Abstract
Introduction
A novel randomized controlled trial tested the efficacy of a technology-assisted case management program in a low income, rural population previously where nurses titrated medication over the phone instead of in a clinic. The primary analysis showed significant improvement in glycemic control at 6 months post-randomization decreasing hemoglobin A1c by 1%. This study aimed to test if the intervention was also effective at decreasing blood pressure without compromising quality of life.
Methods
A total of 113 adults with poorly controlled diabetes (hemoglobin A1c ≥ 8%) were randomly assigned to the technology-assisted case management intervention or usual care. Participants received a 2-in-1 telehealth system to monitor glycemic and blood pressure control, which was uploaded daily to a central server. A nurse case manager was trained to titrate medication under physician supervision every 2 weeks based on the readings. Outcomes were blood pressure and quality of life (12-item Short-Form Health Survey) at 6 months. Baseline adjusted mixed models using a random intercept were used to evaluate change at 6 months for the technology-assisted case management intervention group compared to usual care.
Results
There were no statistically significant differences in systolic blood pressure, physical component of quality of life, or mental component of quality of life between the technology-assisted case management and control group. However, there was a significant change in diastolic blood pressure over time, with the technology-assisted case management group decreasing at 6 months (p = .05), whereas the control group remained stable.
Conclusions
Technology-assisted case management by a nurse with medication titration under physician supervision was efficacious in improving diastolic blood pressure without compromising quality of life in low-income rural adults with diabetes.
Keywords
Introduction
There are more than 34 million individuals in the United States with diabetes. 1 Low-income adults and adults living in poverty have higher odds of having diabetes compared to higher-income adults.2–4 Diabetes is about 17% more prevalent among rural-dwelling adults compared to adults residing in urban areas. 5 Individuals with uncontrolled blood glucose levels are at increased risk of suffering from diabetes-related complications such as neuropathy, kidney disease, heart disease, blindness, and amputations.6,7 Low income is also associated with diabetes related complications and low access to health care.3,8 Diabetes is not only a condition associated with high morbidity and mortality, it is a costly disease, with adults with diabetes having more than two times the healthcare costs of adults without diabetes. Diabetes related costs in the United States have risen from $245 billion in 2012 to $327 billion in 2017, a 26% increase in a five-year period.9,10
It is well known that rural populations experience lower access to health care along the dimensions of affordability, proximity, and quality compared to individuals living in urban areas. 11 Some characteristics putting rural residents at higher risk of death include increased travel distances to specialty or emergency care, higher rates of poverty, and less access to health care. 12 Barriers to accessing health care results in unmet health care need, including a lack of preventive screening services and treatment of illnesses. 11
Chronic disease case management with home telemonitoring is a viable strategy for delivery of care in low-resource areas,13,14 and has been found to be associated with reductions in hospital admissions and emergency department visits among patients with chronic disease. 15 Telehealth strategies may help address increasing healthcare costs while improving access to care. 16 Interventions that empower patients and influence attitudes and behaviors have been found to be most effective at improving clinical outcomes. 14 Additionally, nurse-managed protocols supporting nurse autonomy to titrate chronic disease medications according to structured algorithms have shown favorable results on the biophysical outcomes of patients with chronic disease, including decreases in hemoglobin A1c (HbA1c), blood pressure (BP), and low-density lipoprotein (LDL) cholesterol. 17
Our team previously conducted a novel, two-arm, randomized controlled trial designed to test the efficacy of technology-assisted case management (TACM) program in a low-income, rural population of African-American adults with type 2 diabetes. One nurse case manager titrated diabetes and BP medications over the phone instead of in a clinic for patients in the intervention arm, and results from the primary analysis showed significant improvement in glycemic control at 6 months post-randomization, decreasing HbA1c by 1% and providing a faster rate of decline in HbA1c levels than usual care for low-income patients with type 2 diabetes. 18 A question that remained unanswered is whether TACM is efficacious for BP control and whether quality of life (QOL) may be negatively impacted for individuals participating in the high-intensity TACM intervention compared to those receiving usual care. Therefore, the goal of this analysis was to investigate the efficacy of TACM in low income, rural populations on improving BP control without compromising QOL.
Research design and methods
Study design and participants
All study procedures were approved by the Medical University of South Carolina Institutional Review Board. A partial waiver for the Health Insurance Portability and Accountability Act was approved for use of clinical records, and all participants gave informed consent prior to study enrollment. This study was a two-arm randomized controlled trial (RCT) that included a 6-month follow-up period with participants assessed at baseline, 3 months, and 6 months. Study participants were recruited from eight community-based adult medicine primary care practices within the Franklin C. Fetter Family Health Center, Inc (FCFFHC), a federally qualified health center in South Carolina. Almost three-quarters of the patients receiving service from these clinics were African American and had an income below 100% of the federal poverty level. All study participants had a diagnosis of type 2 diabetes documented in the medical record, and poorly controlled diabetes (HbA1c ≥ 8%). Additional inclusion criteria were being 18 years of age or older, receiving care within the FCFFHC, willing to use the FORA 2-in-1 telehealth system, having a working landline, and ability to communicate in English. Patients participating in other diabetes clinical trials, showing mental confusion suggestive of dementia, alcohol or substance abuse, active psychoses, prognosed to have less than 6 months to live, and pregnant/lactating females were excluded from the study. Study participants were compensated up to $75, $25 for each study visit that was completed (baseline, 3 months, 6 months). Two primary methods of recruitment were utilized with participants being recruited using International Classification of Diseases codes consistent with a diagnosis of type 2 diabetes from clinic billing and laboratory data, and referrals from physicians, clinic staff, or themselves. The protocol for this trial has been previously published. 19
Randomization and blinding
Once eligibility criteria were verified, and informed consent was obtained, the nurse case manager randomly assigned participants (1:1) into one of two study groups. The randomization scheme was developed using a web-based computer-generated sequence. Study sites and research assistants collecting primary data on participants were blinded to randomization. The nurse case manager who delivered the intervention, study participants, and treating physicians were unblinded due to the nature of the intervention. About 50 participants were randomized every 6 months until study enrollment was complete.
Procedures
Participants were randomized to one of two study groups: (a) TACM intervention or (b) usual care. Participants who were randomized to TACM used the FORA 2-in-1 telehealth system by ForaCare® (Moorpark, CA, USA). The FORA system is an inexpensive, off-the-shelf device composed of 2-in-1 blood glucose and BP monitor. Participants were provided with glucose test strips to allow for testing at least once a day, and BP and glucose values collected using the system were uploaded daily to a central server through a modem. The nurse case manager was able to review the uploaded values on a secure website at any time. Study participants randomized to the TACM intervention group were trained how to use the machine and were asked to provide blood glucose and BP measurements once a day using the FORA system (ForaCare® (Moorpark, CA, USA)). An evidence-based treatment protocol approved by the FCFFHC primary care providers was used by the nurse manager to titrate diabetes and BP medication weekly (for patients on insulin), or biweekly adjustments (for patients on oral agents) under the supervision of the study physicians depending on the FORA measurements received.
Patients in the usual care group received the standard of care during their regular scheduled medical visits. The patients’ providers were responsible for determining treatment parameters, making changes to the treatment regimen, or determining follow-up visits. Patients initiated scheduling visits with their provider, and clinic nurses were used to follow-up on patients with complex cases or patients with abnormal results.
Twenty percent of the intervention study charts were selected at random and reviewed along with FORA uploaded BP and blood glucose measurements for the corresponding patients to ensure treatment was completely administered in accordance with the study protocol. The nurse case manager was assessed by using rating forms to determine if medications were titrated appropriately and how well they accomplished relevant tasks for each patient. An optimal level of patient compliance was ensured using five methods: (a) at study enrollment, the importance of testing their blood glucose levels, checking their BP, and uploading the data using the FORA system on a daily basis was emphasized; (b) patients received weekly reminder phone calls to test and upload FORA measurements; (c) the study staff requested the names and telephone numbers of three relatives who would be able to contact the participant if needed; (d) the study staff were flexible in accommodating participants’ schedules; (e) monetary reimbursement was provided for completion of study visits.
Outcomes
BP and QOL were measured and assessed at baseline, 3 months, and 6 months. The outcomes of interests were BP and QOL as measured during the 6-month visit. The OMRON BP742N by © 2021 Omron Healthcare, Inc (Lake Forest, IL, USA) BP monitor was used to measure BP. Patients were seated for 5 min with feet flat on the floor prior to BP measurement. Three BP readings were collected with a 3-min wait period between each reading, and the average of the three readings was documented as the individual's BP reading for the visit.
QOL was measured using the 12-item Short-Form Health Survey (SF-12), a 12-item scale that includes a summary of physical health (PCS), and mental health (MCS) component scores measuring health status and functioning over the past 4 weeks. 20 Items include dichotomous (yes/no) responses and ratings on 5- and 6-point Likert scales. 20 Questions used for PCS asked how often a participant was limited by their physical health when completing activities like climbing stairs or moving a table. 20 MCS items asked how often emotional problems like feeling depressed or anxious led to accomplishing less than one would like. 20 The SF-12 is a valid and reliable scale with an alpha of 0.89 and is regularly used due to its outcome scores being interchangeable with the longer SF-36 version. 20
Statistical analysis
Descriptive analyses of the patient baseline demographics by study group were conducted using mean and standard deviation for continuous variables and frequencies and percentages for categorical variables. Analyses were conducted using the intent-to-treat (ITT) and per-protocol samples. The ITT sample included all randomized patients (total n = 113, TACM; n = 54, usual care; n = 59). Missing data were imputed using multiple imputation methods. 21 Multiple imputations on missing BP and QOL values was performed under the assumption of missing at random using PROC MI (Markov chain Monte Carlo method), and results were combined for final inference using PROC MIANALYZE using Rubin's rule (SAS version 9.4, North Carolina). 22 The variables used in the imputation model were gender, age, marital status, race, insurance status, body mass index, education, smoking, diabetes duration, depression, number of comorbidities, social support, and literacy.
First, comparisons of study group means were made using an independent sample t-test. Second, an analysis of covariance was performed on the ITT data after imputation with baseline levels of BP as a covariate, to determine if there was a significant difference in baseline-adjusted levels of BP at 6-month follow-up between TACM and usual care. Third, a longitudinal linear mixed-effects analysis was used to compare trajectories of BP levels and MCS and PCS over time between the two study groups using a random intercept and slope longitudinal model. The longitudinal model included the treatment group, assessment visit, and the interaction between treatment and assessment visit. A significant interaction would suggest that the trajectories of BP levels over time were different between the two treatment groups. A random intercept and a random intercept and slope based on a likelihood-ratio test using maximum likelihood approximation models were selected.
The magnitude and direction of differences in treatment means were estimated at each time point using 90% confidence intervals (CIs). The order of subtraction was TACM minus usual care, with a positive value indicating a superior mean score for TACM and a negative value indicating an inferior mean score for TACM. SAS 9.4 was utilized for the performance of all analyses and the significance of differences was evaluated using a 2-tailed level of significance of 0.05. Models were not adjusted if the unadjusted models with BP and QOL as outcomes were not statistically significant. If significant differences were detected, a Bonferroni correction was applied to determine if significance was maintained.
Role of funding source
The Department of Defense (Grant # W81XWH-10-2-0057) funded the study. The funding agency did not have a role in the study design, data collection, data analysis, data interpretation, or drafting of this manuscript. The study was approved for 4 years of funding, however the funding agency decided to terminate funding after 3 years due to budgetary constraints. Due to the early termination, the research team was unable to recruit a full sample of 200 participants. After discussions between the funding agency and the study team, it was decided to terminate the study with 113 participants. The decision to terminate allowed 6 months of follow-up within the 3-year revised funding timeframe.
Results
A total of 113 participants were randomly assigned to TACM or usual care study groups between July 1, 2011, and April 30, 2013. Figure 1 shows the flow of patients through the study. Table 1 shows the characteristics of the ITT sample at baseline. Most of the participants were aged 60 and older (33.6%), female (81.4%), non-Hispanic Black (75.2%), earned <$10,000 per year (36.3%), and had fair/poor self-rated health (60.2%). The mean duration of diabetes was 12.2 years and the mean body mass index was 35.6.
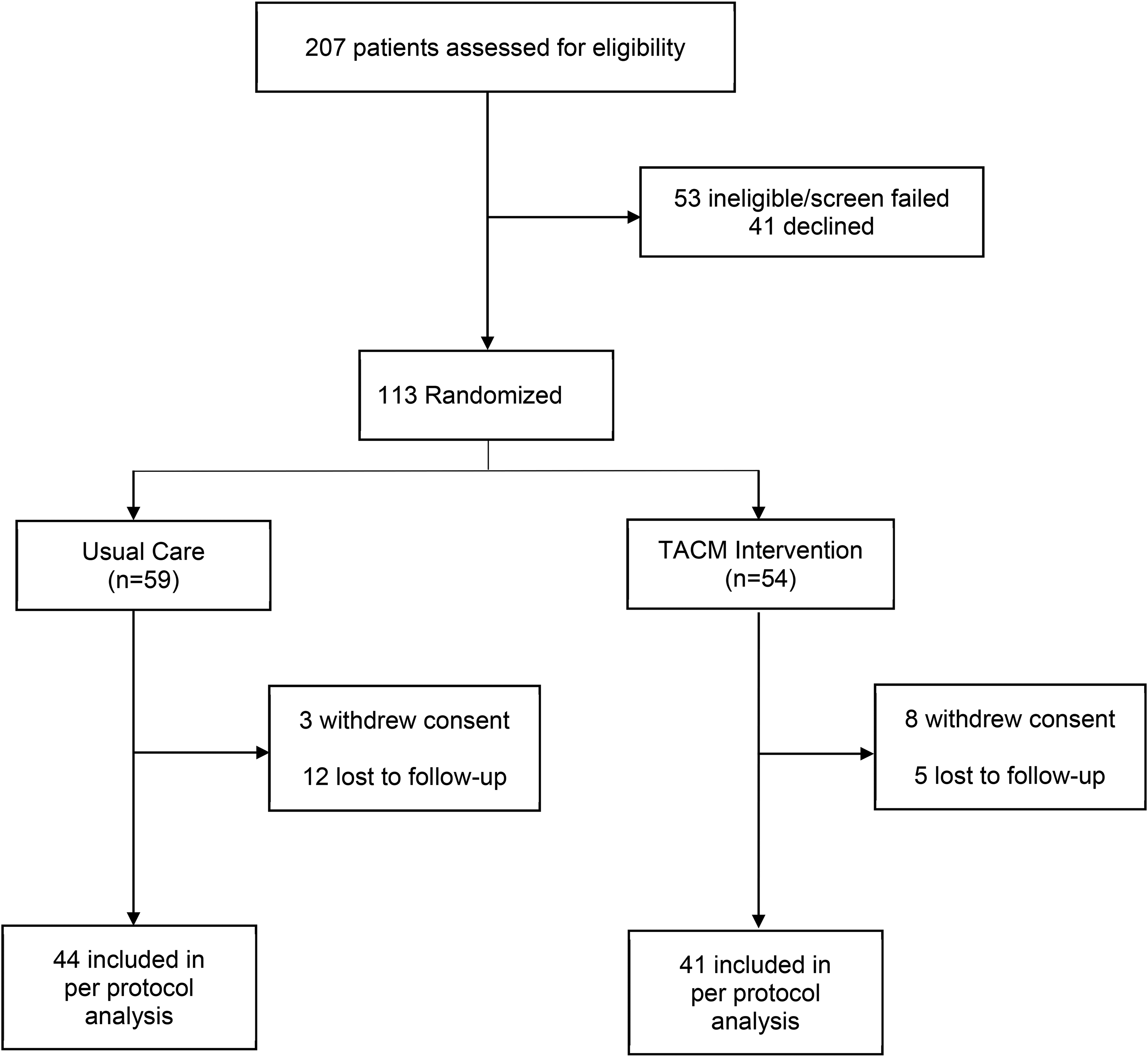
Trial profile, consolidated standards of reporting trials (CONSORT) diagram.
Baseline characteristics by study arm of intention to treat population.
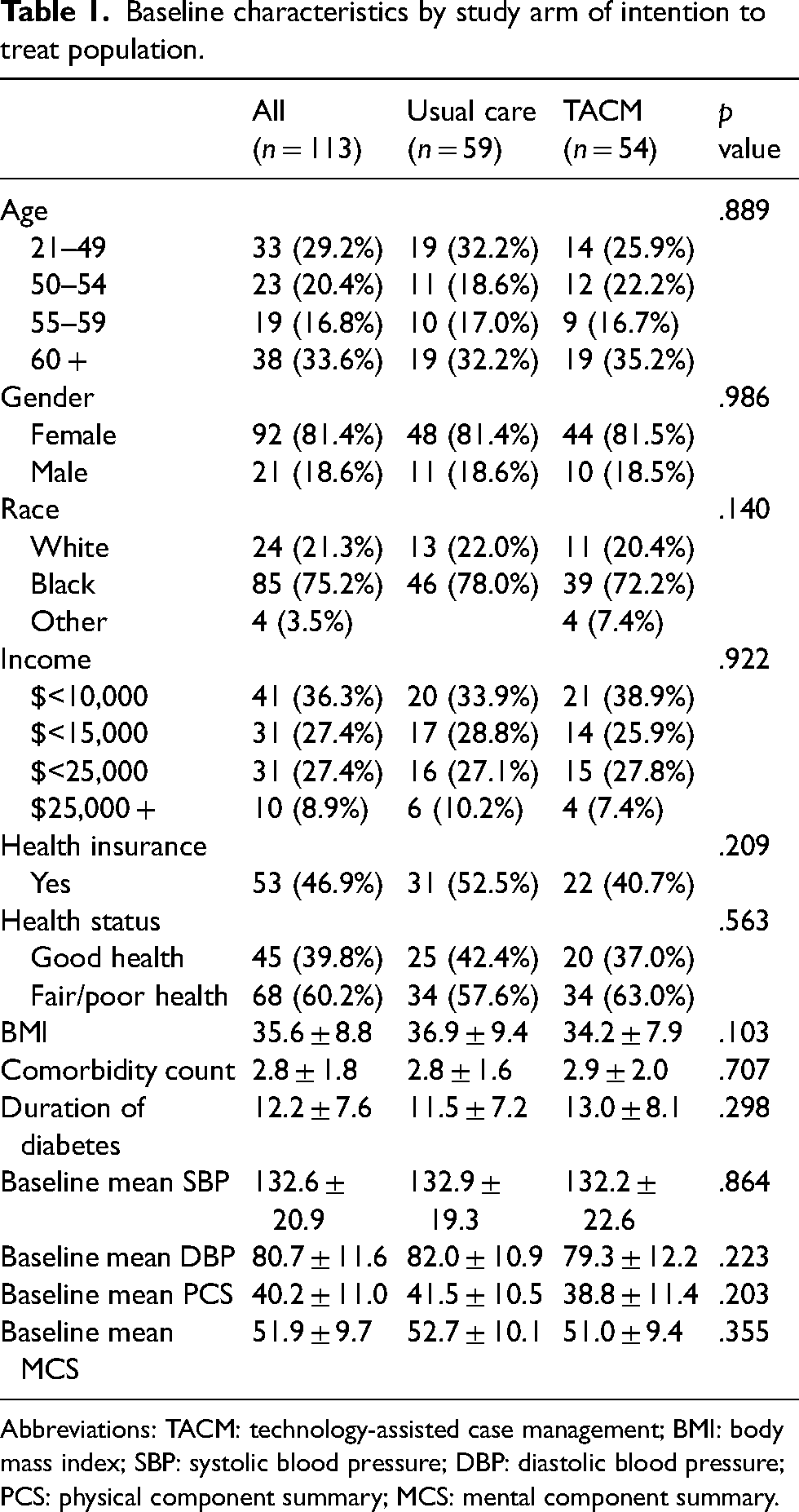
Abbreviations: TACM: technology-assisted case management; BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; PCS: physical component summary; MCS: mental component summary.
Table 2 shows the unadjusted means and differences of BP (diastolic and systolic) measurements and QOL (PCS and MCS) by a study group at baseline, 3 months, and at 6 months. There were no statistically significant differences in systolic BP, physical component of QOL, or mental component of QOL between the TACM and control group. There were no statistically significant differences in BP or QOL between treatment groups at 6 months. However, the significant interaction of diastolic BP and time (p = .05; Table 3) indicates that the trajectories of DBP over time differ by treatment group, as shown in Figure 2.
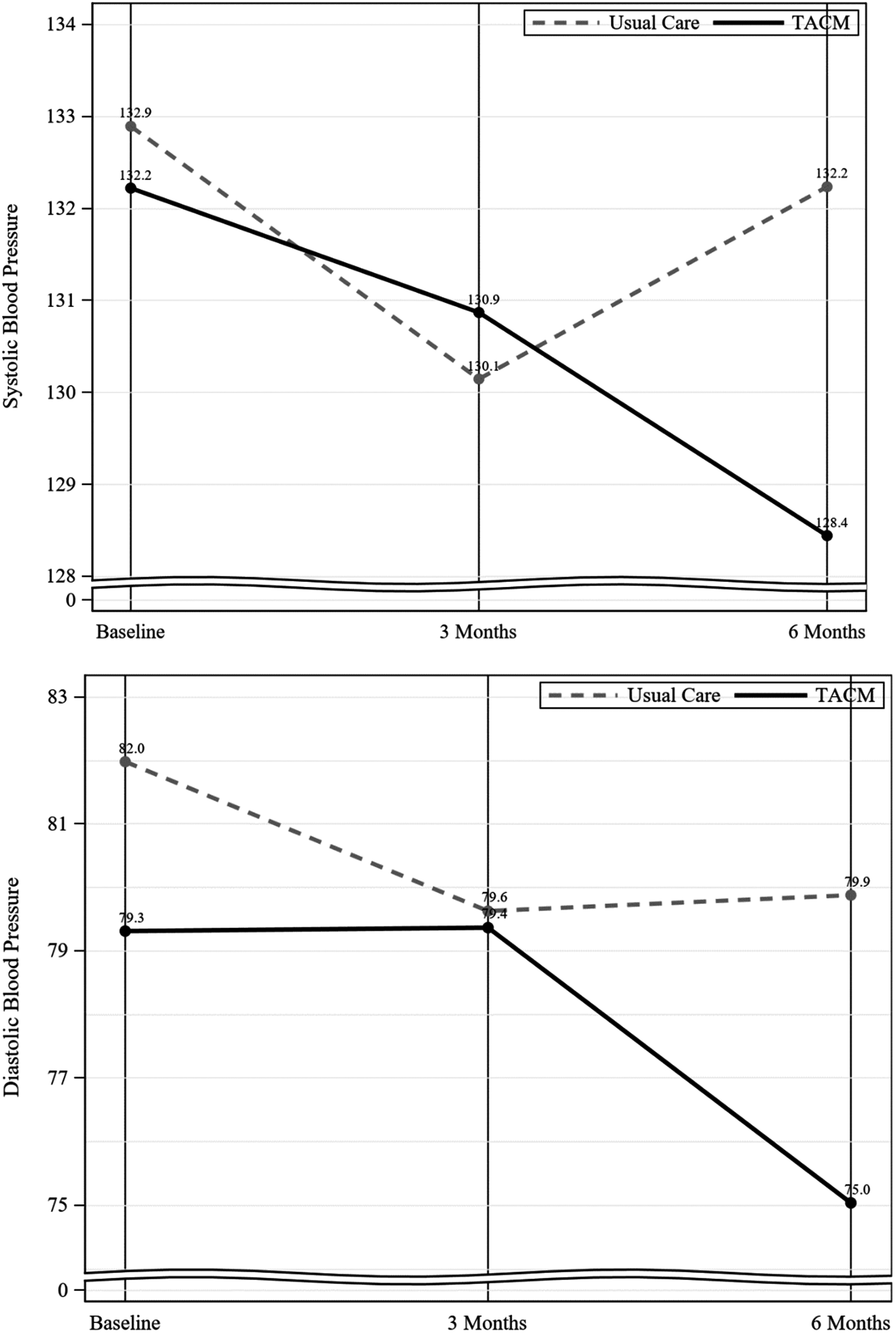
Unadjusted mean levels of systolic and diastolic blood pressure over time by treatment group (intention to treat populations).
Unadjusted means and differences of BP and QOL (MCS and PCS) at baseline, 3 months, and 6 months between treatment groups (intention to treat population).
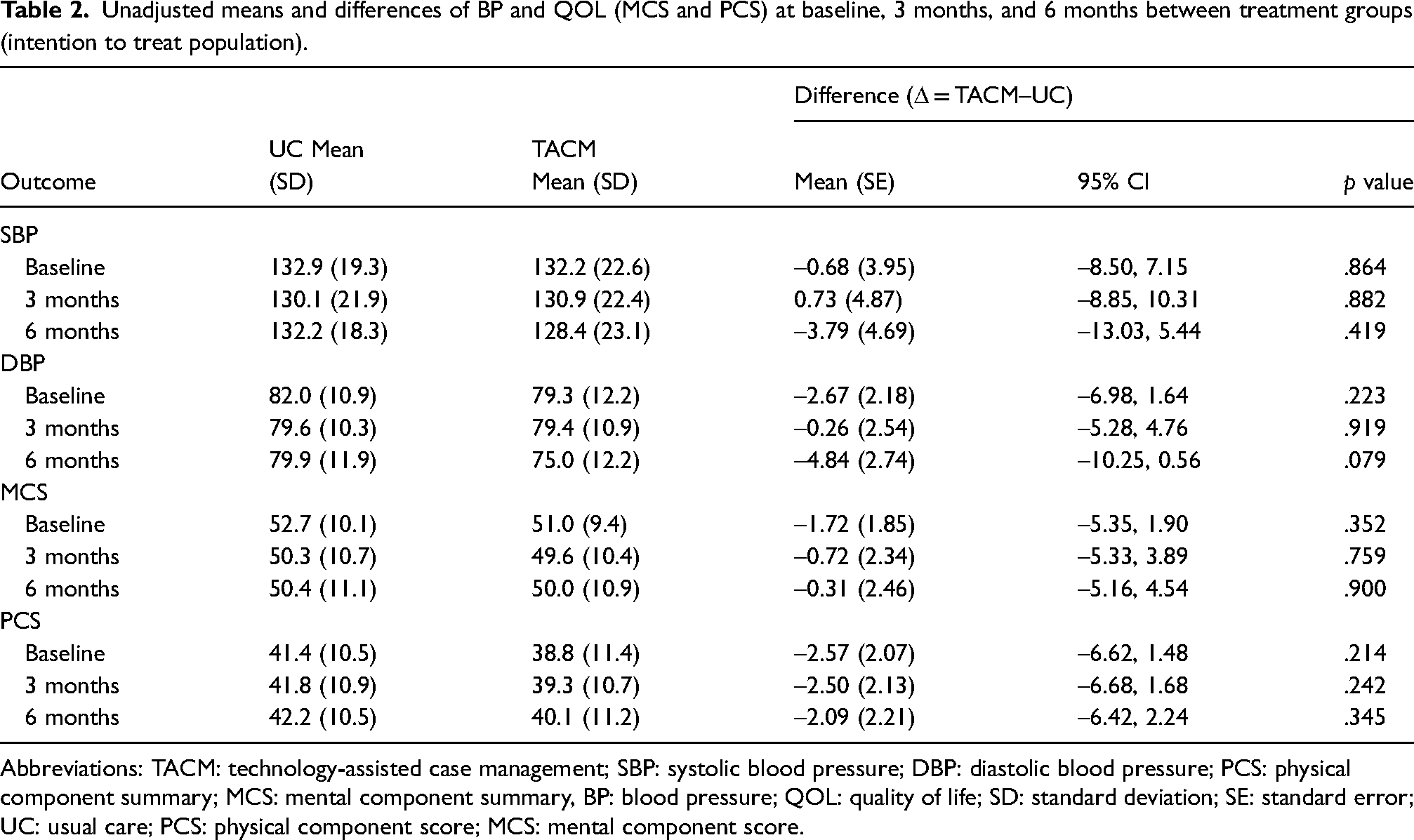
Abbreviations: TACM: technology-assisted case management; SBP: systolic blood pressure; DBP: diastolic blood pressure; PCS: physical component summary; MCS: mental component summary, BP: blood pressure; QOL: quality of life; SD: standard deviation; SE: standard error; UC: usual care; PCS: physical component score; MCS: mental component score.
Longitudinal mixed effects analyses for differences in trajectories over time in levels of BP and QOL between the treatment groups (intention to treat population).
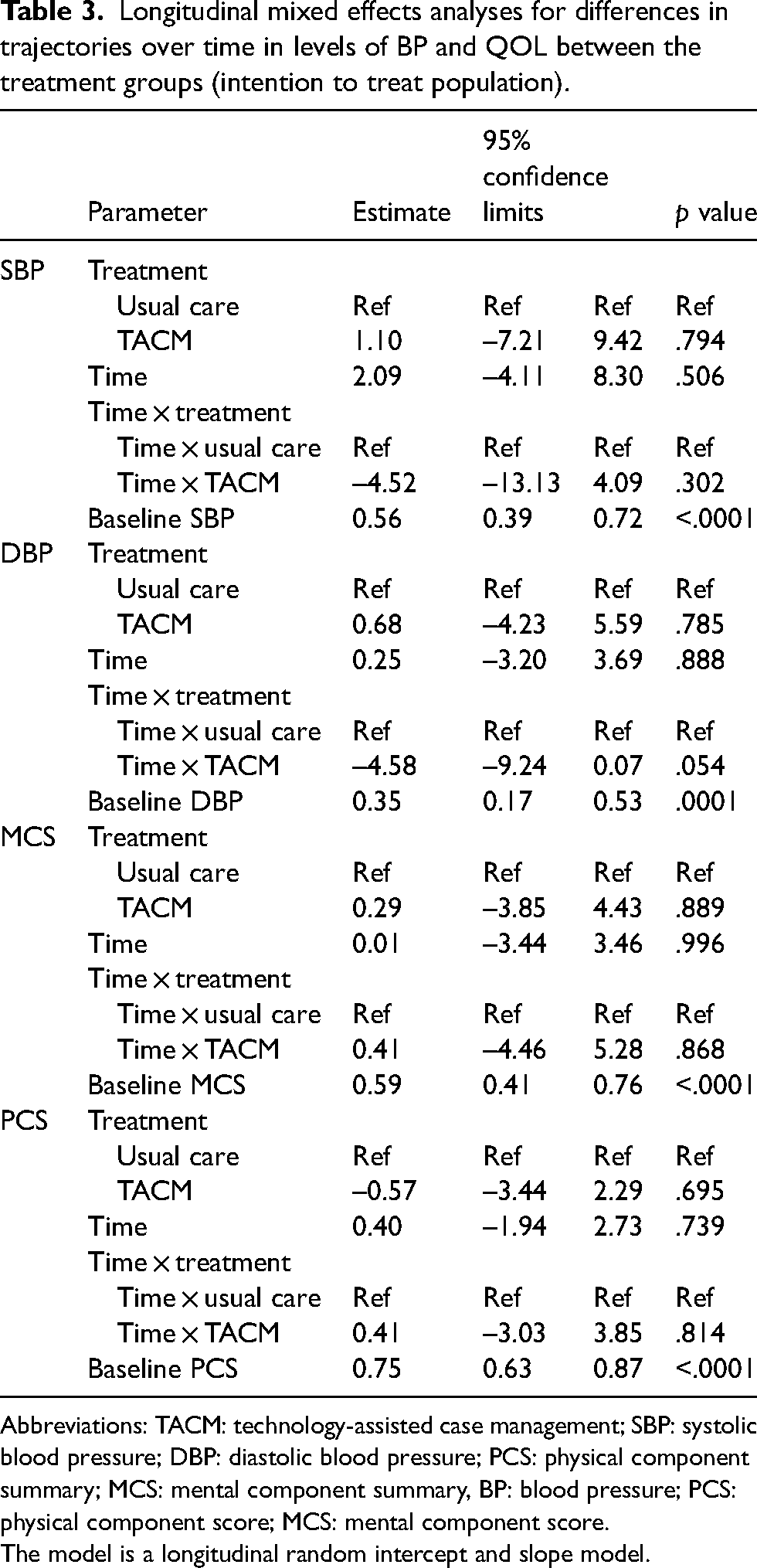
Abbreviations: TACM: technology-assisted case management; SBP: systolic blood pressure; DBP: diastolic blood pressure; PCS: physical component summary; MCS: mental component summary, BP: blood pressure; PCS: physical component score; MCS: mental component score.
The model is a longitudinal random intercept and slope model.
Discussion
Results from this study demonstrated that TACM by a nurse with medication titration under physician supervision was efficacious in improving diastolic BP in this sample of adults with uncontrolled type 2 diabetes. There was no statistically significant change in the MCS and PCS summary score measures of QOL between the two study groups during the 6 months of the study.
The results of the significant interaction of diastolic BP and time, indicating the trajectories of DBP over time differ by treatment group are consistent with the findings previously published examining intervention efficacy on reducing HbA1C, with adults in the TACM intervention group having significantly lower HbA1C levels at 6 months compared to adults in the usual care group (18). Activities associated with participating in the intervention and achieving improved BP and HbA1C levels for individuals with diabetes include daily self-monitoring consisting of daily BP and glucose checks, daily upload of BP and glucose measurements, and medication adherence have the potential to be burdensome for low-income patients managing chronic conditions. It is estimated that adults with diabetes need to spend about 4 h per day to complete recommended diabetes self-care activities which include self-monitoring, recordkeeping of glucose and BP values, taking medications, scheduling medical appointments, and other activities like meal planning, exercising, and problem solving. 23 Using technological devices such as the FORA 2-in-1 telehealth system have been identified as a facilitator of diabetes self-monitoring, where barriers include financial constraints, unrealistic expectations, and other environment-related factors. 24
Given the known burden of completing diabetes self-care activities, it is important to understand if completing activities such as daily self-monitoring, medication adherence, and other components of caring for diabetes negatively impact one's QOL. Surprisingly, results from this study show that there was no difference in the MCS or PCS scores for adults in the TACM and usual care study groups. These findings support the idea that completing activities associated with intervention group participation did not have a negative impact on QOL, and support the hypothesis that TACM is a viable method for caring for low income, adult, patients with uncontrolled type 2 diabetes. This intervention allows nurse case managers to review glucose and BP values more frequently than once every 3 months, the current recommendation for diabetes care, and to titrate medications according to individual patient needs. 25 These modifications to the way care is provided for low-income patients with uncontrolled diabetes are an effective method of providing high-quality care that supports improved glycemic and BP control, all while having no impact on the individual's QOL.
While this study had a significant effect on improving diastolic BP and did not have a negative impact on QOL, the study is not without limitations. First, the study was stopped prematurely due to budgetary constraints and early termination of funding. It is possible that a larger sample may have resulted in different results including larger effect sizes, and possibly statistically significant findings for differences in systolic BP and QOL. Second, participants were recruited from multiple clinics within one health system, therefore these results are not generalizable to the larger U.S. population. Third, attention was not controlled for in the intervention group, therefore it is possible that regular contact with the nurse case manager may have resulted in participant behavior changes that improved BP levels.
Conclusions
In conclusion, results from this study suggest TACM by a nurse with medication titration under physician supervision is efficacious in improving diastolic BP without compromising QOL in low-income rural adults with diabetes. These findings may lead to changes in the way care is provided for low-income rural adults with uncontrolled type 2 diabetes. The use of technology and provision of autonomy to nurses to titrate medications in accordance with a pre-defined algorithm or protocol has the potential to improve health outcomes in a population of individuals who generally experience challenges accessing health care. Improvements in health outcomes may result in decreased healthcare costs, complications, and mortality due to diabetes.
Footnotes
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional Review Board at the Medical College of Wisconsin and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. All procedures were approved by the Institutional Review Board at the Medical University of South Carolina.
Authors’ contributions
LEE obtained funding and designed the study. LEE, RGK, EG analyzed and interpreted the data. LEE, AZD, RJW drafted the article. LEE, AZD, RJW, EG, and RGK critically revised the manuscript for intellectual content. All authors approved the final manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the U.S. Department of Defense (grant no. W81XWH-10-2-0057). Funding organizations had no role in the analysis, interpretation of data, or writing of the manuscript.
