Abstract
Background:
Fatigue is common in multiple sclerosis and may be influenced by cardiovascular comorbidities.
Objective:
To test whether cardiovascular comorbidity burden relates to fatigue at baseline and over time, and to assess dose response.
Methods:
We analyzed prospectively collected data from MS PATHS across 10 centers. Fatigue was measured with Neuro-QoL Fatigue
Results:
Among 5507 participants, 49% had no cardiovascular comorbidity, 32% had one, 16% had two, and 3% had three. In minimally adjusted cross‑sectional models, any comorbidity was associated with higher fatigue (beta = 0.85; 95% CI, 0.18 to 1.53), with attenuation after adjustment for depression, anxiety, and sleep (beta = 0.20; 95% CI, −0.09 to 0.49). A graded dose response was evident across 0–3 conditions. In longitudinal analyses, effect sizes were larger and remained positive after adjustment (Any vs None: beta = 1.06 in minimal models; beta = 0.39 after adjustment), consistent with a greater comorbidity burden associated with higher fatigue over time.
Conclusions:
Cardiovascular comorbidity burden is associated with greater fatigue, with a dose response over time.
Keywords
Introduction
Fatigue is a pervasive and multifaceted symptom of multiple sclerosis (MS) that affects cognitive, physical, and psychosocial domains.1 –3 It shapes daily activities and employment and is not fully explained by neurologic disability or MS disease activity. 4 More than 30 factors, including comorbidities such as depression, anxiety, and sleep disorders, are associated with MS fatigue. 5 Cardiovascular (CV) conditions such as dyslipidemia, diabetes, and hypertension (HTN) are common among people living with MS (pwMS) and have been linked to worsening disability and lower performance on objective physical and cognitive testing.6,7 What is less clear is how these vascular conditions relate to fatigue severity. In this study, we examined the association between CV comorbidities and fatigue in a large cohort of pwMS.
Methods
Study design and data source
We conducted an observational study using prospectively collected clinical and patient-reported data from the Multiple Sclerosis Partners Advancing Technology and Health Solutions (MS PATHS) learning health system. MS PATHS enrolled people with multiple sclerosis (MS) across 10 specialty centers in the United States and Europe and captured standardized information during routine care using the Multiple Sclerosis Performance Test8,9 (MSPT; iPad-based self-administration of questionnaires and quantitative neuroperformance modules).6,10 Core clinical measures, including height, weight, and blood pressure, were abstracted automatically from the electronic medical record according to a uniform workflow across sites. Local ethics committees or review boards at participating centers approved data collection and participants provided informed consent. Biogen funded MS PATHS.
Study population and eligibility
Analyses were performed on harmonized datasets containing Neuro-QoL11,12 patient-reported outcomes and clinic-acquired clinical measurements for the same individuals. Participants were eligible if they had at least one Neuro-QoL Fatigue
Exposure: cardiovascular comorbidities
We examined three cardiometabolic comorbidities, hypertension (HTN), diabetes, and dyslipidemia, 6 and operationalized CV comorbidity in two ways:
Composite (any vs none).
Count (0, 1, 2, or 3 comorbidities); in sensitivity analyses, we collapsed the top categories to 0/1/⩾2.
Hypertension
Primary definition: systolic BP ⩾ 140 mmHg or diastolic BP ⩾ 90 mmHg on ⩾ 2 different calendar dates. Sensitivity definition: systolic ⩾ 130 or diastolic ⩾ 80 on ⩾ 2 dates (reflecting 2017 ACC/AHA “stage 1” thresholds).6,15 Once a participant met the criteria, they were considered hypertensive from baseline onward for all analyses. Only measured BP data were used to define HTN. Diagnosis codes were not consistently available in this research extract, so treated and well-controlled hypertension may be missed.
Diabetes and dyslipidemia
These were identified from medication histories in the MS PATHS feeds consistent with the drug classes cataloged in prior MS PATHS analyses (e.g. biguanides, DPP-4 inhibitors, GLP-1 agonists, SGLT-2 inhibitors, sulfonylureas, thiazolidinediones for diabetes; statins, bile-acid sequestrants, ezetimibe, fibrates, fish oil, niacin, PCSK9 inhibitors for dyslipidemia). 6 If present at any timepoint, the condition was treated as present from baseline onward. As a sensitivity analysis, we redefined comorbidities with a timing constraint anchored to baseline: diabetes and dyslipidemia were counted present only if their first positive record occurred on or before the date of the first Neuro‑QoL Fatigue T‑score or within 1 year thereafter, and hypertension was counted present only if there were ⩾ 2 clinic dates with SBP ⩾ 140 mmHg or DBP ⩾ 90 mmHg and the earliest elevated date fell on or before baseline + 1 year (primary threshold; sensitivity defined analogously at ⩾ 130/80).
Once exposure criteria were met, comorbidity status was carried forward to avoid reverse causation and to align with the pragmatic MS PATHS data structure.
Outcome: fatigue
The outcome was the Neuro-QoL Fatigue
Covariates
We prespecified potential confounders based on prior literature and a directed acyclic graph for comorbidity-fatigue relations: baseline age, sex, race (White, Black, other), ethnicity (Hispanic vs non-Hispanic), BMI (kg/m2), smoking (never, former, current, unknown), PDDS (0 to 8; a patient-reported disability scale), and site. In extended models, we also included Neuro-QoL depression, anxiety, and sleep disturbance
Primary analyses
Cross-sectional (baseline)
We used ordinary least squares (OLS) regression to estimate the association between CV comorbidity and baseline fatigue. All models included site fixed effects and heteroskedasticity-consistent standard errors clustered by site. We fit (1) “minimal” models adjusting for confounders and (2) “extended” models that further included depression, anxiety, and sleep scores. For dose response, the comorbidity count was entered as a categorical factor (reference = 0; 1/2/3) with a complementary linear trend test treating count as a continuous term.
To address residual confounding, we implemented inverse probability weighting (IPW) targeting the average causal effect for the binary composite exposure (having ⩾ 1 CV comorbidity). 16 Propensity scores were estimated via logistic regression including the prespecified baseline covariates (age, sex, race, ethnicity, BMI, smoking, PDDS, and site). Stabilized weights were computed and truncated at extreme values (1st and 99th percentiles, and absolute weight capped at 10) to improve finite-sample performance. Covariate balance was assessed using absolute standardized mean differences (SMD), with |SMD| < 0.10 indicating adequate balance. We also report the effective sample size after weighting (Supplementary Table 1).
Longitudinal
We modeled repeated fatigue scores using linear mixed-effects models (LMM) with a random intercept for participant to account for within-person correlation. Time since baseline (days) was included as a natural cubic spline (knots at the 10th, 50th, 90th percentiles) to allow non-linear trajectories; we tested a time × exposure interaction to assess effect variation over follow-up. Fixed-effects structure: exposure (binary or count), the prespecified confounders, site, and time terms. As a sensitivity check, we fit generalized estimating equations (GEE) with an exchangeable working correlation at the participant level.
Sensitivity and subgroup analyses
Alternate HTN threshold (⩾ 130/80 mmHg on ⩾ 2 dates).
Potential mediator handling: minimal versus extended models.
Dose response recoded as 0/1/⩾ 2 comorbidities.
Effect modification by disability: interaction between exposure and PDDS.
Functional forms: restricted cubic splines for age, BMI, and PDDS.
Depression restriction: exclusion of participants with severe depression (Neuro-QoL T > 60).
Model family: LMM versus GEE for longitudinal correlation structure.
Missing data
For the cross-sectional analysis we used complete-case OLS. Longitudinal LMMs used all available observations per participant under a missing-at-random assumption, conditional on included covariates.
Statistical reporting
Two-sided
Ethics and data governance
MS PATHS data collection was approved by local ethics committees or review boards; all participants provided informed consent. Data are held by participating centers and/or the MS PATHS program and are not publicly available, consistent with prior MS PATHS publications.
Results
Among 5507 participants with baseline fatigue data, 49% had no CV comorbidities, 32% had one, 16% had two, and 3% had three. Median follow-up was 24 months (interquartile range, 13–35). Table 1 summarizes baseline characteristics by comorbidity count. Participants with two or more comorbidities were older (mean age 62.3 years for two and 62.0 years for three vs 49.7 years for zero), had higher BMI, greater disability (median PDDS 2.0 vs 1.0), and a higher prevalence of former or current smoking. The proportion of women ranged from 78% in those with no comorbidities to 68% in those with three. Race distribution was similar across categories, with roughly 11% to 13% identifying as Black and 5% to 8% as other races. Baseline fatigue scores increased monotonically with comorbidity count (mean = 50.1, 50.6, 51.4, and 52.5 for 0, 1, 2, and 3 comorbidities, respectively). Follow-up time and the number of fatigue assessments were similar across categories (Table 1).
Baseline characteristics of all participants and by comorbidity count.
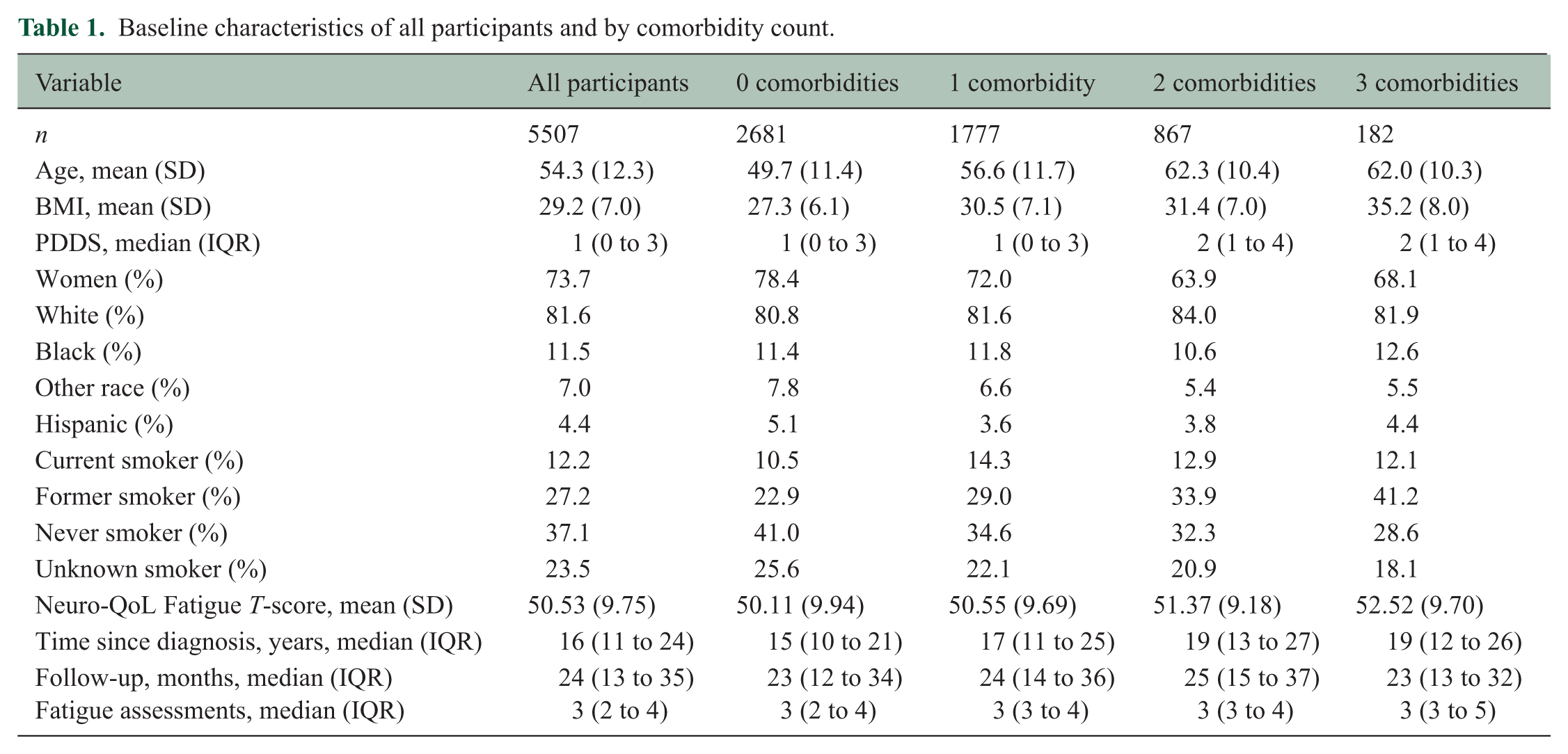
Table 2 presents the cross‑sectional regression results. In the minimal model, participants with any CV comorbidity reported 0.85 (95% CI = 0.18 to 1.53;
Cross‑sectional associations between CV comorbidity and baseline fatigue.
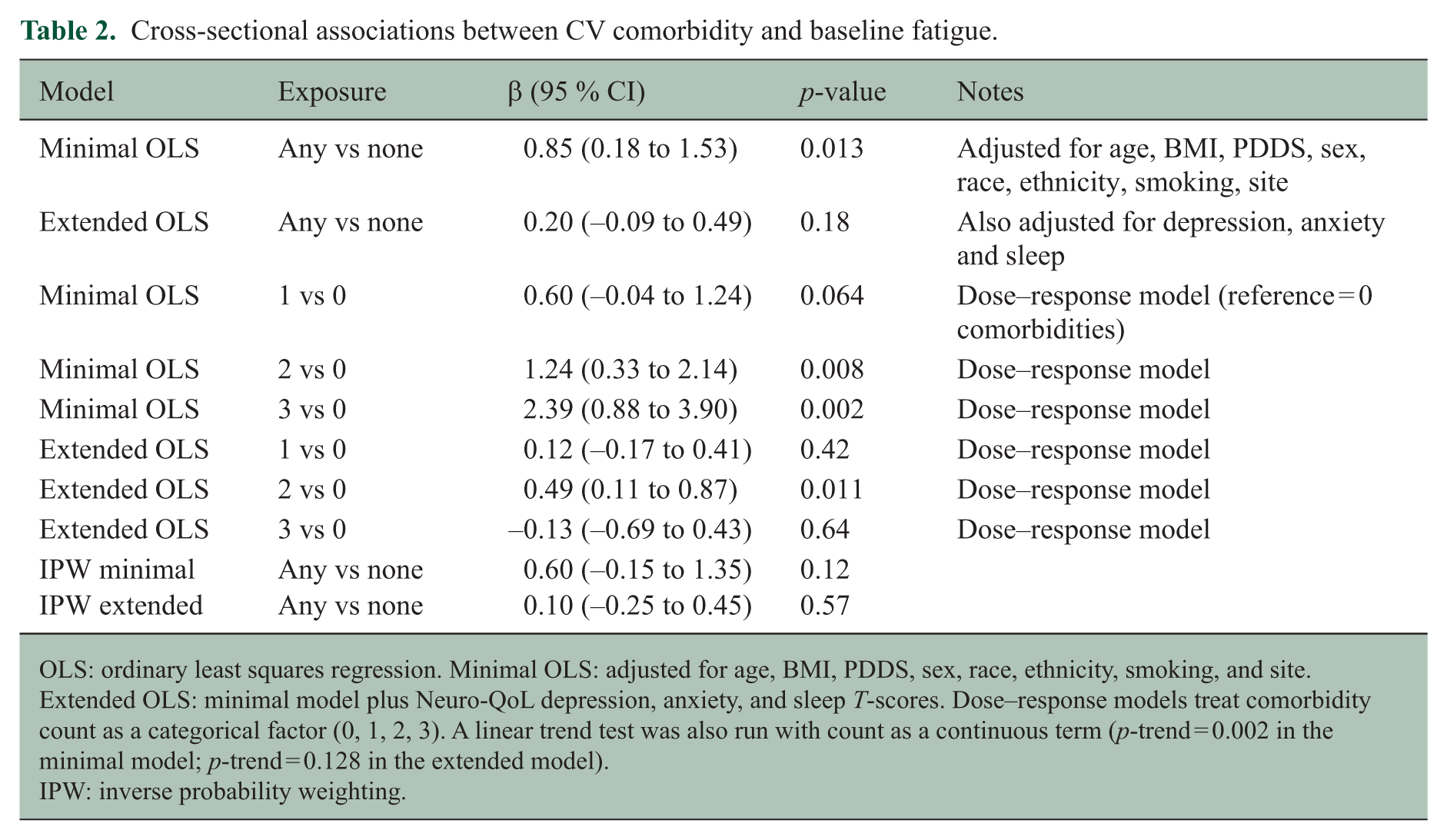
OLS: ordinary least squares regression. Minimal OLS: adjusted for age, BMI, PDDS, sex, race, ethnicity, smoking, and site. Extended OLS: minimal model plus Neuro-QoL depression, anxiety, and sleep
IPW: inverse probability weighting.
Participants contributed a mean of 3.5 fatigue assessments (range, 1 to 15) over a median follow-up of 24 months. In linear mixed-effects models (random intercepts for participant), any CV comorbidity was associated with higher fatigue
Longitudinal associations between CV comorbidity and fatigue.
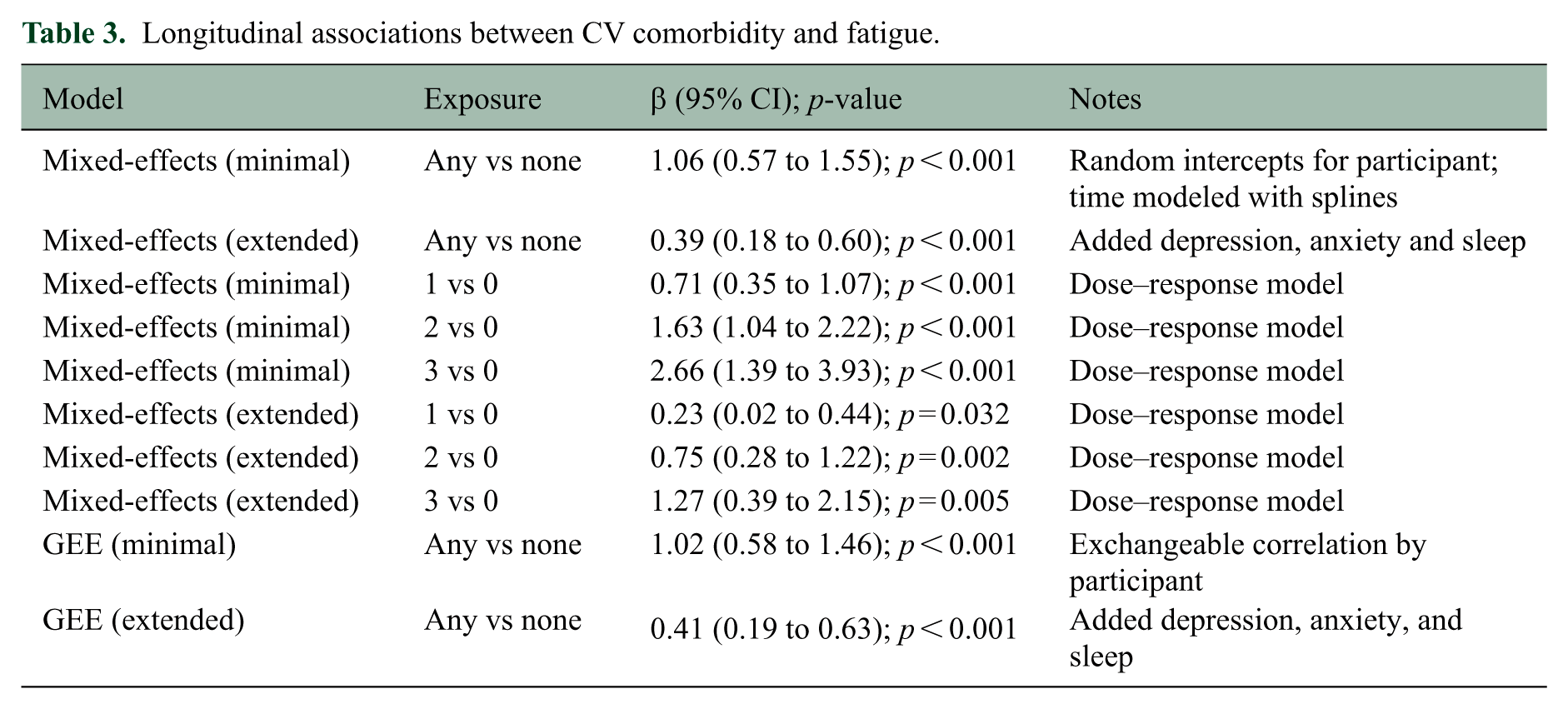
Table 4 summarizes the sensitivity analyses results. IPW reduced covariate imbalance between groups (|SMD|< 0.1 for all variables after weighting) and attenuated the association between CV comorbidity and fatigue. Because IPW re-weights the sample to balance observed confounders, it provides a complementary estimate that accounts for group differences in age, BMI, and other factors; however, it also reduces effective sample size and widens confidence intervals, which limits statistical power. The weighted minimal model estimated a 0.60‑point difference (
Sensitivity analyses (cross‑sectional).
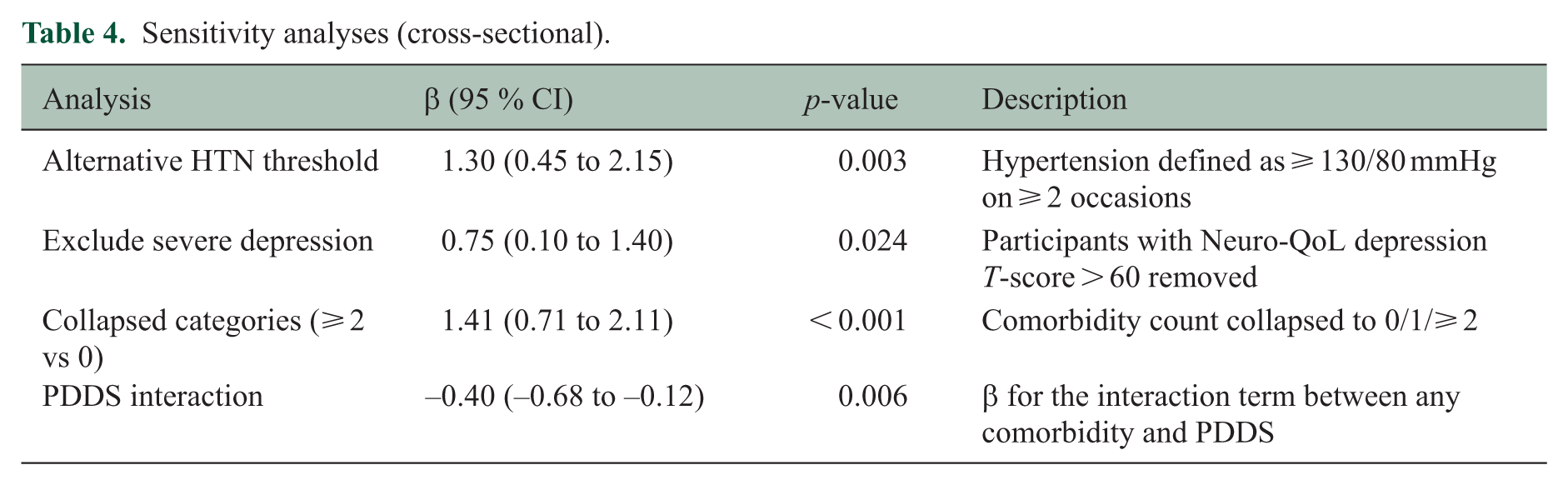
Findings were materially unchanged under the timing‑restricted definition; comorbidity prevalence shifted modestly (fewer participants with ⩾ 2 conditions), but the estimated associations with fatigue in cross‑sectional and longitudinal models were similar in magnitude and interpretation to the primary analyses (e.g. Any vs None minimal model β ≈ 0.83 vs 0.84).
Discussion
In this large, multi‑center cohort of people with MS, we observed that cardiovascular comorbidities were associated with higher Neuro‑QoL fatigue
Our longitudinal analyses corroborated and extended the cross‑sectional findings. CV comorbidities predicted higher fatigue over follow‑up, and the dose–response pattern persisted. The absence of a significant time x comorbidity interaction suggests that the effect is relatively constant over the observed follow‑up. IPW reduced covariate imbalance and attenuated effect sizes, highlighting that some of the associations may reflect differences in age, BMI, disability, and other factors. Importantly, however, IPW targets a different estimand (the average causal effect of the binary composite) and necessarily reduces effective sample size, inflating variance and limiting statistical power, particularly for moderate effect sizes. Because IPW does not accommodate the dose–response structure of the exposure and relies on additional untestable assumptions (e.g. no unmeasured confounding of the exposure‑outcome relationship), we positioned it as a sensitivity analysis rather than the primary approach. The consistency in direction across OLS, IPW, mixed-effects, and GEE models strengthens confidence that the observed association is not an artifact of a single analytic choice. This is consistent with the smaller, but persistent, associations observed in longitudinal models that better account for stable individual differences. We found that the association was weaker among individuals with greater disability which may reflect a ceiling effect. When disability is high, other factors may contribute more heavily to the fatigue experience.
Mechanistically, several pathways might link CV comorbidities with fatigue in MS. Chronic vascular disease can induce systemic inflammation, oxidative stress, and endothelial dysfunction, all of which may amplify central fatigue. 18 Cerebrovascular dysfunction can reduce cerebral blood flow and impair clearance of metabolic by‑products, contributing to fatigue. Autonomic dysregulation and orthostatic intolerance, commonly reported in hypertension and diabetes, may exacerbate fatigue by impairing cardiovascular responses to physical activity. Finally, comorbidity‑related deconditioning may further heighten fatigue. Future studies incorporating biomarkers and imaging of vascular health could delineate these mechanisms.
Cardiovascular comorbidity has been linked to several aspects of MS course. Early registry work showed that people with MS who have vascular comorbidities reach disability milestones sooner, pointing to a faster trajectory of progression in those with hypertension, diabetes, or hyperlipidemia. 21 Multicenter imaging studies reported an association between higher vascular comorbidity burden and smaller brain volumes and poorer objective performance, while a study focused on secondary progressive MS found reduced whole brain volume, particularly cortical gray matter, in those with vascular risk, with limited evidence for short‑term acceleration of atrophy rates.6,22 Cognition fits this pattern as well, as vascular comorbidities are associated with worse neuropsychological performance, partly through structural brain changes. 23 Regarding fatigue, prospective cohort data indicate that a greater comorbidity load is associated with higher and more persistent fatigue, and that the link is not explained solely by depression, even though mood remains an important correlate. 17 Evidence that points specifically to the association of cardiovascular conditions and invisible symptoms is emerging: a recent analysis reported that diabetes and other vascular comorbidities were associated with worse self‑rated functional outcomes, with disability, depression, and fatigue accounting for much of the relationship; 24 related claims work also shows that patients with fatigue tend to carry more comorbidities and are at greater risk of later disability. 25 Study designs, exposure definitions, and adjustment strategies varied among these studies, so results are not perfectly aligned across the literature, yet the overall direction is consistent with cardiometabolic burden association with more disability, less favorable MRI profiles, and worse invisible symptoms, including fatigue.
Key strengths are the sample size, a multicenter cohort with repeated measures, and the use of standardized Neuro-QoL instruments alongside clinic-acquired measurements. We adjusted for site and ran IPW and sensitivity analyses to probe confounding. Several limitations should be kept in mind. CV comorbidities were measured using clinic blood pressure and medication-based proxies for diabetes and dyslipidemia, so misclassification is possible. For example, some agents can be used for weight management or metabolic indications, which can inflate apparent diabetes prevalence. Disease-modifying therapy status and symptomatic fatigue medications were not available in this extract, so we could not adjust for them. Race and ethnicity were captured as harmonized categories in MS PATHS; finer-grained categories were not available in this extract. Last, participants were drawn from specialty MS centers, so the findings may not generalize to settings with different access to care, socio-economic profiles, or racial and ethnic composition.
Conclusion
Cardiovascular and metabolic comorbidities are modestly but consistently associated with greater fatigue in MS, with evidence of a dose–response relationship. While part of this association may operate through depression, anxiety, and sleep disturbance, CV comorbidity burden remains an important factor. Clinicians should assess and manage vascular risk factors in people with MS not only to reduce cardiovascular morbidity but also to potentially alleviate fatigue. Future research should explore mechanisms linking vascular health to fatigue and evaluate whether interventions targeting comorbidities or their sequelae improve patient‑reported fatigue.
Supplemental Material
sj-docx-1-msj-10.1177_13524585261438014 – Supplemental material for Cardiovascular comorbidities are associated with cross-sectional and longitudinal measures of multiple sclerosis fatigue
Supplemental material, sj-docx-1-msj-10.1177_13524585261438014 for Cardiovascular comorbidities are associated with cross-sectional and longitudinal measures of multiple sclerosis fatigue by Mahsa Ghajarzadeh, Ellen M Mowry, Kathryn C Fitzgerald and Bardia Nourbakhsh in Multiple Sclerosis Journal
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.G. and K.F. have no disclosures. E.M.M. has received research support from Biogen and Genentech, and editorial royalties from UpToDate. B.N. has received research support from Genentech and Axsome Therapeutics and consulting honoraria from TG Therapeutics and Alkermes.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The MS PATHS network was funded by Biogen.
Ethical considerations
MS PATHS data collection was approved by the ethics committees or review boards at all participating centers, and all participants provided informed consent, as detailed in the Methods.
Consent to participate
All participants provided informed consent to participate in MS PATHS.
Consent for publication
Not applicable (no identifiable individual data are presented).
Data availability statement
The data that support the findings of this study are derived from the MS PATHS network and were used under a data use agreement. These data are not publicly available and cannot be shared by the authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
