Abstract
Chronic obstructive pulmonary disease is a typical disease among chronic and respiratory diseases. The costs associated with chronic disease care are rising dramatically, and this makes it necessary to redesign care processes, including new tools which allow the health system to be more sustainable without compromising on the quality of the care, compared to that currently provided. One approach may be to use information and communication technologies. In this context, we explored the cost-effectiveness of applying a telemonitoring system to a cohort of chronic obstructive pulmonary disease patients with frequent readmissions (the telEPOC programme).
We conducted an intervention study with a control group. The inclusion criteria used were having chronic obstructive pulmonary disease (forced expiratory volume in the first second/forced vital capacity < 70%) and having been hospitalised for exacerbation at least twice in the last year or three times in the last 2 years. We estimated the costs incurred by patients in each group and calculated the quality-adjusted life years and incremental cost-effectiveness ratio.
Overall, 77 patients were included in the control group and 86 in the intervention group. The raw cost-effectiveness analysis showed that the cost of the telEPOC intervention was significantly lower than that of usual care, while there were no significant differences between the groups in effectiveness.
The incremental cost-effectiveness ratio for the intervention was €175,719.71 per quality-adjusted life-year gained.
There were no differences between the intervention group (telemonitoring) and the control group (standard care) from the cost-effectiveness point of view. On the other hand, the intervention programme (telEPOC) was less expensive than routine clinical practice.
Introduction
Today, it is generally recognised that care for chronic diseases must be focused on the patient (and probably also the caregiver), organised and properly coordinated, ensuring continuity of care between levels and high quality. With these principles in mind, it is important to consider the costs of providing this care, especially given that the burden of chronic diseases is increasing with the steady ageing of our population. In this context, there is a need to develop new processes for integrated care or adapt existing ones in the most prevalent chronic diseases (chronic obstructive pulmonary disease (COPD), heart failure, diabetes etc.), exploiting tools available in other fields, like information and communication technologies (ICTs), in these diseases, seeking to improve resource management and rationalise spending while safeguarding high-quality care. The use of ICTs to monitor patients at distance is defined as telemonitoring.
Telemonitoring as a tool in chronic diseases like cardiovascular diseases and COPD to reduce hospital or emergency admissions has been studied. 1
COPD is a typical chronic disease. In our setting, the aforementioned challenges requiring the assessment of care processes, improvement of the coordination between levels and assurance of the quality of the care are being tackled with various different initiatives.2–4 One of these initiatives involves the application of ICTs with the goal of improving general health outcomes and/or obtaining better results in the assessment of cost-effectiveness. While there are publications that question the effectiveness of such ICTs, particularly telemonitoring, in patients with COPD, 5 it would be fair to say that the existing evidence is inconclusive given the different methodological approaches and the interventions carried out so far. 6 Further, relatively few studies have actually assessed the cost-effectiveness of telemonitoring in COPD.
It is well-known that over 50% of the direct costs in COPD come from hospitalisations for exacerbations. 7 For that reason, our project focused exclusively on COPD patients with frequent admissions. In this research, the hypothesis put forward was that it would be cost-effective to apply a telemonitoring programme to patients with COPD who experience frequent readmissions for exacerbation.
Material and methods
This was a cost-effectiveness study, considering individual costs and outcomes from an intervention study with a control group. We tackled the selection bias common in observational studies by using the propensity score (genetic matching) to achieve a better balance between the two study groups. Between May 2010 and July 2012, patients were recruited from two integrated healthcare organisations (IHOs) (Clinical Trials: NCT02528370).
Participants
During May 2010 and July 2012, COPD patients were recruited in two IHOs in Bizkaia, Spain. The referral hospitals for the patients of these two areas are Galdakao Hospital (Barrualde IHO), with a catchment population of 350,000 (intervention hospital), and Cruces Hospital (Ezkerraldea IHO), with a catchment population of 400,000 (control hospital).
We recruited patients with COPD (forced expiratory volume in the first second (FEV1)/forced vital capacity (FVC) less than 70%) and a history of at least two admissions for exacerbation in the last year or at least three in the last 2 years. The exclusion criteria were the diagnosis of any other related respiratory disease, cancer or terminal illness, and incapacity to complete any of the measurements necessary for the study, as well as refusal to participate or give written informed consent.
The patients were selected from the corresponding database of the participating hospitals. In addition, they were required to have been stable for, at least, 6 weeks before their inclusion in the study (they have to maintain their baseline clinical condition without any exacerbation or change in their baseline medical treatment for COPD). The COPD diagnosis was confirmed at the time of enrolment. All the data compiled were kept confidential and the study was approved by the ethics committees of both hospitals (CEIC Euskadi PI2019038).
Regular medical care
The patients were monitored through check-ups programmed by their primary care doctors and pulmonologists. The monitoring usually includes evaluation of previous exacerbations, the provision of education about COPD (general information about the disease, its treatment focusing on tobacco and inhaled medication, healthy lifestyle habits), and adjustment of the treatment if it was necessary and checking of the inhalation technique. The frequency of these check-ups depended on the severity of their illness. Generally, the medical care by pulmonologists included regular in-person follow-up every 4–6 months.
Intervention
The intervention programme consisted of ‘(1) education about COPD addressed to patients and caregivers (half an hour session to reinforce general knowledge about what COPD is, how to distinguish it and what an exacerbation implies and the importance of inhaled treatment. The inhalation technique was also checked); (2) training in the use of the telemonitoring device and (3) daily phone calls to boost their self-esteem during the first week of telemonitoring (so that they would gain confidence in the management of the smartphone and the use of the application)…’, increasing the intervals between such calls depending on patients’ success in using the telemonitoring system. Once they were using all features of the system properly, calls were only made in the event of an emergency.
The device used for the telemonitoring was a smartphone with specific software that included collection of data on physiological variables (body temperature, respiratory rate, oxygen saturation, heart rate and steps/day) and a clinical questionnaire.
Patients included in the telEPOC program were asked to submit information about their clinical condition on a daily basis. If an alarm was activated the corresponding action plan was triggered. First, a phone call was made by the nurse to the patient or the caregiver. This phone call was used to confirm the alarm. If this action was not considered sufficient to address the alarm, the nurse had two options, depending on the severity of the alarm: waiting for the daily assessment of the patients by respiratory specialists (equivalent to a ward round) or contacting the respiratory specialists directly. At this point, it was the respiratory specialist who decided whether the patient should be referred to their primary care doctor, be assessed by a respiratory specialist of the program, or be sent to the hospital emergency department.
In addition to normal staffing levels, one specialised full-time nurse and one part-time pulmonologist were required to implement to programme. The programme ran from Monday to Friday from 8 a.m. to 3 p.m. From that time to 8 a.m. the following day, plus over weekends and bank holidays the programme was operated through a call centre (staffed by nurses trained in the programme).
Baseline variable assessment
The study protocol, for both groups, included the gathering of data related to the 2 years before their inclusion in the study (number of admissions, lengths of stay, readmissions within 30 days of hospital discharge and number of visits to the emergency department) as well as during the follow-up. We also collected data on sociodemographic characteristics of the patients, COPD-related variables, pulmonary function test, comorbidities assessed by the Charlson comorbidity index 8 and quality of life as assessed by the Short Form 36 Health Survey (SF-36). 9
Cost and effectiveness
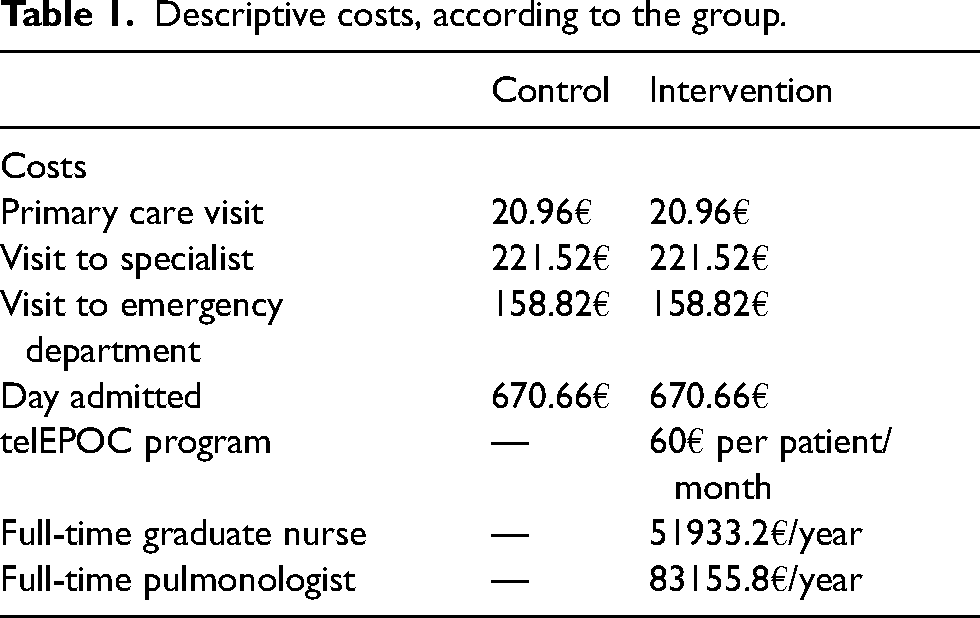
The cost of the individual treatment for each patient during the 2-year follow-up was calculated by summing the cost of the various resources used: costs associated with primary care visits, visits to the specialist, visits to the emergency department and days admitted. The cost of each resource was calculated by multiplying the usage rate by the unit cost The department of analytical accounting provided data on the costs associated with the resources used in 2010–2012 (Table 1). On the other hand, in the intervention group, the cost associated with the telEPOC programme itself was 60 € per person per month, this being adjusted for each patient's length of follow-up, and in addition, we took into account the working time of a full-time graduate nurse and a part-time associate pulmonologist.
Descriptive costs, according to the group.
Effectiveness for each patient was calculated from utility scores of the SF-36 for patients at the three measurement points of the study (baseline, and at 1 and 2 years). SF-6D was used to convert the SF-36 score into a utility measurement, which is a classification for describing health derived from a selection of SF-36 items, composed of six multi-level dimensions and which provides a means for using the SF-36 in economic evaluation by estimating a preference-based single index measurement for health from the data using general population values. 10 First of all, the effectiveness per quality-adjusted life year (QALY) for each period was calculated as the area under the curve through linear interpolation between two consecutive measurement points (between the previous time point and 1 year and between 1 and 2 years). After this, the two ‘effectiveness’ values were summed. In the case of patients who died during the 2-year follow-up, the date of death established the length of the follow-up period and that point was assigned a utility of zero.
Statistical analysis
The design of the study obliged us to ensure that the groups were balanced at baseline and thereby minimise the effect of the possible selection bias. This was carried out through the matching of the cases between the two groups at baseline by applying the ‘genetic matching’ algorithm. Genetic matching is an algorithm that iteratively checks propensity scores, with the aim of adjusting for imbalance or match the groups according to certain important covariates or characteristics.
Once the groups were balanced (adjusted), we performed joint multivariate analysis for the total cost and the QALYs using seemingly uncorrelated regression (SUR). SUR model is a generalisation of a linear regression model that consists of several regression equations, in this case, one for costs and one for QALYs, each having its own dependant variable and different explanatory variables, and error terms are assumed to be correlated across the equations. The group was included as an independent variable and all variables which were statistically significant in the univariate analysis performed after the ‘genetic matching’ were included as covariables. With these models, we calculated the incremental cost-effectiveness ratio (ICER) as the ratio of the coefficients of the group variable in the effectiveness and cost regressions, which are equivalent to the incremental effectiveness (the numerator) and incremental cost (the denominator) respectively. This ICER describes the additional cost for each additional QALY adjusted in line with the factors included in the cost-effectiveness models of the intervention. Further, SUR analysis assesses the uncertainty of the ICER based on the cost-effectiveness plane and the ellipse of the confidence interval, as this incorporates the correlation between the parameters of both regressions (for costs and QALYs) in the analysis using the variance–covariance matrix. The cost-effectiveness plane is a scatter plot on which the vertical axis represents the incremental cost and the horizontal axis the incremental effectiveness. In this plot, the ICER corresponds to the gradient of a line which links any point with the origin. The results of the regressions describe an ellipse which establishes the confidence intervals. When the ellipse crosses the axes, it indicates that the differences are not statistically significant since the confidence interval includes 0 for incremental cost and effectiveness.
All the analyses were performed with R version 4.0.3 (The R Foundation for Statistical Computing).
Results
The cohort of patients diagnosed with COPD included in the study was made up of 163 patients, 86 of whom (52.8%) were in the intervention group. ‘The majority of the participants were men (14% women), and the sample had a mean age of 70 years, FEV1% of 45%, Charlson comorbidity index of 3.5 and BODE index of 3.9’. Table 2 reflects the sociodemographic and clinical characteristics of the patients. In the intervention group 15 patients (17.4%) died and 13 (16.8%) in the control group during the 2 years follow-up.
Descriptive analysis of the sociodemographic and clinical characteristics of the patients, according to the group.
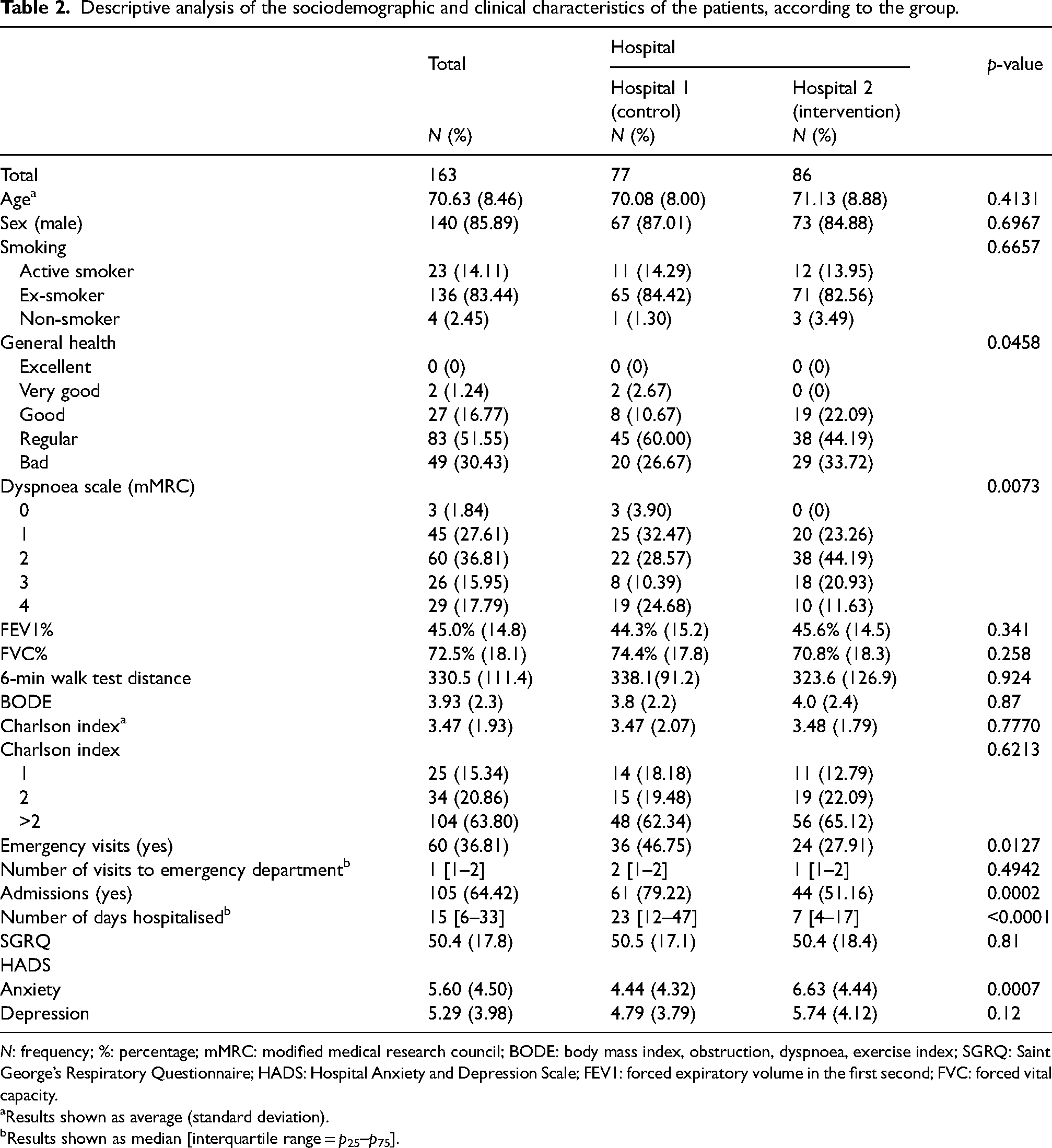
N: frequency; %: percentage; mMRC: modified medical research council; BODE: body mass index, obstruction, dyspnoea, exercise index; SGRQ: Saint George’s Respiratory Questionnaire; HADS: Hospital Anxiety and Depression Scale; FEV1: forced expiratory volume in the first second; FVC: forced vital capacity.
Results shown as average (standard deviation).
Results shown as median [interquartile range = p25–p75].
The raw cost-effectiveness analysis (Table 3) indicates that the cost of the telEPOC intervention was significantly lower than that of usual care whereas the effectiveness was lower in the telEPOC intervention group, albeit not statistically significantly. After applying the genetic matching algorithm, it was observed that the telEPOC intervention prevailed in the group of those younger than 72 years of age and with a Charlson index ≤2 (Table 4). The adjusted analysis showed that the intervention costed less and was less effective, but had a positive high cost per QALY.
Quality of life, QALYs and costs, in relation to the intervention group and control group.
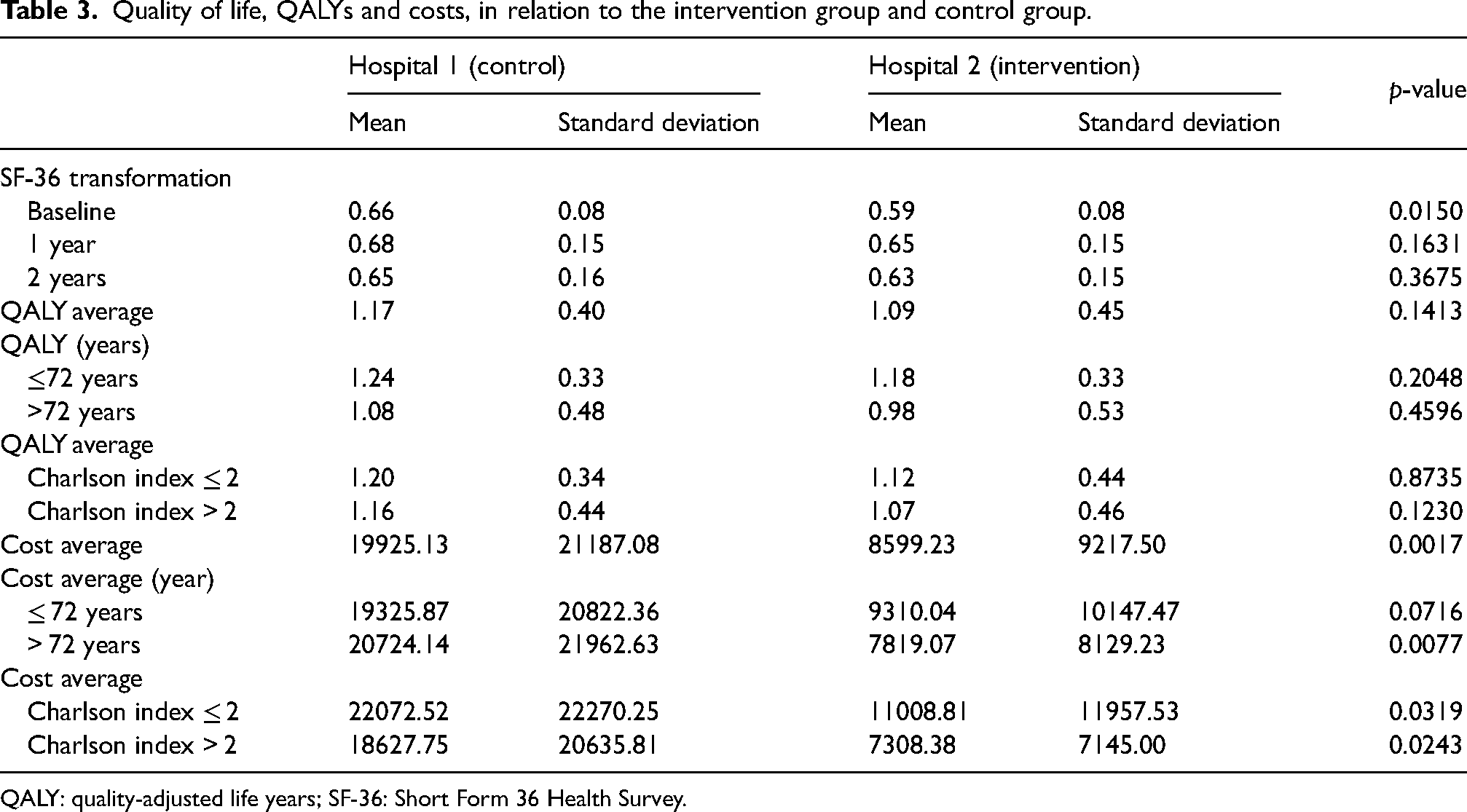
QALY: quality-adjusted life years; SF-36: Short Form 36 Health Survey.
Analysis of cost-effectiveness after genetic matching.
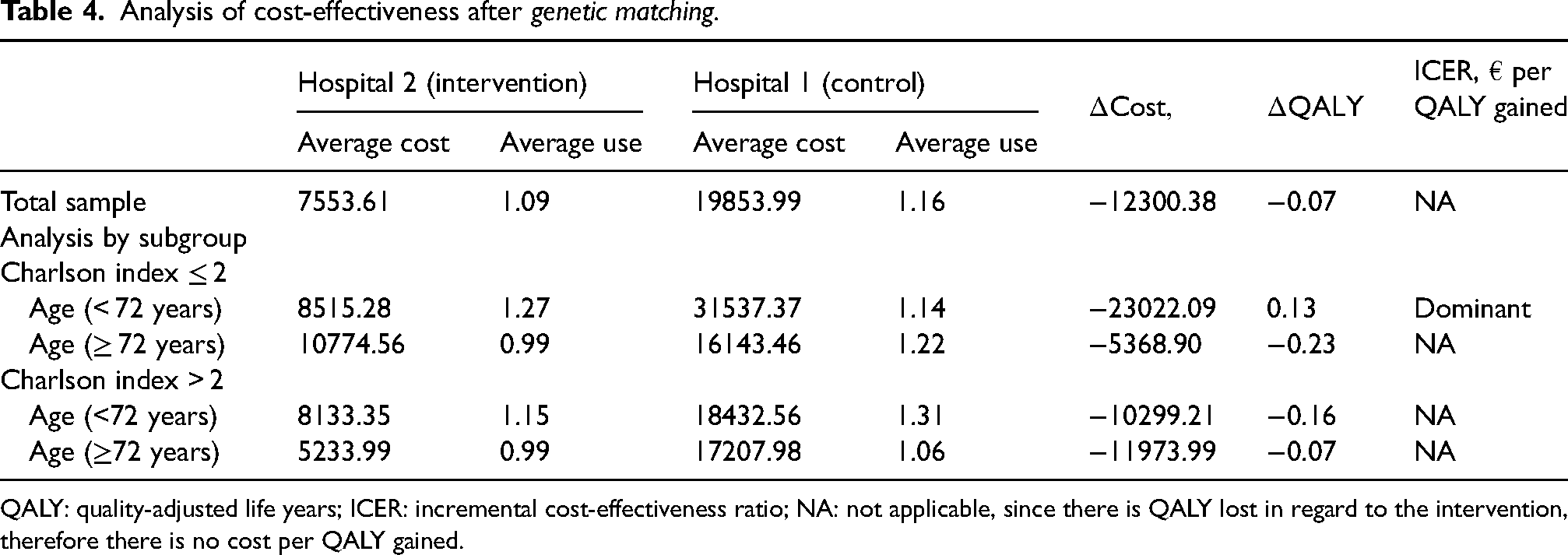
QALY: quality-adjusted life years; ICER: incremental cost-effectiveness ratio; NA: not applicable, since there is QALY lost in regard to the intervention, therefore there is no cost per QALY gained.
The results of the SUR model with interaction terms (Table 5) showed that the costs in the control group were significantly higher than those in the intervention group (telEPOC programme), whereas there were no statistically significant differences between the benefits of the two programmes. Likewise, the cost was higher among women than among men.
Seemingly unrelated regression model with interaction terms for the cost and incremental effectiveness after genetic matching.
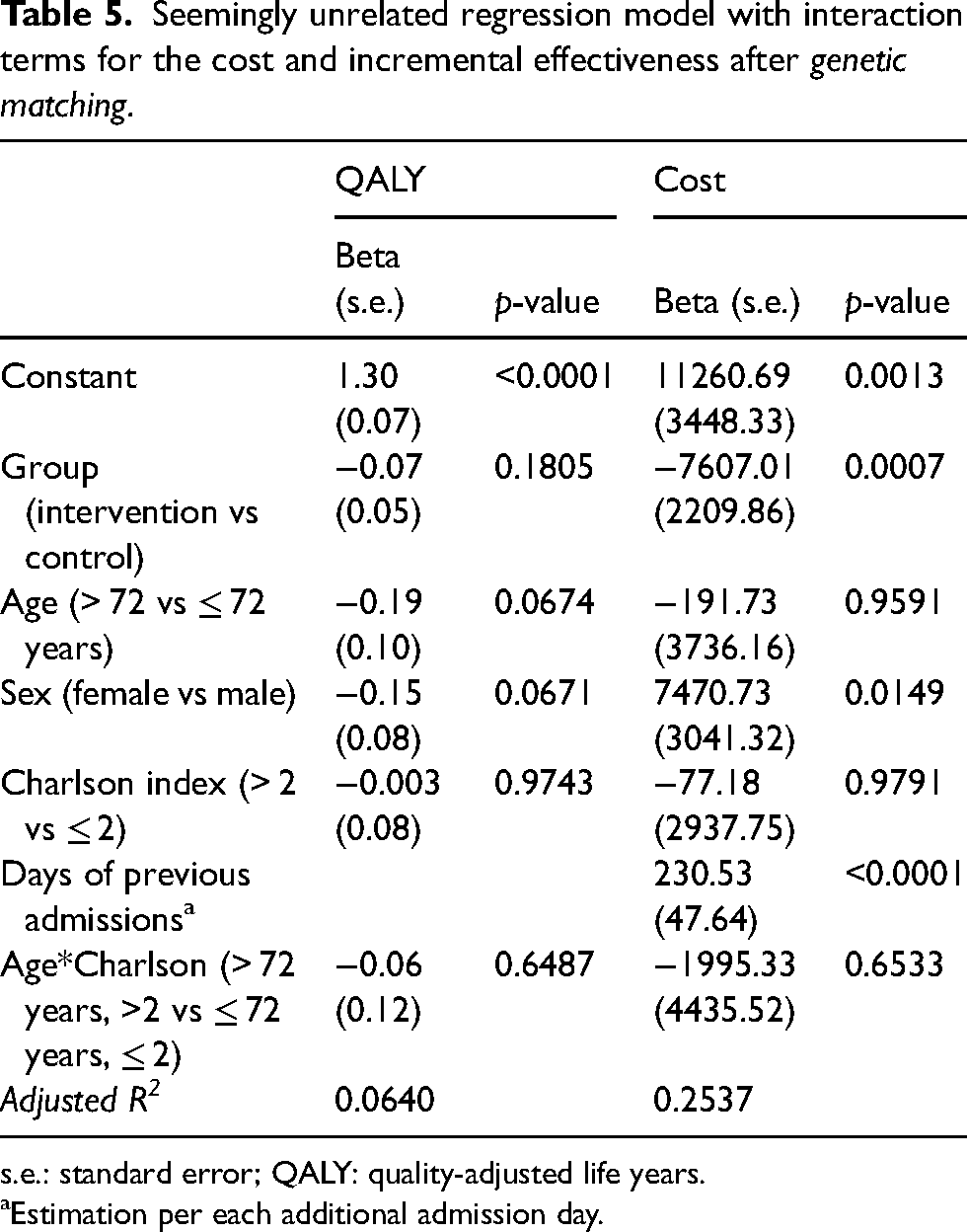
s.e.: standard error; QALY: quality-adjusted life years.
Estimation per each additional admission day.
Figure 1, the cost-effectiveness plane, shows the results graphically. In 3 of the 4 subgroups, the 95% confidence ellipse contains the origin, indicating that neither the incremental cost nor the incremental effectiveness was statistically significant. The telEPOC programme was only significantly related to cost savings in the group of patients younger than 72 years of age and with a Charlson index ≤ 2. On the other hand, the differences in effectiveness were not statistically significant.
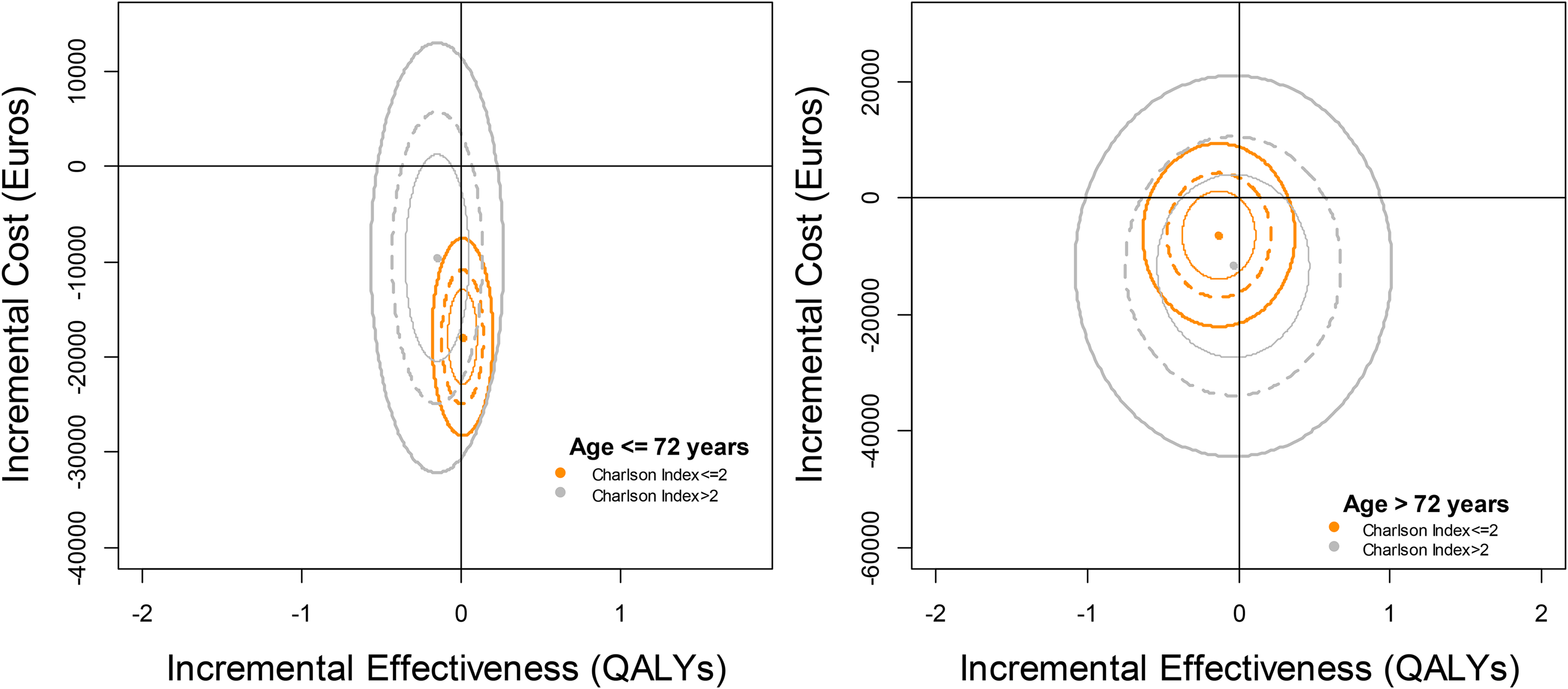
Cost-effectiveness plane including the ellipse of the confidence interval at 95% and considering the interactions between age, Charlson index and the telEPOC intervention.
Discussion
The study has two main findings: (1) there were no differences between the intervention group (telemonitoring) and the control group (usual care) from the cost-effectiveness point of view; and (2) the cost of the intervention programme (telEPOC) was lower than that of standard clinical practice.
A Scottish clinical trial, which included 256 COPD patients (with one hospital admission for exacerbation the previous year as an inclusion criterion) concluded that telemonitoring did not reduce the frequency of admission during the follow-up year,5,11 unlike findings concerning our programme, which was observed to reduce admissions. 12 In the Scottish trial, the cost of telemonitoring (£11,793.12 per patient) was higher than that of usual care, though the difference (£2065 per patient) was not statistically significant. There was also no difference between the two arms in QALYs. 11
Another study, carried out in Denmark, also concluded that a telemonitoring intervention in COPD was not cost-effective. 13 The cost per patient in the intervention group was €8780 during the year of the study, €1540 more than the costs in the control group. The differences in the costs are quite striking, in that they were €836 higher per patient in the intervention group excluding the intervention itself. These differences lay in social-health care provision, with additional costs of €474 for help and care at home and €203 for community nursing in the intervention group, when in theory, these costs should have been similar in the two groups if we really want to be accurate when talking about usual care and thereby limit biases in the analysis. Nonetheless, when subgroups were analysed, they found that the likelihood that the interventions could become cost-effective increased in the GOLD 3 subgroup of COPD patients (due to savings related to admissions). 14 It should be pointed out, however, that in the GOLD 4 subgroup, this premise was not fulfilled, these patients being found to be the group which incurred the highest admission-related expenses and spending also being higher on the intervention group than the control group, which raises serious doubts about the value of the findings in GOLD 3 patients.
In a clinical trial carried out in Northern Ireland, with 110 COPD patients in two arms, the conclusion was that the 6-month intervention managed to improve quality of life and anxiety but was not cost-effective, the ICER being £203,900 per QALY. 15
All the aforementioned three trials11,13,15 were local studies of COPD patients with an FEV1 between 40% and 48%, similar to the values in our study (45%), and the mean age was around 70 years, also similar to that of our patients. On the other hand, 50% were women, markedly different from the sex ratio in the cohorts of our study (14%). Regarding the history of admission, patients had ∼2.5 hospitalisations for COPD in the previous year in the Scottish study and two exacerbations the previous year in the Danish study, while they had just one in the Irish study. In our cohorts, the mean number of hospitalisations was above three but considered the 2 years before the study, to be sure that participants were patients with repeated severe exacerbations. The follow-up was 1 year in the other trials, except the Irish one which only followed patients up for 6 months, while we report 2 years of follow-up. That is, there are marked differences between our study and the previous trials in the nature of the cohorts studied and the research design used. Further, the level of care provided to our patients differed from that offered in the regions and countries in the other studies. We should also note that the costs associated with the intervention were higher than that of usual care in all the other trials, unlike in our study.
Limitations
We understand that the main limitation of our study is that it is a quasi-experimental study, in that when we devised its design, we opted to randomise to hospitals with a similar practice rather than randomising patients because, with that approach, the intervention might have modified usual clinical practice. In the light of the results, the conclusion is clear: the intervention was not cost-effective. Nonetheless, it was associated with in a fall in the costs.
On the other hand, it is worth mentioning that the patients included in the telEPOC programme were patients with marked deterioration (severe obstruction, BODE 4, and 6-min walk test distance of less than 350 m), which reduces the chances of improving their quality of life (HRQoL), and the measurement of HRQoL is an integral part of cost-effectiveness analysis. Further, it remains to be seen whether a generic questionnaire is able to accurately reflect changes in the HRQoL of such patients. In fact, in a previously mentioned study, the HRQoL as measured by the St George’s Respiratory Questionnaire (SGRQ) improved clinically and statistically, whereas no statistically significant differences were shown using the EQ-5D. 15 For that reason, some authors proposed the use of the generic and specific questionnaires simultaneously to capture all aspects of HRQoL. 16 In our study, using the SGRQ, the intervention group improved their score by 2.5 points whereas the control group showed a deterioration of 1.15 points (differences at the limit of statistical and clinical significance) at the end of the 2-year follow-up. 12 Further, other variables like the 6-min walk test distance, functional limitations in daily life as assessed by the London Chest Activity of Daily Living Scale and the Hospital Anxiety and Depression Scale score remained stable in the intervention group, whereas significant clinical and statistical deterioration was observed in the control group. 12 With all these data, we remain unsure about the capacity of the tools used to assess the cost effectiveness to capture potentially subtle changes in people with advanced chronic diseases.
Another limitation is the small number of women included in the programme (15%), which would hinder the generalisation of the results.
It could be argued that our data reflect the reality of 2010. Nonetheless, by analysing changes in costs in various areas, and in particular, in terms of staffing, the increases since 2014 have been 7.8% and 6.7%, respectively for nurses and medical staff in our health system, this translating to a relatively small increase in overall costs (around 1%). On the other hand, once telEPOC had become an organisation-wide programme, the cost per patient was €30/patient/month, down from €60/patient/month at the start of the programme.
Among the strengths of the study, it should be mentioned that it is a 2-year follow-up study. At best, this type of study has been based on a maximum of 1 year of follow-up, and our longer follow-up has allowed us to characterise the patients and their outcomes more comprehensively.
Conclusions
Our study demonstrates that in a carefully selected group of people with COPD it is possible to reduce costs using a properly structured telemonitoring programme (telEPOC). We have not, however, been able to prove that the programme is cost-effective, likely due to the clinical characteristics of the patients studied (advanced COPD, as required by the inclusion criteria), though it is also possible the tools used to assess outcomes are not sufficiently sensitive to detect relevant differences in people with these characteristics.
Future research: new technologies such as machine learning combined with additional data sources should be incorporated into this field for helping in the decision making and cost.
Primary care participants
Abando N, Abasolo A, Abaunza KA, Acaiturri R, Acha A, Albizu A, Aldazabal LM, Antón Diez-Caballero B, Antón Rollán AC, Arraibi I, Balentziaga JL, Bilbao Aguirre F, Bolinaga A, Calvo B, Catania B, Chirapozu JM, Dávila F, De Castro Pelegrin A, De los Bueis G, Delgado Casulleras JM, Durana MA, Echeguibel M, Etxebarri A, Ezquerra JM, Fernandez Martinez A, Gabiña VE, Gago Gómez F, García Hernando MJ, Geijo G, Gerediaga E, Gómez Fuentes L, González Echave MA, González Martin G, González Rodríguez M, González Santiesteban R, González Silvares E, Gorostiza FJ, Gorroño JI, Intxaurza N, Intxausti I, Izaguirre AI, Jaka Z, Jáuregui L, Laboa I, Larrauri M, Lavín A, Lazcano L, López Fernández J, Llama Guerra AM, López Palacios VM, March JF, Marijuan L, Martín Beñaran JR, Martínez de la Cuadra M, Martinez Ortega C, Marzo Jurico E, Matthies N, Mendieta M, Menéndez Gaztan I, Millan MA, Minondo L, Molano MS, Odriozola M, Ortega García MB, Ortego Fernández-Retana A, Ortiz Cobo N, Oteiza L, Paino F, Peña Entrena C, Perez Urquijo AI, Pérez Rodrigo A, Prieto Casado J, Román Cabanillas A, Romaña ML, Rubiera A, Sagarminaga I, Salaverri G, Unzurrunzaga M, Uribarri MV, Urigoitia PL, Urionabarrenetxea I, Zubizarreta E.
telEPOC group
Hospital Galdakao-Usansolo: Tovar MD, Díez R, Aizpiri S, Sainz de Aja L, Gorordo M, Basualdo LV, Aramburu A, Pascual S, Uranga A, Dorado S, Gorordo I, Ballaz A. Egurrola M, García-Loizaga A, España PP, Capelastegui A, Arrizubieta I, Gallardo Rebollal MS, Pulido E.
Hospital Cruces: Tavernero E, Sobradillo P, Insausti M.
Hospital Txagorritxu: Bravo D.
Hospital Basurto: Sanchez-Juez R.
Consejo Sanitario: Roca R.
Osarean: Llano J, Resino S.
Osakidetza: Lopez-Arbeloa P, Rabanal S.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Fondo de Investigación Sanitaria (PI10/01716); Department of Health of the Basque Government (2010111011) and Kronikgune (KRONIK11/020), and the thematic networks – REDISSEC (Red de Investigación en Servicios de Salud en Enfermedades Crónicas) – of the Instituto de Salud Carlos III.
