Abstract
Objectives
Coordinated care plans (CCPs) for high-cost health care system users aim to improve system-level performance. We evaluated health care resource use and costs among CCP patients (enrollees) versus a control group that did not receive coordinated care (comparators) in Southeastern Ontario.
Methods
A difference-in-differences analysis of a quasi-experimental, double propensity score-matched and adjusted cohort was conducted. Linked population-based administrative data were used to measure health care utilization and costs and to identify comparators for two enrollee groups who began CCPs between April 1, 2013, and March 31, 2019. Enrollees were recruited from hospitals in Quinte or community care centres in Rural Hastings/Thousand Islands, and were 1:1 propensity score matched to comparators. Difference-in-differences estimates were calculated using generalized estimating equations for hospitalization rates, homecare visits, primary care visits, other health care resources and total costs.
Results
A total of 558 enrollees in Quinte and 538 in Rural Hastings/Thousand Islands were identified and matched to comparators. Difference-in-differences estimates were significant in both enrollee groups for number of homecare visits ([IRR 1.72; 95% CI (1.44, 2.06)] and [IRR 1.73; 95% CI (1.45, 2.06)], respectively). Number of primary care visits were 1.76 times greater for Rural Hastings/Thousand Islands enrollees versus comparators [IRR 1.76; 95% CI (1.32, 2.35)]; total costs increased by 23% ([IRR 1.23; 95% CI (1.09,1.39)].
Conclusions
Homecare use significantly increased for enrollees versus comparators, indicating specific priority areas of Ontario CCPs were met. However, no reductions were shown for other health system performance indicators. We also showed increased 7-day primary care follow-up visits for community care centre-recruited patients, but not for hospital-recruited patients. Decision-makers may wish to target patients who are less advanced in their chronic disease trajectory.
Introduction
In Canada, approximately 5% of the population incurs 66% of the total health care costs.1–3 These ‘high-cost’ users tend to be elderly patients with multiple chronic conditions, who experience fragmented care and system inefficiencies.4,5 Coordinated Care Plans (CCPs) can support ‘high cost’ users with health care system navigation.1,2,6,7 In 2013, Ontarian CCPs were initiated through a program called ‘Health Links’,6,7 with the aim of improving system-level performance by limiting service gaps for complex patients, increasing communication within and between patients and providers and reducing hospital admissions/readmissions.6,7
Previous evaluations of the efficiency of CCPs for high-cost users have yielded mixed results.1,2,8–12 A meta-analysis of randomized controlled trials showed CCP enrollees had fewer hospitalizations and emergency department (ED) visits than those not enrolled, 11 similarly observed elsewhere.2,10,11 Others have found no significant reduction in the use of those same resources.1,6–10 In Ontario, one study evaluated hospital-led Health Links programs, 10 while another assessed hospital-, primary care- and community care centre-led Health Links without distinguishing between type of lead organization. 9 Health Links operated under a ‘low rules’ approach, whereby organizations (such as primary care practices and community care centres) independently developed patient identification and care delivery strategies.13,14 Therefore, Health Links outcomes should be contextualized within the specific type of organization the program was implemented. Further, Health Links enrollee patient costs have not been studied. Therefore, in order to inform policy decision-making, high-quality, longitudinal data are necessary to understand health care utilization and costs of Health Links led by various organizations. As such, the objective of this study was to evaluate both primary care- and community care centre-led Health Links with respect to health care resources and total costs.
Our study considers the situation in Ontario, a province of Canada. In Ontario, residents receive universal health insurance, which is publicly funded and covers costs of essential medical services. Patients are recommended for enrollment to the Health Link (HL) for a CCP if they are expected to experience many transitions between health care providers, 15 have four or more chronic conditions and/or are negatively impacted by social factors (e.g., socioeconomic status, caregiver status and social connectivity).9,16 The goal of enrollment into HLs is to improve system outcomes and reduce health care costs for complex patients,17,18 by providing patients with early assistance in transitioning between care providers (hospitals, rehabilitation centres, primary care providers) and follow-up care. 14 HL enrollment is initiated with the creation of a CCP developed during a meeting between the patient and their care team (including family members, providers, and care coordinators). HLs operate under a ‘low rules’ approach, whereby organizations leading a program devised unique patient identification, recruitment and care delivery strategies to implement the CCP,13,14 and therefore patient characteristics (e.g., number, type and severity of complex conditions), are expected to differ across HLs.
Methods
This study used a retrospective quasi-experimental difference-in-differences design with double propensity score-adjusted analysis of a cohort of Health Links enrollees and matched comparators.
Study setting: Coordinated care plans in southeastern Ontario
We evaluated the effect of having a CCP and enrollment in a HL in three areas of Ontario: Rural Hastings, Quinte and Thousand Islands. All three areas contain hospital, community care, specialty care, and primary care partners. 3 In Quinte, candidate patients were recruited from hospitals and recommended a primary care-led CCP, and in Rural Hastings/Thousand Islands, candidate patients were recruited from community care centres and recommended a community care centre-led CCP.
Data sources
Records of individual health care system encounters are routinely collected in population-based, deterministically linked health administrative datasets that are anonymized and available from Institute for Clinical Evaluative Sciences (See S1 in the online supplement for a list of ICES databases accessed in this study). These datasets were linked using unique encoded identifiers and analyzed at ICES. ICES is an independent, non-profit research institute, whose legal status under Ontario’s health information privacy law allows it to collect and analyze health care and demographic data, without consent, for health system evaluation and improvement.
Population
Eligible study participants who were enrolled in CCPs between April 1, 2013, and March 31, 2019, comprised the enrollee population. Patients were excluded if they declined to participate in a CCP, had no recorded enrollment date, and/or had missing demographic information. Comparators were Ontario individuals in the Registered Persons Database who met eligibility criteria for HL enrollment. To create the comparator population, the distribution of enrollee index dates (CCP enrollment date) was plotted, and 20% of these dates were randomly selected and assigned to comparators, consistent with techniques used in previous Health Links evaluations.9,10 Similar index date distributions across enrollee and comparator groups helped ensure comparability with respect to exposure to time-related, external environmental variables (e.g., minor changes in health care policy or practice, and economic and social phenomena). This method was used because an Ontario sample of non-enrolled patients is a robust population from which to select controls - choosing controls from the local region would risk selection bias, as those not enrolled locally might be selectively different compared to local enrollees. Inclusion criteria for the comparator group consisted of: being eligible for health care coverage in Ontario; being alive at index date; having complete sociodemographic data; and having complex needs, defined as being a patient with four or more conditions (from 55 conditions identified by the Ontario Ministry of Health and Long Term Care (MoHLTC)). 15
Covariates and outcome measures
Baseline covariates for enrollees were assessed at index date and included age, sex, rurality, geographical area, neighbourhood-level income quintile, primary care model affiliation (non-team based, fee for services or team based, shown to differentially affect patient health care resource utilization), 19 and comorbidity measure based on health care use in the year prior to index (Collapsed Adjusted Clinical Groups [Johns Hopkins ACG Software, version 10]). Health care services use 1 year prior to the index date were also identified.
Health system performance indicators from MoHLTC7,20,21 were selected as outcome measures, and included hospitalization rates (acute care admissions, total length of stay, 30-days readmission rate), ED and primary care visits within 7 days after acute hospital discharge, lab claims, home care visits, and total costs. S2 and S3 in the online supplement show cost break-downs and methods used to derive costs, respectively. Outcomes were measured 1 year pre- and post-index date, respectively, or until date of recorded death. Because participant characteristics may differ systematically between HL groups, data were partitioned into two groups (Quinte HL, and Rural Hastings/Thousand Islands HL), and analyses were conducted separately for each group.
Statistical analysis
A double propensity-score adjustment approach was applied to estimate the causal effect of HL enrollment on study outcomes to avoid incomplete participant matching and imbalanced covariate matching. 22 We matched and adjusted separately for patients with and without acute care hospitalizations within 1 year prior to the index date, as this was an important factor for the HL enrollee population.
First, the probability of HL enrollment (propensity score) was calculated using logistic regression. An index date was randomly assigned to non-participants based on the distribution of participants’ enrollment dates. Next, exact matches on sex, date of birth (+/− 90 days), index date (+/− 90 days) and nearest neighbour-matched propensity scores (based on all above-mentioned covariates assessed at index date) were created. The following covariates were not balanced (standardized differences >0.10) in the Quinte group: 3rd and 4th neighbourhood income quintiles, specialist visits in the first quarter of the year preceding the index date (Q1), acute care admissions in Q1, length of hospital stay (in days) in Q1 and ED visits in Q1–Q4. The following covariates were not balanced in the Rural Hastings/Thousand Islands group: palliative care, propensity score, acute care admissions in Q1 and Q4, length of hospital stay in Q1, and ED visits in Q1 (see S4 in the online supplement for covariate balance and the performance of propensity score algorithm). Study outcomes were then adjusted by propensity scores using regression models. 22 For each outcome, a univariate regression model (with propensity score as the independent variable) was created using only comparator participants in the matched sample. This regression model was then used to predict the potential outcome for all enrollees, had they not been enrolled in a HL. The ‘difference in differences’ estimates were calculated based on the average difference between the outcome observed among enrollees and the predicted potential outcomes.
The difference-in-differences method with generalized estimating equations and robust error variances were used on each outcome. Negative binomial distribution and log link were used to model acute hospital admission, ED visits, and acute care days. A log of person-years offset term was included to account for differences in deaths. A Poisson distribution was used to model hospital readmissions and primary care visits after hospital admission. The total number of hospital admissions was an offset term in the model. A Gamma distribution with a log-link was used to account for rightly-skewed total health care costs. A two-way interaction term, pre and post, and enrollment status were included as binary variables in each regression model. Pre-post differences in HL patients and difference-in-differences between HL patients and the comparator group were obtained. The DID estimator was calculated based on the predicted values from the model in both pre- and post-intervention periods. A visual inspection of time-series graphs for each outcome showed the parallel trends assumption was reasonable for the pre-intervention period (S5 in the online supplement). To account for repeated measures, models used an unstructured correlation matrix. All statistical analyses were performed on SAS Enterprise Guide 7.1 (SAS Institute Inc., Cary, NC, USA).
Results
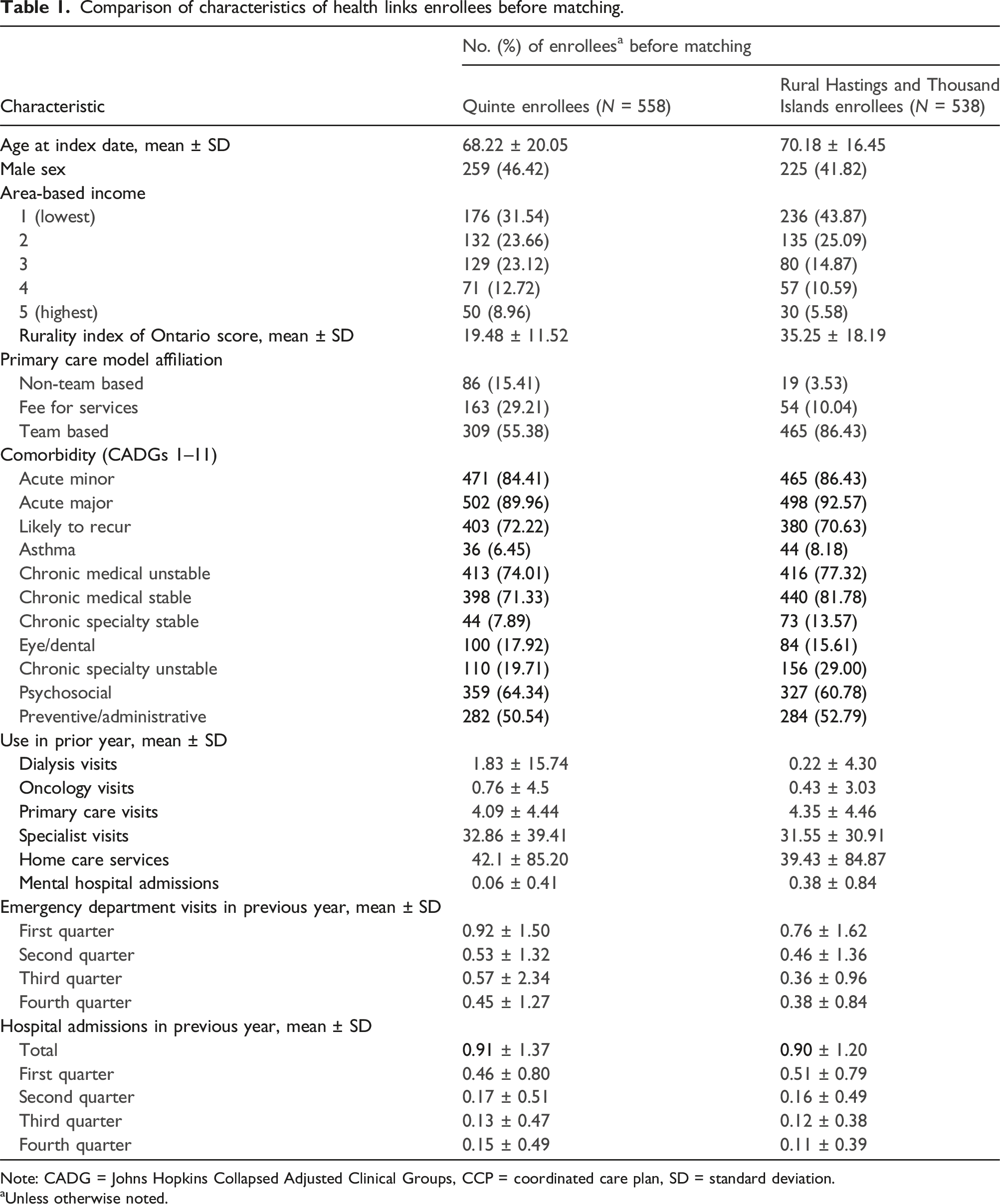
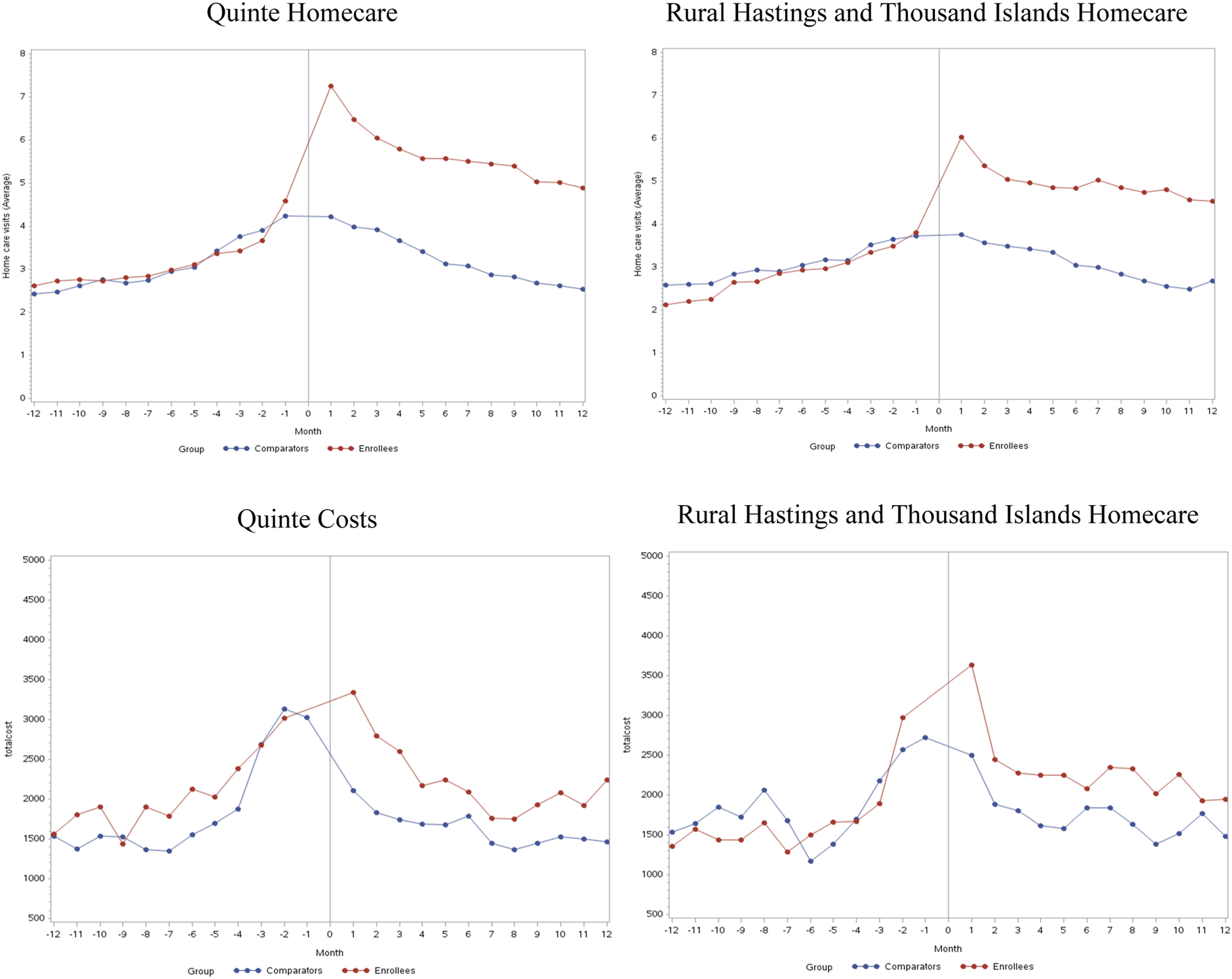
A total of 1096 HL enrollees were identified from Quinte (N = 558) and Rural Hastings/Thousand Islands (N = 538), along with 1,895,065 comparators (Figure 1). All enrollees were matched to comparators. Among enrollees, 1-year mortality after index date was 10.8%. Table 1 shows aggregated baseline characteristics of enrollees in both groups. Figure 2 shows average homecare use and costs sharply increased for enrollees in both Quinte and Rural Hastings/Thousand Islands in the first month post-index date with a gradual monthly decline (see S5 in the online supplement for all monthly resource use for performance indicators). Patient selection. Note: CCP = Coordinated Care Plan; HL = Health Links; ICES = Institute for Clinical Evaluative Sciences; OHIP = Ontario Health Insurance Plan; RIO = Rurality Index of Ontario; SD = standard deviation. Comparison of characteristics of health links enrollees before matching. Note: CADG = Johns Hopkins Collapsed Adjusted Clinical Groups, CCP = coordinated care plan, SD = standard deviation. aUnless otherwise noted. Average monthly resource use for health links enrollees and comparators.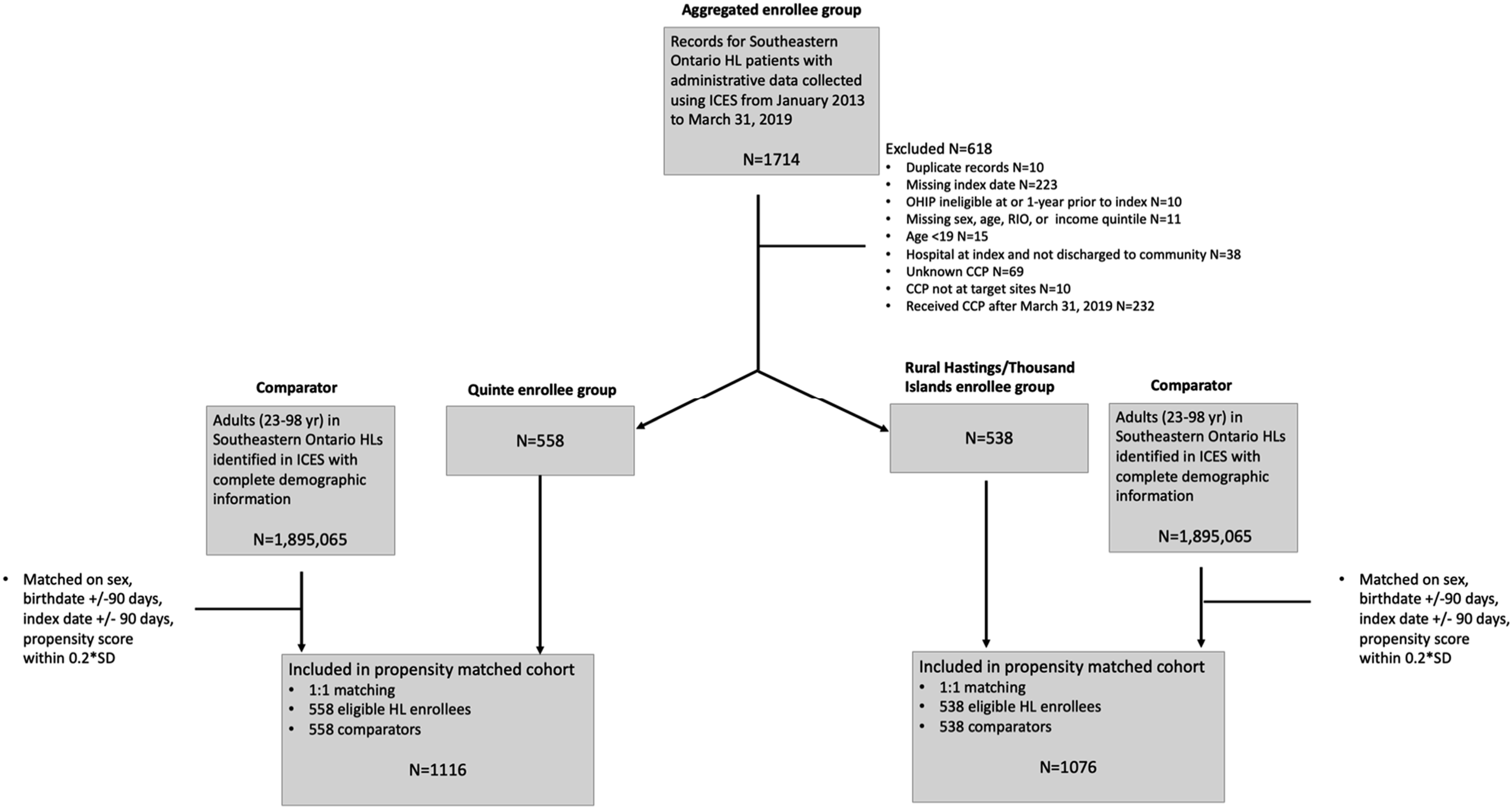
Difference-in-differences analysis results for health system performance indicators.
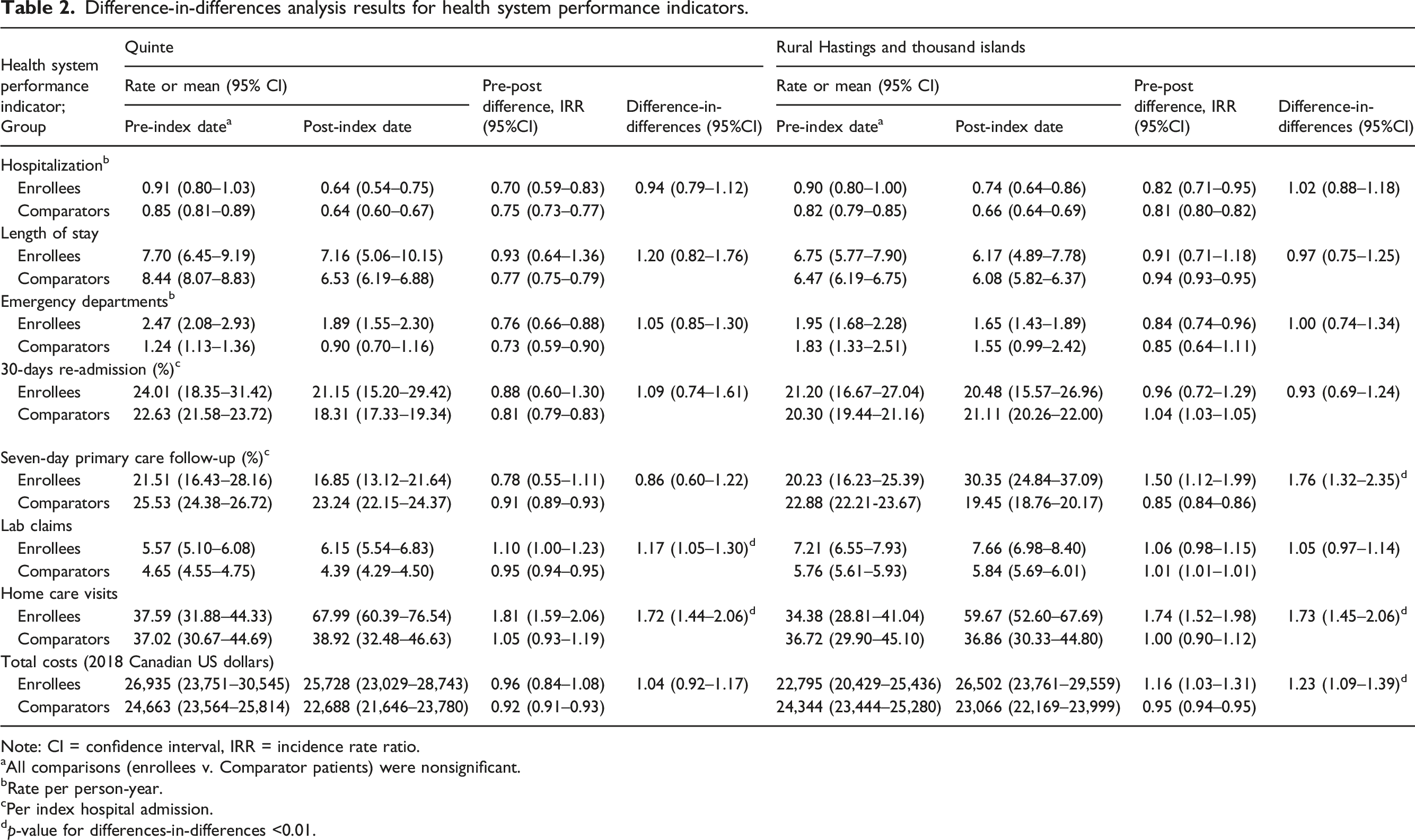
Note: CI = confidence interval, IRR = incidence rate ratio.
aAll comparisons (enrollees v. Comparator patients) were nonsignificant.
bRate per person-year.
cPer index hospital admission.
dp-value for differences-in-differences <0.01.
Cost of enrollees, by cost category.
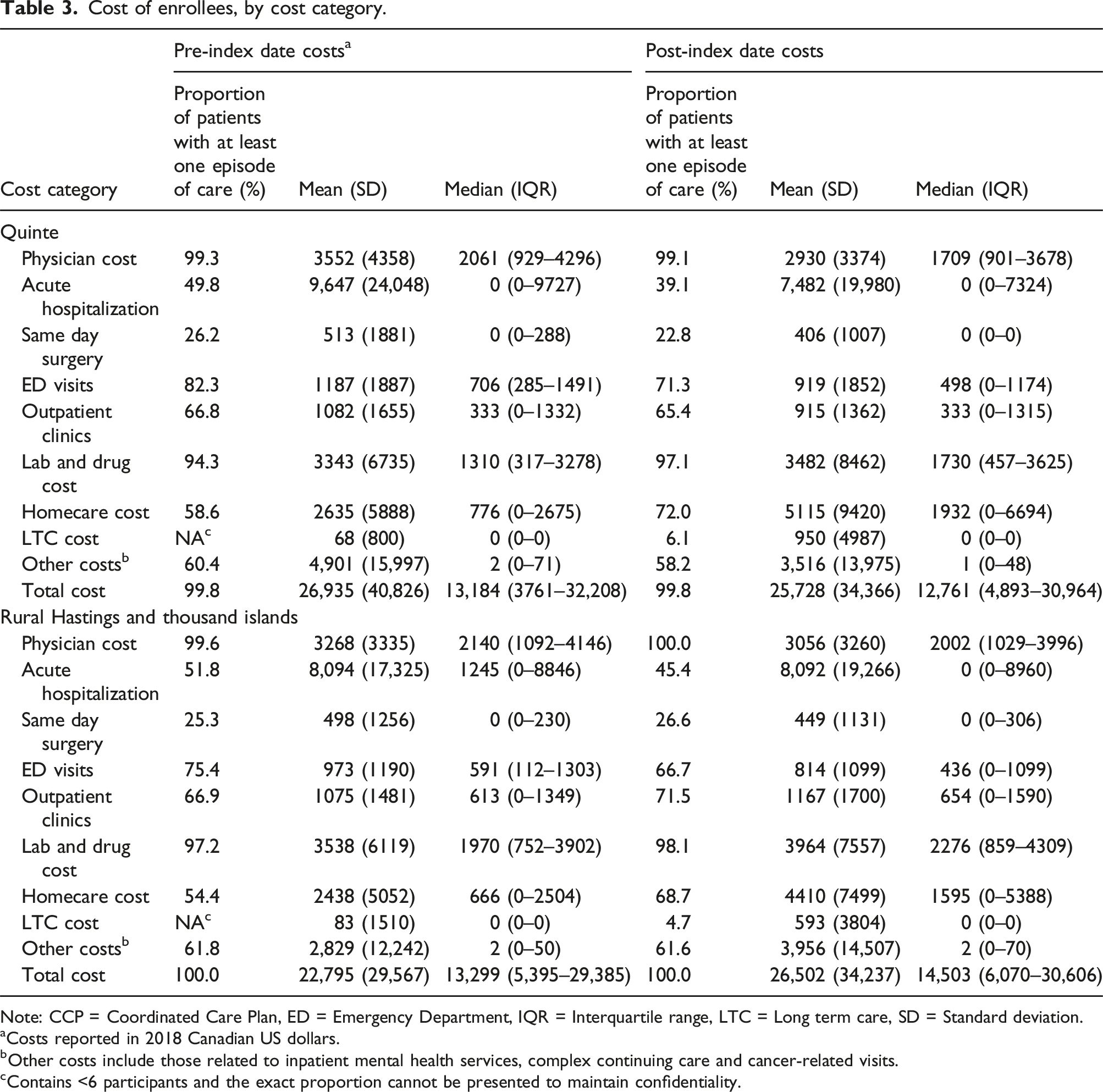
Note: CCP = Coordinated Care Plan, ED = Emergency Department, IQR = Interquartile range, LTC = Long term care, SD = Standard deviation.
aCosts reported in 2018 Canadian US dollars.
bOther costs include those related to inpatient mental health services, complex continuing care and cancer-related visits.
cContains <6 participants and the exact proportion cannot be presented to maintain confidentiality.
Discussion
Using population-level administrative data, we evaluated three Southeastern Ontario CCPs on selected health care system performance indicators and associated costs. No significant reductions in hospitalizations, length of stay, ED visits, or 30-days readmissions were observed for HL enrollees versus comparators. In relation to comparators, Quinte enrollees had 1.17 times more lab claims, and both Quinte and Rural Hastings/Thousand Islands enrollees had approximately 1.70 times greater number of homecare visits. Rural Hastings/Thousand Islands enrollees had significantly higher primary care follow-up visits and total costs.
Findings pertaining to increases in lab claims, costs, homecare and primary care visits are clinically relevant as these increases could indicate that enrollees were able to receive care that may have been previously inaccessible. Similar results pertaining to increases in homecare use have been observed in a randomized controlled trial with community-dwelling older adults, which showed patients receiving integrated care for 22 months had 1.72 times greater odds of accessing homecare than controls. 1 Our results are consistent with evaluations of CCPs in the United States (US): we showed Rural Hastings/Thousand Islands, but not Quinte, had increased costs; similarly, a US study conducted on Medicaid and Medicare patients receiving community-level CCPs found that the former group had per-patient savings of C$1643 per 3 months and the latter did not. 23 Another US study evaluating multiple distinct CCPs versus standard of care found that two out of 15 sites experienced cost savings. 24
Other CCP evaluations have shown mixed results. Similar to our null findings for decreases in enrollee hospital admissions versus comparators, a United Kingdom-based study showed an additional 0.01 monthly admission for multidisciplinary CCP enrollees upon conducting a difference-in-differences analysis. 8 Further, we showed no significant differences in readmission rates or ED visits, consistent with the findings of other research, which evaluated a primary care coordination on 30-days post discharge resource use. 25
Contrary to our null findings for differences in ED visits and hospital admissions for enrollees versus comparators, other studies have shown both significant decreases and increases in these performance indicators. For example, a meta-analysis of RCTs evaluating CCPs in North America, Europe, South Africa, Israel and Australia found 0.69 times fewer ED visits (95% CI 0.54–0.89) and 0.81 times fewer hospital admissions (95% CI 0.72–0.91) for enrollees, 11 similarly observed elsewhere. 2 A partial explanation as to why these findings contrast with our own could be differences in CCP designs, target populations and strategies for patient identification/recruitment into programs. Further, another study showed significantly fewer ED visits for enrollees compared with matched controls 12 months post-index date (IRR 0.88; [95% CI 0.79–0.99]). 10 Another Ontario-based propensity-matched cohort study showed higher rates of enrollee hospital admissions (IRR 1.74; [95% CI 1.40–2.17]) and ED visits (IRR 1.61; [95% CI 1.18–2.20]) versus comparators, 9 as observed elsewhere. 23 Mixed findings across these HL evaluations could be partially attributed to whether investigators excluded people who died in the follow-up period. Similar to our study, two studies did not exclude deceased patients.9,10 Further, we included both community care centre- and primary care-led HLs, while previous HL evaluations either did not distinguish between populations, or only evaluated hospital-led HLs.9,10
Variations in resource use and cost differences among patients belonging to separate HLs may be a consequence of the ‘low rules’ approach guiding coordinated care, whereby organizations leading an HL devise unique patient identification and care delivery strategies in their respective geographic regions. 14 In the present study, such an approach may have resulted in distinct patient populations recruited in each site with respect to disease burden. It could be that Rural Hastings/Thousand Islands patients (recruited from community care centres) were less sick and had fewer health conditions than hospital-recruited Quinte patients, and, upon HL enrollment, had increased primary care follow-up visits. During such visits, Rural Hastings/Thousand Islands patients may have received additional diagnoses, resulting in increased treatment and associated costs. This conjecture is consistent with increased post-enrollment inpatient mental health services cost, complex continuing care and cancer-related services, and could explain why HL enrollees in Rural Hastings/Thousand Islands incurred significantly higher costs than comparators. For Quinte patients, since they were recruited from hospitals, the number of laboratory tests requested at that point may not have been sufficiently conducted. Therefore, upon HL enrollment, access to such services improved, resulting in increased lab claims for enrollees versus comparators.
Knowledge gaps and future directions
Our results show that two priority goals of HLs were met: increased homecare and primary care visits,6,15 while other performance indicators such as hospitalizations and emergency department visits were similar for enrollees versus comparators. One explanation for these null findings may be that targeting the highest cost users is an ineffective strategy for HL patient identification, as these patients could have chronic conditions that have progressed beyond the ability to meaningfully respond to an intervention aimed at reducing resource use. 7 Future studies could be conducted on CCP lived experiences of patients, administrators and care providers and on patients’ access of community-level services. In addition, ministries of health could consider targeting different patient populations (e.g., based on fewer than four to five health conditions, as in HLs) which may result in improvement in program effectiveness.
Similarly, it is important to consider at which point in their disease trajectory patients are recruited (if in the beginning, middle or towards the end). We showed patients recruited from community care centres had increased primary care visits, while hospital-recruited patients did not. This could indicate that patients not yet requiring hospitalization (e.g., the top 10% vs 5% of high-cost health care users) are better targets for coordinated care plans than those who are advanced in their chronic disease trajectory. Likewise, another study 26 investigated the impact of patient characteristics on the ability of preventative care programs to reduce costs. Its authors discussed the importance of having a target population amenable to mitigation strategies, which may exclude patients with the highest risk of poor health outcomes.
Limitations
There are five main limitations with this study. First, the analysis was restricted to three HLs in Southeastern Ontario, and the results of this study may not be generalizable to other CCPs with different characteristics. However, our study assessed both primary care and community-based HLs, giving a novel perspective on the effectiveness of two types of HLs, and additional research could formally compare impacts of primary care- versus community care-centre led programs using statistical tests.
Second, this study investigated the average impacts of the Health Links program relative to a control group, while accounting for patient characteristics including chronic conditions. However, for a given individual, the expected program impact may differ depending on the patient’s specific characteristics, including number, severity and type of chronic conditions. Further, it is possible patients who were excluded from HL were sicker and might have benefitted more from the program than those who were included, and this could have had a conservative downward bias on observed HL program effects. Although it is possible that patients in the comparator group experienced small-scale coordinated care initiatives in their respective regions, there were no comprehensive, government-funded coordinated care programs like Health Links in Ontario at the time of the study. We therefore believe an appropriate control group was identified.
Third, it is possible that program effects on enrollee health care resource use could have extended beyond our 12-months follow-up period, as observed in an England-based evaluation of integrated care programs. 27 However, an Ontario-based Health Links evaluation found reduced ED visits during a 12-months follow-up period, 10 suggesting a year is sufficient to observe program impacts.
Fourth, certain social factors associated with Health Links enrollment (social connectivity and caregiver status) were not available in the ICES data, and comparators possibly had more favorable social circumstances than enrollees. However, socio-economic status was accounted for in matching, which has been shown to be positively associated with both social connectivity 28 and caregiver status. 29 Therefore, accounting for socioeconomic status could have limited the unmeasured confounding of other social factors not included in matching.
Fifth, a regression to the mean effect may have been present, where a patient’s pre-index resource use that was very high or low by random chance may have naturally regressed towards the mean of a given resource in the post-index period. Therefore, observed differences between pre- and post-index resource use may have occurred due to natural variation versus real change due to the impact of the program. 30
Conclusions
The results of this study could inform policy decision-making with respect to future implementation of coordinated care. Specifically, decision-makers may wish to target patients who have fewer than four to five chronic conditions (as in HLs) and are less advanced in their chronic disease trajectory.
Supplemental Material
Supplemental Material - Health care utilization and costs among coordinated care patients in Southeastern Ontario: A difference-in-differences study of a double propensity-matched cohort
Supplemental Material for Health care utilization and costs among coordinated care patients in Southeastern Ontario: A difference-in-differences study of a double propensity-matched cohort by Ana P Johnson, Elizabeth Hore, Walter P Wodchis, Yu Qing Bai, Luke Mondor, Tim Tenbensel, Catherine Donnelly, Michael Green, Michael Spinks, Julia Swedak, Dianne McIntyre and Ashleigh Wolfe in Journal of Health Services Research & Policy
Footnotes
Acknowledgements
This document used data adapted from the Statistics Canada Postal CodeOM Conversion File, which is based on data licensed from Canada Post Corporation, and/or data adapted from the Ontario Ministry of Health Postal Code Conversion File, which contains data copied under license from ©Canada Post Corporation and Statistics Canada. Parts of this material are based on data and/or information compiled and provided by: Ontario Health (OH), MOH, Canadian Institute for Health Information, and IQVIA Solutions Canada Inc. The analyses, conclusions, opinions, results, view and statements reported in this paper are those of the authors and do not necessarily reflect those of the funding or data sources; no endorsement is intended or should be inferred. We thank IQVIA Solutions Canada Inc. For use of their Drug Information File. The authors would like to thank Simone Walters for insights into Southeastern Ontario Health Links. The authors would also like to thank Sunny Cui and Megan Latanik at Queen’s University for assisting with manuscript editing.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received funding from a grant provided by Canadian Institutes of Health Research (165584). Wodchis receives support for a Research Chair from the Trillium Health Partners Foundation. This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health (MOH) and the Ministry of Long-Term Care.
Ethical statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
