Abstract
The isolation of the aneurysm sac from systemic pressure and its consequent shrinkage are considered criteria of success after endovascular repair (EVAR). However, the process of shrinkage does not solely depend on the intrasac pressure, the predictive role of which remains ambiguous. This brief review summarizes the additional pathophysiological mechanisms that regulate the biomechanical properties of the aneurysm wall and may interfere with the process of aneurysm sac shrinkage.
Keywords
Introduction
The criterion of successful treatment of aortic abdominal aneurysm (AAA) after endovascular repair (EVAR) is the isolation of the AAA sac (decrease of the maximum transverse diameter) from the systemic pressure and its shrinkage with absence of endoleaks. An endoleak is defined as persisting blood flow within the aneurysm sac but outside the lumen of a stent-graft. There are four types of endoleak. Type I refers to leakage from the attachment sites, whereas type II is attributed to retrograde flow from side branches (Figure 1). Furthermore, an endoleak caused by fabric tear or disruption, component disconnection or graft disintegration is classified as a type III endoleak, whereas type IV relates to blood flow due to graft porosity. 1

Computed tomography follow-up of a stented aneurysm. The non-contrast-enhanced image helps to detect any calcifications that would obscure the contrast-detected endoleaks (A). The arterial phase contrast-enhanced image allowed the detection of a type II endoleak (lower white arrow) from a patent inferior mesenteric artery (upper white arrow) and a second one (thick black arrow), fed by lumbar arteries (B).
Though the decision of treatment for type I and III endoleaks seems unanimous, the evaluation of the potential risk of rupture regarding type II endoleaks and endotension still poses uncertainties and controversies.2,3 An expanding sac with elevated sac pressure needs treatment, whereas a stable or shrinking sac with a low residual sac pulse pressure does not. However, there is no consensus yet regarding cases that fall between these two extremes. Pressure measurement threshold values (to correlate with sac shrinkage) are based mostly on intraoperative data, and appropriate values to guide follow-up parameters have yet to be derived.4,5
The sac shrinkage process can be a complex procedure, depending on several factors apart from endoleaks, such as the graft porosity and compliance, aneurysm characteristics (aortic neck diameter and angle as well as common iliac artery diameter), properties of the intraluminal thrombus (ILT) and alterations in the mechanical properties of the AAA wall.6,7 This brief review discusses the controversies regarding the role of endoleaks in the process of sac shrinkage and focuses further on the biomechanical alterations documented after EVAR that may have an impact on the process of sac shrinkage.
Evaluating the role of endoleaks
In a recent cohort study, Schanzer et al. reported a 32% overall incidence for the presence of any type of endoleak, while the majority of endoleaks (76%) were evident in post-EVAR imaging during the first year. 7 One-fifth of these endoleaks related to aortic sac enlargement. They also identified predictors of AAA sac enlargement, such as the age (> 80 years) of the patient, the geometric characteristics of the AAA neck and iliac arteries, and the presence of endoleaks.
From the hemodynamic point of view, types I and III endoleaks represent exposure of the sac to systemic pressurization 8 (‘high-pressure’ endoleaks) with sac enlargement and ongoing risk of rupture, whereas the effect of type II on sac shrinkage, mainly those persisting more than 6 months after EVAR, is rather ambiguous. The behavior of these endoleaks can be delineated by hemodynamic information that computed tomography angiography cannot accurately provide. Certain characteristics such as high intrasac velocity and the high pressure of type II endoleaks have been reported to associate with resistance in sac shrinkage, though neither can exclusively predict their impact on sac behavior.2,9,10
Beeman recently reported that the existence of multiple type II endoleaks, as well as the accompanying bidirectional Doppler waveform, predicts more accurately the hemodynamic inhibition to aneurysm sac shrinkage, since this finding reflects the lack of an outflow vessel capable of reducing the flow resistance and, thus, the intrasac diastolic pressure. 11 Interestingly, contrast-enhanced ultrasound (Figure 2) has been identified as a high-sensitive modality for the detection and hemodynamic characterization of type II endoleaks, differentiating sufficiently hypodynamic (prolonged washout time) lumbar endoleaks associating with post-EVAR sac increase.12,13
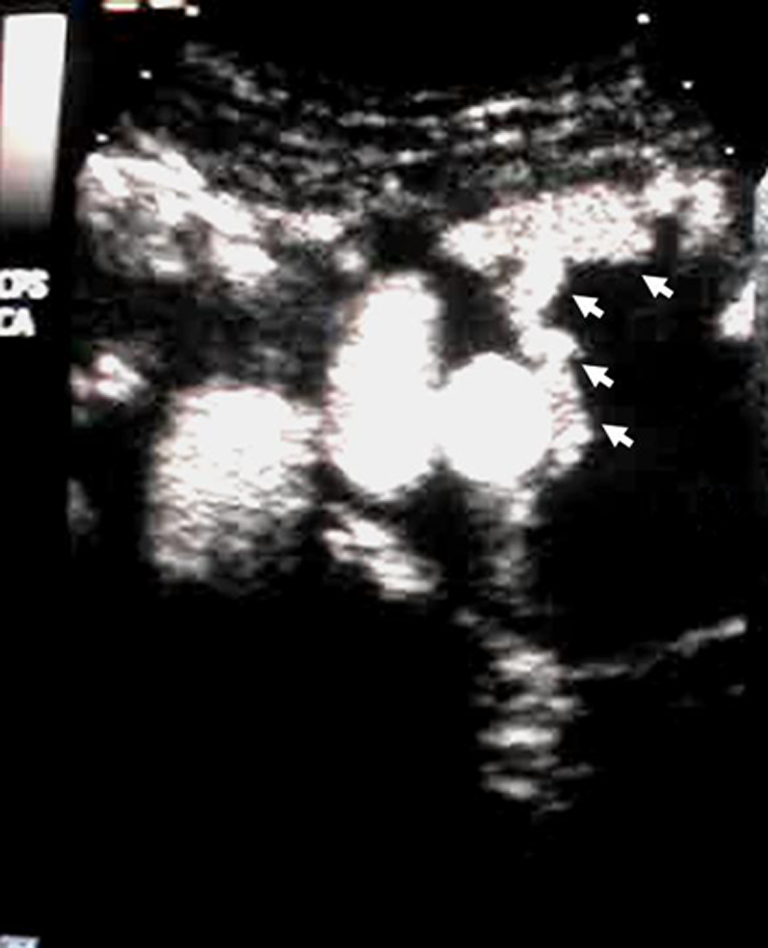
Contrast-enhanced (microbubbles) ultrasound detection of a type II endoleak originating from a patent inferior mesenteric artery (white arrows).
Fairman et al. identified the presence of type II endoleaks as an independent predictive variable for the size change in AAA sac only after the first month post-EVAR, while Greenberg et al. reported a rather moderate effect of type II endoleaks on the rate of aneurysm sac shrinkage.14,15 Of note, aneurysm sac shrinkage can develop despite the presence of a persistent type II endoleak, whereas the absence of an endoleak does not necessarily guarantee the shrinkage of the sac.14,16 Moreover, by comparing the intrasac pressures after EVAR to either the pressure within the sac after open AAA repair or within patent lumbar and inferior mesenteric arteries, Vallabhaneni et al. concluded that retrograde perfusion via patent side branches cannot be the only reason for maintenance of pressurization within the sac after EVAR. 17 Ongoing pressurization in the sac could be attributed to direct transmission of the systemic pressure through the stent-graft material, with the qualitative and quantitative properties of the ILT and the altered sac wall mechanics attributing to variable levels of sac regression and depressurization.
Remodeling of the aneurysm wall
Generally, AAA size change corresponds in consistency with intrasac pressure after EVAR, but not always directly. According to Dias et al., a delay in the sac shrinkage can be noticed in response to the sac depressurization. 18 This implies that AAA sacs of the same maximum diameter that have been excluded successfully with the same type of stent graft, could actually be subjected to a different rate of sac shrinkage.
By computational simulation, Kwon et al. recently showed that the post-EVAR remodeling is strongly influenced not only by the sac pressure but also by the pressure change over a period of time. 19 According to their model, when the intrasac pressure drops drastically immediately after EVAR, the sac continues to shrink, even if the pressure increases thereafter. On the contrary, a pressure value that would not decline below a certain level could not guarantee the effective shrinkage of the sac, despite the fact that there may have been a certain decline in the pressure value after EVAR. 19 The authors concluded that stress-mediated wall cell activity and stress-mediated vascular remodeling of the AAA wall under the change in pressure are presumed to play a key role in the AAA post-EVAR behavior. Molony et al. reconstructed computationally preoperative, postoperative and follow-up models of AAA after endovascular exclusion. 20 They calculated the wall stress values and distribution in each model and showed a redistribution of stress patterns along with decrease in their values. The remodeling of the AAA sac can change the sac volume and configuration, which is associated with reduction and redistribution in wall stress patterns. 20
Yeung et al. studied the sac regression after EVAR in 100 patients, 63 of which presented no endoleak. They reported that the percentage of patients with minimal or no thrombus displaying sac shrinkage was significantly greater than that of patients with increased thrombus volume (75% vs 58%). 21 These findings imply that the presence of ILT could impair the remodeling capacity of the aortic wall. ILT can influence sac remodeling by interfering with the biochemical and biomechanical properties of the AAA wall. Generally, the AAA wall adjacent to thrombus sustains elastic degradation, hypoxia and inflammatory infiltration in a degree dependent on the thickness of thrombus.22–24 These changes result generally in alterations in biomechanical properties such as the compliance or the tensile strength of the AAA wall. 23
Changes in wall mechanics after EVAR
Dias et al. recently reported that in 18 AAAs that had unchanged maximum diameters for more than 1 year after EVAR, invasive intrasac pressure monitoring revealed no postoperative fluid accumulation and no endoleak. 25 Among them, three AAAs expanded, five shrank and 10 remained unchanged during follow-up. Though the samples were small, it was interesting to note that neither intrasac pulse and mean pressure nor the mean pressure index was able to differentiate the last two categories, further implying that the fate of an AAA after EVAR does not solely depend on intrasac pressure levels.
A systematic review by Hinnen et al. concluded that successful stent-graft deployment does not result in immediate reduction of aneurysm sac pressure, even in the absence of any endoleak. 26 Gawenda et al. performed an in vitro experiment with latex aneurysm models of variable compliance. 27 After endovascularly excluding the aneurysm sac and measuring the circumferential compliance, as well as the sac’s mean and pulse pressure relative to the systemic mean pressure, they concluded that the intrasac pressure was influenced inversely by the compliance of the aneurysm wall; a decrease in compliance resulted in an increase of the pressure parameters, showing that stiffer AAAs are associated with higher intrasac pressures, 27 which in turn can influence the sac shrinkage process. The implantation of a stent-graft has been associated with a decrease in the AAA compliance. 20 Van Herwaarden et al. have also reported an increase in AAA wall stiffness after EVAR. 28 This was also confirmed by Long et al., who evaluated the wall movement of the AAA sac after endovascular repair by using tissue Doppler imaging, 29 estimating indices of stiffness, elasticity (pressure strain elastic modulus, Ep) and parameters of sac wall compliance (i.e. maximum systolic / diastolic dilation along a segment in the antero-posterior direction). The compliance parameters decreased and stiffness parameters increased immediately after EVAR but remained stable during follow-up, whereas the sac diameter remained unaltered immediately postoperatively but decreased gradually later, during follow-up. However, no pattern of association between the percentage of compliance loss and the amount of sac shrinkage over time was recorded, postulating that AAA shrinkage after successful EVAR depends on complex interactions between the intrasac pressure, the compliance of the AAA sac and the remodeling of the ILT. The varying mechanical properties of the thrombus account for the large variability in its compressibility, 30 which might also influence the rate of the sac regression. Variations in shear modulus, local thickness and volume of the thrombus modifies the hemodynamic load on the AAA wall. 31
Regarding the configuration of AAA, the preoperative maximum diameter has been also identified as an independent predictor of successful sac regression.21,32,33 The AAA sac configuration seems to play a crucial role in the pattern of the strain distribution and the intrasac pressure, which are decreased in a rather uneven way after the AAA sac exclusion, related neither to the level of systemic pressure nor to the content of the AAA sac. 34 Brekken et al. 35 reported that the cyclic strain (i.e. relative cyclic elongation and contraction of the wall tissue along the AAA circumference) presented an inhomogeneous distribution along the sac circumference, both before and after the endovascular treatment in the absence of endoleak, implying that different segments of the sac possess different elasticity and biomechanical properties, possibly as a result of an uneven repair / remodeling process during the AAA enlargement with variable degrees of stiffness and thickness, leading to different levels of strength (and stiffness). Though the EVAR attenuated the strain in the sac significantly, a certain degree of it was still present, meaning that a small level of the pulse pressure was still transmitted through the stent-graft material and the thrombus. Moreover, van Keulen et al. showed that these uneven postoperative alterations of the sac wall can be reflected by sac volume changes which are not directly related to diameter changes, and vice versa. 36 While the postoperative increase in diameter has been associated with high intra-aneurysm pressure, 18 the clinical significance of the expanded sac volume has not been clearly defined yet. However, such a finding can be related to the wall remodeling, consequently redistributing the mechanical stresses exerted on it. The parameters affecting the AAA sac shrinkage and their interactions are summarized in Figure 3.
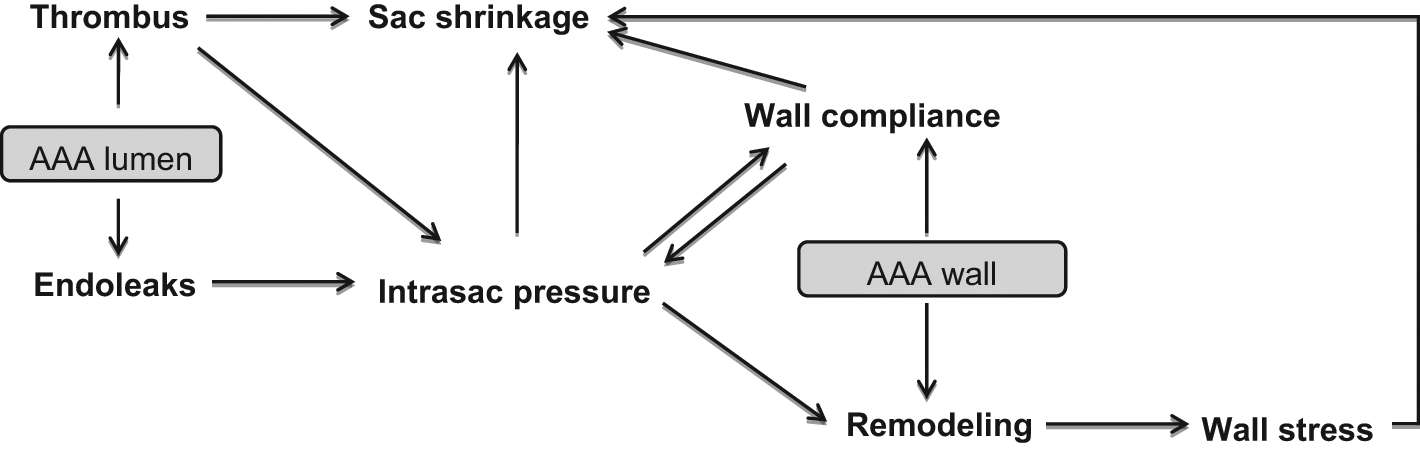
Overview of the parameters influencing the shrinkage of the aneurysmal sac.
Association of biomarkers with hemodynamic changes
The metalloproteinase (MMP) levels mirror a state of increased inflammatory and proteolytic activity and have been implicated in the development of aneurysm formation. 37 A number of studies have reported the use of MMPs after EVAR as enzymatic markers for endoleak.38–40 Sangiorgi et al. reported a significant decrease in plasma MMP-3 and MMP-9 levels for 6 months after successful endovascular repair. On the contrary, an increase of MMP levels in the presence of endoleaks in seven patients was observed, which decreased significantly after successful treatment of these endoleaks. 38 Though there was no direct measurement of intrasac pressures in the above-mentioned cases, it is important to note that two of these patients had an endoleak type Ia (proximal attachment dissociation) and three had an endoleak type Ib (distal attachment dissociation), probably exposing the aneurysm sac to a high level of pressure, thus leading to increasing levels of MMPs. On the contrary, in the series of Lorelli et al., almost all (seven out of eight) patients who presented with endoleak had one of type II, which might have exposed the sac wall to pressure levels generally lower than those of endoleak type I. This could presumably be the reason why the circulating MMP-9 levels remained similar to the preoperative high levels, but with no increasing tendency. 39 Once again, in patients with successful exclusion (no endoleaks), the MMP-9 levels decreased significantly. 40
The association between the MMPs and the endoleak-related pressurization of the sac could possibly be interpreted from the recent findings by Rahman et al., who studied the levels of MMPs in areas of low and high wall stress values in the AAA wall. 41 Finite element analysis was used to estimate the values and distribution areas of low and peak wall stress (PWS) on the wall of AAAs that were about to undergo open surgical repair. These areas were accordingly mapped out and excised intraoperatively, in order to provide tissue samples for MMP analysis. The authors were the first to show that MMP levels were higher at areas of PWS compared to areas of low stress, despite that fact that no statistical significance was reached. The latter could be attributed to a type-2 statistical error, since only 40 patients were studied. The PWS levels and distribution depend on the AAA geometry, shape and sac wall thickness, all of which can be modified in the remodeling process after EVAR.18,20 Furthermore, the presence of an endoleak can influence the applied pressure exerted on the inner surface of the sac, thus modifying the wall stress levels and, consequently, the activity of MMPs. Additionally, areas of high wall stress in the aneurysm wall have been related to inflammation and increased metabolic activity, assessed with 18 F-fluoro-2-deoxyglucose (18-FDG) using positron emission tomography-computed tomography (PET-CT). 42 Therefore, the role of 18-FDG needs to be investigated further in the post-EVAR course, in order to delineate its potential role as a predictor of endoleaks.42,43
Conclusion
The implantation of an endovascular graft in AAAs causes alteration in specific biomechanical characteristics of the AAA wall. These changes can influence the compliance and the remodeling capacitance of the AAA sac, potentially playing a crucial role in efficient AAA sac shrinkage. Measurement of intrasac pressure can be a helpful tool and precise when combined with assessment of the in vivo biomechanical properties of the AAA sac after EVAR. Additional clinical studies are needed to delineate the prognostic value of the parameters discussed with regard to progress of AAA sac shrinkage.
Footnotes
Acknowledgements
The authors would like to gratefully acknowledge the help and comments of Sokrates Tsangaris, from the Laboratory of Biofluid-Mechanics and Biomedical Engineering, National Technical University of Athens, Greece.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
None declared.
