Abstract
Type 2 diabetes mellitus (T2D) is an example of a disease process that results in decrements in function additional to those imposed by the inexorable ‘primary aging’ process. These decrements due to disease, rather than primary aging, can be termed ‘secondary aging’, and include the premature development (as early as adolescence) of asymptomatic preclinical cardiovascular abnormalities (e.g. endothelial dysfunction, arterial stiffness, diastolic dysfunction), as well as impaired exercise performance. These abnormalities are important, as they are associated with greater cardiovascular morbidity and mortality in people with and without T2D. A better understanding of the pathophysiology of secondary cardiovascular aging in people with T2D is warranted, and an evaluation of the benefits of existing treatments for these abnormalities is useful (e.g. exercise training). The focus of this review is to discuss the data relevant to the following key postulates: (a) T2D causes premature cardiovascular aging; (b) in contrast to primary cardiovascular aging, the premature cardiovascular aging of T2D may be modifiable with exercise. The exercise-focused perspective for this review is appropriate because impairments in exercise performance are markers of premature cardiovascular aging in T2D, and also because exercise training shows promise to attenuate some aspects of cardiovascular aging during the preclinical stage.
Keywords
Introduction
Normal or primary aging is an inexorable progress that occurs at a fairly standard rate with increasing chronological age. An example of a manifestation of primary aging is the inevitable decline in maximal heart rate with age (i.e. predicted maximal heart rate = 220 – age [years]). In contrast, effects of secondary aging, defined as impairments resulting from a disease process or other factors, occur at a variable rate and can, in some cases, be modified by behavioral factors or other therapies.1–3 Secondary aging results in impairments in addition to those imposed by primary aging.1–3
The effects of type 2 diabetes mellitus (T2D) on cardiovascular aging are clearly additive to those of the primary aging process.1–4 One example of the secondary cardiovascular aging burden in T2D is the high prevalence of cardiovascular disease (CVD).5–7 Meta-analysis data suggest a comparable cardiovascular mortality risk in people with T2D as compared to people with a history of established coronary heart disease. 6 Factors related to T2D also lead to early development of CVD, including metabolic dysfunction, hypertension, dyslipidemia, adiposity, and inflammation.1–3 Thus, the premature development of CVD in people with T2D appears complex and multifactorial.
While the mechanisms of premature CVD in T2D are complex, the milestones of CVD development are similar in people with and without T2D. Clinically evident CVD is generally preceded by the development of asymptomatic impairments in cardiovascular function (e.g. endothelial dysfunction, arterial stiffness, diastolic dysfunction).2,3,8–10 These asymptomatic impairments in cardiovascular function may be termed ‘preclinical cardiovascular abnormalities’ and occur earlier in life in people with T2D than in non-diabetic people.11–17 Even adolescents with T2D have observable preclinical cardiovascular abnormalities, as well as demonstrating abnormal exercise performance.12–14,18 Premature impairments in cardiovascular function are inherently important because they are associated with greater cardiovascular morbidity and mortality in people with and without T2D.19–23 Recent interest has resulted in better understanding of the pathophysiology of secondary cardiovascular aging in people with T2D, as intervening in this process during the preclinical phase of cardiovascular abnormalities may prevent and/or delay cardiovascular morbidity and mortality.1–3,8,24
The focus of this review is to discuss the data relevant to the following key postulates: (a) T2D causes premature cardiovascular aging; (b) in contrast to primary cardiovascular aging, the premature cardiovascular aging of T2D may be modifiable with exercise and other therapies. The exercise-focused perspective of this review is appropriate because impairments in exercise performance are markers of premature cardiovascular aging in T2D, and also because exercise training shows promise to attenuate some aspects of cardiovascular aging during the preclinical stage.
Although exercise training demonstrates clear cardiovascular benefits, people with T2D are less active than their peers without diabetes, and there are barriers to physical activity for people with T2D that can impede regular physical activity.25–28 In this review, Section II will address the data to support premature worsening of exercise performance in T2D and will discuss potential mediators; Section III will address the data to support preclinical cardiovascular abnormalities in T2D and will discuss potential mediators; Sections IV and V will discuss practical exercise recommendations and challenges to increasing regular physical activity for people with T2D.
Early markers of cardiovascular aging in T2D – abnormal exercise performance
Peak exercise performance levels have generally been ~20% worse for people with T2D as compared to age and sex-matched healthy controls (Table 1). The finding of impaired peak exercise performance has significance for two reasons. First, epidemiologic data strongly suggest that greater levels of physical activity and fitness are associated with less mortality in patients with and without diabetes.23,29,46,47 Although estimates of the efficacy of exercise training to reduce mortality are limited by the lack of RCT-level data, the existing epidemiologic data are very strong (e.g. inverse dose–response relationship between physical activity/fitness and cardiovascular mortality in people with and without T2D, plausible biological mechanisms).23,29,46,47 Also, as discussed further in the section entitled “Effects of exercise training in T2D”, the exercise performance abnormalities and preclinical cardiovascular abnormalities are often amenable to modification, as shown by the joint benefits of exercise training with regard to exercise performance,32,38–43 and preclinical cardiovascular abnormalities (particularly in the earlier stages of disease).48–53
Relationship of exercise performance measures with aging and T2D

T2D, type 2 diabetes mellitus; VO2, oxygen consumption; VO2peak, peak oxygen consumption; tau2, time constant reflecting the time course of the exponential rise of VO2 (2nd phase of VO2 kinetics) until the achievement of steady-state VO2, where slower tau2 denotes poorer cardiorespiratory adjustment.
Decline in exercise performance with usual aging
Exercise performance measures that we will discuss include peak exercise performance (e.g. cardiorespiratory fitness) and submaximal exercise performance. Cardiorespiratory fitness is defined as “the ability to perform large muscle, dynamic, moderate-to-high intensity exercise for prolonged periods”. 30 The accepted gold standard measure of cardiorespiratory fitness is VO2max (or VO2peak, as it is most appropriately referred to in populations with a disease process such as T2D, which may limit exercise performance). 30 VO2peak measures an individual’s maximal or peak ability to utilize oxygen during incremental exercise testing. 30 In contrast, submaximal exercise performance refers to the physiological response to lower intensity work rates, generally measured during exercise at a constant rate workload below the anaerobic threshold, in order to attain a steady-state level of oxygen uptake.
VO2max declines ~10% per decade in healthy individuals.30,31 The effect of age on VO2max is influenced by several factors, some of which are modifiable (e.g. physical activity levels, changes of body composition) and some of which are not modifiable (e.g. maximal heart rate). 31 Maximal heart rate declines at a rate of 3–5% per decade, regardless of exercise training or sex, demonstrating a ‘primary aging’ effect. 31 Changes in body composition (e.g. loss of lean body mass, increasing adiposity) account for a portion of the decline of VO2max with aging in non- athletes, but do not appear to play a significant role in the athletes’ decline of exercise performance with aging.54,55 Although athletes have consistently displayed greater VO2max than their age-matched non-athletic controls, both athletes and non-athletes exhibit declines in VO2max by ~10% per decade.55,56
T2D impairs peak exercise performance
Although it would generally take 20 years of aging for a healthy person to experience a 20% decline in VO2max, both adolescents and adults with T2D demonstrate a ~20% reduction in VO2peak, as compared to healthy controls, even when matched for age, sex, and physical activity levels (Table 1).12,16,17,32–37 A subset of these studies confirmed that even when subjects with T2D have no evidence of prior CVD and normal resting echocardiograms, VO2peak is significantly worse, whether measured in absolute units (l/min) or adjusted for body weight (ml/kg/min).12,16,17,37 Significant impairments in VO2peak have been found in most studies that evaluated exercise capacity in T2D,12,16,17,32–37 although not all.13,57 It is notable that even in the two studies where VO2peak did not significantly differ from controls, concerning preclinical cardiovascular abnormalities were observed in the participants with T2D.13,57
T2D impairs submaximal exercise performance
In addition to impairments in maximal exercise performance described above, abnormalities in submaximal performance have also been demonstrated in people with T2D (Table 1).12,17,58 These include a blunting of the usual rise in VO2 and heart rate per workload increment, as well as slowed oxygen uptake kinetics (VO2 kinetics).12,17,58 VO2 kinetics describe the time course of the exponential rise of VO2 after the onset of exercise until the achievement of steady-state oxygen uptake.59,60 Slowed VO2 kinetics are generally represented by a prolonged time constant (tau2) of the second phase of VO2 kinetics. 60 A prolonged tau2 denotes poorer cardiorespiratory adjustment, due to either factors that impair oxygen delivery to working muscles (e.g. cardiac factors, peripheral autoregulation of blood flow) and/or factors related to oxygen extraction and utilization by working muscles (oxygen diffusion, tissue oxygen consumption). 59 Clinically, a prolonged tau2 may be related to worse exercise tolerance owing to the accumulation of a greater O2 deficit during the transition phase to steady-state VO2. 44
People with T2D generally have slowed VO2 kinetics, as evidenced by a prolonged tau2.12,17,37 In the single study that did not demonstrate a significantly prolonged tau2, the authors hypothesized that the greater oxygen extraction observed in T2D versus control participants may have compensated for abnormalities in microvascular flow related to T2D. 37
Potential mediators of abnormal exercise performance
Likely mediators of the abnormal exercise performance observed in T2D include insulin resistance, skeletal muscle factors, hyperlipidemia, and inflammation. The degree of insulin resistance has been related to the observed reductions in cardiorespiratory fitness in T2D in several studies.12,61–64 Greater insulin resistance has also been associated with lower cardiorespiratory fitness in other medical conditions, such as chronic kidney disease, congestive heart failure, and metabolic syndrome.64–68 Potential mediators related to skeletal muscle factors include impaired mitochondrial content and function,69,70 increased ratio of type IIb-to-type 1 muscle fibers, 71 and greater sarcopenia in people with T2D versus healthy controls. 72 Dyslipidemia, particularly characterized by elevated free fatty acids and low HDL cholesterol, may also mediate impaired exercise performance in populations with T2D, as suggested by such relationships in other insulin-resistant populations.12,68 Inflammatory markers have been correlated with worse measures of cardiorespiratory fitness in people with T2D. 73 Although the diagnosis of T2D hinges on hyperglycemia, several studies have failed to demonstrate any association between exercise performance and either hemoglobin A1c or fasting serum glucose.12,16,34,74 Obesity appears unlikely to be a key mediator of the observed abnormal exercise response, as no relationship has been observed between VO2max and adiposity in people with T2D,12,17 and people with T2D have demonstrated slower VO2 kinetic responses than obese control participants. 17
Existing preclinical cardiovascular abnormalities and clinical microvascular abnormalities may also play a role in the abnormal exercise performance observed in T2D. For example, a transient imbalance of muscle oxygen delivery relative to muscle VO2 appears to play a role in the observed slowed VO2 kinetics. 75 Demonstrating an additional likely cardiac or autonomic component to slowed VO2 kinetics, people with T2D also have slowed heart rate kinetics as compared to controls. 17 Microvascular complications of T2D (e.g. neuropathy) have also been associated with worse VO2peak. 76 Several of the potential mediators listed above also improve with exercise training (i.e. insulin resistance, triglycerides, inflammation, microvascular complications),40,43,49,59,73,77–79 but the mechanisms of these relationships are uncertain. For example, it is unknown if improvements in microvascular complications mediate improved exercise performance, or if exercise training jointly improves microvascular complications and exercise performance through the effects of separate mediators.
Summary – abnormal exercise performance in T2D
In summary, people with T2D demonstrate significantly impaired peak and submaximal exercise responses that highlight the premature cardiovascular aging effects of T2D. The reduction in peak exercise performance associated with T2D (~20% decrease in VO2peak) may be considered to have a similar effect as approximately 20 years of primary aging (10% decrease in VO2peak per decade of life).
Preclinical cardiovascular abnormalities in T2D
Although the mechanisms causing impairments in peak and submaximal measures of exercise performance in T2D are still under investigation, several factors likely play a role. These factors include preclinical cardiovascular abnormalities of vascular function (endothelial function, arterial stiffness, and vascular blood flow), as well as impairments in cardiac function (coronary artery blood flow, and diastolic cardiac function).12–15,17,18,32–35,53,80,81 We review the data supporting the presence of each preclinical cardiovascular abnormality in T2D, and the potential mediators of each abnormality. These cardiovascular abnormalities, even though preclinical, are markers of the secondary cardiovascular aging burden.
Abnormalities of vascular function
Endothelial dysfunction
T2D has a well-known association with endothelial dysfunction.82–85 We and others have shown that both systemic artery and coronary artery endothelial function are ~50% worse in those with T2D as compared with healthy control participants without diabetes.12,35,61,82,84,85 Although the vascular endothelium has a large number of roles, this review will focus on its role in regulating vascular tone and blood flow, particularly during exercise. In response to the increased vascular shear stress induced by exercise, healthy subjects exhibit an acute nitric-oxide-mediated vasodilatory response to normalize this shear stress.86–89 In people with T2D, endothelial dysfunction results in an impaired vasodilatory response to exercise which is partially reversed by exercise training.90,91
The most likely mediators of endothelial dysfunction in T2D are insulin resistance and hyperglycemia. Insulin resistance likely plays a key role. In multivariate regression analyses, worse insulin resistance was the dominant clinical measure associated with worse endothelial dysfunction in a clinical research study of 81 people with T2D. 92 One rationale for why insulin resistance may lead to endothelial dysfunction is the relationship between sustained hyperinsulinemia (found in T2D and other insulin-resistant populations) and impaired nitric oxide production.9,93–95 However, data are mixed on whether insulin resistance is sufficient to induce endothelial dysfunction in populations without T2D.11,85
Mechanistic observations suggest distinct pathways by which insulin resistance and hyperglycemia cause endothelial dysfunction through impaired nitric oxide bioavailability.96,97 In insulin resistance, nitric oxide synthase activation is impaired owing to defective insulin signaling by the phosphatidylinositol-3 kinase pathway, and excessive free fatty acid oxidation also leads to greater superoxide production.96,98–101 In T2D, superoxide production appears to derive from a combination of hyperglycemia-induced increases in the voltage gradient across the mitochondrial membrane, as well as insulin resistance-mediated pathways.99,102–106 Regardless of its origins, excessive superoxide production leads to reduced nitric oxide bioavailability via decreased endothelium-derived nitric oxide production (e.g. reduced nitric oxide synthase) and increased nitric oxide inactivation. The complex mechanisms by which insulin resistance and hyperglycemia lead to endothelial dysfunction have been reviewed more comprehensively elsewhere.97,107
Obesity (as BMI or waist-to-hip ratio) and the inflammatory marker TNF-α are also related to worse endothelial function. 92 However, specific treatments targeting inflammation alone (e.g. therapeutic studies using a TNF-α inhibitor) have not improved endothelial function or insulin resistance in patients with T2D and/or the metabolic syndrome.108–110
Arterial stiffness
Arterial stiffness occurs due to impaired elasticity of the arterial wall,111–113 and is associated with hypertension, 112 endothelial dysfunction,114,115 and increased mortality. 22 Increased arterial stiffness has been demonstrated in adults and adolescents with T2D but without cardiovascular disease, as compared to people without T2D.18,111,116–118 In addition, arterial stiffness was significantly greater in obese adolescents with T2D, as compared to both obese and normal weight adolescents, suggesting an additional impact of T2D beyond the influence of obesity alone. 18
Potential mediators of arterial stiffness in T2D likely include both structural and functional abnormalities of the vasculature, such as increased cross-linking of the vessel wall by advanced glycation end products, increased calcification of the vessel wall, and increased vascular smooth muscle tone related to endothelial dysfunction.52,112,113,118 Improvements in arterial stiffness were significantly correlated with improvements in insulin resistance in a study by Yokoyama et al., 52 suggesting insulin resistance or other factors related to insulin resistance may be possible mediators. The association between glycemic control and arterial stiffness has been inconsistent between studies.111,116,119 The presence of microvascular complications, such as increased albuminuria, has been associated with increased arterial stiffness as well as reduced peripheral arterial blood flow in two studies.119,120 There may also be a relationship between arterial stiffness and either dyslipidemia or inflammatory markers, based on studies in other populations with an increased risk of atherosclerosis (autoimmune disease); 121 however, such relationships have not yet been observed in populations with T2D.
Peripheral arterial blood flow
A small, but compelling body of evidence demonstrates impairments in peripheral arterial blood flow both at rest, and in response to exercise, in people with clinically uncomplicated T2D as compared to people without T2D.61,117–120 Resting popliteal artery blood flow is lower for people with T2D versus no T2D, even in the absence of peripheral vascular disease (ankle–brachial index [> 0.9]).117–120 In addition to resting flow abnormalities, a lower femoral artery flow response to exercise was observed in people with T2D versus controls matched for age, BMI, and peak exercise performance, and in the absence of peripheral vascular disease. 61
Hyperglycemia, insulin resistance, and endothelial function appear to mediate peripheral arterial blood flow response during exercise.84,92,122,123 Insulin resistance remains an important modifier of arterial blood flow even in the setting of good glycemic control.122,123 Abnormal resting arterial leg blood flow in T2D may also be related to arterial stiffness, as arterial stiffness and leg blood flow are significantly associated in people with T2D.117,120,124 Microvascular complications such as albuminuria are also associated with impairments in peripheral arterial blood flow. 119
Abnormalities of cardiac function
Cardiac abnormalities observable only when induced by stress are among the earliest preclinical findings of cardiac dysfunction in people with T2D. The two preclinical cardiac abnormalities most consistently observed in clinical studies of populations with T2D are impaired coronary artery blood flow and impaired diastolic function induced by stress (i.e. exercise or pharmacologic stress).
Coronary artery blood flow
Resting coronary artery blood flow appears to be similar in people with T2D versus age-similar controls, as measured by resting coronary artery blood flow and resting myocardial perfusion.57,81,125–127 However, several studies demonstrate markers of impaired coronary artery blood flow response during exercise or pharmacologic stress (i.e. impaired coronary blood flow reserve and impaired myocardial perfusion reserve) in people with T2D versus healthy non-diabetic control participants.57,81,125,126 These abnormalities have been found despite the apparent absence of flow-limiting stenosis in the coronary arteries (i.e. no ST-segment depression by electrocardiogram at peak exercise, no regional cardiac perfusion deficits, and with flow measurements performed in non-stenotic coronary arteries).57,81,125,126
Endothelial dysfunction appears to be a key mediator for impaired regulation of coronary artery blood flow. This conclusion is based on the specific abnormalities in coronary artery vasodilation with regard to endothelial function-dependent mechanisms and the preserved responsiveness in coronary artery blood flow to pharmacologic agents that act independently of the endothelium.59,125,126 Investigations of other potential mediators are ongoing. To date, randomized controlled trial data in populations with T2D suggest that interventions that specifically improve glycemic control do not improve coronary artery reserve, 128 but a role for insulin resistance is suggested by a study of thiazolidinedione therapy which improved coronary artery reserve. 129
Diastolic dysfunction
A broad spectrum of diastolic functional abnormalities exists in people with T2D, ranging from the earliest and mildest manifestation of asymptomatic diastolic abnormalities induced by exercise to the most severe manifestation of diastolic heart failure.15,21,53,130,131 Asymptomatic diastolic abnormalities are often only revealed during exercise early on in the course of T2D. 59 Exercise requires faster diastolic filling and increased elastic recoil during a shortened myocardial relaxation phase. 132 The stress of exercise is thus able to ‘unmask’ preclinical impairments in diastolic filling. During exercise, markers of left ventricular filling pressure increased disproportionately in people with T2D as compared to non-diabetic controls.57,132,133 These findings suggest preclinical diastolic dysfunction, as diastolic function appeared normal at rest.57,132,133 In the studies reviewed, the markers of left ventricular filling pressure used to approximate diastolic dysfunction were appropriately conservative, as patients with conditions that could otherwise inflate left ventricular filling pressure were excluded (e.g. valvular disease or systolic function abnormalities). 134
Further evidence for cardiac aging in response to T2D comes from the linear worsening in preclinical diastolic dysfunction (resting E/e′ ratio) when plotted against the number of years after T2D diagnosis, despite accounting for other potential clinical predictors of CHF. 15 Although diastolic function also worsens with age in healthy subjects, the observed rate of increase in E/e′ ratio over time was five times worse in a study of people with T2D, as compared to the rate of E/e′ increase in a separate study of healthy individuals.15,135 Another study demonstrated that left ventricular remodeling abnormalities worsened more rapidly over time in people with diabetes, as compared to people without diabetes. 136 Taken together, these findings suggest that in addition to the ‘premature’ or early cardiovascular aging phenotype observed in T2D, there may also be an ‘accelerated’ rate of cardiovascular aging in people with diabetes. Further epidemiologic study using time-series cohort data are warranted to confirm whether there is effect modification by diabetes on the relationship of age and measures of both cardiac and vascular function. Perhaps related to an accelerated worsening of diastolic dysfunction in T2D, the presence of diastolic dysfunction by tissue Doppler techniques has predicted incident heart failure and all-cause mortality in asymptomatic individuals with T2D and normal systolic function.21,137 Thus, even in the absence of symptoms, preclinical diastolic dysfunction portends increased morbidity and mortality in people with T2D.
The potential mediators of diastolic dysfunction in T2D remain unclear. Current epidemiologic evidence supports an association with microvascular complications of autonomic neuropathy and albuminuria131,138 – whether these factors are simply associated with diastolic dysfunction or may share mediators such as oxidative stress or advanced glycation end products is uncertain.53,131,137,139 Short-term glycemic control (by fasting glucose or HbA1c) has not consistently correlated with markers of diastolic dysfunction.13,14,131,139 The relationship between insulin resistance and diastolic dysfunction has not been fully characterized to date, and insulin-sensitizing treatments have had inconsistent benefits on treating diastolic function.140–142 Mediators of preclinical diastolic dysfunction may also include dysfunction or fibrosis of the subendocardium or myocardium, including subclinical impairments in myocardial perfusion induced by exercise.53,57,131,137,139
Summary – preclinical cardiovascular abnormalities in T2D
The presence of T2D is associated with early development of preclinical cardiovascular abnormalities. The causes of preclinical cardiovascular abnormalities in T2D appear multifactorial and complex. A paucity of interventional trials in this area also limits our conclusions from these data. However, some notable themes have emerged regarding potential mediators of the preclinical cardiovascular abnormalities discussed in this review, including the consistency of insulin resistance as a significant predictor of cardiorespiratory fitness, endothelial dysfunction, and arterial stiffness. Additionally, microvascular complications of T2D have been associated with several preclinical cardiovascular abnormalities (e.g. arterial stiffness, impaired popliteal arterial blood flow, diastolic dysfunction) in patients with otherwise uncomplicated and well-controlled T2D,118,119,131,138 which we speculate may indicate a common shared mechanism (e.g. increased advanced glycation end products or oxidative stress). As described in Sections II and III, exercise training improves most of the preclinical cardiovascular abnormalities, with greater benefits seen early on in the disease process. The pleiotropic benefits of exercise are thus of great interest to combat the premature secondary cardiovascular aging observed in T2D.
Effects of exercise training in T2D
The effects of exercise training on exercise performance and premature cardiovascular abnormalities are summarized in Tables 1 and 2, respectively, and we provide further detail with regard to these effects in the following narrative.
Effects of exercise training on premature cardiovascular abnormalities in T2D
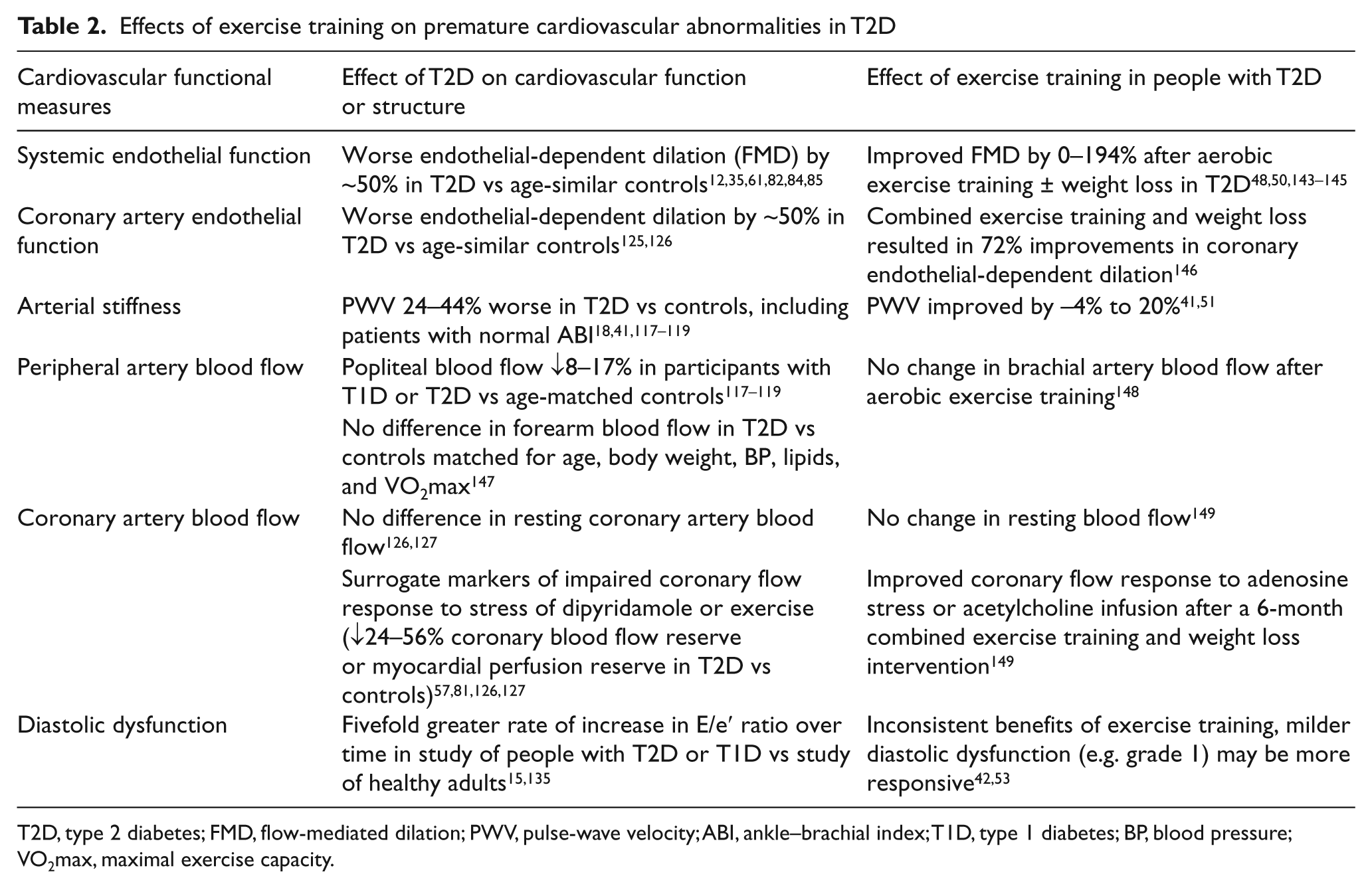
T2D, type 2 diabetes; FMD, flow-mediated dilation; PWV, pulse-wave velocity; ABI, ankle–brachial index; T1D, type 1 diabetes; BP, blood pressure; VO2max, maximal exercise capacity.
Effects of exercise training on exercise performance
In healthy people as well as those with T2D, aerobic exercise training can increase cardiorespiratory fitness across the lifespan. Exercise training has resulted in 5–30% improvements in VO2peak in healthy people,150–152 and 8–41% improvements in VO2peak in people with T2D.32,38–43 In T2D, effective aerobic training regimens were generally performed over 3–6 months, with exercise bouts of ~3 hours per week performed over 3–5 days per week, at an intensity of 60–85% of peak heart rate, peak VO2, and/or age-predicted cardiac reserve.32,38–43 Two aerobic training interventions also included a resistance training component.41,42 Although people with T2D have often responded to a similar aerobic exercise training regimen with a greater relative improvement in VO2peak than their healthy control counterparts, post-training VO2peak remains lower for people with T2D versus healthy controls in most studies. 59
Further study of the effects of exercise training on VO2 kinetics is warranted. Aerobic exercise training has significantly improved VO2 kinetics in one study of adult women with uncomplicated T2D. 32 In addition, significant improvements in cardiac autonomic response have been observed after exercise training. 40
Effects of exercise training on premature cardiovascular abnormalities
Systemic endothelial dysfunction
Effective interventions to improve systemic endothelial dysfunction in T2D include exercise training,48–50 and other therapies.35,62,153–158 In a small randomized controlled trial (RCT) of a 3-month exercise training intervention in people with T2D (n = 38), the intervention group had significantly better end-of-study endothelial function and fewer cardiovascular events as compared to the control group. 48 Other exercise training intervention studies have also demonstrated significant improvements in endothelial function after exercise training in some, but not all studies of people with uncomplicated T2D.49,50,144,145 Of note, exercise training did not improve endothelial dysfunction in patients with T2D complicated by systolic congestive heart failure. 145 This suggests that there may be a window of opportunity for improvement that is lost with the progression of cardiovascular disease heralded by the onset of systolic dysfunction. Thus, systemic endothelial dysfunction frequently improves with exercise training in people with uncomplicated T2D.48–50,144 However, it is unclear to what extent this improvement in endothelial dysfunction is related to concomitant improvements in exercise performance. Further studies are warranted to better characterize the relationship between endothelial dysfunction and impaired peak and submaximal exercise performance in T2D.
Coronary artery endothelial function
To date, we are only aware of one study assessing the response of coronary endothelial function in humans with diabetes – that study demonstrated improvements after 6 months of an intervention of combined exercise training and weight-loss diet that were not initially apparent after 4 weeks of intervention. 149 Exercise training has also improved coronary endothelial function in mice with diabetes, 159 and in humans with coronary artery disease.160,161
Arterial stiffness
Exercise training has significantly improved arterial stiffness in some,51,52 but not all studies of people with T2D. 41 Although Loimaala et al. did not find significant improvements in arterial stiffness from baseline after 12 months of training in an RCT of participants with T2D, arterial stiffness worsened less in the intervention group as compared to the usual care group. 41 The inconsistent effect size may be partly due to variable arterial stiffness measurement techniques between the studies. Given the limitations of the current data available, one can surmise that exercise training may improve arterial stiffness, but the exact relationship between exercise performance and arterial stiffness in people with T2D remains unclear. Although data are limited on the impact of exercise training on arterial stiffness in people with T2D, a study in healthy but sedentary older adults (> 65 years) without diabetes demonstrated that 1 year of exercise training improved arterial elasticity and cardiorespiratory fitness by 19%. 162 Future studies should investigate whether exercise training will have the same effect in people with T2D.
Peripheral artery blood flow
We are only aware of one study examining this outcome in patients with T2D, so this area would benefit from further exploration. In a recent trial, Olsen et al. did not see improved forearm blood flow in patients with T2D after an 8-week aerobic exercise training program of rowing, despite significantly improved post-training forearm glucose disposal rates. 148 Because there are limited data available to assess the effect of exercise training on peripheral blood flow in people with diabetes, the benefits of exercise training and risks of physical inactivity remain uncertain.
Coronary artery blood flow
We are only aware of one study examining this outcome in patients with T2D. Sixt et al. demonstrated an improved coronary artery flow response to either adenosine stress or an acetylcholine infusion after a 6-month combined exercise training and weight loss intervention. 149 Of note, the duration of exercise training was important for the effectiveness of this intervention, as interim measures of coronary artery flow response after 4 weeks of training were unchanged from pre-intervention testing.
Diastolic dysfunction
The question of whether preclinical diastolic dysfunction improves in response to exercise training has not been resolved. One study showed significant improvements in diastolic dysfunction in previously sedentary individuals with T2D after 3 months of thrice-weekly aerobic exercise training for 60 minutes per session at 60–70% VO2peak. 53 Importantly, however, the severity of diastolic dysfunction appeared to predict whether patients would respond to exercise training. People with milder resting diastolic abnormalities (grade 1 diastolic dysfunction) improved uniformly after exercise training, but only one of six subjects with grade 2 diastolic dysfunction improved their diastolic dysfunction with training. 53 Improvements in diastolic dysfunction were also limited to those with milder grades of diastolic dysfunction in a different study population of people with coronary artery disease, only some of whom had T2D. 163 Differing from the above work, a study of 48 men with T2D (diagnosed for ≤ 3 years) did not show any improvement in diastolic dysfunction after 12 months of combined aerobic exercise training (twice weekly at 65–75% VO2peak) and resistance exercise training (twice weekly) with regard to the outcome of end diastolic pressure (E/Ea), despite a significant improvement in VO2peak. 42 Thus, there may be a window of opportunity to slow the progression of diastolic dysfunction in its early preclinical phase that is likely to be lost over time. Of note, the above data also demonstrate that improvements in preclinical diastolic dysfunction are not required for exercise training programs to improve VO2peak.
Exercise training recommendations for people with T2D
Two recent guidelines provide exercise recommendations for people with T2D based on the strong and convincing epidemiologic association of aerobic exercise (or physical activity) with lower cardiovascular disease risk in people with diabetes.164,165 The recent 2010 American Diabetes Association (ADA)/American College of Sports Medicine (ACSM) exercise guidelines recommend 150 minutes of weekly aerobic exercise (i.e. brisk walking or an equivalent activity with intensity ≥ 40% VO2max); and resistance exercise of major muscle groups two to three times weekly on non-consecutive days (ACSM evidence category B, ADA B level recommendation). 164 The ADA/ACSM guidelines also suggest adding unstructured physical activity as much as possible. 164 Before undertaking exercise more intense than brisk walking, sedentary people with T2D should be evaluated by a physician and an exercise trainer. 164 The 2008 Department of Health and Human Service physical activity guidelines for Americans had similar recommendations for adults with T2D – aerobic exercise of moderate intensity for 150 minutes weekly (or vigorous intensity for 75 minutes weekly), and two or more bouts of moderate to high intensity resistance exercise of all major muscle groups. 165
Conclusions: challenges to increase exercise and physical activity for people with T2D
Future challenges in utilizing exercise training (and other treatments) to combat the premature cardiovascular aging effects of T2D are threefold. First, we must better understand the extent to which participant characteristics (age, sex, duration of T2D, extent of complications of T2D) and training program characteristics (intensity, frequency, duration) alter the effectiveness of exercise training programs. Second, we must better understand the mechanisms underlying preclinical cardiovascular abnormalities and impaired exercise performance in T2D in order to identify novel potential treatments. Although we know that routine exercise can improve maximal exercise performance and at least some of the preclinical cardiovascular abnormalities in T2D, people with T2D are less active than their peers without diabetes, and there are barriers to physical activity for people with T2D that can impede regular physical activity, such as greater perceived exertion during submaximal physical activity, and fear of injury.25–28 Thus, the third challenge in utilizing exercise training to combat secondary cardiovascular aging is to identify and overcome barriers to physical activity for people with T2D, so that they are more able to utilize the evidence-based treatment of exercise training.
We already know that interventions that combined behavioral support, diet and physical activity prescriptions can successfully increase physical activity levels for people with T2D (Look AHEAD study) and at risk for T2D.166,167 However, the program components which mediated increased physical activity were combined and thus the individual benefits of each of these program components remain unknown. Given the success of these combined programs, it appears behavioral support and other psychological factors play an important role. However, the abnormal exercise performance and preclinical cardiovascular abnormalities related to T2D may also create physiological barriers to regular physical activity. In order to attenuate the premature cardiovascular aging effects of T2D, we need to identify and overcome the behavioral and physiological barriers to physical activity adherence in T2D.
Footnotes
Dr Huebschmann is supported by the CU Division of General Internal Medicine and the CU Center for Women’s Health Research.
