Abstract
The pathophysiology and time course of an individual converting from asymptomatic peripheral artery disease (PAD) to symptomatic claudication is unclear. The objectives of this study were: (1) to characterize the extent of atherosclerotic disease in individuals with an abnormal ankle–brachial index (ABI), but without claudication; and over 1 year of follow-up to (2) evaluate the progression of PAD using ultrasound imaging, (3) determine changes in the ABI and leg pain symptoms, and (4) correlate PAD progression with changes in the ABI and leg symptoms. We hypothesized that PAD progression would be associated with the development of claudication and changes in the ABI, 6-minute walk distance (6-MWD), and walking quality of life. Individuals with a reduced ABI but without typical intermittent claudication noted on community screening were invited to undergo baseline and 1-year follow-up assessment, including duplex ultrasound. The initial and repeat evaluations included measurement of the ABI, lower extremity duplex arterial mapping, and assessment of leg pain and functional status. Of the 50 people studied, 44 (88%) had significant atherosclerotic lesions in the lower extremity arteries, affecting 80 legs. A total of 33 of 50 individuals (66%) returned for the 1-year follow-up visit. On ultrasound examination, two of 18 normal legs developed PAD, and in 48 legs with PAD at baseline, 17 legs (35%) developed new or progressive lesions. Thirteen legs developed new claudication. Overall, there was no significant worsening in the ABI, 6-MWD, or the Walking Impairment Questionnaire (WIQ). However, legs with new lesions or lesion progression were significantly more likely to develop claudication, and the 13 legs (seven subjects) developing claudication showed a significant decline in the 6-MWD. In conclusion, these data indicate that a significant number of people with asymptomatic PAD show progression over 1 year, that such individuals are more likely to develop claudication, and that those developing claudication have a significant decrease in their 6-MWD.
Introduction
The PARTNERS study showed that the prevalence of peripheral artery disease (PAD) in an at-risk population approached 30%. 1 In that study, using a hand-held Doppler device in the office setting, less than 10% of patients with PAD had typical claudication symptoms based on the San Diego Claudication Questionnaire (SDCQ), indicating that evaluating people with the ankle–brachial index (ABI) provides additional diagnostic benefit beyond the history of claudication symptoms. 2 The natural history of patients with PAD is poorly understood. Epidemiological studies indicate that approximately 75% who have claudication remain stable, without progressive leg deterioration, throughout their lifetime. 3 However, one study indicated that functional decline was more common than originally thought, even though such individuals may not be cognizant of lifestyle impairment. 4
The pathophysiology leading to development of claudication and decline in functional status is thought to be due to progression of the underlying lower extremity atherosclerosis. 1 Cardiovascular risk factors such as smoking, diabetes mellitus, hypertension and dyslipidemia, along with genetic factors, predispose to development of atherosclerosis in the leg arteries, with a sharp increase in prevalence with age. 5 Few patients go from being asymptomatic to symptoms of sudden acute arterial occlusion; rather, most have an insidious development of claudication. The natural history of atherosclerosis in the extremities involves plaque development in the intimal layer of the artery, typically in susceptible regions where turbulent blood flow occurs, such as the superficial femoral artery at Hunter’s canal. 6 There are few published prospective studies evaluating the progression of disease using an imaging method such as ultrasound.
Patients with a reduced ABI have occlusive disease present in their lower extremity, but may not have typical claudication symptoms and are therefore often considered asymptomatic. Lower extremity arterial duplex ultrasound mapping combined with ABI can assess the location and hemodynamic severity of PAD. Although still unclear, there is some evidence that atherosclerotic disease progression involves plaque hemorrhage or ulceration leading to vessel stenosis or occlusion, similar to that which occurs in the coronary arteries. 7 Duplex ultrasound may allow for tracking of PAD progression.
The objectives of this study were: (1) to characterize the extent of PAD in individuals with an abnormal ABI > 0. 60 and < 0.90, but without claudication; and over 1 year of follow-up to (2) evaluate the progression of PAD using ultrasound imaging, (3) determine changes in the ABI, leg pain symptoms, 6-minute walk distance (6-MWD), and the Walking Impairment Questionnaire (WIQ), and (4) correlate PAD progression with changes in the ABI, leg pain, 6-MWD, and the WIQ. We hypothesized that individuals showing ultrasound PAD progression would be more likely to develop claudication, as well as changes in the ABI, 6-MWD, and WIQ.
Methods
Study design
This was a two-site study with the aim of evaluating progression of PAD over a 1-year time period. The institutional review board of each of the respective institutions approved the study. Study subjects were recruited from university clinics and a community screening program when they were found to have an abnormal ABI. After signing an informed consent, patients previously found to have a reduced ABI were invited to undergo repeat ABI measurement and bilateral lower extremity arterial duplex ultrasound mapping, and the 6-MWD test. 8 Also, individuals were asked to complete the SDCQ and the WIQ.2,9 These tests and questionnaires were repeated 1 year after enrollment. A 6-month visit or teleconference was also done to assess for any interim questions or problems and served to promote retention in the study.
Inclusion and exclusion criteria
The inclusion criteria were subjects with PAD defined as an abnormal ABI in any of four ABI measurements (right posterior tibial [PT], left PT, right dorsalis pedis [DP] or left DP), defined as > 0.60 and < 0.90, and subjects could not have complaints of typical claudication on the SDCQ. Also, subjects with an ABI > 1.30 had the toe–brachial index (TBI) measured and were included if a TBI was < 0.70. Three individuals had at least one ABI measurement which was > 1.30; in two of those individuals, there was also an ABI measurement that was < 0.90. In the third person, the PT ratios were both above 1.3 and the DP pulse was not detected bilaterally, but both TBIs were < 0.70. Other inclusion criteria included an ability to perform a 6-MWD test. An exclusion criterion was an inability to lay supine for the ultrasound study.
Ankle–brachial index measurement
The ABI (and if necessary TBI) was measured using standardized methods. 10 Brachial and ankle (right PT, left PT, right DP and left DP) systolic pressures were measured with appropriately sized cuffs with attached sphygmomanometers. Return of flow was determined using a continuous wave Doppler. If an ABI was > 1.30, great toe pressures were measured using a 2.5 cm cuff and return of flow was determined using photoplethysmography – IMEX 9000). Each ankle pressure (or the toe pressure) divided by the highest brachial pressure was used to calculate each ABI (or TBI). Thus, each participant had four ABIs, two left and two right, and a right and left TBI if necessary. The index ABI was defined as the lower of the two leg ABIs.
Lower extremity ultrasound study
All patients underwent a lower extremity duplex ultrasound evaluation of the arterial system and plaque was used as the measure of disease progression. The evaluation began at the inguinal ligament level and included evaluation of the common femoral, superficial femoral, popliteal, and posterior tibial arteries, as per the Inter-societal Commission for Accreditation of Vascular Laboratory guidelines. The peroneal arteries were not evaluated with ultrasound due to difficulty in visualizing these vessels in most individuals. Vessels were recorded as having no lesions, stenotic or occluded. A completely occluded artery was defined as having no blood flow on color and spectral imaging. A plaque was defined as a focal region that protruded into the lumen > 1.5 mm and was distinct from the adjacent boundary. In each artery, PAD progression at follow-up was conservatively defined as either a new stenosis or progression of a baseline stenosis to an occlusion.
Six-minute walk test and functional questionnaires
For the 6-MWD test, subjects were asked to walk in the hall for 6 minutes as per the previously published method. 8 They were asked to cover as much distance as possible during the 6 minutes and were also told to rest and resume walking or cease walking if needed. The primary unit of analysis was the total distance walked (in meters) regardless of whether the subjects stopped or not during the conduct of the test.
The SDCQ 2 is a modification of the WHO claudication questionnaire, and yields five mutually exclusive leg pain categories: no pain, pain at rest, non-calf pain, atypical calf pain, and classic claudication. For leg-specific analyses, we collapsed the initial three categories into ‘no claudication’ and the final two categories, both of which involve calf pain on exertion that does not begin at rest, into ‘claudication’.
The WIQ is a validated measure, which has questions regarding walking distance, walking speed and stair-climbing. 9 The walking distance questions concern the degree of physical difficulty walking distances ranging from indoors around the home to 1500 feet (five blocks; 457 meters) without stopping to rest, with answers ranging from no difficulty to inability to walk that distance. The walking speed questions concern the degree of physical difficulty walking one city block without stopping at speeds ranging from slowly to running or jogging, with answers ranging from no difficulty to inability to move at that speed. The stair-climbing questions range from one to three flights without stopping to rest, with answers again ranging from no difficulty to inability to climb flights of stairs.
Statistical analysis
Data were entered into a relational database created in Microsoft Access with screens that reflected the paper forms. The screens included range and validity checks. The data were converted into a file usable by SAS 9.2, which was used for all statistical analyses. Data were checked and edited. Descriptive analyses were conducted to characterize the subjects at baseline. The follow-up analyses were performed on available data from subjects completing the study and therefore excluded the baseline data from subjects not available at follow-up. Correlations among variables were examined to assist in assessing issues of co-linearity. Bivariate analyses compared mean and standard deviation for continuous variables sorted by binary or categorical variables, with t-test for difference in means. General linear models were used for analyses of change data. All p-values reflect two-tailed tests.
Results
Subject characteristics
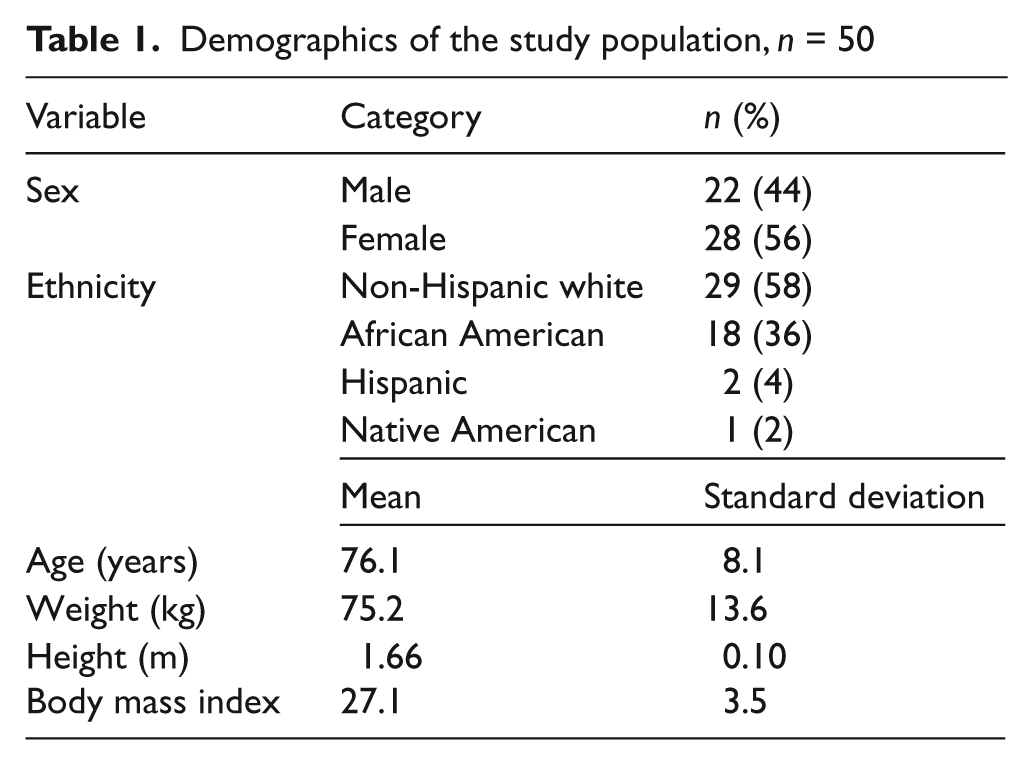
A total of 50 subjects with abnormal ABI on community screening underwent repeat ABI testing and duplex ultrasound in the vascular laboratory (Table 1). There were six subjects without PAD in the study population. Therefore, out of the 50 subjects, 44 had objective evidence of PAD defined as an abnormal ABI and atherosclerotic plaque on ultrasound. The population was an older age group with 58% of non-Hispanic white ethnic origin. The comorbidities included hypertension (n = 36), hypercholesterolemia (n = 27), and diabetes (n = 15), with all defined as being on prescription medication(s) for the disease. Eight subjects had a previous myocardial infarction and three had a previous cerebrovascular accident. Three deaths occurred during the study: one secondary to spontaneous bacterial peritonitis, one due to cardiorespiratory arrest following a stroke, and one due to complications from ischemic cardiomyopathy in the setting of colon cancer.
Demographics of the study population, n = 50
Ankle–brachial index and SDCQ
The ABI and SDCQ results are given in Table 2 and are depicted in sequential columns for all subjects at baseline, for study completers at baseline, for study completers at follow-up, and for the change over time study completers. The mean ABI in each leg was consistent with mild–moderate PAD. Study completers had similar ABIs at baseline to the non-completers, with no significant differences in any ABI measurement between the two groups, and the ABI in completers actually increased slightly. When the original 50 subjects were evaluated by leg, 80 legs had PAD and 20 legs did not have PAD. Ten legs had claudication and 90 did not have claudication. After excluding the six subjects without PAD, there were 78 legs without claudication (Table 2). The 10 legs with claudication were included because the symptoms had changed from the initial eligibility screening, and four of these legs were studied at follow-up. Thirteen of 50 legs (seven subjects) developed claudication at follow-up as per SDCQ.
Ankle–brachial index (ABI) and leg pain category
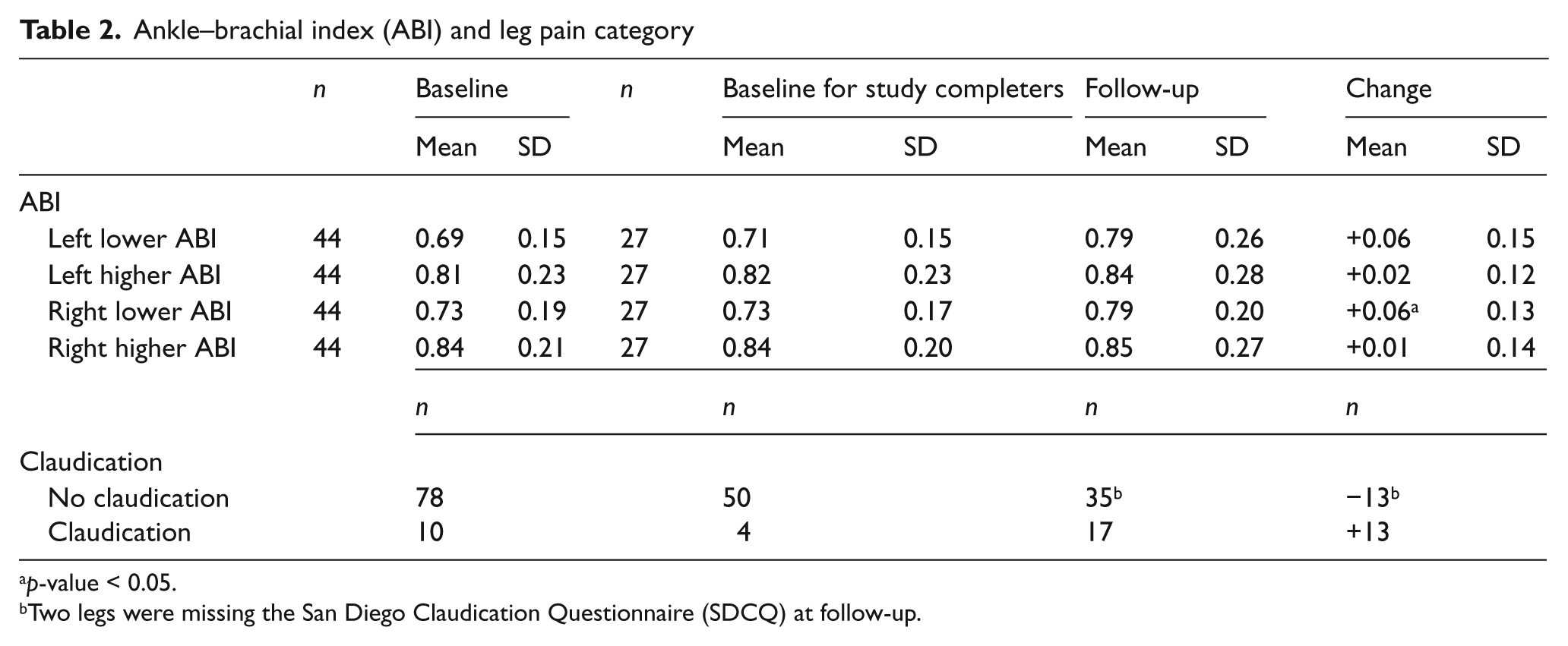
p-value < 0.05.
Two legs were missing the San Diego Claudication Questionnaire (SDCQ) at follow-up.
6-MWD and WIQ
Table 3 uses the same format as Table 2. There were no significant differences between completers and non-completers at baseline (p-values for 6-MWD = 0.19; WIQ distance = 0.62; WIQ speed = 0.93; WIQ stairs = 0.55). Although walking distance declined about 20 meters overall, this change was not statistically significant (excluding the two non-walkers at the 1-year follow-up, the p-value was 0.21; including them with values of 0, the p-value was 0.06). At baseline the WIQ demonstrated that completing patients had moderate severity of functional limitations in walking distance (60%), speed (45%), and stair-climbing (56%) scores. None of the WIQ scores showed significant changes over 1 year (p-values were 0.89, 0.94 and 0.70, respectively).
Six-minute walk distance (6-MWD) and Walking Impairment Questionnaire (WIQ)
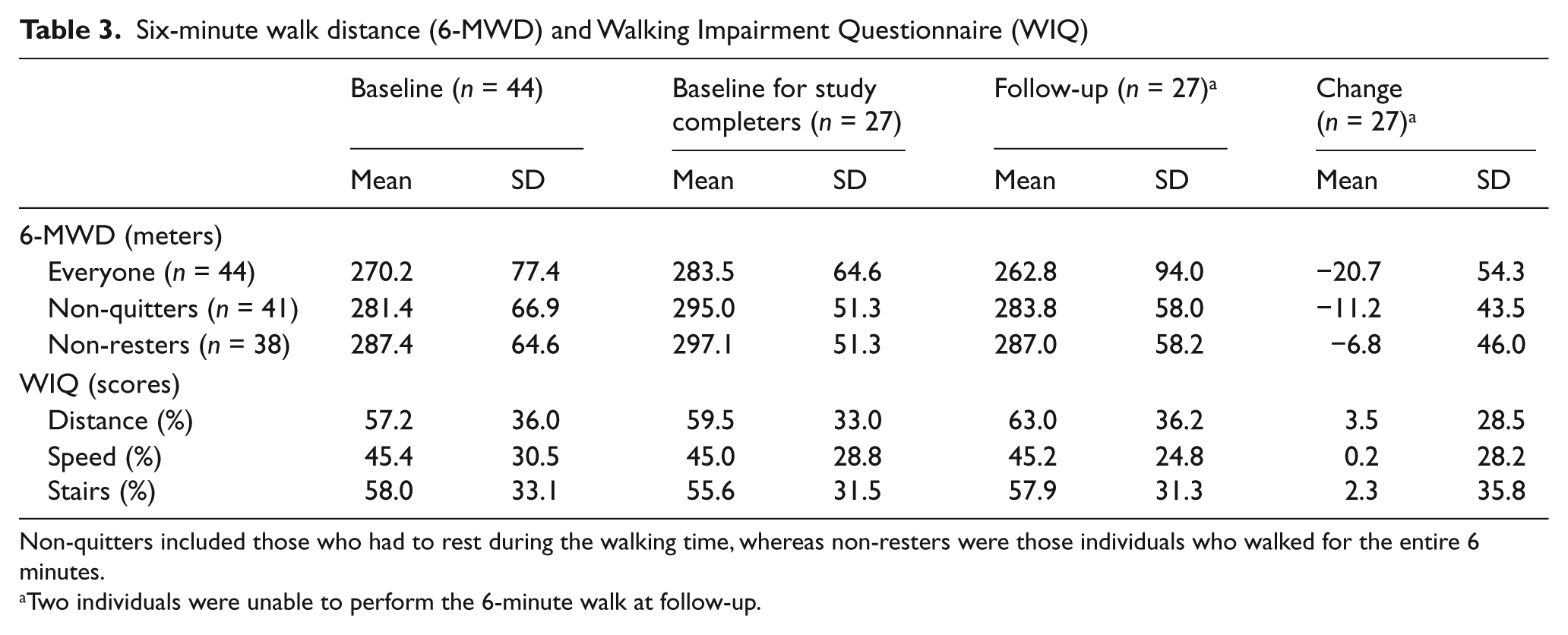
Non-quitters included those who had to rest during the walking time, whereas non-resters were those individuals who walked for the entire 6 minutes.
Two individuals were unable to perform the 6-minute walk at follow-up.
Lower extremity artery lesions and PAD progression on ultrasound
At baseline, atherosclerotic lesions that involved arteries of both lower extremities from the common femoral to posterior tibial level were identified by duplex ultrasound. Forty-four subjects had significant atherosclerotic lesions in the lower extremity arteries, with 80 affected legs. The majority of complete vessel occlusions were in the posterior tibial artery. Of the 13 individuals with occlusions in the posterior tibial arteries, nine had diabetes mellitus. Sixty percent of diabetics (9/15) had posterior tibial occlusions, in contrast with 11% of non-diabetics (4/35) (p = 0.009). All common femoral artery waveforms were triphasic, making significant more proximal disease unlikely.
In study completers, PAD was present at baseline in 48 of the 66 legs. Over 1 year of follow-up, two of the 18 normal legs at baseline developed PAD, and of the 48 legs with PAD at baseline, 17 developed new or progressive lesions. Two legs developed two separate lesions. The leg-specific anatomical location of these 21 lesions in 19 legs of 14 subjects is shown in Table 4. Changes were observed for both above and below-knee vessels, with seven new lesions in the popliteal artery, six in the superficial femoral artery (SFA), four in the common femoral artery (CFA), and four in the posterior tibial artery (PTA). Table 5 shows the characteristics of subjects who had lesion progression compared to those who did not have lesion progression. Although the numbers are small in each group, those who progressed tended to be male, have a higher percentage of hypertension, diabetes and high cholesterol and had more smoking pack years. The cardiovascular disease co-morbidities were similar except for an excess of carotid occlusion.
Lower extremity PAD progression according to anatomical site on ultrasound over 1 year
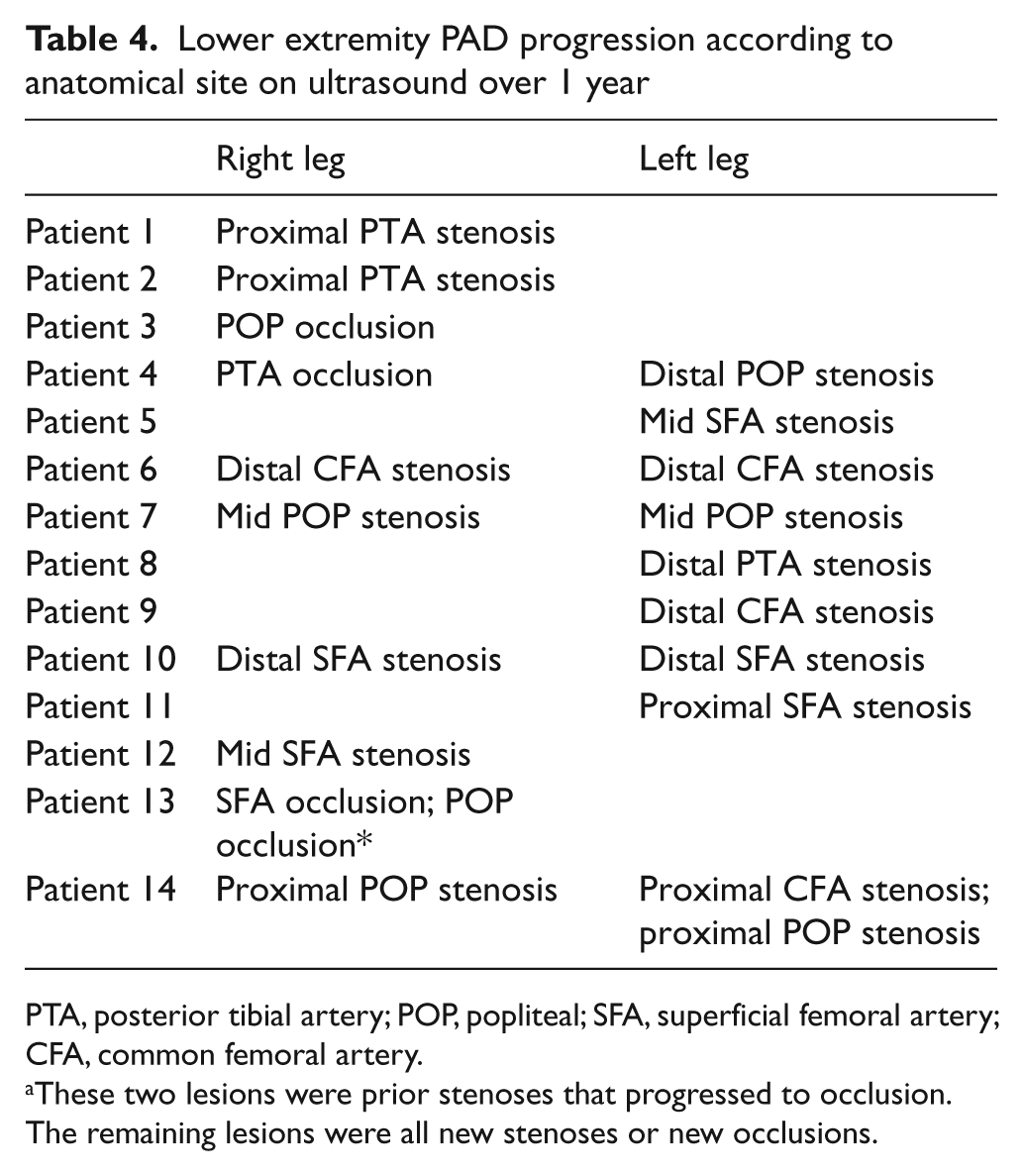
PTA, posterior tibial artery; POP, popliteal; SFA, superficial femoral artery; CFA, common femoral artery.
These two lesions were prior stenoses that progressed to occlusion. The remaining lesions were all new stenoses or new occlusions.
Subject characteristics according to lesion progression
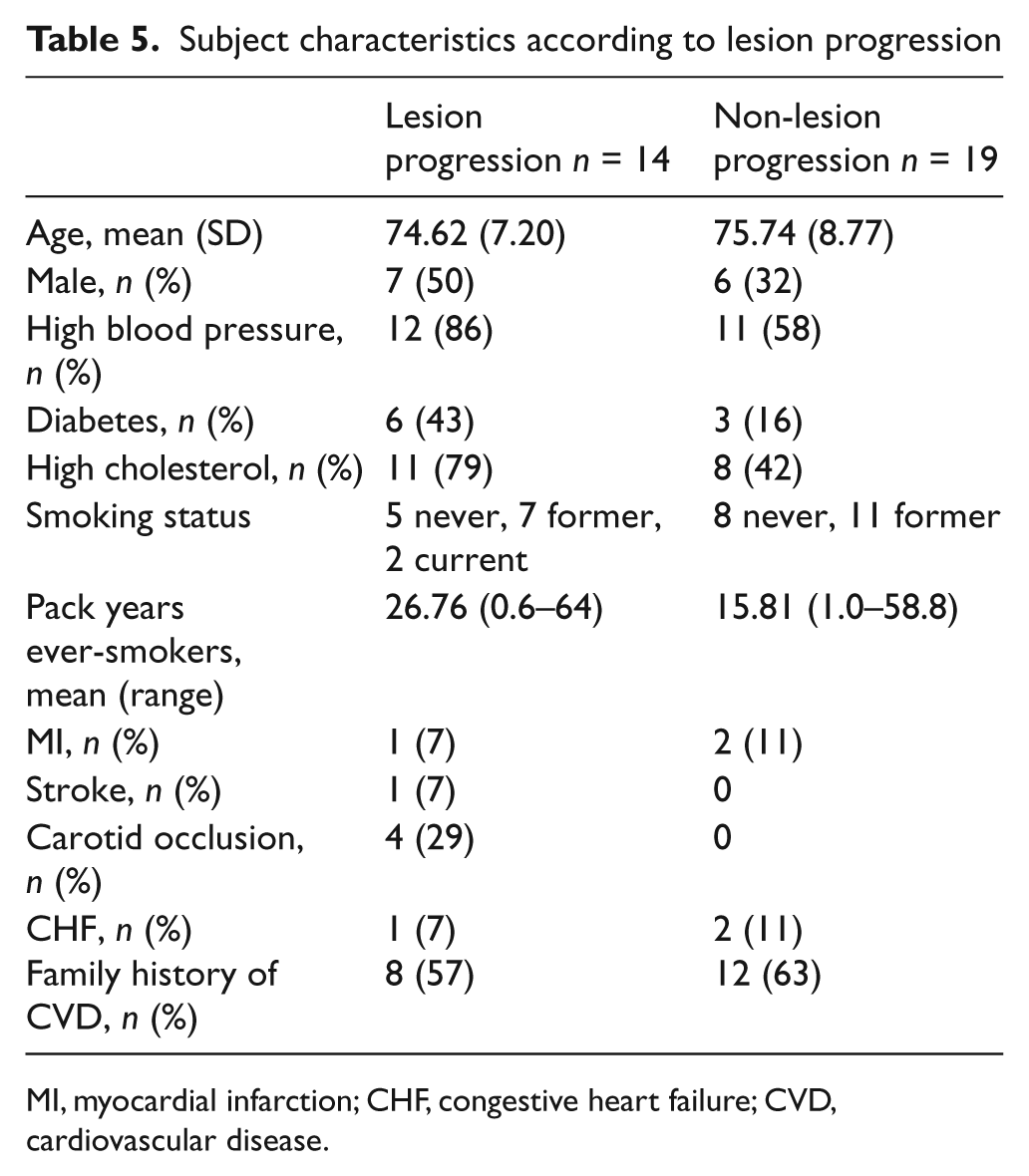
MI, myocardial infarction; CHF, congestive heart failure; CVD, cardiovascular disease.
PAD progression on ultrasound and change in claudication, ABI, 6-MWD, and WIQ
Compared to the 47 legs without ultrasound changes, the 19 legs with PAD progression on duplex ultrasound showed a significantly higher rate of incident claudication: 43.8 versus 13.6%, p-value = 0.012 (Table 6). Legs with PAD progression also showed a smaller increase in the PT and AT (anterior tibial) ABIs, a slightly greater decline in the 6-MWD, and decline rather than increase in the WIQ distance, speed, and stair-climbing scores. However, except for incident claudication, none of these differences was statistically significant. The DP change and the PT change were highly correlated (r = 0.58, p-value < 0.0001), but neither was significantly correlated with walking distance change (r = 0.05, p-value 0.73; r = −0.23, p-value = 0.10, respectively).
One-year claudication, ABI, 6-MWD, and WIQ change by change in PAD progression status
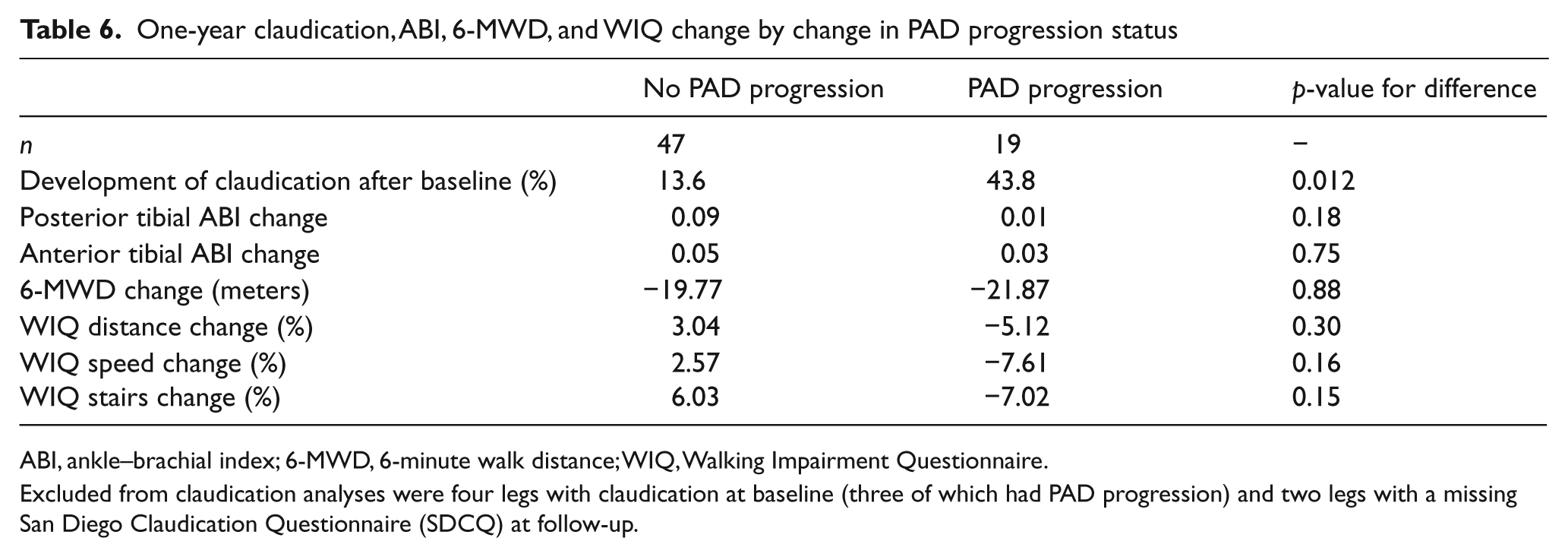
ABI, ankle–brachial index; 6-MWD, 6-minute walk distance; WIQ, Walking Impairment Questionnaire.
Excluded from claudication analyses were four legs with claudication at baseline (three of which had PAD progression) and two legs with a missing San Diego Claudication Questionnaire (SDCQ) at follow-up.
Claudication change and 6-MWD change
Legs of study completers could be grouped into three categories based on their SDCQ results at baseline and follow-up: Group 1 – continued no claudication (n = 35 legs of 18 subjects); Group 2 – no claudication converting to claudication (n = 13 legs of seven subjects); and Groups 3 – continued claudication (n = four legs of two subjects). Figure 1 shows that change in distance on the 6-MWD was −5.3 meters for Group 1 (reference), −36.6 meters for Group 2 (p = 0.05 vs Group 1), and −99.1 meters for Group 3 (p = < 0.001 vs Group 1 and p = 0.03 vs Group 2).
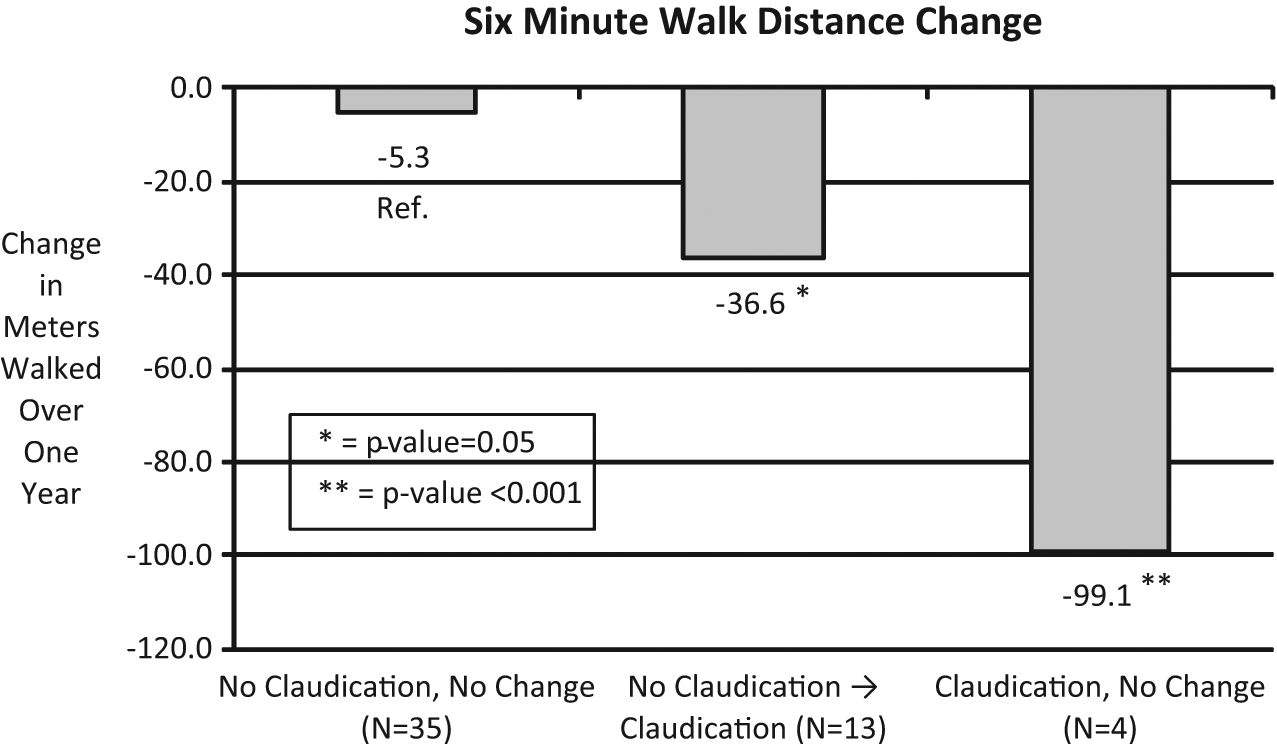
Six-minute walk distance change according to claudication group at 1 year of follow-up (p-values are comparison against the no claudication, no change group).
Discussion
This study demonstrated that patients without claudication but with atherosclerotic lesions on ultrasound had lesion progression over 1 year, and that progression occurred in a variety of vessels. Claudication was more likely to develop if there was concomitant lesion progression, and new claudicants had a significant decrease in the 6-MWD. However, overall there was minimal change in the ABI, 6-MWD, or WIQ over 1 year.
Several studies, beginning as early as the 1940s, have reported the progression of claudication symptoms in patients with PAD who had classic symptoms of claudication at study initiation.11,12 These early studies mainly focused on the development of need for surgery, foot ulceration, and gangrene but also asked patients if claudication symptoms had worsened over time. Clinical risk factors such as smoking and diabetes were correlated with increased risk for progression of disease. One study of 15 patients evaluating progression of disease on serial angiography in patients with claudication found over a 5-year period reported progression of claudication symptoms (patient’s subjective report) in two patients and progression of disease in 14 legs. 13 A publication from the Edinburgh Artery Study, a large-scale cohort study of 695 subjects (5.1% with PAD) aged 55–74 years, reported claudication developing in 179 cases over a 12-year follow-up. 14 The progression of atherosclerotic disease occurred more rapidly in the leg with the higher ABI than in the leg with the lower ABI. In a study evaluating the natural history of PAD using angiography and ultrasound, superficial femoral artery lesions progressed more rapidly among patients whose contralateral superficial femoral artery was occluded. 15 In our study, the conversion from no claudication to claudication was a bilateral phenomenon in six out of the seven patients with symptom progression.
The ability of the ABI to predict disease progression in the lower extremity appears less sensitive than ultrasound in the present study, although the follow-up was limited to 12 months. A study by McLafferty et al. of 193 extremities in patients with prior lower-extremity revascularization, found that the ABI was relatively insensitive in identifying disease progression as demonstrated on angiography or duplex ultrasound scanning. 16 Similarly, despite PAD progression and development of claudication, overall neither the 6-MWD nor the WIQ changed significantly over 1 year.
Limitations
The aorto-iliac vascular territory was not evaluated directly in this study and thus proximal disease cannot be completely ruled out in subjects. However, the spectral Doppler waveform in all subjects was triphasic at the proximal common femoral artery, indicating no significant proximal disease in subjects. There was some attrition that occurred throughout the course of the study, including three subjects who died. This attrition may have introduced a type 2 statistical error for questionnaires. The baseline ABIs of non-completers was not statistically different from the ABIs of completers (Table 2). It is unclear how their non-participation might have affected the progression data. Mortality in this study most likely minimized disease progression, since disease progression has been independently related to mortality. 17 Another potential limitation is that there were some instances where the same ultrasound technician at the University of Pennsylvania site did not conduct the follow-up ultrasound study; theoretically that could introduce variability.
Conclusions and clinical implications
There is a population of subjects with no classic claudication symptoms who have both reduced ABI and significant atherosclerotic lesions in the lower extremity arteries. A significant number of legs (21%) developed claudication over 1 year. These data support the hypothesis that a significant number of asymptomatic patients with PAD and abnormal ABI progress relatively rapidly. Those progressing were more likely to develop claudication, and new claudicants had a decrease in 6-MWD. This population deserves more study to determine if risk factor modification inhibits atherosclerotic lesion progression and development of symptoms.
Footnotes
This study was supported by an unrestricted grant from Bristol-Myers Squibb and Sanofi-Aventis pharmaceutical companies.
A portion of Dr Mohler’s salary was supported via NIH National Heart, Lung, and Blood Institute grant K12 HL083772-01. Dr Mohler received an unrestricted research grant from the Bristol-Myers Squibb and Sanofi-Aventis partnership to conduct the study. The other authors do not report any relevant disclosures.
