Abstract
The pathogenic mechanisms of thromboangiitis obliterans (TAO) are not entirely known and the imbalance of matrix metalloproteinases (MMPs) plays a role in vascular diseases. We evaluated the MMP-2 and MMP-9 circulating levels and their endogenous tissue inhibitors of metalloproteinases (TIMP-1 and TIMP-2) in TAO patients with clinical manifestations. The study included 20 TAO patients (n = 10 female, n = 10 male) aged 38–59 years under clinical follow-up. The patients were classified into two groups: (1) TAO former smokers (n = 11) and (2) TAO active smokers (n = 9); the control group included normal volunteer non-smokers (n = 10) and active smokers without peripheral artery disease (n = 10). Patient plasma samples were used to analyze MMP-2 and MMP-9 levels using zymography, and TIMP-1 and TIMP-2 concentrations were determined by enzyme-linked immunosorbent assays. The analysis of MMP-2/TIMP-2 and MMP-9/TIMP-1 ratios (which were used as indices of net MMP-2 and MMP-9 activity, respectively) showed significantly higher MMP-9/TIMP-1 ratios in TAO patients (p < 0.05). We found no significant differences in MMP-2/TIMP-2 ratios (p > 0.05). We found higher MMP-9 levels and decreased levels of TIMP-1 in the TAO groups (active smokers and former smokers), especially in active smokers compared with the other groups (all p < 0.05). MMP-2 and TIMP-2 were not significantly different in patients with TAO as compared to the control group (p > 0.05). In conclusion, our results showed increased MMP-9 and reduced TIMP-1 activity in TAO patients, especially in active smokers compared with non-TAO patients. These data suggest that smoke compounds could activate MMP-9 production or inhibit TIMP-1 activity.
Keywords
Introduction
Thromboangiitis obliterans (TAO) or Buerger’s disease is a distinct disease that often leads to vascular insufficiency and is characterized by chronic inflammation and acute thrombosis of small- and medium-caliber arteries in the arms and legs, particularly tibial and radial arteries, with involvement of the veins and nerves of the extremities.1–4 It has been reported that approximately 40% of patients with TAO develop superficial thrombophlebitis and approximately 70% experience sensory abnormalities. It is these additional features that help to differentiate TAO from atherosclerosis. 4
A reaction to the constituents of tobacco cigarettes is recognized as a factor in the initiation, progression, and prognosis of TAO. Additionally, genetic modifications or autoimmune disorders are essential etiologic factors.5–7 Peripheral endothelium-dependent vasodilatation is impaired in the non-diseased limb of TAO patients, and this type of vascular dysfunction may contribute to segmental proliferative lesions or thrombus formation in peripheral vessels. 8
The matrix metalloproteinases (MMPs) and their tissue inhibitors of metalloproteinases (TIMP) have an essential function in the restructuring of extracellular matrix and hence tissue composition and are thought to play a pivotal role in angiogenesis, a process that is vital for new blood vessel formation. Additionally, MMPs are a family of structurally related zinc-dependent enzymes involved in the degradation of many components of the extracellular matrix during both physiological and pathological processes. 9 Increased expression and activity of gelatinases (MMP-2 and MMP-9) have been reported in a variety of pathological conditions.10–12 Several studies have shown that circulating levels of MMPs have been associated with the risk of atherosclerotic events and can be used as a biochemical marker for cardiovascular risk, 13 even for individuals without clinical disease.14,15 Thus, increased levels of MMP-9 were associated with rapid progression of coronary artery disease and fatal cardiovascular events. 16 Still, the increased movement of MMP-9 was associated with a higher incidence of acute cardiovascular events and/or hypertension in people without cardiovascular disease. 17 The activity of MMPs can be modulated by endogenous TIMPs (small proteins, 23 kDa) and TIMP-1 expression is induced by a variety of different stimuli, including pro-inflammatory cytokines 18 and angiotensin II, 19 while TIMP-2 expression in the heart is constitutive. 20
Considering that changes in the levels of MMP have been described in cardiovascular disorders,13,20 and the mechanisms involved in the development of TAO are not fully understood, the present study analyzed the circulating levels of MMP-2, MMP-9, TIMP-1, TIMP-2, and the MMP-9/TIMP-1 and MMP-2/TIMP-2 ratios in TAO and normal patients.
Our rationale for conducting this study was based on observations that MMPs are widely expressed in the vascular tissues, and MMP-2 and MMP-9 have been implicated in many cardiovascular diseases.1,2 While a growing number of studies are now showing imbalanced MMP proteolytic activities in cardiovascular diseases, no previous study has examined MMP levels in patients with TAO. Thromboangiitis obliterans is a segmental inflammatory disorder of the small and medium arteries and veins of the extremities that is most often associated with tobacco use. This evidence led us to explore the connection of MMPs and their inhibitors with TAO. Since the pathophysiology of TAO is complex and incompletely understood, the current study was carried out to demonstrate imbalances in MMP and TIMP levels, which are important inflammatory biomarkers. In addition, metalloproteinase inhibition has been considered as therapeutic and preventive targets for cardiovascular disease.21,22
Methods
Subjects
The study included 20 TAO patients (n = 10 female, n = 10 male) aged 38–59 years under clinical follow-up at the University Hospital of the Ribeirão Preto Faculty of Medicine, University of São Paulo, Brazil. The TAO diagnosis was based on the Shionoya or the Olin criteria that were routinely used in our vascular division. The five classic Shionoya criteria include history of tobacco abuse; onset of symptoms before the age of 50 years; infrapopliteal arterial occlusive disease; either upper limb involvement or thrombophlebitis migrans; and the absence of atherosclerotic risk factors other than smoking. 23 The Olin criteria consider the onset of disease before the age of 45 years; current tobacco use; distal extremity ischemia (infrapopliteal and/or infrabrachial), such as claudication, rest pain, ischemic ulcers, and gangrene documented with non-invasive testing; laboratory tests for the exclusion of autoimmune or connective tissue diseases and diabetes mellitus; the exclusion of a proximal source of emboli by means of echocardiography and arteriography; and demonstration of consistent arteriographic findings in the involved and clinically non-involved limbs. 4 All selected patients reported the use of cigarettes for more than 20 years, and TAO was diagnosed at a mean age of 40 years. A total of 90% of the patients exhibited evidence of critical limb ischemia and 60% presented leg amputations (below or above-knee amputation) in the contralateral leg. Thus, the patients were classified into two groups: (1) TAO former smokers (at least 10 years of abstinence) with clinical remission (n = 11) and (2) TAO active smokers with clinical exacerbation (n = 9); the control groups included normal volunteer non-smokers (n = 10) and active smokers (n = 10).
The standard therapy was applied to all TAO patients: antiplatelet therapy with aspirin (100 mg/day), pain control with anti-inflammatory and opioid drugs, and instructions for the immediate cessation of smoking. Patient plasma samples were used to analyze MMP-2 and MMP-9 levels using zymography, and TIMP-1 and TIMP-2 concentrations were determined by enzyme-linked immunosorbent assays.
Informed consent was obtained from all of the patients, and the study protocol was approved by the Ethics Committee of the University Hospital, Ribeirão Preto Faculty of Medicine, University of São Paulo, Brazil (no. 12810/2008).
Blood collection
A trained biomedical technician collected a 10 ml venous blood sample from each participant. Blood samples were collected in trace metal-free tubes (BD Vacutainer®; BD, Franklin Lakes, NJ, USA) that contained heparin as an anticoagulant. Then, 2 ml of blood were pipetted into an Eppendorf tube previously cleaned in a Class 100 Cleanroom and immediately frozen at –70°C before analysis.
Sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gelatin zymography of MMP-2 and MMP-9
Gelatin zymography of MMP-2 and MMP-9 from plasma samples was performed as previously reported.9,13 Briefly, plasma samples were diluted in sample buffer (2% SDS, 125 mM Tris-HCl [pH 6.8], 10% glycerol, and 0.001% bromophenol blue) and subjected to electrophoresis on 7% SDS-PAGE gels co-polymerized with gelatin (1%) as the substrate. After electrophoresis was complete, the gel was incubated for 1 h at room temperature in a 2% Triton X-100 solution and incubated at 37°C for 18 h in Tris-HCl buffer (pH 7.4) that contained 10 mmol/l CaCl2. The gels were stained with 0.05% Coomassie Brilliant Blue G-250 and then destained with 30% methanol and 10% acetic acid. Gelatinolytic activity was detected with densitometry as unstained bands against a background of Coomassie Blue-stained gelatin using a Kodak Electrophoresis Documentation and Analysis System (EDAS) 290 (Kodak, Rochester, NY, USA). The pro-forms of MMP-2 and MMP-9 were identified as bands at 72 and 92 kDa, respectively, using the relationship between log Mr and the relative mobility of Sigma SDS-PAGE LMW marker proteins.
Determination of TIMP-1 and TIMP-2 concentrations in plasma
Plasma TIMP-1 and TIMP-2 concentrations were measured using the sandwich enzyme-linked immunosorbent assay (ELISA) with reagents from R&D Systems (Minneapolis, MN, USA) following the manufacturer’s instructions. The reaction was evaluated using a μQuant™ Microplate Spectrophotometer (BioTek Instruments, Inc., Winooski, VT, USA).
Statistical analysis
The biochemical parameters were compared using the Mann–Whitney test, and values of p < 0.05 were considered statistically significant.
Results
The gelatin zymography of plasma samples revealed that all forms of MMPs are normally found in human plasma, including the 180 kDa, 130 kDa and 92 kDa forms of MMP-9, and inserted into the 72-kDa form of the MMP-2 (Figure 1). We found high levels of MMP-9 in patients with TAO, especially in the active smoker group, compared with the other groups (all p < 0.05). MMP-2 was not significantly different in patients with TAO in comparison with the control group (Figures 1 and 2; all p > 0.05). In addition, we found decreased levels of TIMP-1 in TAO patients compared with controls (p < 0.05), but no difference in the levels of TIMP-2 between the groups (Figure 3).
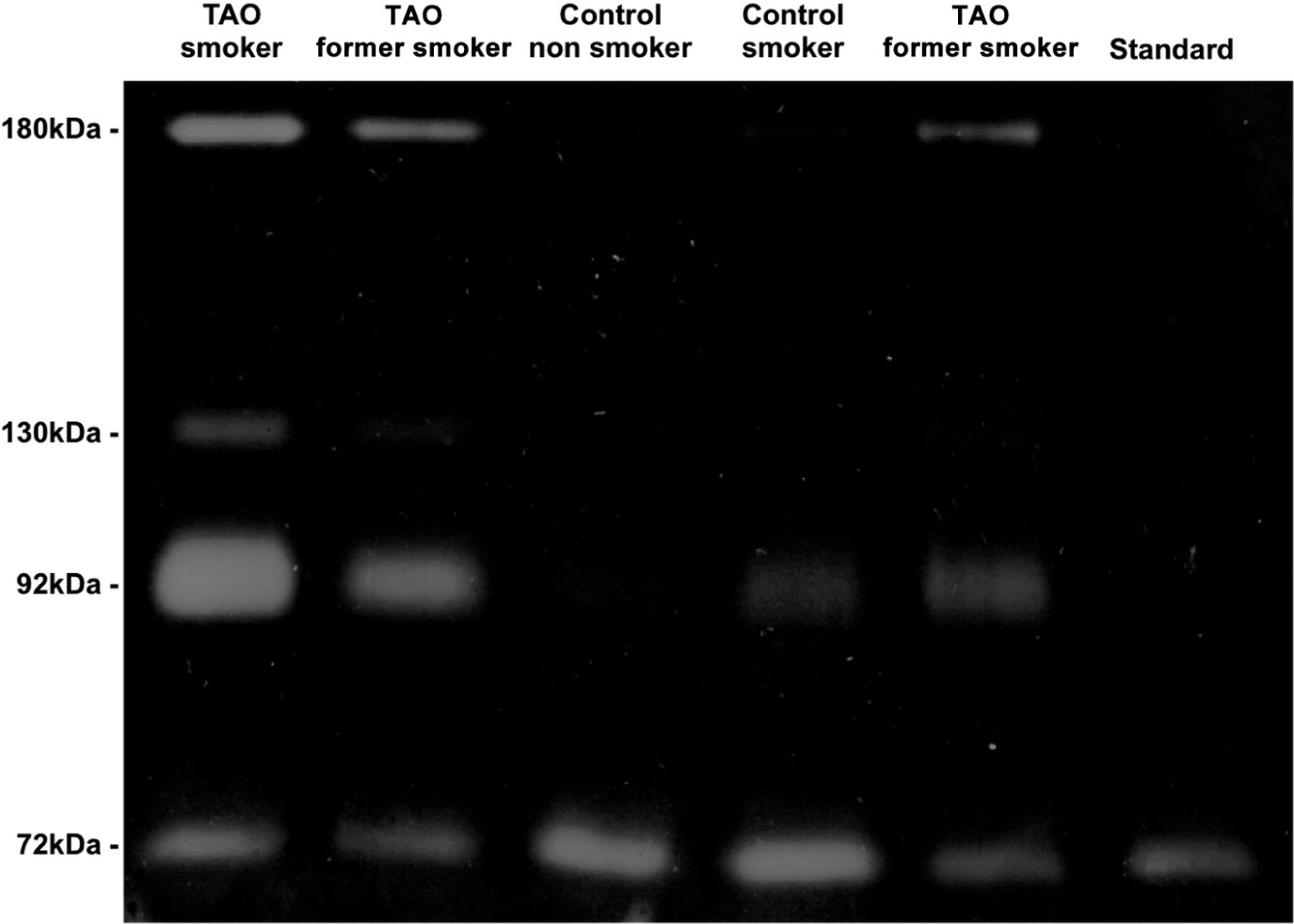
Representative zymogram of plasma samples (control, ischemia, TAO former smokers and TAO active smokers). The ‘marker’ lane shows the bands corresponding to gelatinases (180 kDa, 130 kDa, 92 kDa and 72 kDa) from whole blood. ‘Standard’ shows the 72 kDa band (pro-MMP-2) from fetal bovine serum, which was used as standard to normalize the data from all the gels, thus allowing comparisons between gels.

Plasma MMP-9 and MMP-2 concentrations in control non-smokers (n = 10), control active smokers (n = 10), TAO former smokers (n = 11) and TAO active smokers (n = 9). (A) MMP-9, 180 kDa; (B) MMP-9, 130 kDa; (C) MMP-9, 92 kDa; and (D) MMP-2, 72 kDa. Results expressed as median (arbitrary units). (*Significant differences [p < 0.05].)
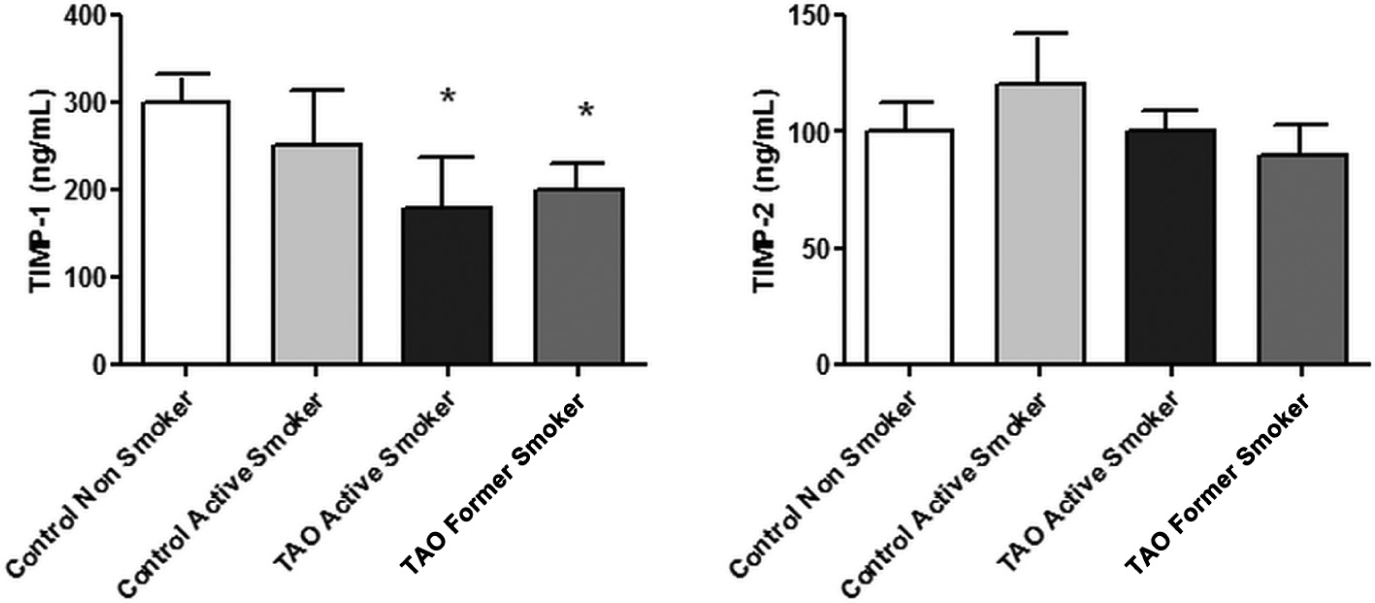
TIMP-1 and TIMP-2 activity in control non-smokers (n = 10), control active smokers (n = 10), TAO former smokers (n = 11) and TAO active smokers (n = 9). Results expressed as median (ng/ml). (*Significant differences [p < 0.05].)
The analysis of MMP-2/TIMP-2 and MMP-9/TIMP-1 ratios, which were used as indices of net MMP-2 and MMP-9 activity, respectively, showed a significantly higher MMP-9/TIMP-1 ratio in TAO patients (Figure 4; p < 0.05). We found no significant differences in the MMP-2/TIMP-2 ratio (p > 0.05).
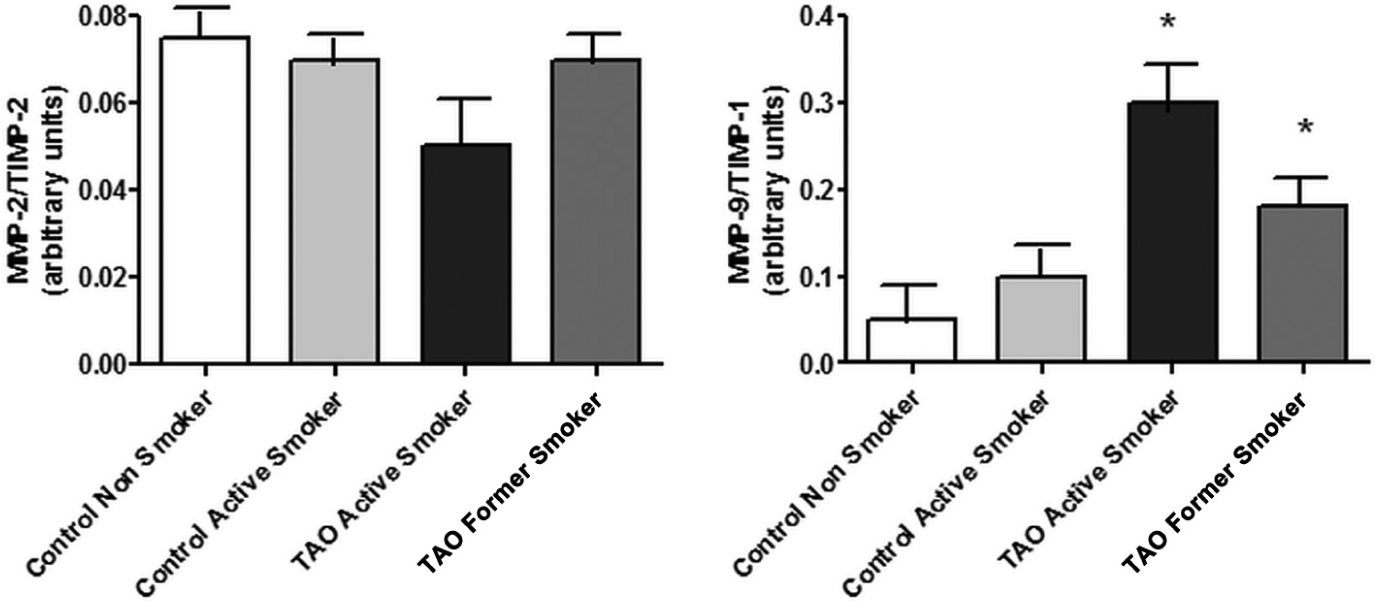
The MMP-9/TIMP-1 and MMP-2/TIMP-2 ratio in control non-smokers (n = 10), control active smokers (n = 10), TAO former smokers (n = 11) and TAO active smokers (n = 9). Results expressed as median (arbitrary units). (*Significant differences [p < 0.05].)
Discussion
Matrix metalloproteinase plays an important role in cardiovascular disease,8,9 including atherosclerosis,24,25 dilated cardiomyopathy, 26 and myocardial infarction. 27 The extracellular matrix plays a key role in the proper function of the various organs of the human body, including the heart and blood vessels. Changes in the extracellular matrix have been implicated in the pathogenesis of several cardiovascular conditions, including atherosclerosis, aneurysms, post-angioplasty restenosis, and heart failure.28,29
Different studies have shown that smokers have increased plasma levels of MMP-9 and TIMP-1 or only MMP-9.30–34 In this study, high levels of MMP-9 were found in TAO patients, mainly in active smokers. The differences between TAO active smokers and TAO former smokers (Figures 2 and 4b) confirm the close relationship between the TAO pathophysiology and tobacco use.
Our hypothesis is, that during the inflammatory phase of healing in TAO patients, rapid activation of MMPs may degrade the vascular extracellular matrix and the matrix fragments could act as a potent pro-inflammatory mediator enhanced by the use of cigarettes; new studies are necessary to confirm this idea.
TIMPs are often up-regulated when MMP activity increases. 20 In this study, we also measured the concentrations of TIMP-1 and TIMP-2 to evaluate the MMP-9/TIMP-1 and MMP-2/TIMP-2 ratios, which may provide information about net MMP activity. Curiously, increased pro-MMP-9 levels and pro-MMP-9/TIMP-1 ratios were found in TAO patients (both smokers and non-smokers). However, we have no precise explanation for these conflicting results that may be attributable to differences in the methods (zymography vs ELISA) used to measure circulating MMP-9 levels. Also, this explanation may not be probable because both methods have been well standardized.35–37 Another explanation may be based on the imbalance of MMPs and TIMPs; specifically, the MMP-9/TIMP-1 ratio in TAO patients.
In this study, we report for the first time increased levels of MMP-9 and the MMP-9/TIMP-1 ratio in the plasma of patients with TAO, suggesting that the pathogenesis may also be related to an imbalance of metalloproteinases. The increases in MMP-9 activity and decreased TIMP-1 reported here may lead to increased susceptibility to cardiovascular disorders, as suggested by growing evidence from previous studies. Metalloproteinase inhibition has been considered as therapeutic and preventive targets for cardiovascular disease. So, even in the absence of clinical or experimental data, it is reasonable to speculate that MMP-9 or TIMP-1 could be therapeutic targets.21,22 Another speculative idea, but an attractive one, is to investigate whether the ratio of MMP-9 to TIMP-1 could be used as part of the evaluation of patients with suspected TAO.
Study limitations
Often, the clinical challenge in the diagnosis of TAO comes from distinguishing this disorder from atherosclerotic arterial occlusive disease. Unfortunately, we do not have access to MMP and TIMP levels from smoking and non-smoking patients with atherosclerotic arterial occlusive disease. It would be of great interest to demonstrate whether there are differences in MMP and TIMP levels between patients with TAO or atherosclerotic arterial occlusive disease. The study has another limitation with respect to the statistical accuracy: we did not perform power analysis. The groups were defined previously and blood samples were gathered as the patients were scheduled for their medical appointments. Considering the adopted criteria for including patients, using the previously defined investigative groups, obtaining the blood samples took a long time and we decided to present our results based on the observed levels of statistical significance (p < 0.05) applying the Mann–Whitney test.
Conclusion
The results showed increased MMP-9 and reduced TIMP-1 activity in TAO patients, especially in active smokers, compared with non-TAO patients. These data suggest that compounds in the smoke could activate MMP-9 production or inhibit TIMP-1 activity in these patients.
Footnotes
This work was supported by FAEPA (HCFMRP/USP) and FAPESP.
None declared.
