Abstract
Old and frail patients undergoing vascular surgery seem at great risk of developing postoperative delirium (POD). The aim of this review was to identify risk factors for the development of POD in vascular surgery patients. Different aetiological factors have been identified, such as increased age, excessive blood transfusion, preoperative cognitive impairment and depression. Mounting evidence supports a role for inflammation and tobacco exposure in the development of POD. Vascular surgery patients differ from the general surgical population because they suffer from both loco-regional and systemic atherosclerosis. Although current scientific evidence cannot fully link both entities, evidence is growing that suggests a relationship between systemic and cerebrovascular atherosclerosis and the development of POD.
Introduction
Cardiovascular disease is a common problem in the general population, affecting an essential segment of the adult population over the age of 60 years. The lifetime risk for individuals at the age of 40 is 49% in men and 32% in women, making it a substantial healthcare burden. 1 The main process in the development of cardiovascular disease is atherosclerosis, which starts as early as adolescence and may cause plaque formation and thromboembolic events later in life. Among the multiple risk factors established for cardiovascular disease, high age remains one of the strongest predictors for a cardiovascular event.2,3 Furthermore, there is a strong relationship between the number of vascular beds affected and the risk for additional atherosclerotic events. For example, patients with a low ankle–brachial index (< 0.9) also have a significantly increased risk for fatal myocardial infarction, emphasizing that atherosclerosis is, and acts like, a systemic disease. 4
For elderly patients admitted to hospital, postoperative delirium (POD) is a common and important complication. It is defined as an acute disorder of attention and cognition and is characterized by fluctuating symptoms of inattention, disturbance of consciousness and disorganized thinking. 5 Not only is it the most common complication in elderly (> 65 years) patients, affecting about 11–60% during hospital admission, POD is also associated with short-term effects such as prolonged hospitalization and institutionalization (due to functional loss, loss of independence and the inability to return to one’s home) and increased medical costs, but also long-term effects such as persistent functional decline and death.5–12
With the elderly population increasing at an unprecedented rate, more elderly patients are being considered for surgery, which inevitably will result in an increased incidence of POD in the near future. In current literature, various risk factors for the development of POD in the elderly have been identified. General risk factors are mainly age-related and include high age, pre-existing comorbidities, pre-existing dementia or cognitive impairment, and a history of prior delirium.10–12 The overall incidence of POD is estimated at 11–24%, with an incidence up to 60% in high-risk geriatric patients. There is a widespread variation in incidence between different types of surgery, with few outliers carrying an increased risk, including cardiac, orthopaedic and vascular surgery. In a recent systematic review, addressing preoperative risk assessment for POD after non-cardiac surgery (gastrointestinal surgery not included), the highest incidences were found in abdominal aortic surgery (46–52%). 5 POD and atherosclerosis often occur together in elderly patients, but it is questionable whether high age alone is the common denominator. We therefore hypothesized that atherosclerosis, as a systemic disease, is the main reason for the increased incidence of POD in vascular surgery patients when compared to the general surgical population. The purpose of this study was to identify prognostic factors for the occurrence of POD within the field of vascular surgery and to find out whether atherosclerosis causes an increased risk.
Incidence and risk factors of postoperative delirium in vascular surgery
Vascular surgery patients are among patients at highest risk for developing POD. At first this seems to be caused by the fact that vascular patients are both old and frail and usually present with multiple comorbidities, including cerebrovascular disease. Yet notwithstanding these known risk factors, an evident pathogenesis of POD is still unclear. 13 Inouye et al. was able to identify four baseline risk factors for the development of POD in a general population which included impaired vision, severe illness, cognitive impairment and a high blood urea nitrogen/creatinine ratio. However, a prospective study specifically focusing on risk factors and outcome in POD among elderly patients was unable to identify an underlying cause in 88% of subjects who developed POD.11,14 These results further underline the heterogeneity of POD and its challenges in treating it solely based on these biological factors. With regard to the surgical population, there are a number of perioperative factors that influence the incidence of POD. Both the amount of intraoperative blood loss and intravenous fluid infusion and a poor preoperative nutritional status and low body mass index (BMI) are associated with a significantly higher incidence of POD and a longer hospital stay.15–18 Second, one of the most potent risk factors which may be prevented is pain, which seems to be directly associated with both POD and functional decline.19,20 No difference exists between intravenous or epidural analgesic modalities on POD, but postoperative opioid administration is associated with an increased risk of POD in elderly patients.21,22 Furthermore, in an emergency setting, the incidence of POD significantly increases, suggesting an even greater awareness of possible POD is needed under these conditions.5,23
Few studies focus selectively on risk factors and POD prevention among vascular surgery patients15,24–30 (Table 1). A recent review on POD after elective vascular surgery was able to identify various aetiological factors, including age, preoperative cognitive impairment, depression, and excess interoperative blood transfusion. 28 Unfortunately, due to small sample sizes, heterogeneity and differences in confounding variables, a proper meta-analysis could not be performed. Nevertheless, the influence of inflammation and tobacco exposure emerge from the current data that appear to be particularly applicable to patients who undergo vascular surgery procedures, both of which we will therefore further discuss in detail.
Studies on incidence of POD in elective vascular surgery
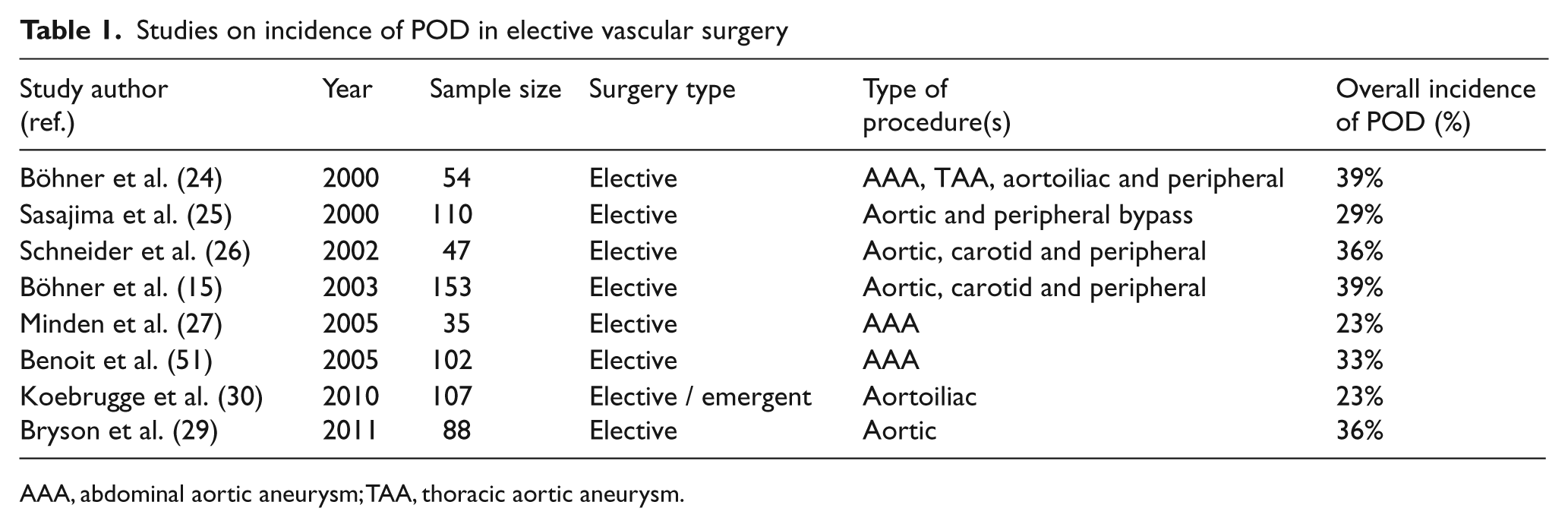
AAA, abdominal aortic aneurysm; TAA, thoracic aortic aneurysm.
Inflammation
In a comparative study in patients with asymptomatic peripheral artery occlusive disease (PAOD), an elevated C-reactive protein (CRP) blood level turned out to be a significant negative predictor in multiple cognitive tests and an impaired visuoconstructive performance suggested the presence of a relationship between POD and inflammatory processes. 31 The only study to date that focuses on POD and critical limb ischaemia found a highly significant relationship between critical limb ischaemia and POD (OR = 3.8; 95% CI 1.3–10.9). 25 Unfortunately, the expected processes in critical ischaemia, such as necrosis with concomitant elevated inflammatory markers, were not further analysed with regard to the risk of POD and thus the relationship is merely suggestive. In view of a potential intervention strategy for POD, Böhner et al. determined a prediction model based on both somatic and psychiatric risk factors in vascular surgery patients and also found a preoperative elevated (> 3 mg/l) CRP level to be associated with an increased probability for the development of POD. 15 Furthermore, vascular surgery patients who developed POD scored higher preoperative depression scores, had decreased psychosocial functioning and more cognitive impairment, suggesting the presence of some predisposing factors within this group.
Although various studies have found high levels of CRP to independently predict the incidence of delirium, the apparent cause was not clear.32,33 Even more indicative of a causal relation between POD and inflammation is the substantial evidence that more refined inflammatory markers such as cytokines play a vital role. 34 Whereas CRP is an acute-phase protein, and a non-specific marker for inflammation, cytokines are far more sensitive as they are specific mediators of the immune system. Current data suggest a pathway of inflammation that culminates in higher concentrations of various markers in peripheral blood.35,36 Various studies focusing on inflammatory markers as predictors for delirium showed that patients who developed a delirium had significantly elevated levels of interleukins 6 and 8.37–39 A recent review, investigating the role of inflammation in the pathogenesis of delirium, further confirmed the role of these inflammatory processes in the development of delirium. 34 Despite the selective regulation of the blood–brain barrier, the brain is in fact directly influenced by peripheral cytokines through activation of vascular endothelial cells and perivascular cells in the brain. By propagating the inflammatory cascade, this may then lead to neuron injury. 34 This neuroinflammatory response causes neurocognitive behavioural abnormalities. 40 One of the studies focusing on CRP and activation of vascular endothelial cells also found a close correlation between the two systems, further supporting the concept that inflammatory processes indeed interact with vascular cells. 41 There is increasing evidence that aging is associated with increased neuroinflammation, manifested as increased levels of activated microglia, the immune cells of the brain. 42 This increased baseline inflammation in the elderly has a priming effect resulting in an enhanced response to peripheral stimuli. 43 This priming of the aging brain can be a plausible explanation for the increased response to stimuli associated with surgery.
In view of the proposed inflammatory responses, current insights even suggest that atherosclerosis constitutes an autoimmune disease and shares similarities with chronic autoimmune diseases such as systemic lupus erythematosus (SLE), rheumatoid arthritis and vasculitis. So it seems that both auto-antigens and peripheral lymphocytes play an important role in the development and regulation of atherosclerotic lesions. 44
Tobacco
Nicotine consumption is a well-known risk factor within the multifactorial pathogenesis of delirium. Particularly within an intensive care population, smoking significantly increases the risk for delirium.45–47 Patients suffering from vascular disease are known for their nicotine abuse and thus seem to be at increased risk of POD because of this. The causal relation between nicotine abuse and POD is not fully understood but seems twofold. First, cigarette smoking effects are believed to induce neuro-adaptive changes in the brain, especially after cessation. Although these changes are complex, they are related to an increase in neurotransmitter function which shares a common pathway with delirium. 46 Second, atherosclerosis and microvascular changes in the brain caused by smoking can lead to impaired cognitive functioning and reserve. 47 Although a different entity, there is a significant relationship between the increased risk on the development of Alzheimer’s disease, vascular dementia, cognitive decline and smoking.48–50
Clinical proof for these theories is based on several studies. Patients who undergo elective abdominal aortic aneurysm repair are already susceptible for the development of POD. Additionally, there appears to be a strong association between tobacco exposure and POD (p = 0.001) in this specific group of vascular surgery patients. 51 Patients developing POD were taking significantly more psychoactive medication, had lower mental status scores and had a significantly higher number of smoking pack-years. With age being equally distributed between the studied groups, age ≥ 80 years was also a risk factor for POD. In a second study, focusing on vascular risk factors for POD, the indication for vascular surgery and tobacco exposure again proved to be independent risk factors for POD. 52 After adjustment for age, the associated risk factors for tobacco exposure and vascular surgery were RR 1.6, 95% CI 1.0–2.6 and RR 2.7, 95% CI 1.7–4.2, respectively. Patients with both vascular risk factors and impaired cognitive performance were at double the risk for POD development (RR = 3.2; 95% CI 2.1–4.9), an effect the authors refer to as ‘a double gradient effect’. Nevertheless, the above-mentioned results point more towards lifetime tobacco exposure than to current smoking behaviour, suggesting the presence of smoking-induced (micro) vascular changes rather than a direct toxic effect. Further support for this theory comes from a study focusing on smoking and dementia. Compared with never smokers, current smokers had an increased risk of Alzheimer’s disease (RR = 2.72; 95% CI 1.63–5.42) and vascular dementia (RR = 1.98; 95% CI 1.53–3.12) after adjusting for age, sex, education, blood pressure, and alcohol intake. 53 In non-demented elderly individuals, smoking may also accelerate cognitive decline. 54 Whether this is related to POD and causes an increased risk remains unclear.
Atherosclerosis and cognitive dysfunction
Cerebral atherosclerosis and cognitive dysfunction
Atherosclerosis is a systemic disease. Consequently, patients presenting with PAOD are at a higher risk for cere-brovascular disease.55,56 Atherosclerosis in the carotid arteries may lead to both decreased cerebral perfusion and/or microemboli, suggesting that a decreased cognitive function might be causal to this disease and therefore itself a potential risk factor for POD. One of the studies supporting this hypothesis compared the cognitive performance of PAOD patients with healthy individuals and stroke patients. PAOD patients had a diminished performance of cognitive function and seemed, independent of age, at risk for a continuum of cognitive impairment. 55 A large, prospective, population-based study (The Leiden 85-plus study), used to asses the relationship of generalized atherosclerosis and cognitive decline, annually scored a large community-dwelling elderly population with a predetermined atherosclerotic burden on various cognitive domains. This study showed that in old age generalized atherosclerosis is indeed associated with cognitive decline.57,58 In contrast to the expectations, there was no association between generalized atherosclerosis and depressive symptoms, a finding that conflicts with the vascular depression hypothesis.59,60 This hypothesis proposes that vascular disease underlies mood disorders in some older adults and that vascular disease is associated with a greater risk of cognitive decline and dementia. Rudolph et al. found that mild cognitive impairment and predisposed vascular risk factors, such as hypertension, diabetes, congestive heart failure, previous myocardial infarction and the need for vascular surgery, all independently contribute to the occurrence of POD. 52 Unfortunately, all other research conducted in the area of atherosclerosis and POD was done after coronary artery bypass surgery and most papers in this field focused on aortic atheroma burden and the risk of aortic clamping leading to iatrogenic thromboemboli, which makes comparison with general (peripheral) vascular surgery difficult.61–67 It seems likely that these patients have a similar high burden and distribution of atherosclerosis. Although both carotid artery and intracranial stenosis have been associated with worse cognitive outcome, 62 the literature is contradictory on this point. Whereas Rudolph et al. found a significant association between a higher atherosclerosis score and the incidence of POD, Bar-Yosef et al. found no relationship between aortic atherosclerosis and postoperative cognitive dysfunction.61,64 The former uses a 0–3 scale, points given for carotid stenosis > 50%, three or more affected cardiac vessels and moderate or severe ascending aortic plaques; the latter focuses solely on the proximal aorta atheroma burden, which therefore seems less relevant to the general vascular surgery population.
Thromboembolic events
Although the relationship between atheroma burden (%) of the proximal aorta and the development of POD has been clearly demonstrated, interestingly the current results contribute to the lack of a clear consensus on its true pathophysiology. 63 Although the relationship between the degree of cerebral microemboli and aortic atheroma is evident, its influence on cognition remains unclear. 65 Whether the development of POD is related to thromboembolic events, due to aortic atherosclerosis, remains therefore also unanswered. A recent study, designed to determine the association between cerebral microemboli and POD in cardiac surgery patients, found no significant difference after short-term follow-up between the microemboli count and the occurrence of POD. 66 It was concluded that the long-term clinical impact of cerebral microemboli remains unknown. The increased risk of POD after coronary artery bypass graft surgery may possibly be caused by direct manipulation of the aortic arch, which may lead to disrupted plaques and thrombi-emboli. Whether the current results also apply to general vascular surgery patients is therefore difficult to estimate. Although hypothetically the perioperative stress in general vascular surgery patients could lead to similar plaque disintegration, instability and thrombi, this can not be reliably determined.
Discussion
The development of POD in vascular surgery patients seems not just a process of combined comorbidities and high age but more so the inability to adjust to changes in stress, physiology and environment. We have explored the incidence and the multifactorial pathogenesis of POD in vascular surgery patients and attempted to demonstrate a relationship between atherosclerosis and POD. The known literature on delirium and POD is overwhelming and confirms the complex composition which leads to POD. In the vascular surgical population, there are several general factors, such as blood loss, use of analgesics, emergency surgery and nutritional status that could potentially be affected by preventive measures by incorporating them into risk-reduction strategies of postoperative POD.
Our study has identified groups of patients that are at increased risk for POD development. First, there appears to be an association between the emergence of POD and smoking. Although current literature falls short in giving a clear statement, the relationship with the number of pack-years smoked clearly suggests (micro) vascular damage as a causal factor, although neuroadaptive changes may also play a role. With the combination of vascular surgery and tobacco exposure, as independent risk factors for POD, it is highly suggestive that an increased atherosclerosis burden is the causal factor. Whereas smoking cessation was formerly advised in favour of wound healing, patients with vascular disease should also cease smoking in time to reduce the chance of POD at time of surgery.
Another interesting observation is an apparent relationship between inflammatory processes and the risk of POD. The origin of these elevated inflammation markers seems twofold. The first, and most obvious reason, is ischemia-related necrosis due to PAOD, which is frequently seen in vascular patients, especially in advanced disease. Second, there is increasing evidence that atherosclerosis acts as an autoimmune disease, in which both auto-antigens as well as peripheral lymphocytes play an important role in the development and regulation of atherosclerotic lesions. As elderly patients already have increased neuroinflammation, this can lead to an amplified inflammatory response to peripheral and central stimuli. However, it is very likely that both mechanisms co-exist and both can be a potential target for intervention.
Vascular surgery patients should be considered a highly vulnerable population and are more susceptible for postoperative complications, such as POD, than their counterparts in general surgery. This increased vulnerability is frequently referred to as frail, which is defined as the risk for adverse outcomes due to losses in different domains of functioning, related directly to these adverse processes. 68 Robinson et al. already suggested that it is more a matter of physiological vulnerability and that baseline frailty characteristics play a vital role in the development of POD. 11 At our centre, the Groningen Frailty Indicator (GFI) has been developed to pre-emptively identify patients at risk for POD and other adverse outcomes. Although primarily designed for oncogeriatric patients, it is already validated among different patient groups in various publications.69–72 A recent review on delirium prevention and management stresses the importance of secondary prevention of POD, including early detection and treatment. 73 Unfortunately, very few studies focus on risk factors and POD prevention among vascular surgery patients and an accurate pre-screening tool is currently lacking. Although specialized geriatric wards and multicomponent interventions have been proven to prevent POD and improve outcome, in many hospitals this is probably not feasible.14,74 Recently, a prospective study was conducted at our hospital that demonstrated that the GFI is a significant predictor for the occurrence of POD. 75 The GFI can be helpful in the early identification of a select group of high-risk patients with respect to the development of POD after vascular surgery. By identifying these patients during the preoperative outpatient evaluation, appropriate preventive arrangements, such as preoperative geriatric consultation, can be implemented. This seems particularly important because proactive geriatric consultation has been proven to reduce both delirium incidence and severity after surgery.76,77 To date, there is no consensus regarding the efficacy of pharmacological treatment for delirium prevention. Although prophylactic low-dose haloperidol may reduce the severity and duration of delirium episodes, there is no effect on the POD incidence. When POD still occurs, the pharmacological treatment is primarily based on antipsychotics. 78 In particular, low-dose haloperidol may be effective in decreasing the degree and duration of delirium in postoperative patients. 79
POD is associated with various long-term effects such as persistent functional decline, and death. Although it is generally assumed that atherosclerosis is a risk factor for depressive symptoms, ‘the vascular depression hypothesis’, this could not be demonstrated in The Leiden 85-plus study. POD, on the other hand, is statistically significantly associated with long-term psychosocial distress with symptoms of depression and/or anxiety. 80 Interestingly, preoperative anxiety and depressive symptoms are not associated with POD. 81
In conclusion, although the results of the studies published thus far have not consistently identified the aetiology between atherosclerosis in vascular surgery patients and POD, a subgroup of individuals can be identified with clear predictive parameters (smoking and elevated inflammatory markers) for whom a preoperative intervention strategy could be necessary.
Footnotes
Acknowledgements
The authors thank Dr Gerbrand J Izaks, MD, PhD for critically revising the manuscript and providing relevant suggestions for improving the final manuscript.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
None.
