Abstract
Endovascular intervention for obstruction to venous drainage of the head and neck is an established treatment for disorders such as superior vena cava syndrome. Some patients with multiple sclerosis have been observed to have anomalies of the veins draining the head and neck. It is possible that some symptoms associated with multiple sclerosis may be secondary to disturbed venous flow. In an uncontrolled clinical series of 40 patients who had been previously diagnosed with multiple sclerosis, anomalies of the venous drainage of the head and neck were observed, including venous stenoses of the internal jugular veins. In 38 of 40 patients, venous stents were placed with restoration of luminal dimensions and abrogation of the venous pressure gradient. The angiographic and hemodynamic improvement was associated with improvement in symptomatology, most particularly in cognitive and constitutional symptoms that may be related to cerebrovenous flow. Serious complications included death in one subject and stent embolization requiring open heart surgery in another. In conclusion, in this series, endovascular intervention to correct venous stenosis associated with multiple sclerosis was associated with improvement in symptoms possibly related to disturbed venous hemodynamics. However, given the serious adverse events in this small series, a randomized clinical trial is required to confirm these findings, and to determine if the procedure has any effect on the progression of multiple sclerosis, or untoward long-term adverse effects.
Introduction
Anatomical anomalies in the extracranial veins draining the central nervous system (internal jugular, vertebral and/or azygous veins) have been reported in patients with multiple sclerosis (MS).1,2 Duplex ultrasound studies have suggested that these anomalies may alter cerebral venous hemodynamics, causing alterations in venous pressure and flow patterns. The association of anatomical cerebrovenous anomalies with altered cerebrovenous hemodynamics has been termed chronic cerebrospinal venous insufficiency (CCSVI).1,2
It seems likely that individuals with CCSVI as manifested by hemodynamically significant venous obstruction, might have neurological symptoms related to abnormal venous outflow and insufficient venous collateral formation. Accordingly, these patients may benefit from endovascular reconstruction to restore normal venous drainage from the head and neck (much as patients with superior vena cava syndrome benefit from endovascular correction of venous obstruction 3 ). Accordingly, one of us (MD) treated a series of patients with CCSVI associated with MS using percutaneous venous intervention. We hypothesized that correction of the abnormal venous drainage that can be associated with MS would improve the constellation of symptoms that might be directly related to disturbed cerebral venous flow (chronic fatigue, heat intolerance and impaired cognition). Accordingly, we performed a retrospective review of this innovative clinical therapy to determine the immediate and long-term effects of the treatment on venous hemodynamics and symptomatology.
Methods
This is a retrospective review of clinical care, approved by the Stanford IRB (Protocol #18203; Data collection for endovascular correction of cerebrovenous anomalies in patients with Multiple Sclerosis). We analyzed the results of an uncontrolled clinical series of 40 patients with MS who were assessed from April to October 2009 (Table 1). In these patients, the form of MS was characterized (by at least one neurologist) as relapsing-remitting in 23, secondary progressive in 10, and primary progressive in seven. Patients were also categorized by the authors, using the Expanded Disability Status Scale (EDSS), which quantifies disability in eight functional systems. 4 The disability scores were characterized prospectively, based on patient-reported data. As this was not a clinical research study, but rather innovative clinical care, no strict inclusion criteria were used in the selection of the subjects. Accordingly, all patients referred with MS (relapsing-remitting, primary progressive, secondary progressive) were accepted for evaluation and treatment.
Patient demographic and clinical characteristics
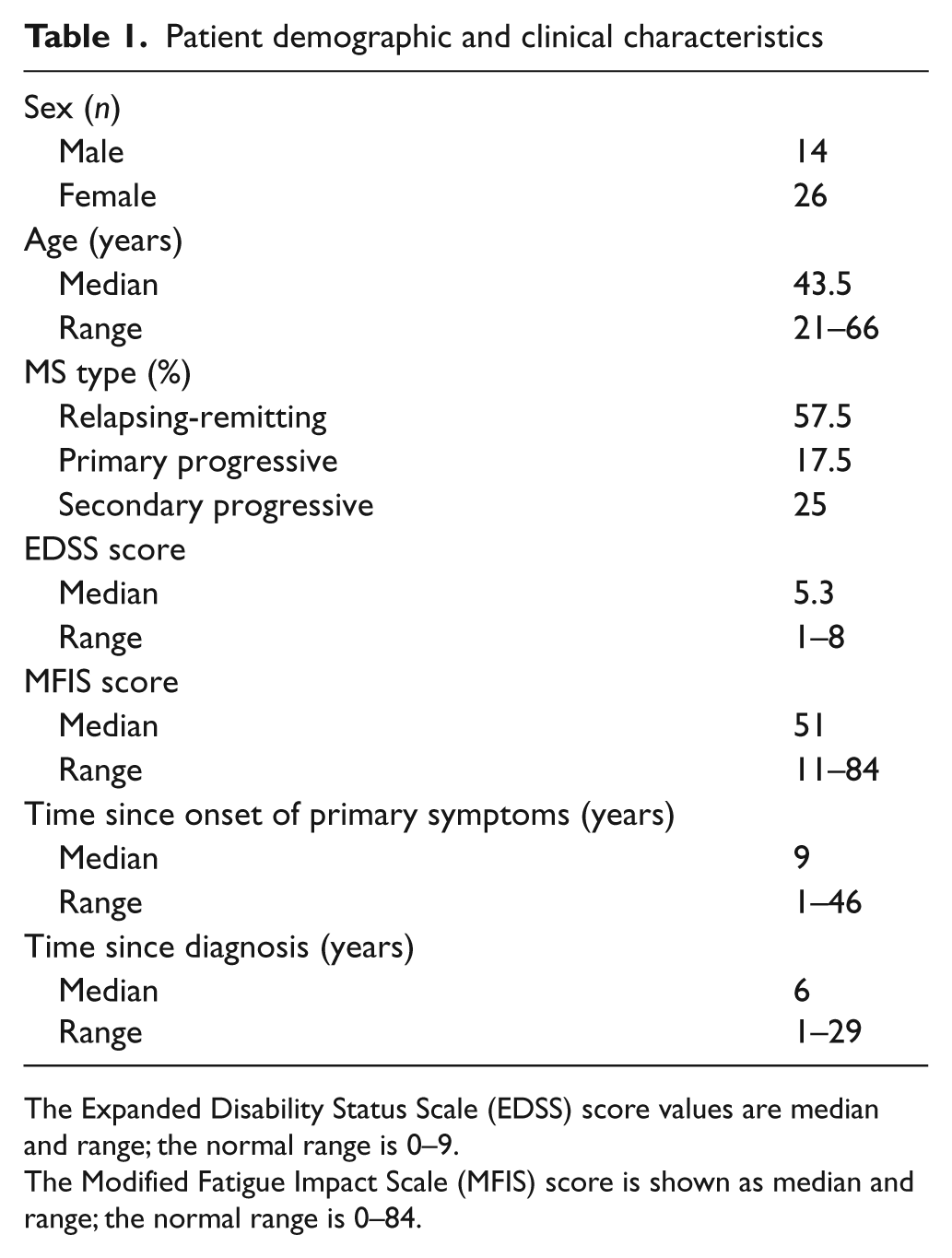
The Expanded Disability Status Scale (EDSS) score values are median and range; the normal range is 0–9.
The Modified Fatigue Impact Scale (MFIS) score is shown as median and range; the normal range is 0–84.
We assessed the patients’ symptoms using the Modified Fatigue Impact Scale (MFIS). The MFIS is a multidimensional scale developed to assess the perceived impact of fatigue on a variety of daily activities, and consists of 21 items. The items of the MFIS can be aggregated into three subscales (Physical, Cognitive, and Psychosocial), as well as into a total MFIS score. 5 All items are scaled so that higher scores indicate a greater impact of fatigue on a patient’s activities. The Total MFIS score can range from 0 to 84, as computed by adding scores on the Physical, Cognitive, and Psychosocial subscales. The MFIS was obtained prior to any procedure, and at 2 months and 1 year following venoplasty and stenting.
Magnetic resonance venography (MRV) was performed at Stanford University in all patients. In all cases, MRV revealed venous anomalies, which included internal jugular vein stenoses, venous valvular dysfunction or external compression due to soft tissue or bony anatomic variations. In most cases these venous abnormalities were associated with prominently enlarged venous collaterals, indicating the hemodynamic significance of the MRV findings (Figure 1). Patients provided informed consent for the imaging and the endovascular intervention, which specified that these procedures were not directed toward therapy of MS.
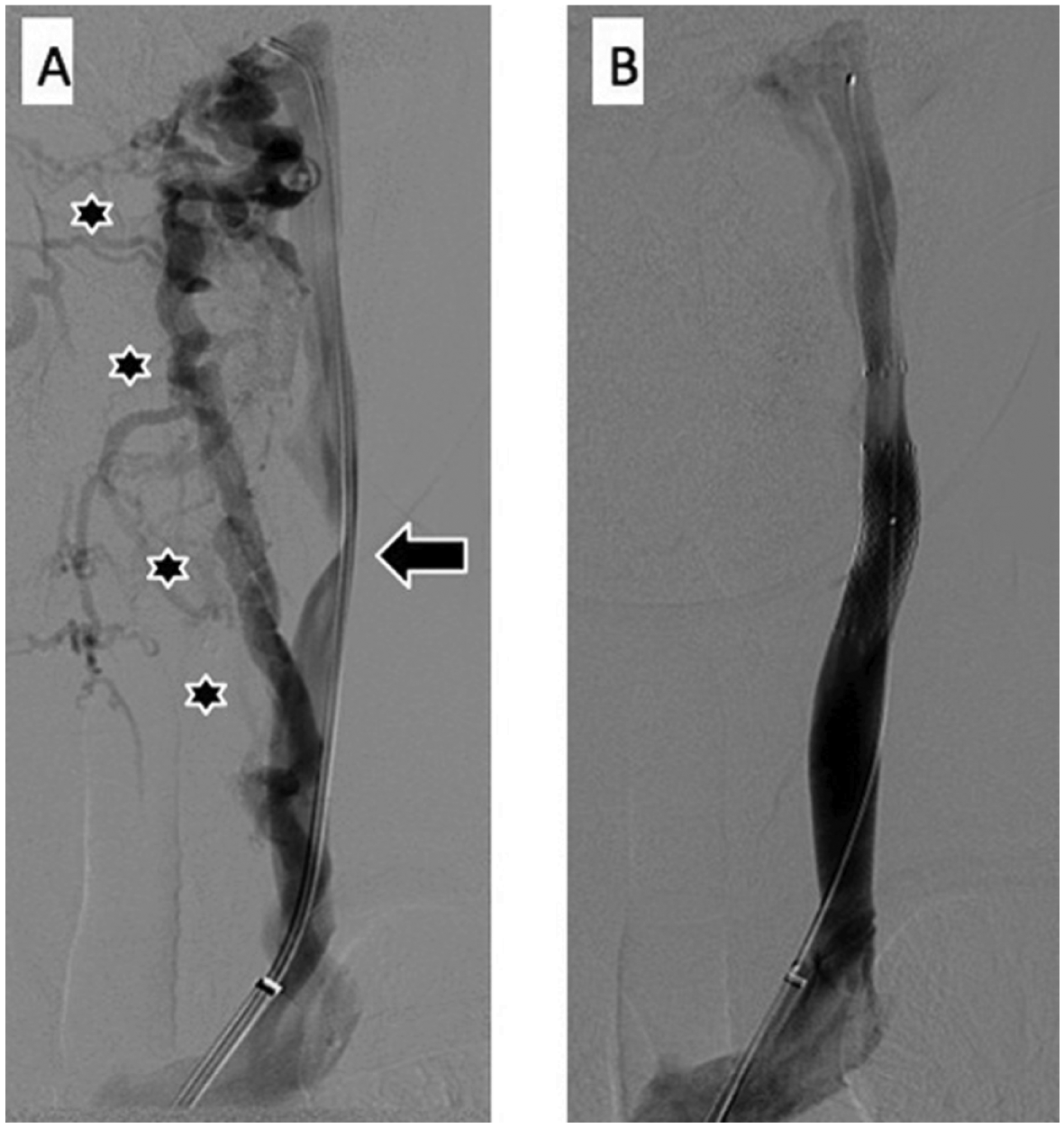
Frontal venograms of the left internal jugular vein before (A) and after (B) endovascular intervention in a 46-year-old woman with a history of multiple sclerosis and symptoms of extreme fatigue, heat intolerance, and cognitive difficulties. She underwent venography after initial non-invasive imaging evaluations showed bilateral internal jugular narrowing and a prominent network of collateral veins in the neck. (A) The diagnostic venogram of the left internal jugular vein performed with the catheter tip in its upper segment demonstrates an obstruction of the mid-jugular consisting of a parabolic medial narrowing of the contrast column caused by an ectatic carotid artery bifurcation (arrow). Also noted are large, medial collateral veins bypassing flow around the internal jugular (stars). The narrowing is extrinsic and was associated with a mean trans-stenotic pressure gradient of 4 mmHg. (B) Venographic image after placement of a self-expanding stent across the obstruction shows resolution of the abnormality and no filling of the previously conspicuous collaterals. In addition, the pressure gradient was obliterated (0 mmHg) after the intervention. In follow-up at 2 months, all of her symptoms had improved and there was a marked resolution of her fatigue.
The indication for therapy was the presence of extracranial venous obstruction on non-invasive imaging with MRV, which was then confirmed by catheter venography. If both imaging modalities suggested venous obstruction, the patient underwent intervention. Initial intervention consisted of percutaneous transluminal angioplasty (PTA) alone. If PTA was successful, no further intervention was indicated. In the event of suboptimal or failed PTA, defined as residual stenosis of the vein of 30% or greater, or no change in the pre-existing pressure gradient, stenting was then performed. Subsequently, venous balloon angioplasty, followed by endovascular placement of stents to address persistent venous stenosis, was performed in 38 of 40 patients (two patients were not treated). On average, 1.8 veins were treated per patient—including left and right jugular and/or azygous veins. A post-procedure venogram and, in most cases, measurement of the venous pressure gradient, were performed immediately thereafter, documenting an improvement in venous blood flow and pressure (Figure 2). Each patient had magnetic resonance imaging (MRI) of the brain with and without gadolinium before the procedure and 2 months after the procedure. MRI with and without gadolinium performed 2 months after the procedure showed no new or enlarging lesion(s).
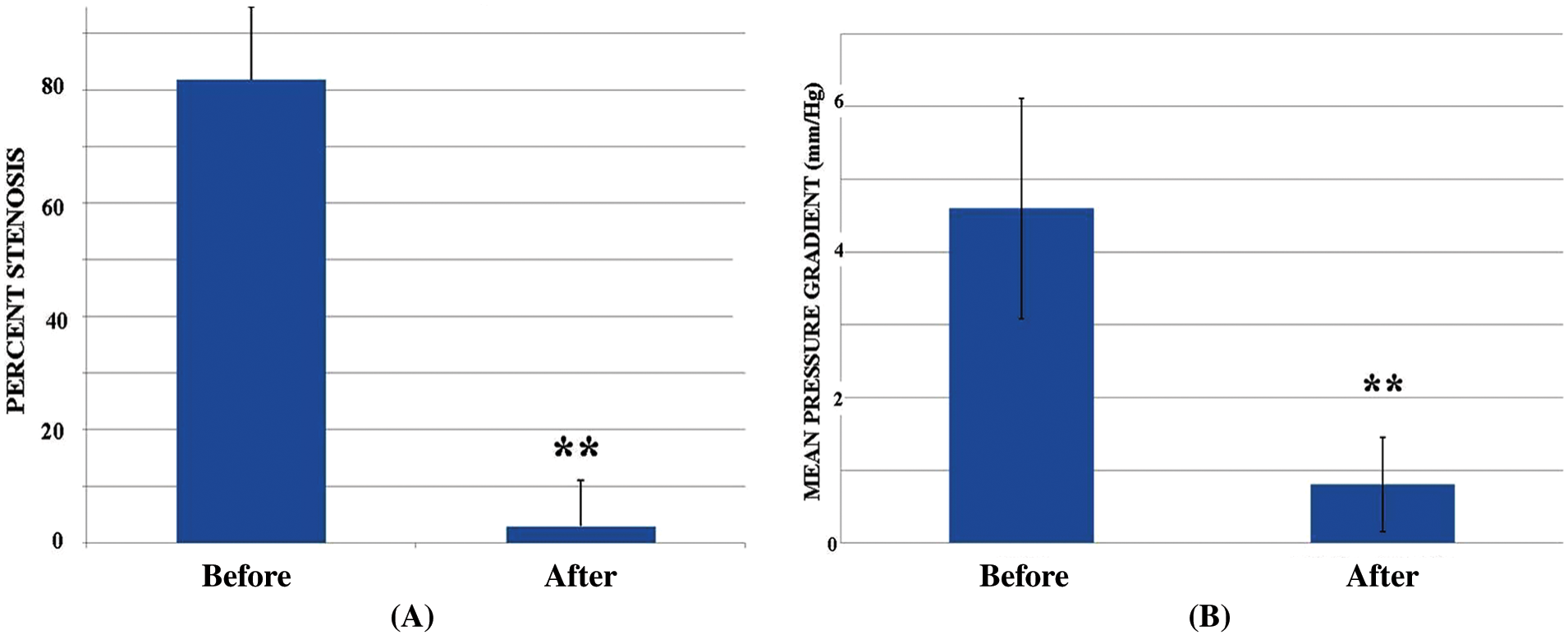
(A) The degree of venous stenosis (mean ± SD) before and immediately after stenting of 92 lesions in 38 patients. **p < 0.001, paired t-tests before and after the intervention. (B) The venous gradient (mean ± SD) before and immediately after stenting in the treated patients. Venous gradients were measured across 81 of the 92 lesions in 38 patients. **p < 0.001, paired t-tests before and after the intervention.
Results
During initial patient interviews, 22/40 patients reported cognitive impairment; 24/40 patients reported heat intolerance; and 40/40 patients reported fatigue. Fatigue was further assessed using the MFIS. Please note: no scoring system was used to assess heat intolerance or cognitive impairment. In total, 38/40 patients underwent a procedure. Following the procedure, patients noted subjective improvement in cognition, heat intolerance, and fatigue. Of these individuals, 71% described improvement in heat intolerance post procedure.
No patients were lost to follow-up. At 2 months of follow-up, the MFIS documented a substantial improvement in fatigue (Figure 3). At 1 year, this benefit appears to be maintained.
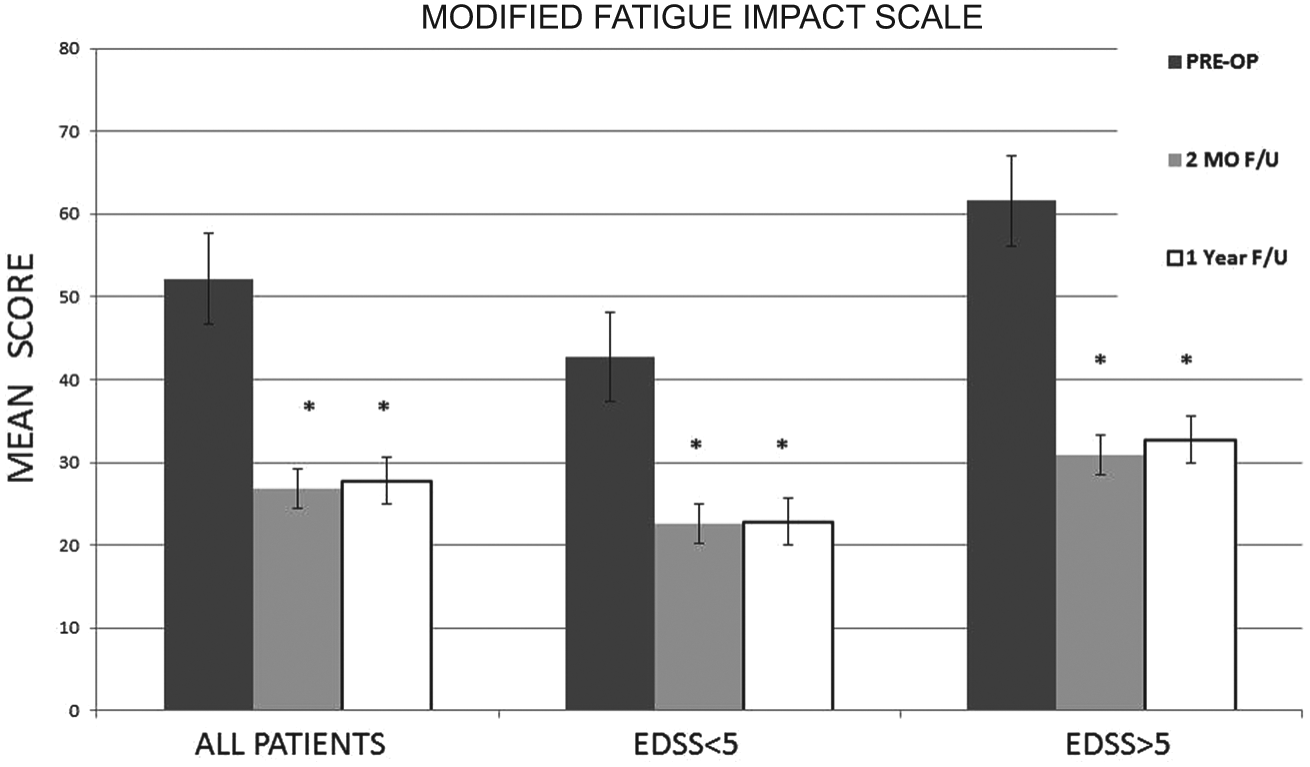
Improvement in the Modified Fatigue Impact Scale (MFIS (mean ± SD); *p < 0.01) in treated patients at 2 months and at 1 year of follow-up. In aggregate, patients improved considerably after the procedure. The improvement was maintained at 1 year. There was no difference in the degree of symptomatic improvement in patients who were more (EDSS > 5) or less (EDSS < 5) limited by their symptoms of multiple sclerosis (dns). (EDSS, Expanded Disability Status Scale.)
Adverse effects of the procedure were common. Approximately 80% of subjects had some degree of local neck discomfort after stent placement. A subset of these individuals noted discomfort with swallowing and/or occipital headaches. For most individuals, these local adverse effects spontaneously resolved in 4–5 days. Another 12% of subjects noted shoulder muscle weakness, sometimes associated with pain, presenting 7–10 days after the procedure. In some cases these symptoms persisted for up to 3–4 months, but also resolved with time. Two serious adverse events were observed. One patient incurred stent migration from the internal jugular vein to the tricuspid apparatus, requiring thoracotomy and surgical stent extraction. The patient recovered uneventfully. One patient suffered a fatal intracerebral hemorrhage on warfarin 10 weeks after the procedure.
Discussion
Chronic cerebrospinal venous insufficiency and multiple sclerosis
Zamboni and colleagues have marshaled evidence for the entity of CCSVI. Using Duplex ultrasonography and transcranial Doppler studies, they have documented the association of abnormal venous hemodynamics with MS. In one study of 109 MS patients, and 177 age- and sex-matched controls, subjects underwent a blinded assessment of five parameters related to venous outflow hemodynamics using transcranial and extracranial color-Doppler sonographic examination (TCCS-ECD). In controls, only 2.7% of the measurements were anomalous, whereas in MS patients 47% of measurements were abnormal. 1 In a study comparing duplex ultrasound with contrast venography, 40–70% of MS patients had evidence of flow disturbances and/or venous stenosis by TCCS-ECD. Of these patients, 86% and 91% had obstructive disease of the azygous or internal jugular veins, respectively. 2
Recently, several clinical imaging studies using MR or ultrasound have questioned the high degree of association between CCSVI and patients with MS, reported initially by Zamboni.6–8 For example, Tsivgoulis and colleagues performed a color-coded Doppler sonography case–control study in an attempt to validate the association of MS with CCSVI. 7 They observed no evidence of stenosis or non-detectable Doppler flow in cervical veins in patients (n = 42) and controls (n = 43), and found internal jugular vein reflux in a few subjects in both groups. Sundstrom and colleagues performed MR imaging in a case–control study and claimed to find ‘no evidence confirming the suggested vascular multiple sclerosis hypothesis’, although their imaging revealed internal jugular vein stenosis in several of the cases, and none of the controls. 8 Critics of these negative studies respond with counter arguments that question the criteria and techniques applied to diagnose CCSVI and how these may differ from the original approach by Zamboni and colleagues. What is becoming increasingly clear from this debate is the realization that imaging studies alone are limited and most likely insufficient to understand the physiology of abnormal extracranial venous drainage.
There is an emerging recognition that it is critical to assess the physiological relevance of any anatomic luminal narrowing in order to confidently diagnose an abnormality. MR or CT imaging alone appears subject to high false- positive and false-negative interpretations. Ultrasound examinations are better at evaluating intraluminal lesions and the valvular anatomy, but these are operator dependent, time intense, hard to blind, and difficult to reproduce. Catheter venography is invasive and evaluations of vein morphology may vary depending on the patient’s head position, phase of respiration, and degree of neck muscle contraction. Furthermore, in cases where a malformed valve may present resistance to venous flow, the catheter itself may transiently relieve (and therefore obscure) the hemodynamic impairment when passed through the valve. Thus, it is important to acknowledge that at this time there is not a single simple test that can adequately evaluate the anatomy and physiology of extracranial venous drainage to diagnose unequivocally CCSVI to the satisfaction of all interested parties.
Based upon our experience, and the observations of others, 9 the entity of CCSVI appears to encompass a spectrum of extracranial venous lesions that perturb venous drainage. Venous narrowing and flow disturbances may be situated at any location within the jugular or azygous systems. Certain locations appear to be more commonly involved. The area of the jugular valve adjacent to the brachiocephalic confluence is frequently involved in CCSVI. Our imaging studies suggested that the valve itself may be malformed or associated with a diaphragm or aberrant cul de sac. These abnormalities may represent examples of congenital truncal venous malformation. In addition to intrinsic venous pathologies that may be congenital or acquired, other jugular sites may be susceptible to extrinsic compression. Alternatively, venous narrowing may be secondary to inflammatory processes. Suffice to say, the patterns of cerebrovenous disease are not fully characterized and the underlying mechanisms remain to be elucidated.
In our experience, proximal jugular lesions were more common than those affecting the cephalic jugular segments. The jugular veins were much more frequently involved than the azygous vein in our series.
Symptoms related to CCSVI
In the subset of MS patients with CCSVI, a disturbance of cerebrovenous hemodynamics could be responsible for some symptoms and findings that are not well explained by an inflammatory response. Notably, dynamic susceptibility contrast perfusion MR imaging shows prolonged mean transit times of blood flow through the brain in patients with MS compared with those measured in control subjects.10,11 Impaired tissue perfusion would be expected to occur in regions of impaired venous outflow, associated with a reduced arteriovenous pressure gradient. Impaired cerebral perfusion might explain the generalized mild impairment in cognitive function described by some MS patients.
Vascular inflammation and MS
In addition to explaining some of the poorly understood symptoms of MS, an impairment of venous hemodynamics could also participate in the inflammatory pathophysiology of MS. It is known that alterations in shear stress and cyclic strain can induce the expression of adhesion molecules and chemokines that participate in vascular inflammation. 12 Accordingly, it is possible that alterations in venous hemodynamics could participate in MS by increasing the expression of endothelial adhesion molecules, chemokines, and cytokines that alter endothelial adhesiveness and permeability. These alterations in vascular homeostasis might facilitate the adherence of immune cells and their infiltration into the surrounding tissue. A predisposition to autoimmunity against an antigen in the brain parenchyma could thus be facilitated by obstruction to venous flow.
We speculate that chronic venous diseases (CVD) of the lower extremities, and CCSVI associated with MS, have many similarities in their molecular and physiological mechanisms. This speculation is supported by the fact that perivenular inflammation is a common histopathological finding in patients with CVD, as well as in those with MS. In the leg veins, disturbed venous flow increases endothelial adhesion molecules (e.g. ICAM-1), vascular permeability, edema, infiltration of inflammatory cells, insudation of plasma proteins, and causes hemosiderin deposition.13,14 The latter is due to erythrocyte extravasation. Iron overload promotes oxidative stress, 15 which further increases endothelial adhesiveness and inflammation. 16 Whereas normal levels of blood flow provide for endothelial shear stress that reduces the expression of adhesion molecules and chemokines,17,18 disturbed flow increases endothelial adhesiveness and interaction with immune cells. 12 The role of inflammation in CVD is consistent with the clinical observation that individuals with this condition may obtain partial relief of their symptoms with anti-inflammatory agents. However, in patients with CVD, complete relief of symptoms, and modification of the progression of disease, requires treatment of the underlying venous insufficiency. 19
Notably, histology confirms a close relationship between dilated cerebral veins and inflammatory lesions in MS. 20 Within the MS plAQue can be found perivenular deposits of iron which may be due to erythrocyte extravasation. 21 Imaging studies also implicate a vascular component in the pathophysiology of MS. Increased brain iron is observed by MRI, which could reflect elevated venous pressure and venular permeability. 22 Furthermore, MRI with gadolinium reveals that some MS lesions enhance and some do not. Experimental evidence indicates that the enhancing lesions are associated with active inflammation and increased vascular permeability. 23 The histological and MRI evidence for cerebrovenous permeability and hemorrhage is directly supported by observations of retinal venular permeability and hemorrhage in MS patients.24,25
There is another intriguing parallel between individuals with lower extremity venous insufficiency and those with CCSVI associated with MS. In patients with more severe CVD, the high venous pressure reduces the pressure gradient for microvascular flow, leading to ischemia. Ischemia, oxidative stress and inflammatory cytokines cause cell death and fibrosis in the dermis, which is manifested as atrophie blanche. These lesions are small, acellular regions of fibrosis that at least superficially resemble the acellular plaques of MS.
Finally, it is also possible that CCSVI represents an entirely different disease that mimics MS. In this case, a vascular anomaly has been misdiagnosed (and quite possibly inappropriately treated) by neurologists.
Adverse effects of stent placement for venous stenosis associated with MS
Adverse events were common with stent placement. The majority of patients had some local discomfort, probably related to inflammation of the vein wall after stent implantation. These symptoms usually resolved completely within a few days of the procedure. In a small subset of patients, shoulder weakness was reported about 1 week after the stent placement. These symptoms were usually related to the placement of a high jugular stent. It is possible that the local inflammation and/or pressure may affect the function of the accessory nerve (cranial nerve XI). Stent migration is a known complication of venous stent implantation. In one case, stent migration required surgical extraction.
Some of these complications would be avoided by performing balloon venoplasty in the absence of stenting. However, many of the lesions encountered did not respond to venoplasty alone. Furthermore, the long-term patency of venoplasty alone is likely limited by elastic recoil, myointimal hyperplasia and/or negative remodeling, as we and others have observed with venous disease affecting the lower extremities. Indeed, Zamboni and colleagues recently reported a restenosis rate of 50% in the internal jugular veins after venoplasty in CCSVI patients. 26 Notably, those without internal jugular vein restenosis were not observed to have relapses of their MS in the 18-month follow-up period.
Anticoagulation has been suggested as a potential therapeutic approach for MS. 27 However, one of our patients suffered a fatal intracerebral hemorrhage on warfarin anticoagulation. The risks and benefits of anticoagulation in MS, particularly in the setting of endovascular intervention, need further study.
Limitations
Many symptoms of MS fluctuate and are largely subjective. As this is an uncontrolled clinical series, the apparent benefit of the procedure could be due to clinical (or statistical) regression to the mean. Because all patients underwent extensive clinical testing and intervention, the improvement in symptoms could reflect a strong placebo effect induced by the technology and the clinical team. Furthermore, in the context of this clinical care, neurological evaluations and imaging information were not systematically collected.
Despite the flaws in this observational experience, the biological plausibility linking cerebral venous congestion to reduced cerebral perfusion, impaired thermoregulation, and inflammation requires serious consideration. This innovative clinical care has generated controversy, but it has also generated sufficient experience that a clinical trial can be designed. After careful review of this clinical series, the authors and the leadership of Stanford School of Medicine have agreed to conclude this observational series, and have determined that a randomized clinical trial should be the next step. 28
Conclusions
There is a strong rationale for endovascular treatment of stenoses in large veins draining the head and neck, based on long experience and success in treating superior vena cava syndrome. In this series, endovascular intervention to correct venous stenosis associated with MS was associated with improvement in symptoms possibly related to disturbed venous hemodynamics. However, given the serious adverse events in this small series, a randomized clinical trial is required to confirm these findings, and to determine if the procedure has any effect on the progression of MS, or untoward long-term adverse effects. Until then, our recommendation is that this procedure should not be performed outside of a clinical trial. The observations made in this uncontrolled clinical series will be invaluable in designing a proper randomized clinical trial to determine the efficacy and safety of endovascular intervention for CCSVI associated with MS.
Footnotes
This work was funded in part by the National Heart, Lung, and Blood Institute [K12 HL087746].
The authors have no relevant financial or personal relationships with people or organizations that could inappropriately bias this work. The authors report no further disclosures.
