Abstract
Background
Adherence to continuous positive air pressure (CPAP) in patients with obstructive sleep apnoea (OSA) has remained invariably low over the last decades. Remote monitoring of the nocturnal CPAP treatment, within telemedicine (TM)-based follow-up programs, in these patients has been suggested as a potential tool to improve adherence and release the workload of sleep units. The aim of this study was therefore to assess whether a follow-up program carried out by a Remote Medical Care Centre (RMCC), outside the sleep unit, improves adherence to CPAP in the short and long term in patients with OSA.
Methods
In this pilot protocol, we enrolled 37 patients starting CPAP in our Sleep Centre (SC). After three months of standard care in our SC, patients initiated a six-month remote follow-up carried out by the RMCC, functioning as an intermediary between patients and SC. Monthly reports and indication for face-to-face visits were sent to the SC for six months. After this period patients returned to usual care for one year. Results were compared with those obtained in 38 patients (controls) followed with usual care over the same time range.
Results
Mean nightly use of CPAP increased from 3.2 ± 2.4 h pre-RMCC to 5.2 ± 1.9 h post-RMCC (p < 0.0001). Nights/month of CPAP use improved from 19.8 ± 9.2 to 25.2 ± 2.5 (p < 0.05) and nights/month with CPAP use >4 h from 12.5 ± 10 to 21.03 ± 8.9 (p < 0.05). This improvement remained stable after 12 months from the return of patients to usual care. No significant changes in CPAP use were observed in controls over the time.
Conclusion
A six-month follow-up through a remote facility can significantly improve adherence to CPAP in the short and long term. This pilot study provides a solid base for the design of multicentre randomized trials focusing on new models which are able to increase the long-term efficacy of TM programs.
Introduction
Obstructive sleep apnoea (OSA) is a major public health issue affecting almost one billion people worldwide.1,2 Untreated patients with OSA consume a disproportionate amount of healthcare resources, and healthcare expenses decrease significantly after treatment. 2 Continuous positive airway pressure (CPAP) is the most prevalent and effective treatment for OSA, improving symptoms and reducing the risk of cardiovascular morbidity and mortality in regular users. 3 However, many patients struggle and have difficulty using the device so that adherence to CPAP has remained low in the last decades. 4 Obtaining a constant and sufficient use of CPAP is extremely challenging for sleep Specialists, particularly in the last years when the demand for sleep care has increased exponentially, reducing resources dedicated to each patient.
Telemedicine (TM) refers to the distance exchange of data between patients and health professionals aimed at improving management particularly of those living in remote areas. 5 Although in the field of sleep medicine TM has been studied for long time, its use has increased during the last three years driven by the COVID-19 pandemic. 6 Telemedicine has been implemented mainly for the follow-up of patients on CPAP treatment and telemonitoring systems have been approached as a promising technology to detect problems associated with sub-optimal CPAP usage. 7
Most of the new generation CPAP devices, through a built-in telemonitoring module, can transmit nightly treatment data to cloud-based platforms accessible to health professionals who will be able to timely intervene if problems occur providing quality care to patients at a distance. 8 Although some positive effects of telemonitoring have been already described, data are still inconsistent mainly due to a number of different approaches and protocols.7–10 In fact, it has been pointed out that the technology behind TM is often less of a consideration than strategic design of the TM practice such as organization, logistics and communication. 11
Although TM reduces time and costs related to face-to face visits, telemonitoring requires proactive interventions and therefore it still consumes resources, particularly in terms of staff responsible for checking data, usually within the sleep unit, where the workforce is often insufficient to meet the demand of the large population of patients. Therefore, for a successful implementation, understanding the best way to integrate TM in OSA management is fundamental.11,12 In addition, as the number of patients with access to TM is increasing constantly, telemonitoring suffers from the limitation that it leads to data overload when trying to manage a large population. 13
Recently, traditional centre-to-home models have expanded to include not only the patient and the sleep unit but also an external home care provider. 8 This role is often entrusted to companies specialized in home respiratory devices. In redesigning the management of OSA, the role of these companies could not be limited to technical support and collection of remote data, but should be extended to active monitoring, data integration and interaction with patients, functioning as an intermediary between the patient and the specialist. This system could theoretically reduce the workload of the sleep unit while producing timely assistance to patients.
In Italy, a number of private remote clinical centres for the management of OSA, run by home care companies, are now operating, but the advantage of integrating such services as a supporting tool within the National Health System is still under evaluation.
We therefore performed a pilot study aimed to assess whether a follow-up program carried out by a Remote Medical Care Centre (RMCC) may improve adherence to CPAP treatment in the short and long term in patients with OSA.
Methods
This is prospective mono-centric pilot study conducted in the Sleep Centre (SC), Respiratory Unit, “Policlinico-S. Marco” University Hospital of Catania, Italy. The study has been approved by the local Ethical Committee and signed consent has been obtained for all participants.
Study population
Study group
We enrolled patients >18 years with a diagnosis of OSA (apnea–hypopnea index [AHI] > 10) who initiated treatment with nocturnal CPAP and completed the first month of treatment adaptation/titration. Exclusion criteria were: (1) discontinuation of the treatment before completion of the first month of titration; (2) presence of other respiratory diseases associated with OSA (e.g., chronic obstructive pulmonary disease, chronic respiratory failure); (3) requirement of oxygen supplementation during the night; (4) presence of known psychiatric disorders or history of alcohol or drug addiction; (5) OSA uncontrolled by CPAP therapy; (6) pregnancy; and (7) presence of any behavioral or social difficulty that could hamper understanding or cooperation.
Control group
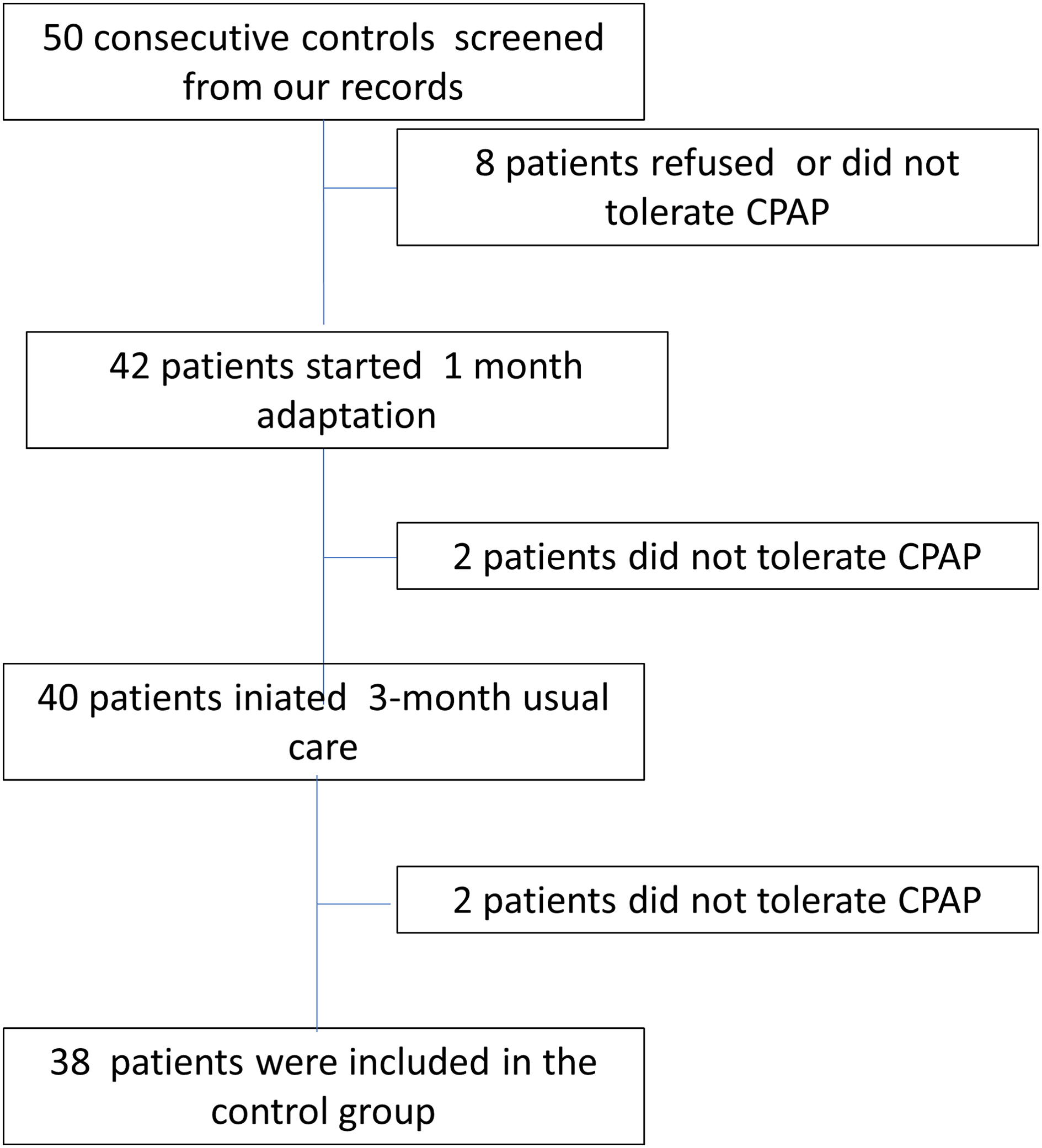
In order to make a comparison between patients undergoing the experimental RMCC follow-up (study group) and patients fully managed with our Usual Care (UC) protocol, we selected a control group by screening patients managed in our SC in the months prior to the study initiation. We screened a total of 49 consecutive patients on CPAP treatment in the age range of the study group. No other matching criteria were needed as patients affected by OSA are usually quite homogeneous for demographic and clinical history. Inclusion and exclusion criteria were identical as for the study population. We finally selected 38 patients to be matched as controls.
Study design
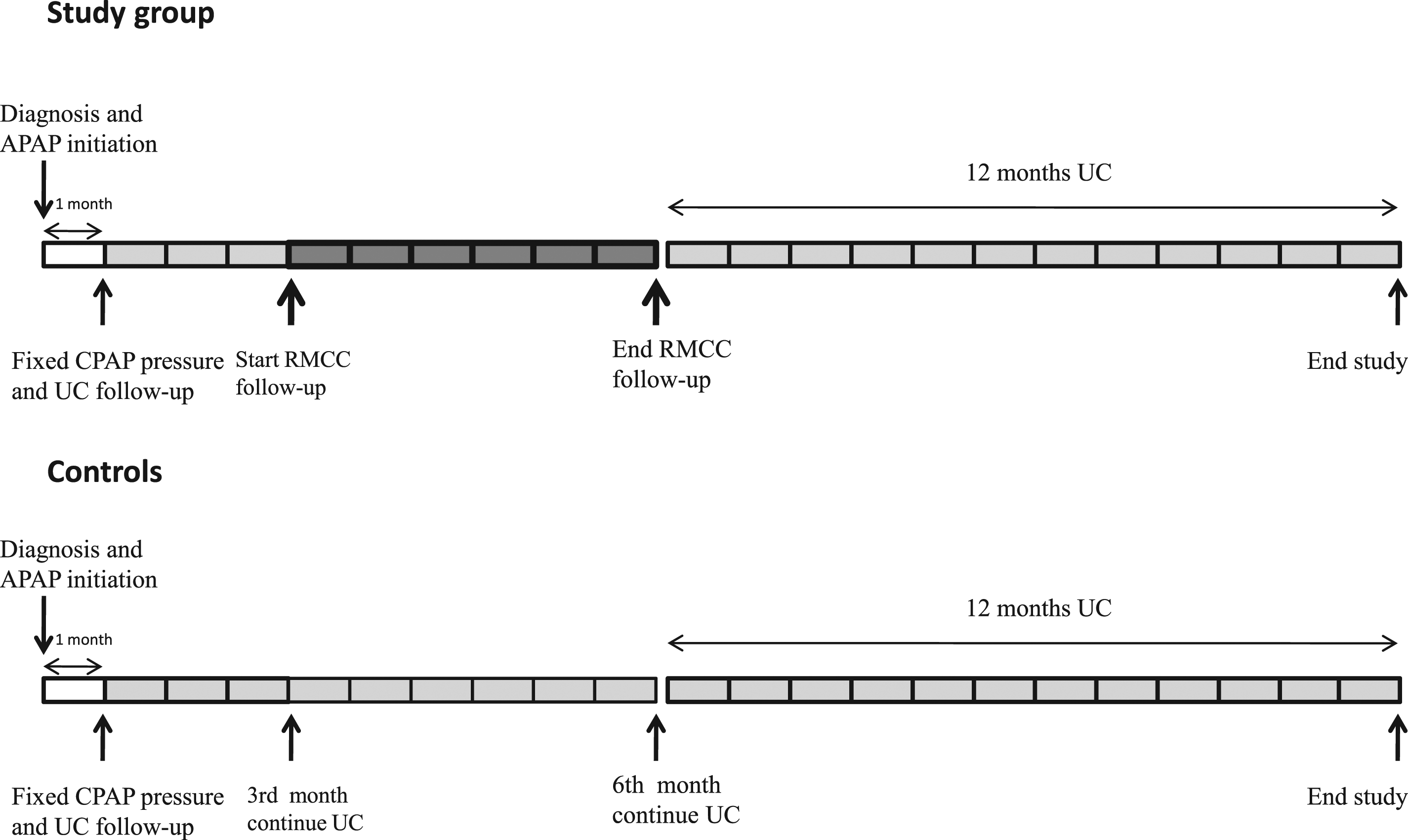
The study protocol consisted of five consecutive phases (Figure 1):
(1) Diagnosis of OSA and CPAP prescription; (2) CPAP titration, according to standard practice; (3) Three months of CPAP treatment with UC follow-up; (4) Six months of CPAP treatment with experimental follow-up by the RMCC; (5) Return to UC consisting in a face-to-face visit in our SC with a review of data on CPAP use.
Study flow chart. Controls were retrospectively analyzed from our records.
Interventions
Diagnosis of OSA and CPAP prescription
Procedures for diagnosis of OSA and CPAP initiation have been described in detail elsewhere. 14 In brief, diagnosis of OSA was obtained through home cardiorespiratory polygraphy using a Vital Night Plus device (Vitalair, Italy). Continuous positive air pressure (ResMed Airsense 10 Autoset) was prescribed to patients with AHI > 15 or with AHI between 10 and 15 in the presence of symptoms or high cardiovascular risk. Treatment data were available by the CPAP smart card or directly by the AirView platform (© ResMed 2019).
First month of adaptation to CPAP and pressure titration
After CPAP was prescribed and the best fitting mask chosen, all indications and instructions were given to patients by our qualified staff. According to our standard practice, the adaptation period lasts one month. During adaptation, we set an auto-CPAP mode and after one month we choose a fixed pressure (mean 95th percentile pressure). Face-to face visits are usually scheduled after the first week and after the first month.
Follow-up with UC for three months
According to our standard protocol, after each patient was adapted to CPAP, he continued his treatment without active control from the SC. Patients were provided with telephone numbers/e-mails and strongly advised to contact us in case of problems. After three months from titration, patients were evaluated in the SC to assess symptoms and underwent a polygraphy during CPAP.
Follow-up through the RMCC for six months
The RMCC, owned by the Vivisol Home Care Services ® (Italy), was based in Milano (Italy). All services were offered to our Institution free of charge for the entire duration of the study. The RMCC was provided with its own staff including nurses, doctors operating at distance and technicians operating at distance or on site if necessary. Remote monitoring of patients was carried out using the ViviCheck® platform (Vivisol, S.r.L Italy). This is a digital platform for the comprehensive management of patients in home treatment, through which healthcare professionals can run a number of functions including enter patient's clinical history, questionnaires, treatment progress in order to acquire information on the patients’ condition and manually add biomedical parameters. The platform allows acquisition and storage of data on CPAP use by each patient nightly and distance adjustment of parameters. Data on CPAP use by each patient were transmitted nightly and included: hours of CPAP use, residual AHI, pressures and leaks. Patients were also provided with an app (Vivisol My Care), containing general advices, instruction manual, scheduled replacements of disposals and RMCC contact.
In brief, according to an established protocol specific for this study the RMCC operated as follows:
First week: checked data on CPAP use transmitted and stored in the ViviCheck® platform. First month: checked data on CPAP use and contacted each patient to assess any kind of problem and the occurrence of sleepiness. Successively: checked monthly CPAP use and contacted the patient until the end of follow-up.
In those patients showing a low adherence during the first week of follow-up, data check and contacts were made weekly until adherence reached the established target (>4 h/night) or improved and remained stable or the patient abandoned the treatment. Interventions from the RMCC were carried out remotely (phone calls or tele-visits) with the exception of technical interventions made in presence when required. The patient was instructed to contact the RMCC for any need. If a problem required an intervention from the specialist, an alert would be sent to the SC in order to schedule a face-to-face visit of the patient. For each patient, a monthly report was sent to the SC containing the following information: (1) summary of adherence to CPAP and efficacy of the treatment; (2) problems detected and interventions made. Direct login into the ViviCheck platform to get details on treatment was allowed.
End of the RMCC follow-up and return to UC
After six months of follow-up in the RMCC patients returned to the UC protocol. No active follow-up was performed. A face to-face visit in the SC, with a review of data related to the use of CPAP was scheduled after one year from RMCC follow-up termination.
Data collection
Data collected included demographics, clinical history, symptoms and severity of sleepiness assessed with the Epworth Sleepiness Scale. During the period of follow-up in the RMCC, data were obtained from each monthly report sent to our SC. Data on CPAP usage included: (1) mean hours/night; (2) mean nights/month; (3) mean of the nights/month using CPAP for >4 h and (4) mean residual AHI.
For the control group treated with UC, all data were retrieved by our clinical records and records of CPAP treatment data stored in the AirView platform. As shown in Figure 2 time points of data collection for controls were the same as for the study group.
Study protocol.
Outcomes
Change in nightly and monthly use of CPAP at the end of the RMCC follow-up compared to the pre-RMCC period and to the control group.
Change in nightly and monthly use of CPAP after one year from cessation of RMCC follow-up and return to UC.
Statistical analysis
Data were analyzed using SPSS 20 software (StataCorp, College Station, Texas, USA). Continuous data are presented as mean ± standard deviation, categorical data as frequency and percentage.
Comparison between RMCC and control groups was performed using an appropriate Student's t test. To compare categorical variables, we used the chi-square test (χ2). P value <0.05 was considered to indicate statistical significance.
Results
Study population
We screened a total of 48 consecutive patients to whom CPAP was prescribed. Of these, seven refused or discontinued the treatment within the first month of titration; 38 patients initiated follow-up with UC for three months, among these, one patient abandoned the treatment on the third month. A total of 37 patients were therefore enrolled in the study and initiated the RMCC follow-up program (Figure 2). Patients were mostly middle-aged, predominantly males (72%), obese (BMI 32.7 kg/m2) with a mean AHI of 45.1 events/h (Table 1). The control group was similar to the study group for age, BMI and severity of the disease (Table 1).
Demographic and clinical data of the population.
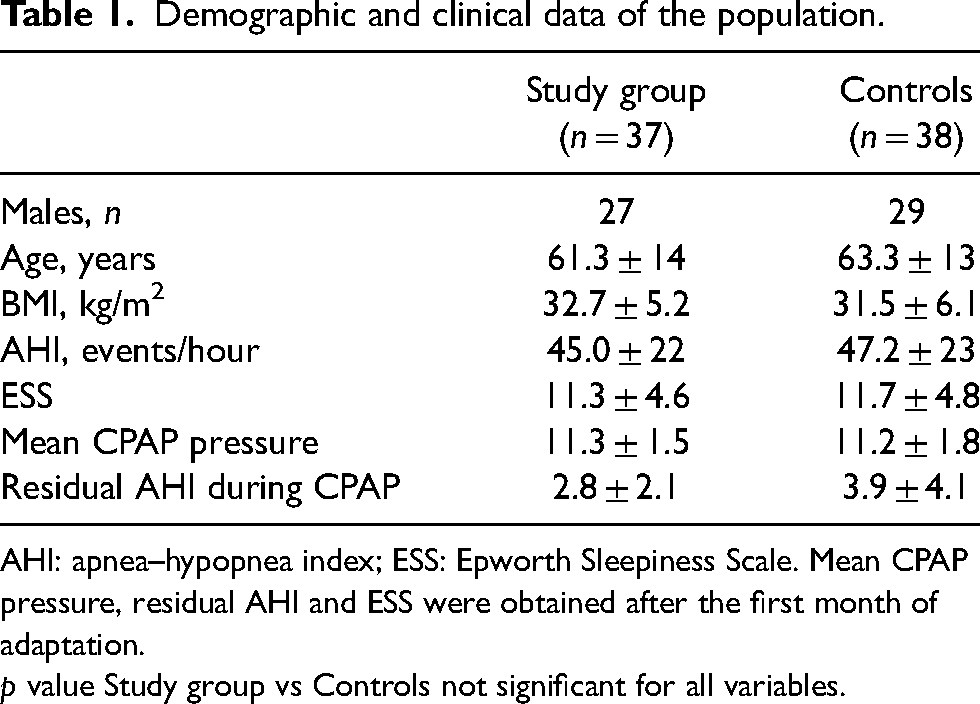
AHI: apnea–hypopnea index; ESS: Epworth Sleepiness Scale. Mean CPAP pressure, residual AHI and ESS were obtained after the first month of adaptation.
p value Study group vs Controls not significant for all variables.
Continuous positive air pressure use during three months of UC
The study group was followed for three months with UC, before initiating RMCC follow-up. In all patients, obstructive events were normalized (AHI < 5/h) by CPAP treatment (Table 1). The monthly adherence to CPAP was similar over the course of three months of UC, on average, the nightly use of CPAP was 3.2 ± 2.4 h, for 19.8 ± 9.2 nights/month and 12.5 ± 10 nights/month with a CPAP use >4 h. In controls, the nightly use of CPAP was 3.1 ± 2.0 h, for 20.8 ± 8.5 nights/month and 13.9 ± 9.3 nights/month with a CPAP use >4 h.
Continuous positive air pressure use during the RMCC follow-up program
After three months of UC, the study group initiated the RMCC follow-up, lasting six months. The term “pre-RMCC” refers to the months before initiation of RMCC follow-up, while the term “post-RMCC” refers to the last month of RMCC follow-up. In the six-month period, we observed a gradual significant improvement in adherence to CPAP, with a plateau reached after five months (Figure 3). The mean nightly use of CPAP significantly increased from 3.2 ± 2.4 h pre-RMCC to 5.2 ± 1.9 h post-RMCC (p < 0.0001). Also nights/month of CPAP use improved from 19.8 ± 9.2 to 25.2 ± 2.5 (p < 0.05), and nights/month with CPAP use >4 h improved from 12.5 ± 10 to 21.03 ± 8.9 (p < 0.05) (Figure 3).
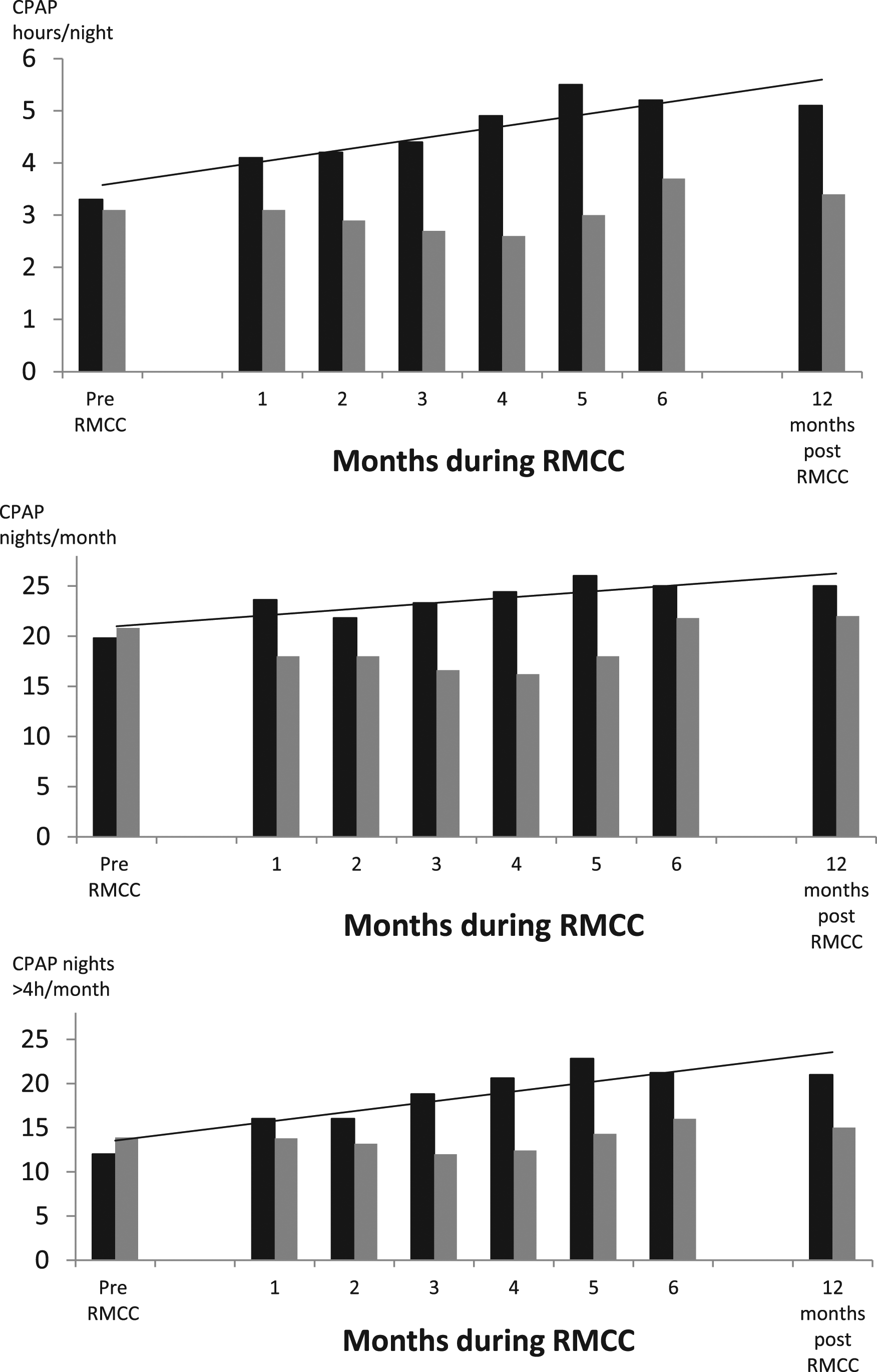
Changes in variables related to CPAP treatment in the study group from the period pre-RMCC to the end of RMCC follow-up and after one year post-RMCC. The differences between pre-RMCC and end-RMCC or 12 months post-RMCC were significant for all variables (see text for p values).
Five patients (13.4%) abandoned CPAP during RMCC follow-up. According to a widely acknowledged definition a patient is considered “adherent” to the treatment when using CPAP for >4 h/night for more than 70% of nights. The number of patients defined adherent, among those who continued the treatment changed from 11 of 37 pre-RMCC (29.7%) to 18 of 32 (56.2%) post-RMCC, but statistical significance was not reached. All patients who abandoned the study were non-adherent at initiation of the study.
The control group was managed only with our standard UC. In this group, we analysed changes in monthly CPAP use from the first three months of UC (corresponding to pre-RMCC) to the ninth month of UC (corresponding to post-RMCC). In the three months of UC, adherence in controls was similar to the study group and variables remained unchanged during the following six months (Figure 3). The mean nightly use of CPAP during the sixth and ninth month was, respectively, 3.09 ± 2.0 h vs 3.7 ± 2.3 h (p = NS), nights/month of CPAP use were 20.8 ± 8.5 vs 21.8 ± 10.3 (p = NS) and nights/month with a CPAP use >4 h were 13.9 ± 9.1 vs 16.8 ± 10 (p = NS) (Figure 3 and 4). Eight out of 38 patients (21.0%) abandoned CPAP between the third and ninth month of UC.
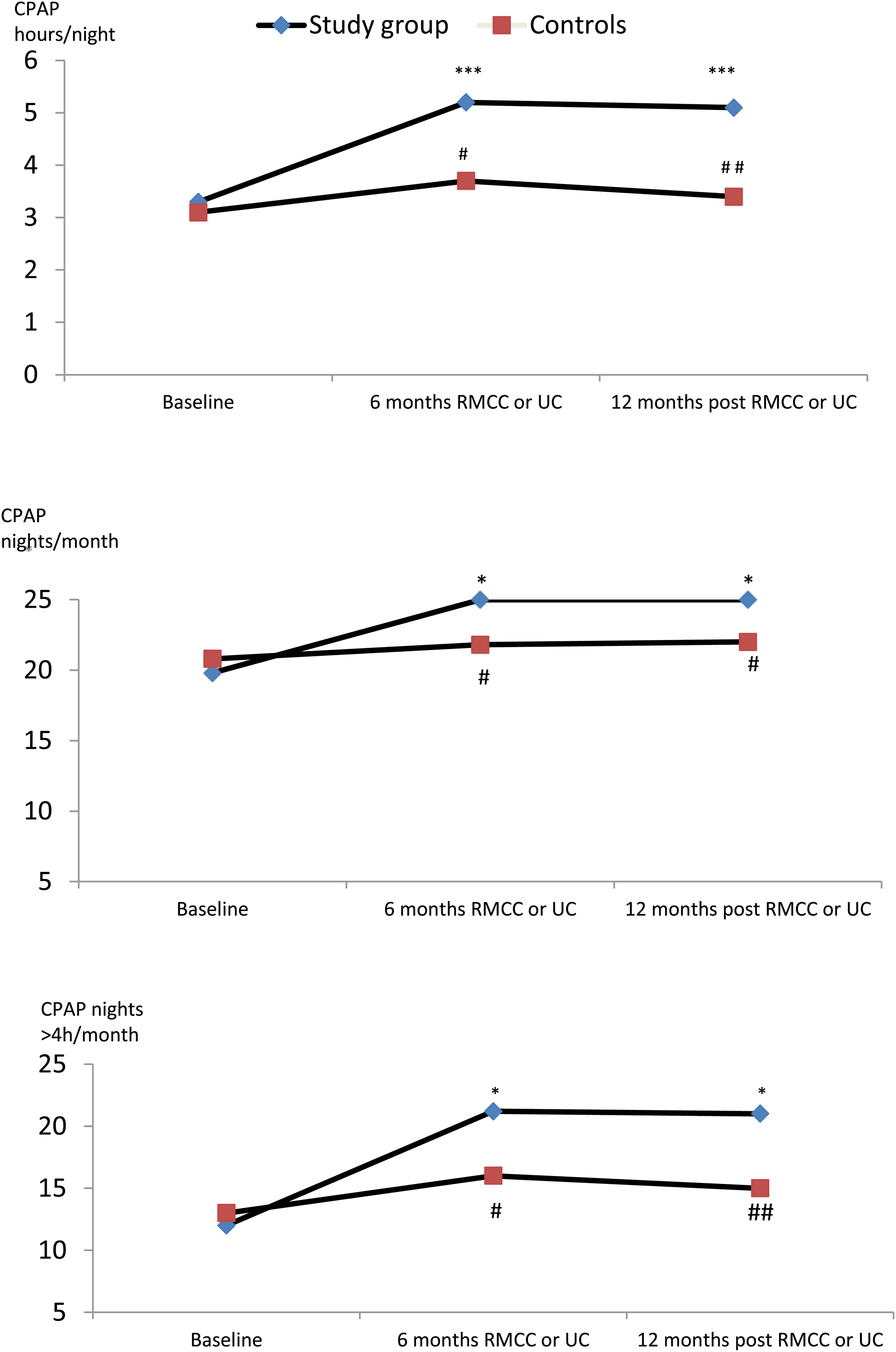
Changes in variables related to CPAP treatment in the study group and in controls. Baseline: three-month UC follow-up for both study group (pre-RMCC) and controls. Sixth month: the end of RMCC follow-up for the study group, continuation of UC for controls. 12th month: the study group was on 12th month after termination of RMCC corresponding to 18th month from initiation of CPAP; controls were on 18th month from initiation of CPAP, always on UC. ***p < 0.0001, * p < 0.05 vs baseline. # p < 0.05, ## p < 0.001 controls vs study group.
Changes in CPAP use in adherent and non-adherent patients
In order to understand which patients may take more advantage from the RMCC program we analyzed data dividing patients in two groups, those who were adherent (n = 15) and those who were non-adherent (n = 22), before initiation of RMCC program, according to the above definition. In patients who were non-adherent, the mean nightly use of CPAP changed from 2.0 ± 0.6 h pre-RMCC to 4.4 ± 3.3 post-RMCC (p < 0.0001). Nights/month of CPAP use increased from 3.9 ± 4.4 to 22.8 ± 6.3 (p < 0.001) and nights/month with a CPAP use >4 h increased from 4.6 ± 3.7 to 18.6 ± 7.4 (p < 0.001) (Table 2). In patients who were adherent pre-RMCC treatment, data remained unchanged post-RMCC and the same occurred in controls (Table 2). As shown in Table 2, no significant changes were observed in the same time window in controls.
CPAP use in non-adherent and adherent patients, over the time, in the study group and controls.
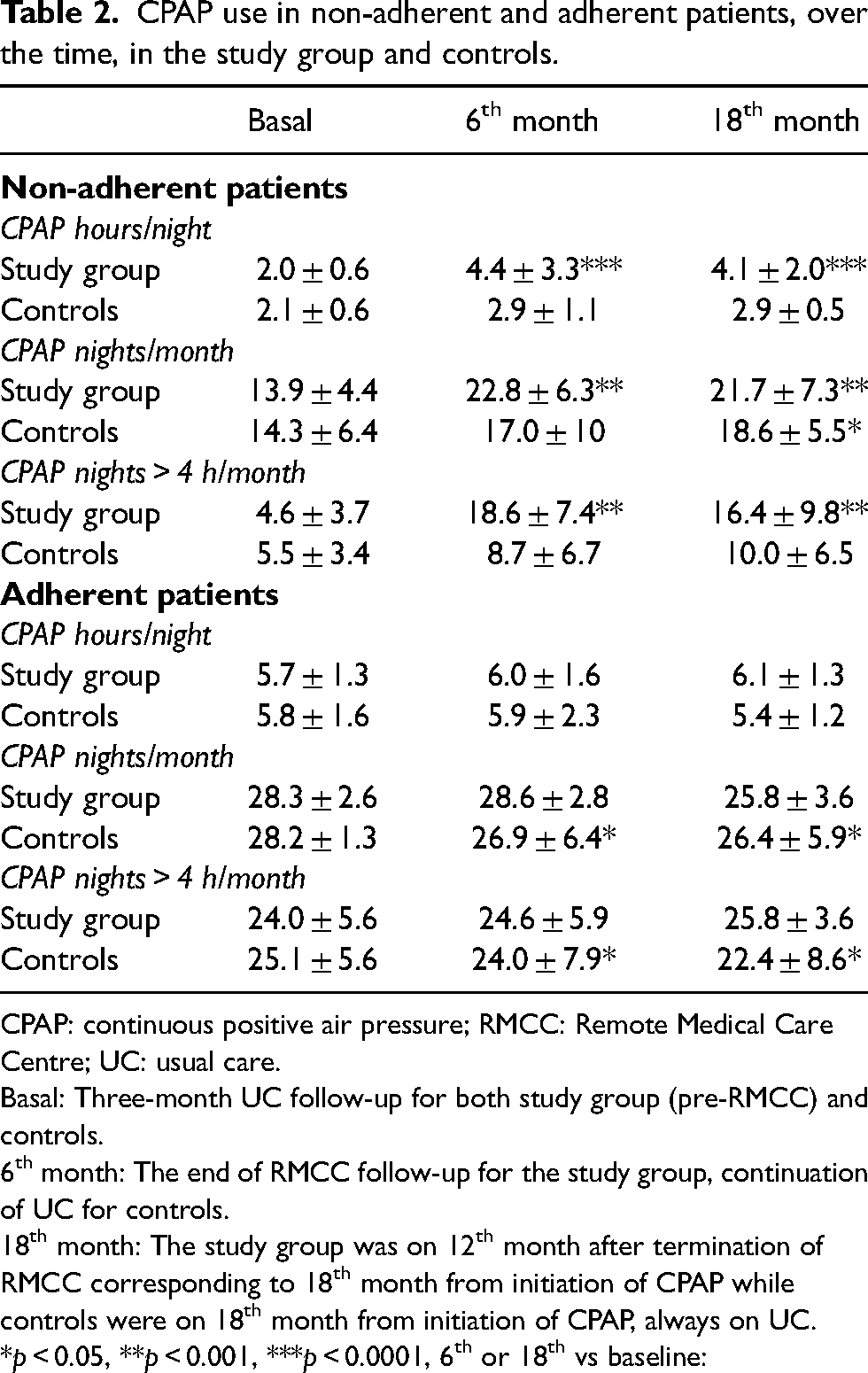
CPAP: continuous positive air pressure; RMCC: Remote Medical Care Centre; UC: usual care.
Basal: Three-month UC follow-up for both study group (pre-RMCC) and controls.
6th month: The end of RMCC follow-up for the study group, continuation of UC for controls.
18th month: The study group was on 12th month after termination of RMCC corresponding to 18th month from initiation of CPAP while controls were on 18th month from initiation of CPAP, always on UC. *p < 0.05, **p < 0.001, ***p < 0.0001, 6th or 18th vs baseline:
Interventions by the RMCC and interaction with the SC
Regular reports were sent monthly from the RMCC to our SC. A total of 23 interventions to solve problems were reported, 16 of which in the first two months of follow-up. These included mainly problems with mask fitting and temperature regulation of the humidifier. However, none of the patients needed face-to-face visit during this period of follow-up.
Effect of the RMCC follow-up on long-term adherence to CPAP
Continuous positive air pressure use was evaluated after 12 months from the end of RMCC follow-up (totally 21 months from initiation). Of 37 patients enrolled 30 (81%) continued to use CPAP after 12 months from the end of the intervention. Over this period, during which patients returned to UC and no active follow-up was performed, CPAP use remained unchanged. Mean nightly use was 5.2 ± 1.9 h post-RMCC and 5.1 ± 2.2 h 12 months later (p = NS), mean nights/month and nights/month with a CPAP use >4 h also remained unchanged, 25.2 ± 2.5 vs 25.6 ± 2.6 (p = NS) and 21.03 ± 8.9 vs 21.6 ± 8.3 (p = NS), respectively (Figures 3 and 4).
In the control group at the same time points, variables related to CPAP adherence remained unchanged and lower compared to the study group (Figure 4).
Discussion
This pilot study indicates that a six-month period of follow-up through a remote medical centre, serving as an intermediary between the patient and the sleep unit, significantly improves adherence to CPAP in patients with OSA, mainly in those who have adaptation difficulties and high risk of quitting treatment. The beneficial effects of remote follow-up persisted unchanged after 12 months. Our follow-up (21 months from initiation of CPAP) is the longest available in literature among clinical studies on TM, while longer periods are available only from big data studies.
This study provides information on two significant aspects of the management of patients with OSA. These include the usefulness of the remote monitoring of CPAP treatment in the short- and long-term, and the feasibility of a follow-up program managed by a home care provider via a remote clinical centre. This model of TM, already in use by a few European countries, has the potential to become an effective way to both reduce the burden on sleep units and improve treatment outcomes.
Efficacy of the TM intervention
As a dose–response relationship exists between nightly use of CPAP and the improvement in health outcomes, increasing the hours of use in patients with OSA is fundamental. 15 A number of randomized controlled trials (RCTs) have been published on TM interventions for CPAP users and included in meta-analyses.10,16,17,19 These latest suggest a trend towards a greater adherence in patients followed with TM compared to standard care.10,16,17 The most recent meta-analysis including 16 RCTs on any kind of TM intervention showed that TM on average improved CPAP usage by 29.2 min/night, with better results obtained by those devices with a built-in software transmitting information wirelessly. 10 A major issue is that these studies vary greatly for the duration and intensity of the monitoring program, telemonitoring systems, intervention techniques and outcomes. It is therefore difficult to compare ours with previous data. Furthermore, most of the TM programs have been tested during the first 1–3 months of treatment, as adherence during this period predicts long-term adherence. 20 It has been shown, although not unanimously, that a three-month TM program, improves compliance compared to UC.9,18,21–23 With early initiation of TM, the first intervention for technical problems occurs in an early phase improving outcomes. 24 However, how long the benefits produced by this relatively short period of telemonitoring last is unknown.
In our study, the duration of the TM program (six months) was longer, and few studies have examined the effect of TM programs lasting more than three months.9,10 In one RCT, a six-month multimodal TM program positively affected CPAP compliance compared to UC (TM 5.28 h per night vs UC 4.75 h). 25 Nilius and coworkers in patients with stroke and OSA found that after six months compliant patients were 57.3% in the TM group vs 27.5% in the standard care group, and the daily CPAP use time was, respectively, 4.4 h and 2.1 h. 26 Similarly, in our study TM improved the nightly use of CPAP by about 2 h. This improvement was gradual, and reached its maximum on the fifth month, indicating that 5–6 months may be an optimal time to positively affect adherence. Accordingly, in one study a short-lasting TM program did not affect compliance after six months. 27
Maintaining adherence to CPAP over the long term should be the ultimate aim of TM, but data are few and inconclusive. An important finding of our study is that our TM program had long-lasting effects as adherence variables were unchanged one year after termination of the intervention.
In one study, initiating TM after the habituation phase and terminating it after achievement of the treatment goal did not affect adherence after one year, compared to standard care. 28 Similarly, Contal and collegues found that a three-month TM program during the habituation phase did not affect daily CPAP use after 12 months. 23 In the HOPS study, the increase in compliance obtained with a three-month TM program was maintained after one year, but statistical significance was not obtained due to the high drop-out rate. 29 Therefore, establishing the proper duration of a TM intervention is an important issue to address for long-lasting results.
Another important aspect is to understand which patients can benefit more from TM, in order to concentrate resources on these patients (patient-centred approach) avoiding misuse. We found that non-adherent patients used CPAP for about 2 h/night. These patients showed the largest benefits, doubling the hours of nightly use during TM, while those using CPAP for about 5 h/night did not significantly modify their habit. Interestingly, a recent RCT showed no effect of TM in patients using CPAP for 5–6 h/night. 30 Therefore, one possible approach could be to extend TM programs over the time only in poorly adherent patients.
It is noteworthy that in our study after the RMCC intervention the number of patients who could be defined fully adherent nearly doubled compared to baseline and the drop-out rate in the study group was lower compared to controls. However, as this was a small sample, statistical significance could not be used to confirm this trend.
Use of a remote medical clinical centre
A novelty of this study is that we entrusted a commercial company, specialized in home care, to take full charge of those aspects of patients’ care not necessitating an expert health professional. These included technical and educational issues, contact with patients and non-specialist medical evaluation. Indeed, an integration of these services in a remote facility releases the pressure on sleep units, allowing a limited number of specialists to serve a broader patients population. 11 While some European countries, where health care is based on private insurance reimbursement, are already implementing this model, in Italy, and in other countries, where costs are covered by the National Health System, these models of care are still under evaluation, so that studies are fundamental to understand benefits and limitations. The promotion of a model of care in which sleep specialists, patients, primary care providers and other members of the health team aims to improve the value of healthcare delivery in a coordinated fashion has been recently emphasized by the Academy of Sleep Medicine. 31 Of course, a number of issues hamper this kind of approach, for example, the integration of different kind of data on adequate platforms. 13 In our study, the use of a platform allowing the integration of clinical and treatment data appears as an important step for the implementation of TM programs.
Study limitations
The main limitation of this study is the cost of such TM model. In fact, we did not evaluated costs, which are however under evaluation for the design of our future trials. Though a number of studies have already reported that TM in OSA is cost-effective when calculating global costs, not only related to procedures and technology but including for example patients’ travel expenses or days off-work.5,11 As investments in telehealth are rapidly expanding, digital health companies are providing outcomes and costs validation in order to clarify this aspect. 11 Another limitation of this study is that, including a small number of patients, we could not evaluate other potential outcomes affected by the TM program. In addition, as this is a single centre study, it could be difficult to generalize results. However, as such kind of research may be expensive, our aim was to understand whether this kind of telecare approach deserves further exploration with larger multicentre studies.
Conclusion
Our study provides evidence that a TM program via a remote clinical centre outside the sleep unit is a valid resource, to improve adherence to CPAP in patients with OSA, particularly in those with adaptation difficulties. If the duration of the program is adequately long (5–6 months), the benefits persist for at least one year after its termination. We believe that this pilot study provides a solid base for the design of RCTs focusing on new models to increase the long-term efficacy of TM programs, that are necessary but still lacking.
Footnotes
Acknowledgments
The authors thank Vivisol Italy, for providing us free services from their remote clinical centre. The authors are particularly grateful to Claudio Garbellini, Teresa Iovino, Nicolò Zanetti, Francesca Brescia and Michela Fumagalli.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics
The study has been approved by the local committee and has been performed in accordance with the ethical standards laid down in the 1964 Declaration of Helsinki and its later amendments. All persons gave their informed consent prior to their inclusion to collect data and to publish.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
