Abstract
Clinically evident and subclinical peri-procedural bleeding following interventional therapies are associated with adverse cardiovascular outcomes. The risk factors for clinically evident bleeding have been well described. Despite the well-documented association of adverse outcomes for patients with a subclinical peri-procedural hemoglobin drop, the clinical predictors have not yet been defined. We identified 1176 consecutive patients with a subclinical drop in hemoglobin (fall of ≥ 1 g/dl in patients without clinical bleeding) following percutaneous coronary interventions (PCI) and peripheral vascular interventions (PVI). Multivariate logistic regression analysis was performed. A subclinical peri-procedural hemoglobin drop ≥ 1 g/dl was identified in 41% (400/972) of PCI and in 49% (213/435) of PVI. More than one access site predicted a higher risk of a subclinical drop in hemoglobin in both groups. A body mass index ≥ 30 predicted a lower risk of a subclinical drop in hemoglobin in both groups. For PCI, creatinine clearance < 60 ml/min was associated with a higher risk of a subclinical drop in hemoglobin. In conclusion, clinically silent peri-procedural hemoglobin fall ≥ 1 g/dl is common in patients undergoing both coronary and peripheral percutaneous intervention. Predictors identified in our study will need prospective validation.
Introduction
Peri-procedural bleeding has emerged as a common complication of percutaneous coronary interventions (PCI) that negatively impacts a patient’s prognosis.1,2 In addition to an increased mortality risk, other potential deleterious consequences of bleeding, including a hypercoagulable state, interruption of anti-platelet therapy, exacerbation of coronary ischemia and the harmful effect of a blood transfusion.3–6
Predictors of PCI-related peri-procedural bleeding include increasing age, 7 female sex, 8 lower body weight, 9 chronic renal impairment, 10 history of hypertension, 11 baseline anemia, 7 glycoprotein 2b3a inhibitor, 7 low molecular weight heparin, 7 activated clotting time > 250 seconds,12–14 femoral as compared to radial access, 15 intra-aortic balloon pump, 7 larger sheath diameter, 16 delayed sheath removal,15,17 and procedure time greater than 60 minutes. 17 These reports include patients with both clinically evident and clinically silent (subclinical) peri-procedural bleeding events.1,7,8,11,12,18–21 Recent studies have shown that clinically silent peri-procedural blood loss is strongly associated with increased mortality and contrast-induced nephropathy.22,23
The predictors of a clinically silent or subclinical peri-procedural drop in hemoglobin have not been defined. We performed a retrospective analysis of a large cohort of patients undergoing both coronary and peripheral vascular percutaneous interventions to identify predictors of a subclinical drop in hemoglobin.
Methods
We reviewed the records of patients undergoing PCI and peripheral vascular interventions (PVI) from January 2007 to September 2009. The Institutional Review Board approved the study protocol. Consecutive patients, with a documented baseline and post-procedural hemoglobin were identified from the John Ochsner Heart and Vascular Institute’s catheterization laboratory database. We excluded patients with clinically evident bleeding noted in the chart, such as a hematoma, retroperitoneal hemorrhage, gastrointestinal bleeding, hemoptysis, hematuria, those with transfusions and patients who underwent next-day surgery.
Baseline clinical and demographic information as well as details of the percutaneous interventions were extracted from the electronic medical record system. PVI included cerebral, renal, mesenteric, and upper and lower extremity interventions. A history of chronic kidney disease was defined as an estimated glomerular filtration rate of < 60 ml/min [Modification of Diet in Renal Disease study formulae was used: glomerular filtration rate in ml/min per 1.73 m2 = 175 × serum creatinine (exp[–1.154]) × age (exp[–0.203]) × (0.742 if female) × (1.21 if black)].24 All patients received aspirin and Plavix post-procedure.
Our primary outcome was a post-procedural hemoglobin fall ≥ 1 g/dl without a clinically evident reason. We divided the study population into a PCI group (n = 972) and a PVI group (n = 435). Patients who underwent combined coronary and peripheral interventions at the same sitting (n = 52) were excluded for the purpose of this analysis. The entire study population was followed via an electronic medical record and the social security death index was used for the 6-month adverse cardiovascular outcomes (all-cause mortality, myocardial infarction and stroke).
Statistical analysis
Demographic characteristics of the coronary and peripheral groups were analyzed using the Student’s t-test for independent continuous data and the chi-square test or Fisher’s exact test for categorical data. Some patients had more than one record in the database so generalized estimating equations (GEE) regression methods were used to derive the logistic regression models, corrected for multiple records. Separate models were generated for the PCI and PVI groups following the same strategy. We used non-directional stepwise modeling methods to find the best prediction models. Univariate analyses were used to determine whether the baseline clinical, demographic, and procedural variables listed in Tables 1 and 2 were associated with a hemoglobin drop ≥ 1 g/dl, using a liberal criterion of p < 0.20. Those variables associated with the outcome were then used as possible predictors in a non-directional stepwise procedure. All analyses were performed with SAS statistical software (version 9.1; SAS Institute Inc., Cary, NC, USA).
Baseline clinical and demographic characteristics
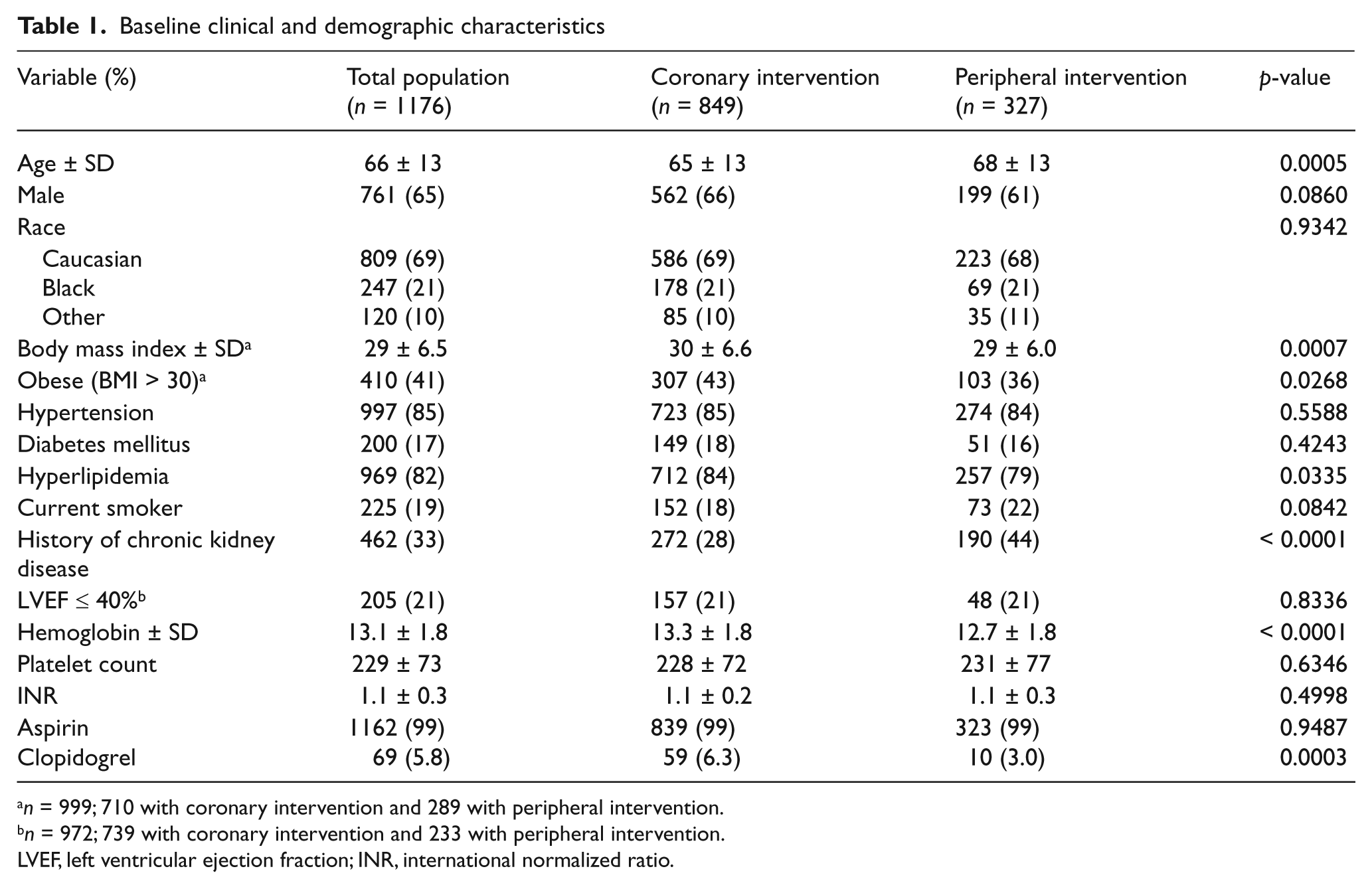
n = 999; 710 with coronary intervention and 289 with peripheral intervention.
n = 972; 739 with coronary intervention and 233 with peripheral intervention.
LVEF, left ventricular ejection fraction; INR, international normalized ratio.
Baseline clinical and demographic characteristics between patients with a hemoglobin drop more than or equal to 1 g/dl versus less than 1 g/dl
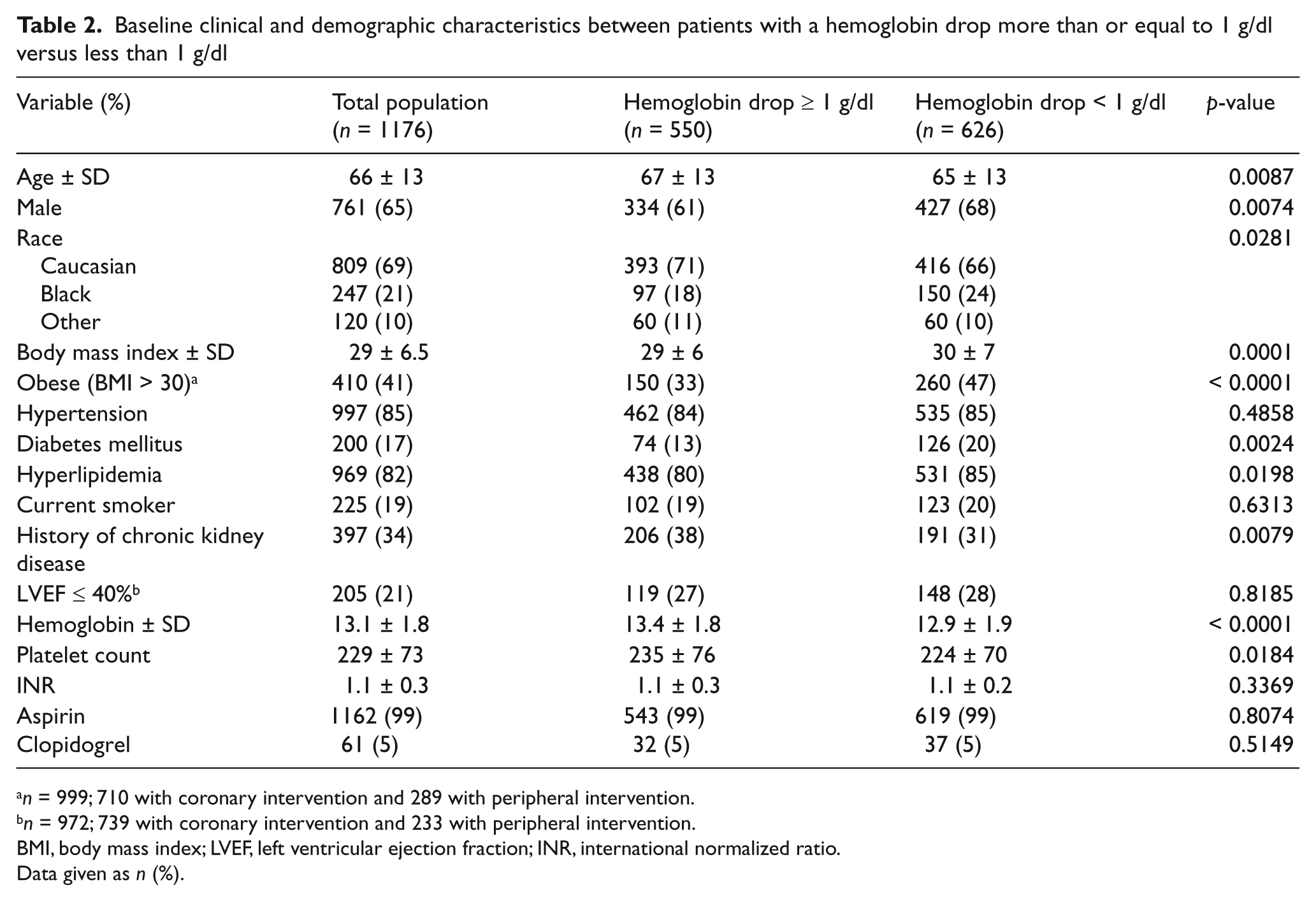
n = 999; 710 with coronary intervention and 289 with peripheral intervention.
n = 972; 739 with coronary intervention and 233 with peripheral intervention.
BMI, body mass index; LVEF, left ventricular ejection fraction; INR, international normalized ratio.
Data given as n (%).
Results
There were 849 patients who underwent 972 PCIs and 327 patients with 435 PVIs. The average time to obtain the post-procedural hemoglobin was 21.6 ± 9.7 hours. A post-procedural hemoglobin fall of ≥ 1 g/dl occurred in 44% (613/1407) of all interventions: 41% (400/972) among the PCI group and 49% (213/435) in the PVI group. Baseline clinical and demographic characteristics of the entire study population, and the PCI and PVI subgroups are shown in Table 1. Compared to PCI patients, the PVI patients were older (68 ± 13 versus 65 ± 13, p = 0.0005), had a lower pre-procedural hemoglobin (12.7 ± 1.8 versus 13.3 ± 1.8, p < 0.0001), a higher prevalence of chronic kidney disease (44% versus 28%, p < 0.0001), and a lower body mass index (29 ± 6.0 versus 30 ± 6.6, p = 0.0007).
Among PCI patients, 35% (340/972) underwent interventions for acute coronary syndromes (ACS) and 65% (632/972) were for other indications. Among PVIs, 23% were cervicocerebral (99/435), 14% (60/435) were visceral and 63% (276/435) were extremity arterial interventions. A comparison between patients with a hemoglobin fall of more than or equal to 1 g/dl versus less than 1 g/dl is shown in Table 2. Compared to PCI, those undergoing PVI more frequently had more than one access site, more catheter exchanges, larger sheath sizes, longer procedural times and more frequently received manual compression to achieve access site hemostasis. Other procedural differences are shown in Table 3. The majority of patients had a hemoglobin fall between 1 and 2 g/dl (84% PCI, 86% PVI). Significant predictors of a subclinical drop in hemoglobin for PCI and PVI procedures by logistic regression analysis are shown in Table 4.
Procedural characteristics
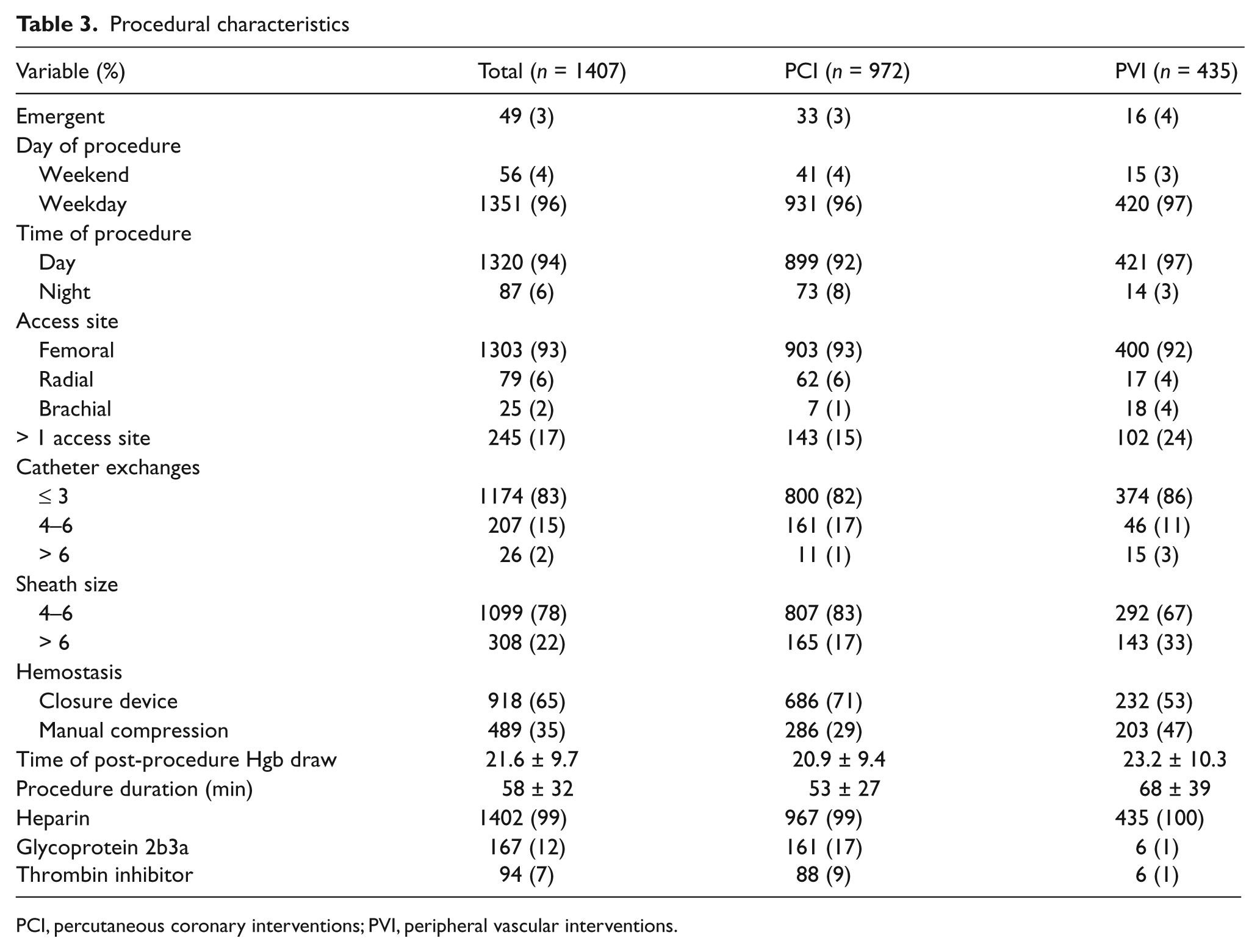
PCI, percutaneous coronary interventions; PVI, peripheral vascular interventions.
Logistic regression analysis for predictors of a hemoglobin fall ≥ 1 g/dl
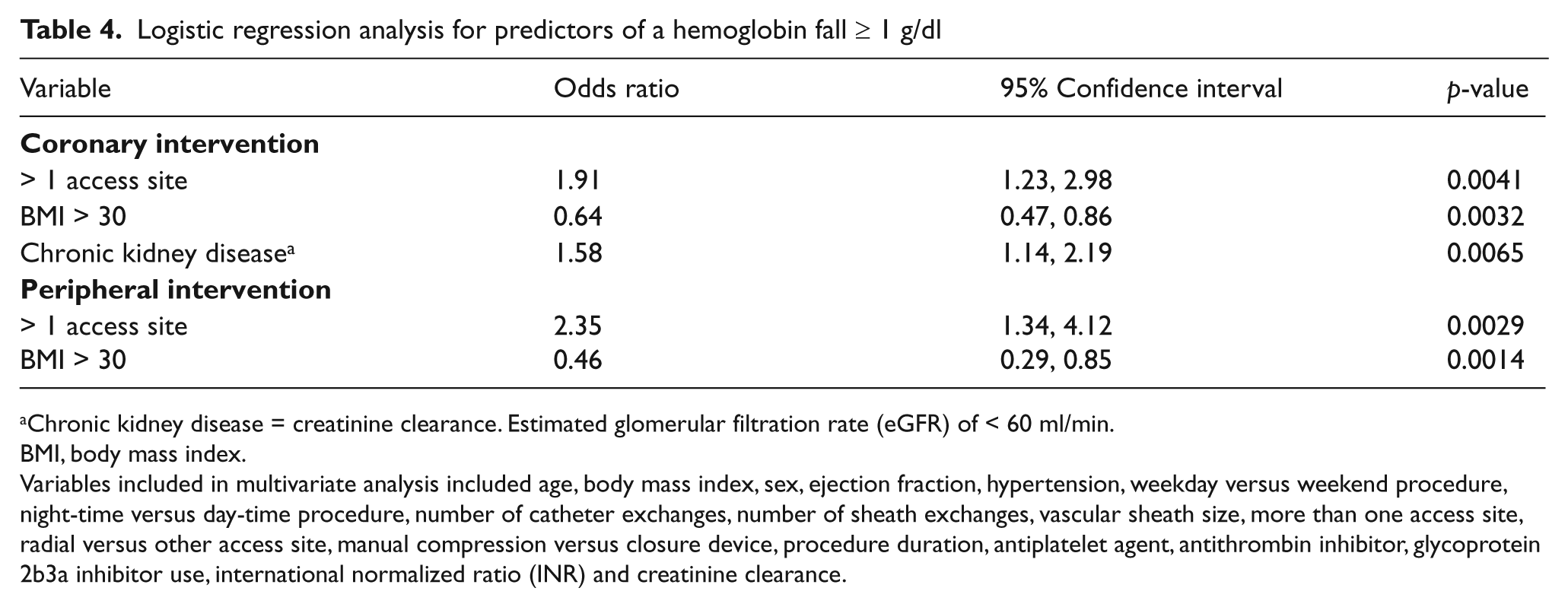
Chronic kidney disease = creatinine clearance. Estimated glomerular filtration rate (eGFR) of < 60 ml/min.
BMI, body mass index.
Variables included in multivariate analysis included age, body mass index, sex, ejection fraction, hypertension, weekday versus weekend procedure, night-time versus day-time procedure, number of catheter exchanges, number of sheath exchanges, vascular sheath size, more than one access site, radial versus other access site, manual compression versus closure device, procedure duration, antiplatelet agent, antithrombin inhibitor, glycoprotein 2b3a inhibitor use, international normalized ratio (INR) and creatinine clearance.
Discussion
Our results corroborate previously published data on subclinical bleeding. 22 In our study, 41% of PCI and 49% of PVI had a peri-procedural hemoglobin fall ≥ 1 g/dl in the absence of clinically evident bleeding. More than one access site predicted a higher risk of a subclinical drop in hemoglobin in both groups. Similar to recently published data on clinical bleeding, a body mass index ≥ 30 kg/m2 predicted a lower risk of a subclinical drop in hemoglobin in both groups. For patients undergoing PCI, chronic kidney disease increased the risk of a subclinical drop in hemoglobin.
Published data demonstrate a U-shaped relationship between serum hemoglobin levels and mortality in patients undergoing PCI, with a 20% increase in cumulative 2-year mortality per 1 g/dl change in hemoglobin. 25 Identification of risk factors for clinically evident peri-procedural blood loss allows preprocedural assessment of the patient’s bleeding risk, which provides an opportunity to modify procedural variables. One example would be choosing a radial artery access in a high-risk patient, rather than a femoral artery access site. 26 With recent data demonstrating an increase in the risk of adverse outcomes in patients undergoing PCI with a peri-procedural drop in hemoglobin,22,23 identification of predictors of a clinically silent procedural drop in hemoglobin will impact daily practice.
The increased prevalence of medical comorbidities in patients undergoing PVI when compared to PCI, highlights that patients presenting for a peripheral arterial intervention are often sicker. PVI, when compared to PCI, had a longer procedural time and required larger sheaths. Both these variables have been previously identified to increase the risk of a clinical bleeding event,16,17 underscoring the risk of peripheral vascular interventions.
In patients undergoing PCI, the two predictors for a subclinical drop in hemoglobin were similar to those identified in prior studies on clinical bleeding.7,10,27 Chronic kidney disease was associated with a higher risk of a subclinical drop in hemoglobin7,10 and an increased body mass index (> 30 kg/m2) was associated with a lower risk of subclinical bleeding. 27 For the PVI subgroup, one or more access sites predicted an increased risk and, similar to the PCI subgroup, a body mass index > 30 predicted a decreased risk of a subclinical drop in hemoglobin. Recently published data on clinical bleeding and body mass index discusses the existence of a bleeding–obesity paradox. 27 The pathophysiology behind this paradox remains unclear. An alteration in circulation coagulation factors has been suggested, with obesity being associated with higher levels of multiple coagulation factors such as factor VII, VIII, fibrinogen and plasminogen activator inhibitor-1. 28
Limitations
Our study is limited by its single center retrospective study design. In addition, we lack data on peri-procedural intravenous fluid administration (hemodilution could possibly affect the results), undocumented access site blood loss and activated clotting time values.
Conclusion
Ours is the first study to address a subclinical peri-procedural hemoglobin drop in patients undergoing PCI and PVI. We found that almost half of the patients undergoing these interventions have a subclinical peri-procedural hemoglobin drop of ≥ 1 g/dl. Predictors identified in our study will need prospective validation.
Footnotes
The research was internally funded by the John Ochsner Heart and Vascular Institute.
None of the authors has any conflict of interest related to this manuscript.
