Abstract
Isolated peripheral pulmonary artery stenosis presenting in adulthood is an uncommon cause of right ventricular hypertension and may be misdiagnosed as primary or thromboembolic pulmonary hypertension. Treatment consists of balloon angioplasty and stenting of the affected vessels, although the procedure can be complicated by reperfusion pulmonary edema.
Clinical summary
Diagnostic challenge
A 39-year-old primiparous woman with progressive dyspnea over 18 years presented for termination of pregnancy at 16 weeks’ gestation. She had been diagnosed with severe pulmonary hypertension 6 years earlier based on an echocardiogram and a cardiac catheterization and was advised against pregnancy. The coronary arteries and left ventricular function were normal, but pulmonary artery (PA) pressure was elevated (100/11 mmHg), pulmonary capillary wedge (PCW) pressure read 20/25 (mean 16) mmHg and right ventricular (RV) pressure 100/12 mmHg. Pulmonary angiography was not performed during the initial workup. The presumed diagnosis was severe primary pulmonary hypertension (PHtn). The patient had received no therapy for PHtn and had experienced a gradual increase in dyspnea, which worsened rapidly during pregnancy.
Physical examination revealed no dysmorphic features. She was afebrile, the heart rate was 80 bpm, blood pressure was 110/70 mmHg, respiratory rate 16/min, and pulse oximetry measured 100% breathing room air. The lungs were clear. Cardiac auscultation disclosed accentuation of the pulmonic component of the second sound and a harsh upper left parasternal systolic murmur that radiated to the back. The electrocardiogram showed normal sinus rhythm, RV hypertrophy, right axis deviation and a right atrial abnormality. Transthoracic echocardiography showed normal left ventricular systolic function, severe RV hypertrophy and systolic dysfunction, and an estimated RV systolic pressure of 122 mmHg, but the peripheral pulmonary arteries were not visualized. Radionuclide perfusion imaging and computerized tomography were deferred because of pregnancy. Right heart catheterization demonstrated a PCW pressure of 10, PA pressure 102/15 (mean 44), RV pressure 102/3, and right atrial (RA) pressure 3 mmHg, and a cardiac index (CI) of 2.5 l/min/m2. Pulmonary angiography (Figure 1A,B) revealed severe stenosis of the distal right and proximal left pulmonary arteries as well as more peripheral branches as the cause of proximal PHtn.
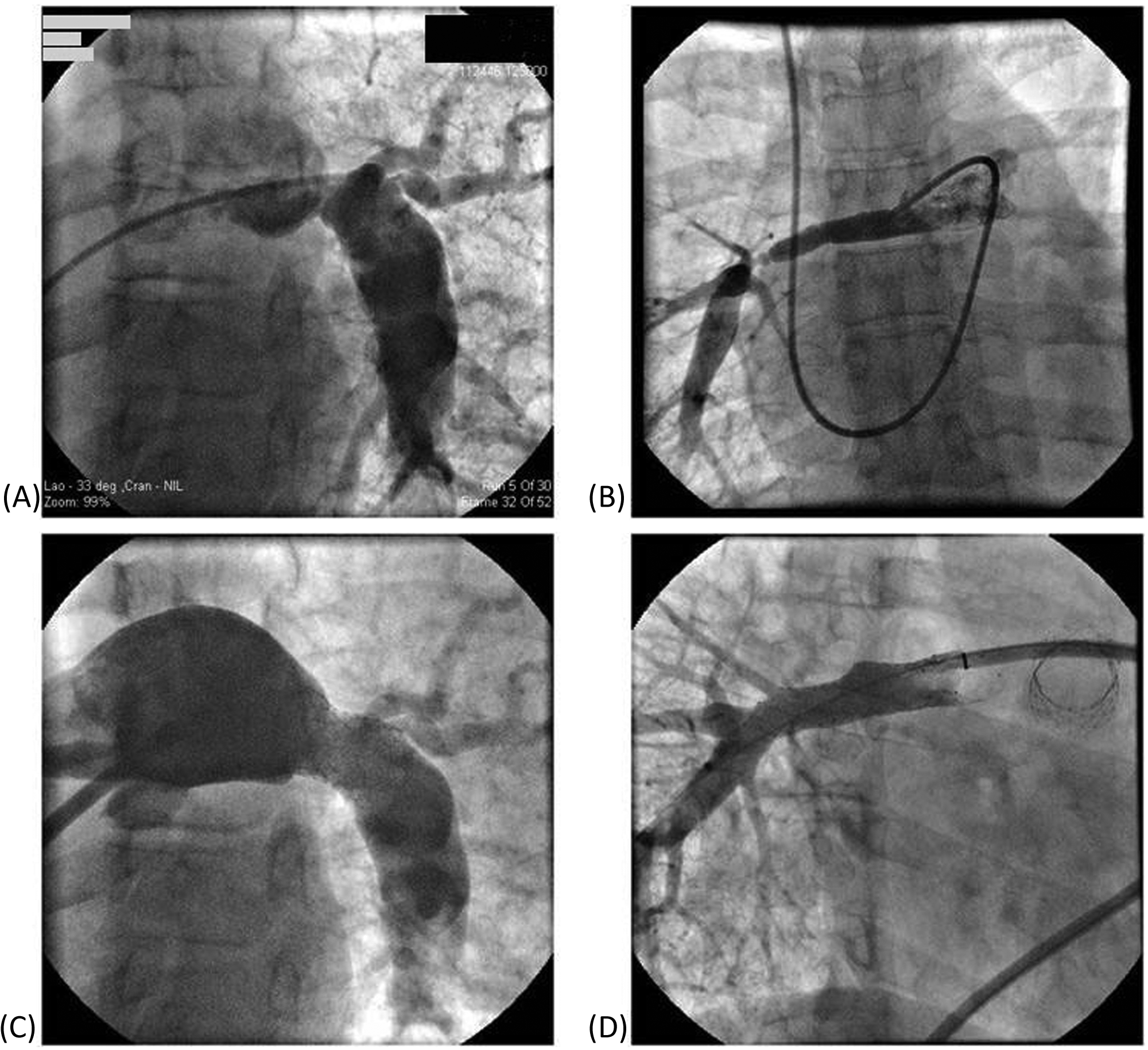
(A) Left pulmonary arteriogram showing severe stenosis of the left proximal pulmonary artery, dilated main pulmonary artery and post-stenotic dilatation of the left pulmonary artery. (B) Right pulmonary arteriogram showing stenosis of the right distal pulmonary artery and its branches. (C) Left pulmonary arteriogram after balloon angioplasty and stenting of the left proximal pulmonary artery. (D) Right pulmonary arteriogram after balloon angioplasty and stenting of the right distal pulmonary artery and balloon angioplasty of its branches.
Therapeutic challenge
The patient underwent staged balloon angioplasty. The first procedure involved stenting of the proximal left PA to reduce RV pressure (Figure 1C). Three days after stenting, however, she became febrile to 38.3°C and hypoxemic to 80% saturation with pulmonary edema (Figure 2A). Emergent angioplasty and stenting of the right PA (Figure 1D) was followed by resolution of pulmonary edema (Figure 2B). The proximal PA pressure improved to 54/12 (29) mmHg, RV pressure 56/13, and RA pressure 7 mmHg. Despite improvement in pulmonary status and because the patient also had three large uterine fibroids, she was advised by the obstetrical team to terminate the pregnancy. The patient proceeded with elective termination of the pregnancy. Follow-up perfusion imaging revealed 39% of total perfusion to the right lung and 61% to the left. The patient was discharged home, reporting resolution of dyspnea and much improved exercise tolerance. Echocardiography 1 week after the final procedure found the RV systolic pressure less than half systemic with normal systolic function.
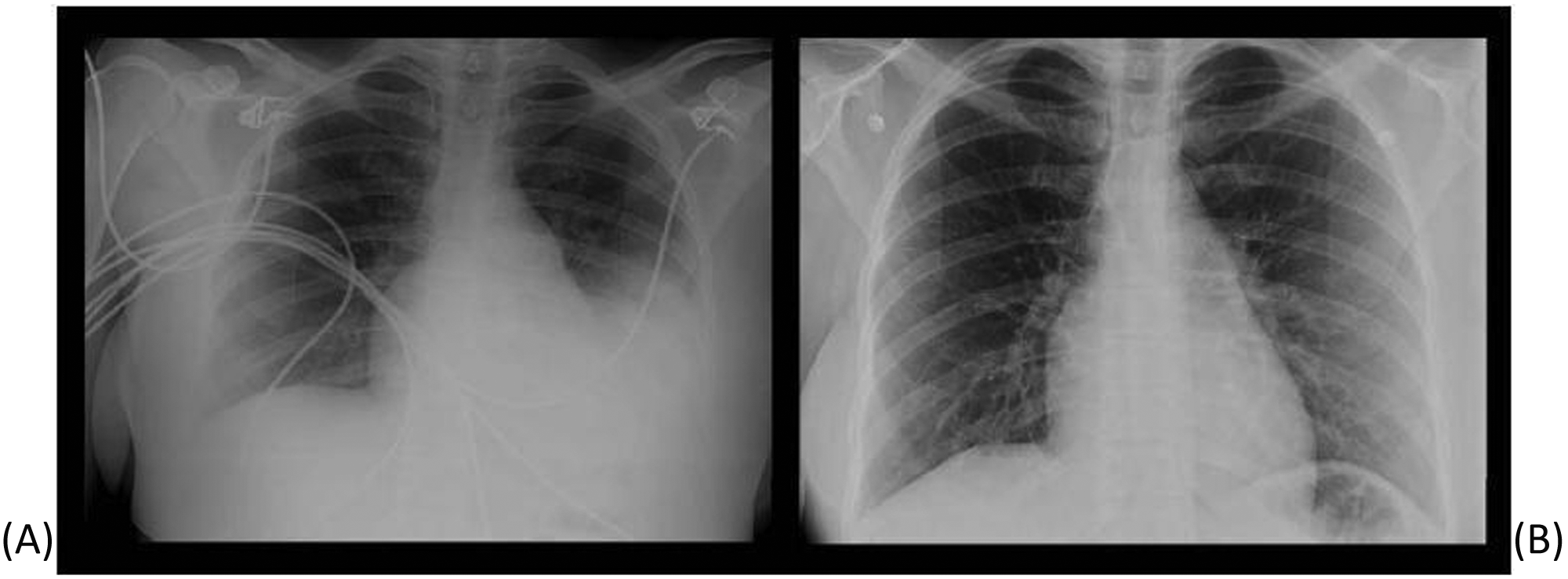
(A) Chest X-ray after balloon angioplasty and stenting of the left proximal pulmonary artery showing left pleural effusion. (B) Chest X-ray after balloon angioplasty and stenting of the right distal pulmonary artery showing resolution of left pleural effusion.
Discussion
Diagnostic challenge
Pulmonary artery hypertension is grouped as (I) idiopathic or due to disease of the pulmonary muscular arterioles (due to connective tissue disease, HIV, congenital heart disease, or other disorders); (II) due to left heart disease; (III) a consequence of lung disease or chronic hypoxemia; (IV) secondary to thromboembolic occlusion of the pulmonary vasculature; or (V) due to miscellaneous other mechanisms. 1 The diagnostic algorithm begins with a detailed history and physical examination, chest X-ray and electrocardiogram, followed by transthoracic echocardiography (TTE). Exercise-echocardiography or transesophageal echocardiography (TEE) may be helpful in certain cases. If the echocardiogram suggests left heart disease, management proceeds accordingly. If not, the patient’s symptoms should guide further testing to determine the etiology of PHtn. Radionuclide ventilation–perfusion imaging followed by pulmonary angiography, CT angiography, and tests of coagulation may suggest thromboembolism. Spirometry and oximetry contribute to the assessment of pulmonary pathology. Other laboratory testing and polysomnography may identify sleep-disordered breathing or medical conditions such as HIV infection. Finally, right heart catheterization and responses to exercise or volume loading confirm the diagnosis of PHtn and delineate the hemodynamic profile. 1 In the patient described, the PA and PCW pressures on initial catheterization were reportedly elevated. The PCW pressure may have been inaccurately measured by a ‘partially wedged’ catheter, resulting in a reading higher than the true wedge pressure. The elevated PA pressure may have been measured proximal to the site of stenosis. Otherwise, a drop in the PA pressure as the catheter passed the site of stenosis would have raised the suspicion of stenosis, rather than primary PHtn.
Isolated peripheral PA stenosis does not fall directly under any of the above-mentioned categories. It is unusual in adulthood and frequently misdiagnosed, especially as group IV PHtn. It can appear as an isolated congenital problem in childhood or more commonly seen as part of Noonan’s, Alagille’s or William’s syndromes.2,3 We are aware of few cases of acquired isolated peripheral PA stenosis in adulthood unrelated to specific congenital etiological factors.2,3 Single or multiple stenoses may be confined to the PA trunk or main PA branches, or affect more peripheral segmental branches. Branch PA stenosis can occur in adults with systemic inflammatory disease, such as Behçet’s disease, in association with PA aneurysmal disease, 3 or as a complication of thromboembolism, but can also occur in isolation. 2 The slow, non-uniform progression to segmental vascular obstruction due to isolated PA stenosis is typically associated with progressive dyspnea and fatigue in the second decade of life. 3 Lung perfusion imaging may reveal segmental defects but diagnosis typically requires direct arterial imaging by contrast-enhanced CT, magnetic resonance angiography or catheter-based arteriography.
Therapeutic challenge
Branch PA stenosis is difficult to treat surgically. Catheter-based angioplasty is the preferred, albeit not always successful, therapy. Stenting of the more proximal PA bed is generally reserved for adults and older children to obtain optimal vessel caliber.4,5 Stents implanted in children for PA stenoses provide long-term improvement, 5 although the durability of the procedure in adult patients has not been reported.
A possible early adverse consequence of stenting is acute pulmonary edema attributed to a sudden increase in perfusion of downstream (unconditioned) pulmonary vessels and a subsequent acute increase in distal PA pressure. 6 This likely occurred in our patient due to redistribution of blood to the left lung from the right lung after successful left PA stenting. The etiology and predictors of reperfusion pulmonary edema after PA angioplasty are not well understood, although it has been proposed that a greater increase in vessel diameter and distal PA pressure after ballooning leads to more significant reperfusion pulmonary edema. 6 We propose that more significant inequity in stenoses bilaterally predisposes to reperfusion pulmonary edema upon re-establishing blood flow to the side subtended by the more significant PA stenosis. Perhaps if both sides were re-perfused during a single procedure, this complication could be avoided.
Conclusions
We report an unusual case of bilateral peripheral PA stenosis in an adult causing proximal PA and right heart hypertension. The patient had developed dyspnea 18 years earlier and without therapy experienced only a mild decline in function, which would be unusual for untreated primary PHtn. In cases of atypical presentation of PHtn, diagnostic assessment should include direct arterial imaging. In patients with PA stenosis, balloon angioplasty and stenting provide symptomatic relief, but survival advantage is less certain, since prognosis may vary depending upon etiology. Pregnancy is not contraindicated in patients with PA stenosis, especially if the stenosis is managed and there is not significant RV strain that would predispose to heart failure or arrhythmias. In the patient described, long-standing stenosis likely lead to redistribution of blood flow to relatively few pulmonary segments, raising regional vascular resistance in these segments despite intervention, and increasing the risk during pregnancy. Reperfusion pulmonary edema after intervention is a potentially life-threatening complication and additional studies are needed to determine whether this can be avoided through specific therapeutic maneuvers.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
The authors listed do not have any conflicts of interest to declare.
