Abstract
Purpose:
We conducted a systematic review to examine the use of spinal cord (SC) magnetic resonance imaging (MRI) in clinical trials for multiple sclerosis (MS) and discussed the significant challenges that impede its widespread implementation.
Major findings:
We identified 291 phase II and phase III clinical trials. Of these, 228 (78.4%) included at least one MRI outcome. Eight clinical trials (2.7%) were planned to include at least one SC MRI outcome, but only three (1.0%) reported related methodologies or results. The MRI metrics considered in these studies included cervical SC cross-sectional area (1.0%) and SC T2-lesion number (0.3%).
Conclusion:
A very small proportion of clinical trials have used SC MRI metrics as outcome measures. Given the substantial impact of SC disease on disability, it is essential to standardize SC imaging, post-processing, and analytical methods across centres to enhance the effectiveness and applicability of SC MRI in future clinical trials.
Introduction
Magnetic resonance imaging (MRI) of the brain and the spinal cord (SC) is a vital instrument in the process of diagnosing and monitoring people with multiple sclerosis (PwMS). 1 MRI enables in vivo identification and quantification of both focal lesions and diffuse injury in normal-appearing tissue. 2
Compared to metrics of brain diseases, measures of SC damage show stronger correlations with physical disability. 3 However, SC MRI-related outcome measures have rarely been incorporated into phase II and phase III clinical trials, even in those focusing on people with primary progressive (PwPPMS) or secondary progressive MS (PwSPMS), who carry a heavier burden of SC damage.
In this work, we examine the findings of these clinical trials, highlighting the challenges associated with integrating SC MRI into both single-centre and multi-centre clinical trials. We then discuss the knowledge gaps that must be bridged to enhance the utilization of SC imaging in clinical research.
Methods
Search methods, inclusion criteria and study selection
We searched clinicaltrials.gov without any restrictions on the start date and set 7 May 2025, as the end date. The search terms we used included ‘multiple sclerosis’ or ‘MS’, ‘clinically isolated syndrome’ or ‘CIS’ and ‘radiologically isolated syndrome’ or ‘RIS’. We included all registered clinical trials, regardless of their status, whether active or inactive, and whether they were recruiting or had been terminated. As clinicaltrials.gov was launched on 29 February 2000, two additional searches were performed using the search terms (‘multiple sclerosis’ or ‘MS’, ‘clinically isolated syndrome’ or ‘CIS’ and ‘radiologically isolated syndrome’ or ‘RIS’) AND ‘trial’ AND ‘spinal cord’. The first search was conducted on PubMed and Embase, filtering the results before 2000, and without a limitation on the start date. The second search targeted journals that publish conferences proceedings without constraints to the publication year, including Multiple Sclerosis Journal (the journal of the European and American Committees for Treatment and Research in Multiple Sclerosis – ECTRIMS and ACTRIMS), the Annals of Neurology (the journal of the American Neurological Association – ANA), the European Journal of Neurology (the journal of the European Neurological Association – ENA) and Neurology (the journal of the American Academy of Neurology – AAN).
In all searches, we excluded articles not written in English, Spanish or Italian, as well as reviews, commentaries, registries, observational studies and clinical trials focused on symptomatic pharmacological or non-pharmacological treatments for MS (such as exercise techniques or meditation). The inclusion criteria were as follows: (1) phase II or phase III clinical trials; (2) any disease phenotype, including RIS, CIS, relapsing–remitting MS (RRMS), SPMS and PPMS; (3) any disease-modifying treatment (DMT) and (4) at least one SC MRI-related outcome.
Data extraction
We imported the search results into Rayyan, 4 a tool for screening articles for systematic reviews. We compiled a list of clinical trials based on their titles and abstracts. The first author (A.A.T.) exported these data to an Excel spreadsheet and conducted a full-text review of the trials, discussing the findings with the last author (F.B.). For each study, we extracted the authors’ names, year of publication, involved countries, study design, methods, treatment arms, primary, secondary and exploratory endpoints, and results.
Results
Search results
Our primary search yielded 2223 records, which we screened based on their titles and abstracts. We excluded 1932 of these records because they were observational studies or clinical trials that did not assess the efficacy of a DMT. The supplementary search on PubMed and Embase yielded 207 articles; 68 abstracts were retrieved from ACTRIMS and ECTRIMS, 76 from AAN, 89 from ANA and 63 from ENA proceedings. All these additional articles and abstracts were excluded because they were observational studies, did not assess the efficacy of DMTs, or were already included in the primary search.
The remaining 291 clinical trials were reviewed in full text by the first author (AAT). Of these, 63 (21.6%) did not include any MRI outcomes, while 228 (78.4%) included at least one. Of the 291 clinical trials, eight (2.7%) were planned to include at least one SC MRI outcome, as indicated on clinicaltrials.gov; however, only three performed that SC MRIs and reported the related outcome (1.0%). Table 1 summarizes the clinical trial designs and the planned endpoints, while Table 2 details the SC MRI acquisition and post-processing methods proposed in these eight clinical trials.
Design and results of clinical trials using SC MRI metrics as measures of outcome.
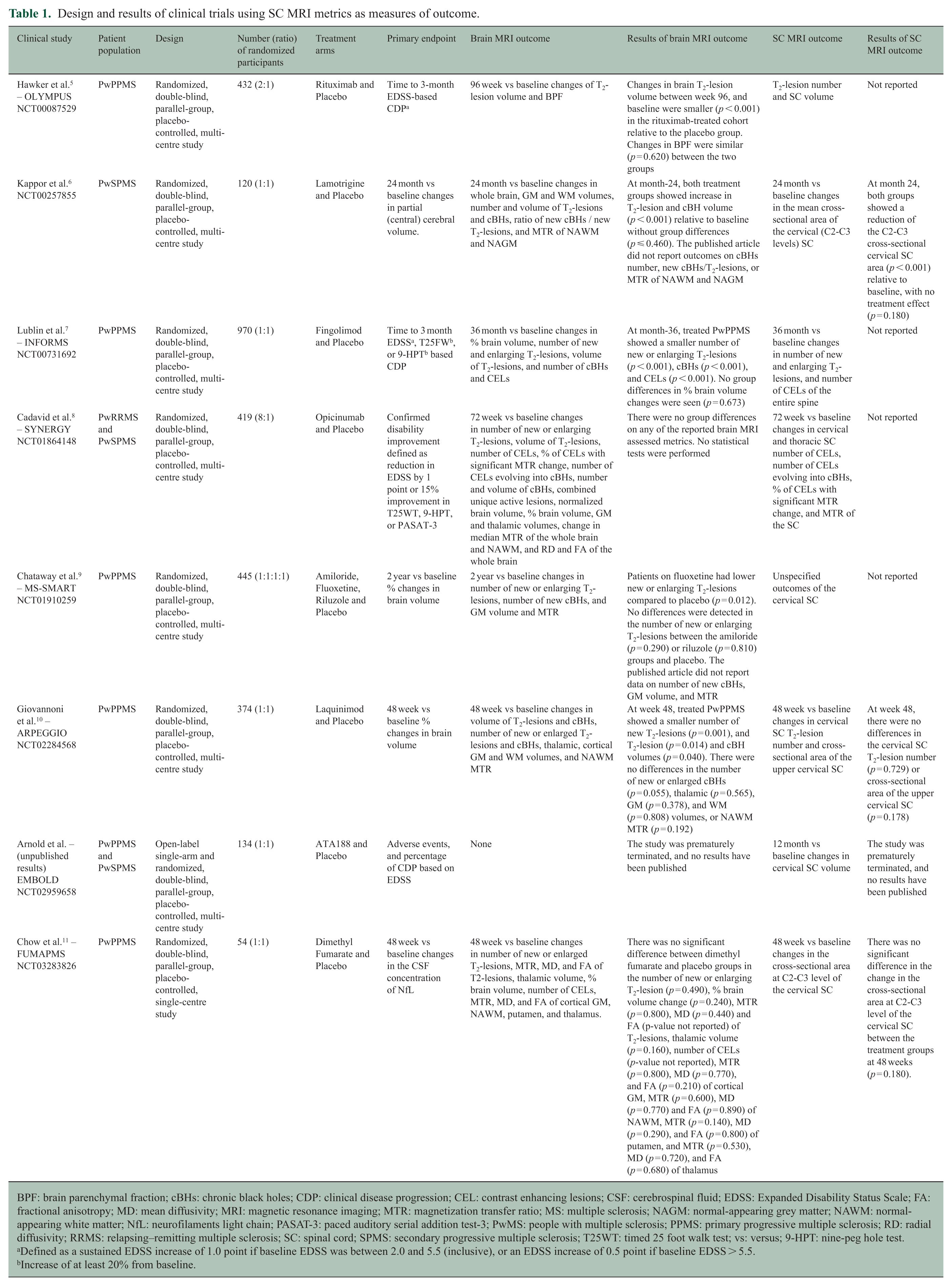
BPF: brain parenchymal fraction; cBHs: chronic black holes; CDP: clinical disease progression; CEL: contrast enhancing lesions; CSF: cerebrospinal fluid; EDSS: Expanded Disability Status Scale; FA: fractional anisotropy; MD: mean diffusivity; MRI: magnetic resonance imaging; MTR: magnetization transfer ratio; MS: multiple sclerosis; NAGM: normal-appearing grey matter; NAWM: normal-appearing white matter; NfL: neurofilaments light chain; PASAT-3: paced auditory serial addition test-3; PwMS: people with multiple sclerosis; PPMS: primary progressive multiple sclerosis; RD: radial diffusivity; RRMS: relapsing–remitting multiple sclerosis; SC: spinal cord; SPMS: secondary progressive multiple sclerosis; T25WT: timed 25 foot walk test; vs: versus; 9-HPT: nine-peg hole test.
Defined as a sustained EDSS increase of 1.0 point if baseline EDSS was between 2.0 and 5.5 (inclusive), or an EDSS increase of 0.5 point if baseline EDSS > 5.5.
Increase of at least 20% from baseline.
SC MRI acquisition and postprocessing protocols.
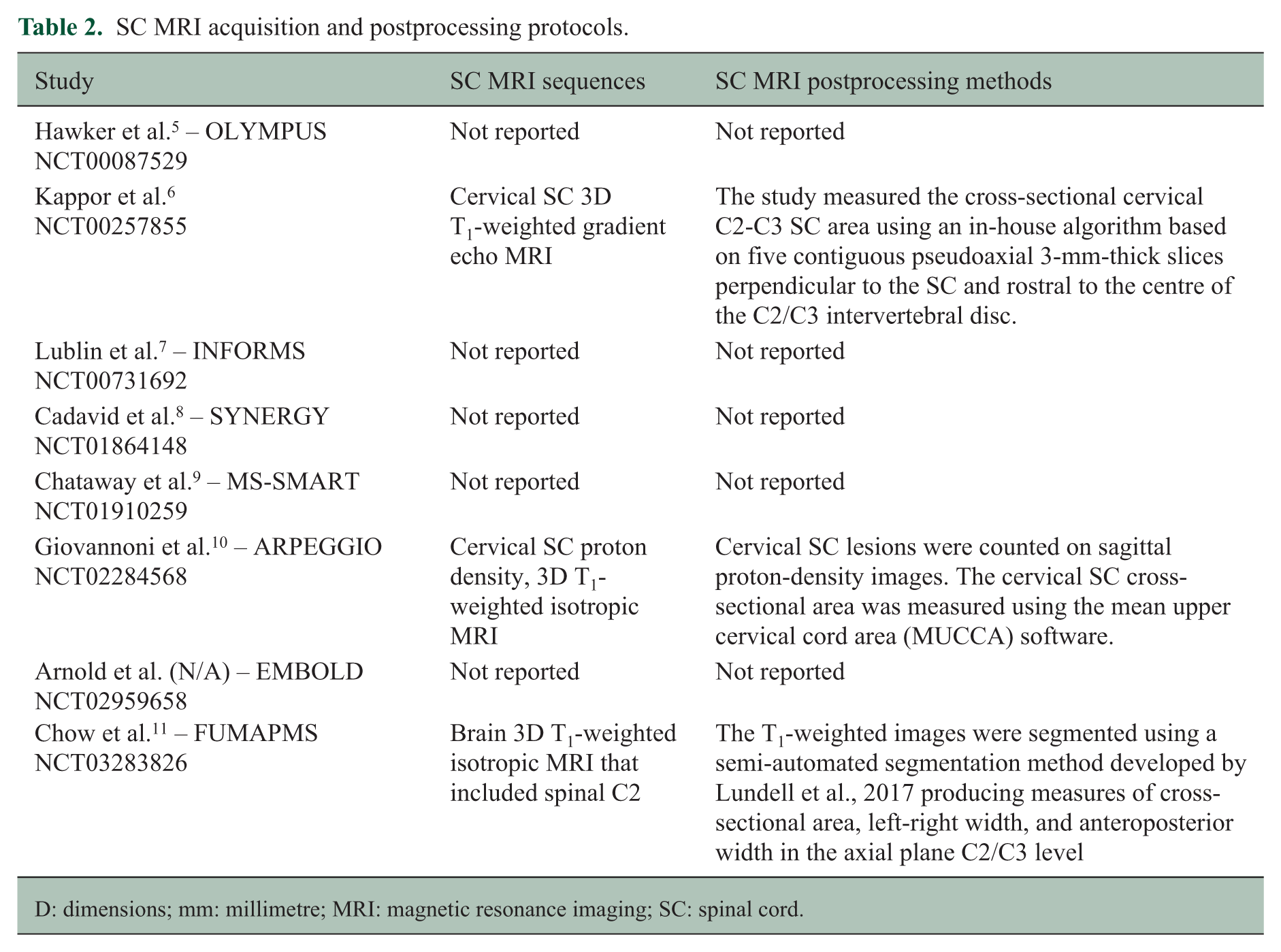
D: dimensions; mm: millimetre; MRI: magnetic resonance imaging; SC: spinal cord.
Analysis of individual clinical trials
Table 1 summarizes the results of the eight clinical trials that planned to or included SC MRI-related measures in chronological order, and we here expand upon these studies.
OLYMPUS (NCT00087529) 5 was a 96-week-long phase II/III clinical trial in which PwPPMS received either 1000 mg rituximab or placebo intravenous infusions at baseline and week 2, 24, 26, 48, 50, 72 and 74 thereafter. The clinical trial did not meet its primary endpoint. Although SC-related outcomes (T2-lesion number and SC volume) were listed on clinicaltrials.gov, the authors did not report SC MRI data in the methods or results sections.
The second clinical trial was a 24-month-long phase II clinical trial investigating the potential neuroprotective effect of lamotrigine (NCT00257855). 6 PwSPMS received either 400 mg of lamotrigine or a placebo orally once daily for 24 months. The participants underwent two cervical SC MRIs, including a three-dimensional (3D) T1-weighted gradient echo sequence at baseline and after 24 months. The clinical trial did not meet its primary endpoint. Regarding SC metrics, the authors measured the cross-sectional SC area at the cervical levels 2 and 3 (C2-C3) using an in-house algorithm based on five contiguous pseudo axial 3-mm-thick slices perpendicular to the SC and rostral to the centre of the C2/C3 intervertebral disc. They reported a significant decrease (p < 0.001) in the C2-C3 cross-sectional SC area at month 24 compared with baseline in both groups. Specifically, reductions of 2.51% and 1.99% were measured in the treated and placebo group, respectively. The magnitude of these changes, however, did not differ between the two groups (p = 0.180).
INFORMS (NCT00731692) 7 was a phase III clinical trial in which PwPPMS received either 0.5 mg oral fingolimod or a placebo once daily for at least 3 years and a maximum of 5 years. The clinical trial did not meet its primary endpoint; however, it showed several between-groups differences in brain MRI focal disease metrics. As shown in the OLYMPUS clinical trial, SC-related outcomes (number of new and enlarging T2-lesions and contrast-enhancing lesions across the entire SC) were listed on clinicaltrials.gov, but not reported in the final manuscript.
SYNERGY (NCT01864148) 8 was a 72-week-long phase II clinical trial in which PwRRMS and PwSPMS received either four different doses of opicinumab (3 mg/kg, 10 mg/kg, 30 mg/kg, or 100 mg/kg) or placebo intravenously once every 4 weeks for 72 weeks. The clinical trial partially met its primary endpoint finding a higher likelihood of confirmed disability improvement in one of the four treatment regimens (30 mg/kg group) relative to placebo (p = 0.022). SC-related outcomes were listed on clinicaltrials.gov, but the authors did not report the acquisition and post-processing methods or results.
MS-SMART (NCT01910259) 9 was a 96-week-long phase II clinical trial in which PwPPMS received amiloride (5 mg), fluoxetine (20 mg), riluzole (50 mg), or a placebo orally twice daily for 96 weeks. The clinical trial did not meet its primary endpoint. SC-related outcomes (cervical SC imaging) were listed on clinicaltrials.gov; however, the authors did not report the SC MRI acquisition and post-processing methods or results.
ARPEGGIO (NCT02284568) 10 was a 48-week-long phase II clinical trial in which PwPPMS received oral laquinimod (0.6 or 1.5 mg) or placebo for 48 weeks. Patients underwent three cervical SC MRIs, including proton density and 3D T1-weighted isotropic sequences at baseline, week 24 and week 48. Cervical SC lesions were counted on sagittal proton-density images. The cervical SC cross-sectional area was measured using the mean upper cervical cord area (MUCCA) method. 12 The clinical trial did not meet its primary endpoint. At week 48, the mean SC T2-lesion count was 2.9 in the laquinimod group and 3.0 in the placebo group, with no significant difference between the two (p = 0.729). The mean change in upper cervical SC area was −1.54 in the laquinimod group and −0.87 in the placebo group with no significant difference between the two groups (p = 0.178).
EMBOLD (NCT03283826) 13 was a phase I and II open-label single-arm dose determination in its first part and a double-blinded parallel-arm randomized trial in its second part. PwPPMS and PwSPMS received either intravenous ATA188 or placebo for 4 years in the first part and once yearly for 5 years in the second part. The clinical trial was discontinued earlier than planned due to a lack of efficacy, and results had not been published at the time of writing this report.
FUMAPMS (NCT02959658) 11 was a 48-week-long clinical trial in which PwPPMS received either 240 mg of oral dimethyl fumarate or a placebo twice daily for 48 weeks. Patients underwent two 3D T1-weighted isotropic MRIs of the brain, extending to the C2 level of the SC, at baseline and at 48 weeks. These images were segmented using a semiautomated segmentation method 14 yielding measurements of the C2/C3 level cross-sectional area, left-right and anteroposterior widths on the axial plane. The clinical trial did not meet its primary endpoint. Regarding SC data, the authors reported only results for changes in the C2-C3 SC cross-sectional area. They found a significant difference in the C2-C3 SC cross-sectional area between baseline and week 48 in the treatment (mean change = −1.614 and 95% confidence interval: −2.880 to −0.348) but not in the placebo (mean change = −0.488 and 95% confidence interval: −1.758 to 0.782) group. These longitudinal differences were similar between the two groups (p = 0.18).
Discussion
The first MS clinical trial dates back 1970, 15 with more than 2000 studies conducted over the past 55 years. However, only eight clinical trials planned to use SC MRI measures. Of these, only one planned to use thoracic SC-related measures, and only three clinical trials reported results for SC-associated outcomes; none included thoracic SC data.
At least in part, these findings may be because assessing brain disease alone has been sufficient to establish the efficacy of DMT with anti-inflammatory properties. Brain MRI is less challenging than the SC MRI, and expertise in it is more widespread.
Unfortunately, this evidence is in contrast to the recognized importance of SC disease. Although more clinically impactful and prominent in PwPPMS or PwSPMS, whose disability and outcome heavily depend on it, SC involvement occurs in all MS disease phenotypes.16,17 For example, 25% of PwRRMS may develop SC lesions within a relatively short time. 18 When this occurs, patients are more likely to experience a clinical relapse. 18 In PwRIS, the presence of SC lesions predicts the development of clinically definite MS.19,20 In PwCIS or with early RRMS, SC lesions are associated with a higher risk of relapses, disability progression and conversion to SPMS.21,22 SC atrophy is also a very important metric in MS. Analyses based on all MS phenotypes combined show that SC atrophy correlates with physical disability as measured using the Expanded Disability Status Scale (EDSS) 23 and contributes to smouldering disease. 17 These findings are also observed in PwRRMS alone for whom SC atrophy predicts progression to SPMS and progression independent of relapse activity (PIRA) events, 24 and appears to be a complementary predictor of disability worsening (along with paramagnetic rim lesions). 25
Some SC regions are more affected than others. A previous multi-centre study demonstrated that central SC and lateral corticospinal lesions are more common in PwPPMS than in PwRRMS. 26 In addition, lesions in the above locations are associated with greater SC atrophy and are significantly correlated with EDSS scores. 26 Both white matter and grey matter tissues are vulnerable to MS injury, 27 but grey matter lesions tend to associated with progressive forms of MS and its associated disability. 28
Therefore, assessing SC microscopic and macroscopic changes using both clinical and quantitative MRI to evaluate the neuroprotective and anti-inflammatory effects of any experimental molecules is undoubtedly informative and clinically relevant. Yet, several technical, logistical and statistical challenges hinder the widespread utilization of SC MRI.
Technical challenges associated with SC imaging
At the acquisition side, SC imaging faces several technical challenges related to image resolution, signal-to-noise ratio (SNR) and artefacts. 29 Those challenges are even more critical for the thoracic and lumbar tracts of the SC, given their thinner size relative to the cervical SC.29,30
First, achieving high resolution is limited by hardware capabilities like magnetic field gradients and radiofrequency coil design, as well as by the need to maintain adequate SNR. 29 The small size of the SC and its surrounding complex anatomy require fine voxel sizes, but smaller voxels reduce signal strength, leading to longer scan times and potential motion artefacts. 29 Pulse sequences, such as T2-fast spin echo, are commonly used to balance contrast, resolution and scan speed. Although some sequences, such as the fluid-attenuated inversion recovery, are effective for brain imaging, they are less reliable in the SC due to motion and poor cerebrospinal fluid suppression. 29 SNR is further limited because the SC occupies a small portion of the imaged volume, and surface coils used for spine imaging, while improving SNR locally, can introduce inhomogeneity. 29 Artefacts such as magnetic susceptibility (from tissue or metal), truncation (Gibbs artefact), motion and slice saturation are common, and each requires specific mitigation strategies. 29
Second, structures with different magnetic susceptibility properties, such as bones, soft tissues and air, surround the SC. This heterogeneity of tissue composition easily leads to image distortions and signal loss. To overcome this challenge, most MRI protocols implement shimming procedures. These procedures effectively resolve spatial inhomogeneity but tend to fail in settings with slight and localized variations, such as cartilaginous discs between vertebral bodies. 31
Third, the SC moves in conjunction with respiration, movements of the heart, lungs, surrounding visceral organs and vessels, and the cerebrospinal fluid pulsation. 32 These movements may degrade image quality with motion artefacts. 33 The MRI acquisition needs to respect the directionality of these anatomical structures to achieve the highest resolution and quality.
Challenges related to SC MRI post-processing: Atrophy and lesion burden measurements
Challenges arise at the post-processing stage as well. Given the notion that tissue loss is an essential component of disease pathology in MS and a significant determinant of physical impairment, 16 SC cross-sectional area measurements have been used as indirect estimators of SC atrophy. Several methods have been developed; however, irrespective of the methodology, several challenges are associated with SC cross-sectional area measurements. First, rater bias can affect manual segmentation of the cross-sectional area. Second, the SC cross-sectional area has a high degree of variability in morphology among individuals, that varies by age, sex, height, among others, with a standard deviation as high as 9%34,35 in contrast to the anticipated atrophy changes of 2% per year due to MS. 24 For example, Kappor and collaborators 6 measured a 2.5% change in the cervical SC cross-sectional area over 2 years, and the FUMAPMS clinical trial 11 reported a 1.6% change. Third, pseudoatrophy may affect SC area measurements. Pseudoatrophy is attributed to the early pseudo reduction of SC volume after initiation of DMT, due to a reduction of inflammatory components (oedema and cells). 36 Last, SC measurements of the cross-sectional SC areas remain limited to a specific SC region.
To overcome these limitations and improve longitudinal sensitivity, advanced semi-automated and automated techniques have been established. The active surface method (as implemented in Jim software) uses localized surface evolution to quantify atrophy with high reproducibility, demonstrating superior sensitivity to longitudinal changes and reduced sample size requirements for clinical trials compared to manual techniques.37,38 Furthermore, state-of-the-art registration-based methods, such as the generalized boundary shift integral (GBSI) 39 and SIENA-SC, 40 now allow for the direct quantification of voxel-wise volume changes over time, offering robust solutions for monitoring neurodegeneration in progressive MS cohorts. More recently, deep learning frameworks like DeepSeg 41 and contrast-agnostic models42,43 have automated segmentation across varying MRI contrasts, though monitoring for morphometric drift due to algorithmic changes and updates remains essential in longitudinal applications. 43
SC lesion burden is another outcome measure utilized in clinical trials. However, SC lesions are smaller than brain lesions. 44 Therefore, their manual segmentation is time-consuming and has significant inter- and intra-rater variability. 45 Automated lesion segmentation offers a potential solution to this problem, 46 but most algorithms are unable to produce reliable results in the SC due to the small size of the lesions relative to the surrounding structures. Even when feasible, the inter-centre reliability of these algorithms still needs to be evaluated.
Statistical challenges
The literature indicates that approximately 80% of PwMS develop SC lesions; thus, about one in five study participants would not have overt SC disease. 47 When present, the SC MS disease burden tends to be less extensive compared to that of the brain. 48 These two factors can significantly affect power analyses, necessitating a substantially larger sample size. Similarly, SC cross-sectional area changes over time are typically small, with studies reporting an average annual change of 2%. 24 To be measurable, the magnitude of such changes requires large cohorts monitored for relatively long periods. While large, long studies can be expensive and challenging, underpowered studies pose a risk of failure due to statistical limitations and difficulties interpreting the data.
Logistical challenges
Several logistical challenges also impact the utilization of SC MRI in clinical trials. First, financial constraints can explain the low use of SC MRI, as the cost of SC MRI is higher compared to that of brain MRI, 49 and implementing both can significantly impact clinical trials’ budgets. For the same reasons, authors may not feel compelled to pursue SC planned analyses when the primary (brain MRI or disability-related) endpoint is not met. Scan time is also another limiting factor, as adding SC imaging would triplicate it. 49 Finally, the generally lower expertise in SC imaging limits its utilization in clinical trials. 50 Together, these factors can result in the inability of some institutions to participate in a clinical trial or to recruit an adequate number of participants.
General implications of our findings and future directions
The absence of SC implementation in clinical trials prevents assessment of changes in the structural integrity of regions that directly affect the motor disability of PwMS. It remains to be determined whether the drug effect detected on SC MRI translates directly into improved motor disability outcomes, or whether brain disease changes alone can still serve as a surrogate measure. Discussions should be held among clinicians, scientists and industry representatives to determine whether resolving the current challenges associated with SC MRI should be a priority.
Should a consensus be reached on this goal, a multi-step parallel approach must be undertaken to advance the field.
At the acquisition side, improvements in acquisition protocols (including reduced field-of-view strategies, 51 accelerated imaging sequences,52 –54 advanced hardware and higher field strengths)55,56 have increased SNR and enhanced spatial resolution. However, new imaging methods are needed to increase further SNR and spatial resolution, which advances in coil design, shimming methods and motion artefact correction can achieve. 57 To this end, a partnership between academia and industry (MRI manufacturers) is fundamental. Although incorporating SC imaging into clinical trials may present particular challenges, designing time-efficient acquisitions that enable both lesion count and atrophy measurements would be of substantial benefit.
Regarding SC outcome measures, dedicated pipelines now incorporate motion correction, 58 distortion correction59 –61 and artefact mitigation tailored to the SC. However, harmonizing quantitative MRI methods across centres is urgently needed. Notably, the emergence of standardized toolboxes 58 and template-based frameworks 62 enable anatomically consistent region-of-interest definition and reproducible feature extraction across individuals and sites,63,64 but additional studies are needed to assess the variability and accuracy of these metrics within subjects and across centres. Last, authors and sponsors should feel encouraged to pursue SC analysis and report data even when a clinical trial does not meet the primary or brain MRI-related endpoints. An improved report of clinical trial results, specifying the circumstances that led to the non-performance or non-reporting of SC-related analyses, can be very instructive for understanding the challenges and designing mitigation strategies.
Conclusion
SC clinical and quantitative MRI techniques can provide insight into SC microstructure, function and integrity; however, their implementation in clinical trials remains limited. Studies harmonizing acquisition and post-processing methods within centres and across institutions are urgently needed to facilitate the clinical translation and widespread use of SC MRI in clinical trials. The implementation of these methods would benefit PwMS, whose disability is extensively explained by SC disease, and the design of proof-of-concept clinical trials assessing the neuroprotective effects of experimental molecules.
Footnotes
Acknowledgements
This manuscript is dedicated to the sweet memory of Mr Oscar Castro.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: Support includes the Veterans Health Administration (I01CX002160-01A1: A.A.T. and F.B.) and the Voros Innovation and Impact Fund (F.B.).
Data Availability
Data are available to share upon reasonable request to the corresponding author.
