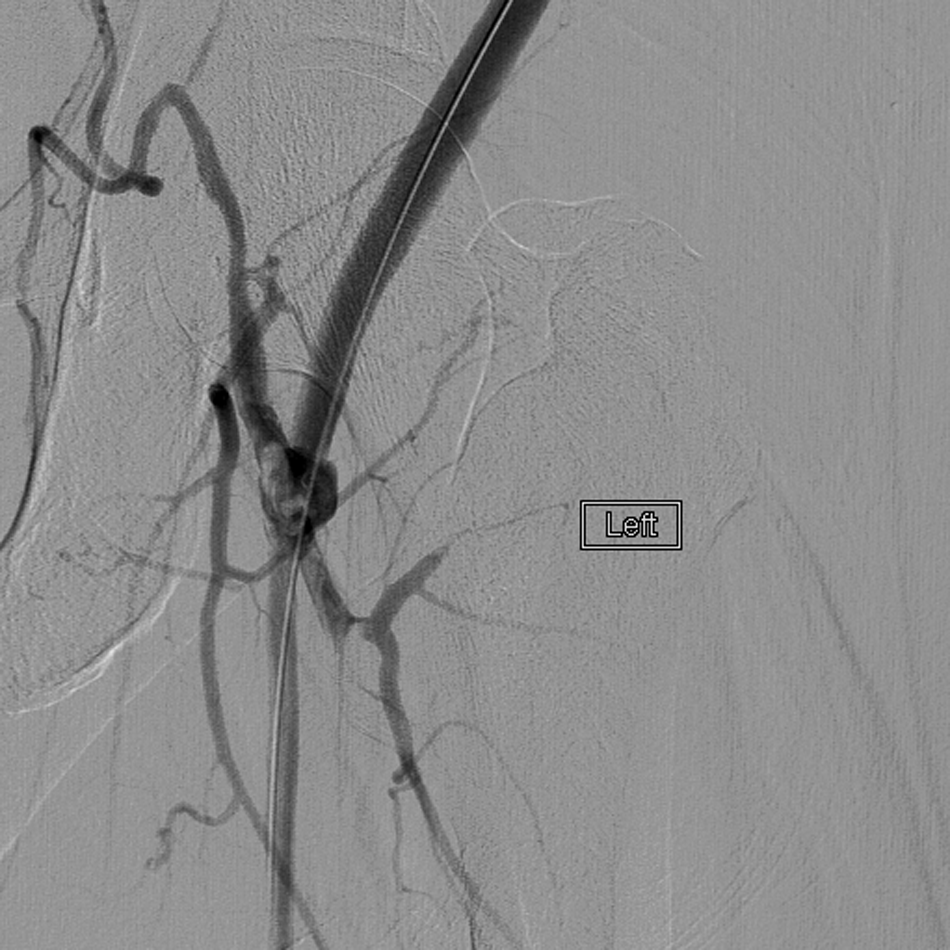
Restoration of flow in the distal AXF graft (before distal stent placement).
Discussion
Traditionally, surgical thrombectomy and bypass surgery has remained the mainstay of treatment for acute limb ischemia. However, some patients tend to have aggressive atherosclerotic disease and are therefore faced with the prospect of multiple target vessel revascularization procedures. Repeat surgeries is usually not an option in a majority of these patients in light of high surgical risks, lack of adequate grafts and diffuse atherosclerosis precluding optimal distal graft targets. Hence, in recent years, percutaneous options have emerged as excellent alternatives for restoring graft patency. In fact, studies have shown that catheter-directed thrombolysis (CDT) alleviated limb ischemia and reduced the need for surgical intervention, limb loss or mortality. 9 Nevertheless, thrombolysis comes with its own inherent risks of bleeding and need for prolonged infusions. 9 Percutaneous mechanical thrombectomy (MT), especially the AngioJet® rheolytic thrombectomy system (Possis Medical, Inc., Minneapolis, MN, USA), has displayed varying degrees of success in recanalizing endografts based on previous case series,10,11 but has the disadvantages of hemolysis, hyperkalemia and bleeding (when used with thrombolytics). Other limitations of rheolytic thrombectomy include the potential for distal embolization (especially when used without embolic protection) and the inability to remove chronic or insoluble thrombus. 12 Studies on venous interventions have revealed that combining MT with CDT (i.e. pharmaco-mechanical thrombectomy (PMT)) has the potential to achieve more complete clot removal as well as to decrease the dose and duration of thrombolytic therapy than either therapy alone. 13 Two primary PMT techniques that are being utilized for venous interventions are the AngioJet rheolytic thrombectomy system using the power-pulsed spray technique (Possis Medical, Inc.) and the Trellis thrombectomy system (Covidien). Limited data are available on the comparative efficacy of these two devices in peripheral arterial interventions. Though the Trellis thrombectomy system is preferred at our institute, physician preference usually determines which device is used in peripheral arterial interventions.
The choice of treatment depends on the length of the diseased segment and the duration of occlusion. Surgical treatment is usually favorable in long diseased segments (> 30 cm) and with chronic occlusions (> 14 days).14 –16 Catheter-based treatments are usually preferred for patients with acute occlusions and short segments of diseased endografts. Patients unsuitable for intra-arterial thrombolytic therapy (active internal hemorrhage, known bleeding diathesis, rapidly progressing sensory and motor deficits, and pregnancy) are usually the ones who fare better with surgical management. Patients like ours have diffuse atherosclerotic disease precluding good surgical targets, a lack of good quality grafts (in light of previous surgeries), and often have poor surgical risks for a repeat revascularization procedure after an occluded AXF graft. Hence, salvaging such bypass grafts is of paramount importance. Slovut and Bacharach 15 utilized rheolytic thrombectomy along with prolonged thrombolytic infusion via the Unifuse® catheter (Angiodynamics, Queensbury, NY, USA) for the percutaneous treatment of an AXF bypass occlusion with high residual clot burden.
The Trellis thrombectomy system has displayed good success in the treatment of symptomatic deep vein thromboses (DVTs). 17 In addition to this, few case reports have alluded to significant success in the treatment of localized thrombi of native arteries,18,19 aortofemoral bypass graft occlusion, 20 and in a bifurcated aortic endograft occlusion. 20 However, this was the first time that the Trellis thrombectomy system was successfully used in the recanalization of an AXF endograft. We believe that the novel design of the Trellis thrombectomy system helped us treat extensive clot burden without the need for post-procedure prolonged thrombolytic infusion (via the Unifuse catheter) as was needed in the case by Slovut. 15 Also, the distal balloon in this system prevented distal embolism of the clot during the pharmaco-mechanical thrombolysis of the occluded AXF graft.
The Trellis device is a 6-Fr multi-lumen dispersion catheter with treatment zone lengths ranging between 10 cm and 30 cm. Two of the five ports within this system are used to inflate proximal and distal compliant balloons, which when inflated isolate the treatment zone, maintain concentration of the thrombolytic agent, localize thrombolytic delivery, and prevent distal embolization. The fourth port or the proximal end-hole directs the dispersion wire (nitinol cable with a sinusoidal-shaped region) into the central lumen, where it resides within the isolated treatment zone. The dispersion wire is connected to a hand-held oscillation drive unit, which serves to oscillate the dispersion wire (can be confirmed fluoroscopically), thereby optimizing mechanical dispersion of the thrombolytic agent as the clot is fragmented. 20 After 10 minutes, the liquefied thrombus is aspirated through a fifth port, and the balloons subsequently deflated. The Trellis thrombectomy shortens treatment duration by combining mechanical and pharmacological lysis, allowing the use of a lower dose of thrombolytics, less systemic release of thrombolytics, and less distal embolization. 20 In our patient, owing to the extensive length of the occlusion throughout the endograft, we used this device twice to completely recanalize the AXF bypass endograft. We believe this was the first case of successful percutaneous recanalization of an AXF endograft by sequential use of the Trellis thrombectomy system.
Mechanisms of AXF graft occlusion include stenosis at the proximal or distal anastomosis, graft kinking, and progression of arterial occlusive disease distal to the bypass. 21 Unsuspected subclavian or axillary disease also may cause graft failure. The mechanism of AXF graft failure in our patient remains unknown. However, we believe that a combination of medication non-compliance (previously prescribed regimen that included aspirin, clopidogrel, and atorvastatin), aggressive atherosclerotic disease and distal anastomotic disease might have contributed to the occlusion of this patient’s AXF graft. General principles regarding prevention of graft occlusion include treatment of inflow and outflow disease and aggressive medical management. 21 Our patient had no significant inflow disease but did have some stenosis at the distal anastomotic site; hence, the placement of the nitinol stent in our patient.
Conclusion
In patients with diffuse aortoilliac disease, hostile abdomen, high cardiovascular risks, and an occluded AXF bypass graft, the options for revascularization are often limited. Although the Trellis thrombectomy system has been successfully used before in occluded veins, native arteries and aortofemoral grafts, this case represents its first successful use for pharmaco-mechanical thrombectomy in an occluded AXF bypass endograft. However, it is imperative to delineate the cause of such endograft occlusions in each patient. Hence, assessment of inflow and outflow disease followed by appropriate intervention should always be undertaken post-recanalization to prevent future recurrences.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors report no financial relationships or conflicts of interest regarding the content herein.
