Abstract
Background:
Neurological care and disease-modifying therapies (DMTs) are central to multiple sclerosis (MS) management, but how comorbidities like cancer affect them remains unclear.
Objective:
We assessed the impact of cancer on MS-related healthcare and DMT use.
Methods:
This cohort study accessed population-based data from France (2009–2021) and British Columbia, Canada (1991–2020). Cases were individuals with MS diagnosed with an incident cancer. The 4-year study period encompassed the 2 years before and after the cancer diagnosis. Each case was matched to two cancer-free individuals with MS (i.e. controls). Outcomes were neurologist visit rates, MS hospitalization rates, and percentage of DMT users. Mixed-effects models were used with a knot at cancer diagnosis. Incidence rate ratios (IRRs) were estimated pre- and post-cancer (cases vs. controls) and compared using a slope ratio (SR).
Results:
In total, 6902 cases were matched to 13,804 controls. There was a negative effect of cancer on DMT use (SR = 0.36 [0.19–0.70]), especially among those on chemotherapy (SR = 0.24 [0.14–0.40]). Neurologist visit IRRs did not differ pre- and post-cancer (SR = 0.96 [0.91–1.005]). Similarly, MS hospitalization IRRs did not differ (SR = 1.06 [0.84–1.33]).
Conclusion:
Following cancer, DMT use declined sharply. However, there was no evidence of a strong impact of cancer on neurologist visits and MS hospitalizations.
Introduction
People with multiple sclerosis (pwMS) are living longer, although their life expectancy remains 5–10 years shorter than the general population.1 –3 In France and North America, the average age of pwMS has increased, and the majority are aged 50 years or older.4 –7 Consequently, the prevalence of age-related comorbidity is increasing in this population, particularly cancer. However, how this dual diagnosis is being navigated by health professionals and patients and its impact on MS-related care remains to be determined.
Several studies have examined cancer risk in MS, but little is known about cancer effect on MS-related care.8 –13 A mixed-methods study looked at the clinical course of MS after a breast cancer diagnosis and found that about half the patients who were on a disease-modifying therapy (DMT) at cancer diagnosis discontinued or changed their DMT. It also reported that about half of the cohort continued a stable MS management routine, whereas 25% switched focus on the cancer, putting MS care aside. 14 However, this study was limited to 43 participants, focused only on one cancer type, and did not include a comparison with a representative cancer-free MS population.
Another smaller study on 16 individuals with MS and central nervous system (CNS) cancer found reduced inflammatory activity in patients undergoing chemotherapy compared with those who did not. 15 In some older studies from the 1990s, radiotherapy for CNS tumors using radiation techniques not typically used at present was linked to MS exacerbations; however, this may not be comparable to conformal or stereotactic radiotherapy techniques.16,17
Studies on comorbidities in MS have focused on cardiovascular, autoimmune, and mental diseases and failed to include cancer.18 –21 Moreover, narrative reviews have shown that there is no unified practice regarding MS management after a cancer diagnosis and is highly dependent on the practitioner’s judgment and experience as well as the type, severity, and treatment of cancer.22 –24 The French MS society recently released guidelines on the topic, highlighting that evidence on MS care pathways after cancer diagnosis remained sparse. 24 Research is needed to accurately capture the impact of cancer on MS-related care in order to identify current practices and potential disparities in access to care.
Here, we assessed the impact of cancer on MS-related care and DMT use in a multinational matched population-based retrospective cohort study using administrative data.
Materials and methods
Data source
Population-based health administrative data from France (SNDS) and the province of British Columbia (BC) in Canada were used. Both study regions (total = 73 million residents) retain electronic records of sociodemographic data and health service use for nearly all residents (~99%). Information on dates of birth and death, sex, and place of residence is available, as well as on hospitalizations, prescriptions filled, and physician visits. 25
In both regions, each hospitalization is associated with an admission and discharge date, one principal discharge diagnosis code, and multiple associated diagnosis codes from the International Classification of Diseases (ICD; France = ICD-10; BC = ICD-9 or 10). Each physician visit is associated with a visit date and the physician’s specialty. Only in BC, physician visits are associated with up to five ICD-9 diagnoses. In addition, in France, all medical procedures (e.g. chemotherapy or radiotherapy) are recorded. Moreover, patients with a chronic disease that requires either long or expensive treatments can obtain the long-term disease (LTD) status (from a specific list, including MS and cancer).
MS participants
PwMS were identified through a healthcare use-based algorithm in both regions. In BC, MS was defined as ⩾3 MS ICD codes (ICD-10:G35/ICD-9:340 in hospitalizations or physician visits) or ⩾1 filled prescription of an MS-specific DMT (Supplemental Table 1). After identification, the MS diagnosis date was defined as the date of the first MS or demyelinating-related diagnostic code or the first MS-specific DMT prescription filled (Supplemental Tables 1 and 2).
In France, as records do not include diagnosis codes of physicians’ visits, pwMS were identified using an algorithm developed in collaboration with neurologists and medico-administrative database experts, that had been extensively used in previous studies.4,26,27 MS was identified through MS hospitalizations, MS-specific drug reimbursements, or MS-LTD status. 4
Study design and population
This multinational cohort study investigated the impact of cancer on MS-related healthcare use. The study population comprised two population-based cohorts of pwMS. Data were available: 1 January 1991 to 31 March 2020 in BC and 1 January 2009 to 31 December 2021 in France.
Cases were defined as ⩾18 year-old pwMS with an incident cancer. The index date was defined as the first cancer-related event (Supplemental Tables 3 and 4). A cancer was considered incident if there was no evidence of cancer in the 3 years pre-index date. Each individual’s follow-up period included the 2 years before and after the first cancer-related event (index date) (Figure 1). Therefore, cancer cases were included from 1 January 1994 to 31 March 2018 (BC) and 1 January 2012 to 31 December 2019 (France). For both cases and controls, MS onset had to be at ⩾2 years pre-index. Each case was matched to two randomly selected controls from the MS population without replacement. Controls were assigned the same index date and study period as their matched cancer case and were without evidence of cancer pre-index date of their case and in the 2 years following. Matching criteria were calendar year, sex, birth year (±3 years), place of residence (France = department/ecological deprivation quintile; 28 BC = first three digits of the postal code), MS duration (defined as the time between the first recorded MS event and the index date), and DMT use in the 12–24 months pre-index (France = none; ⩾ 1 moderate efficacy, no high efficacy; ⩾ 1 high efficacy; BC = none, ⩾ 1 moderate or high efficacy). In addition, DMT use was studied according to each region’s prescription practices. During the study period, off-label drugs (i.e. DMT without formal regulatory approval to treat MS) could be identified as part of MS management in France but not in BC. Therefore, off-label drugs were only included in the French matching/subgroup analysis. In the absence of an exact match, residence criteria were gradually relaxed to become less stringent (Supplemental Figure 1).
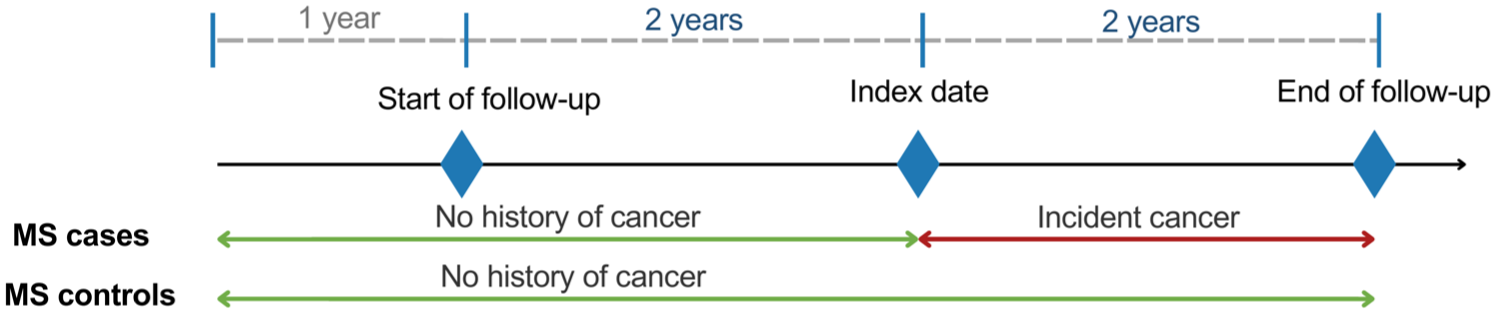
Study design.
Cancer
In BC, cancer cases were identified using a validated definition (⩾1 cancer-related hospitalization or physician visit claims). 29 Although this previous study focused on the most common cancer types (breast, lung, prostate, and colorectal), this definition was applied to all cancer types. In France, cancer cases were identified using a previously used definition developed with the French Cancer Institute and based on hospital records, chemotherapy/radiotherapy use, and LTD status.12,30 Information on cancer treatment was only available in France.
Outcomes
MS-related healthcare use was determined through three outcomes, using administrative data: (1) neurologist visits, (2) MS hospitalizations, and (3) DMT use. Overnight MS hospitalizations had a principal diagnosis code of MS (ICD-10:G35/ICD-9:340). DMT use was defined as filled prescriptions of (1) MS-specific DMTs (Supplemental Table 1) and (2) non-specific/off-label drugs used to treat MS (Supplemental Table 2).
Current treatment for MS was based on each DMT and off-label drug used during the study period grouped as: high efficacy (alemtuzumab, fingolimod, natalizumab, ocrelizumab, cladribine, siponimod), moderate efficacy (daclizumab, dimethyl fumarate, glatiramer acetate, interferon beta, teriflunomide) or off-label/non-specific to MS. For all treatments, except anti-CD20 drugs (i.e. ocrelizumab, rituximab), participants were considered treated for 90 days, and discontinuation occurred if there was no dispensation for ⩾ 90 consecutive days, plus a 30-day grace period. For anti-CD20, participants were considered treated for 12 months and for 6 months, respectively. Similarly, there was a 30-day grace period. Each of these DMT and off-label drugs was considered discontinued if no further prescription was filled. There were no cladribine or alemtuzumab prescription fills in the study population, so we did not include additional specific treatment window considerations.
Statistical analysis
For each follow-up year, neurologist visits and hospitalizations were assessed as yearly rates and as percentages of individuals with at least one encounter during the year. Periods of hospitalization were discounted when computing individual neurologist visit rates because physician visits were not consistently recorded during hospitalizations. DMT use was described as a percentage of individuals on treatment at each time point.
Mixed-effect models were used to assess the impact of cancer on MS management in cases compared with controls, and time was modeled using a piecewise linear spline with a knot at the time of cancer diagnosis. The inclusion of the knot allowed fitting two slopes (pre- and post-cancer) that could then be compared through a ratio of the estimates, a “slope ratio” (SR). An SR < 1 indicates that the pre-cancer estimate was higher than the post-cancer estimate. The type of mixed-effect model used differed depending on the outcome structure: neurologist visits and MS hospitalizations were modeled using a Poisson regression yielding incidence rate ratios (IRRs) and DMT use using a logistic regression yielding odds ratios (ORs).
The three following additional analyses were carried out in the French cohort only. DMT use models were stratified by cancer treatment type (≥1 chemotherapy sessions, ≥1 radiotherapy sessions without any chemotherapy, cancer surgery only, other). Neurologist visits and MS-related hospitalization models were stratified by type of DMT used in the 24–12 months pre-index. To explore a potential period effect, all three outcomes were stratified into cancer diagnosis years (2012–2017 vs. 2018–2019).
As per data access and privacy agreements, analyses were performed separately in each region, with aggregated results pooled. The I2 statistic was computed; if ⩽ 50%, this suggested no evidence of regional heterogeneity, and estimates were pooled by meta-analysis. Otherwise, more conservative pooling estimates were derived from mixed models. Analyses were performed using SAS (v.8.3.7) and R (v.4.1.2).
Results
A total of 20,706 pwMS were included 6902 MS cases (France = 4555; BC = 2347) matched 1:2 to 13,804 MS controls (France = 9110; BC = 4694) (Figure 2). Their characteristics at the index date are described in Table 1. Most were female (France 71.6%; BC 75.9%) and mean age at index = 58.6 years (SD = 12.5) in France and 55.9 years (SD = 11.6) in BC. The most common cancer types were breast (females only, France = 36.4%; BC = 23.9%), prostate (males only, France = 26.1%; BC = 19.8%), and colorectal cancer (France = 10.3%; BC = 10.9%).
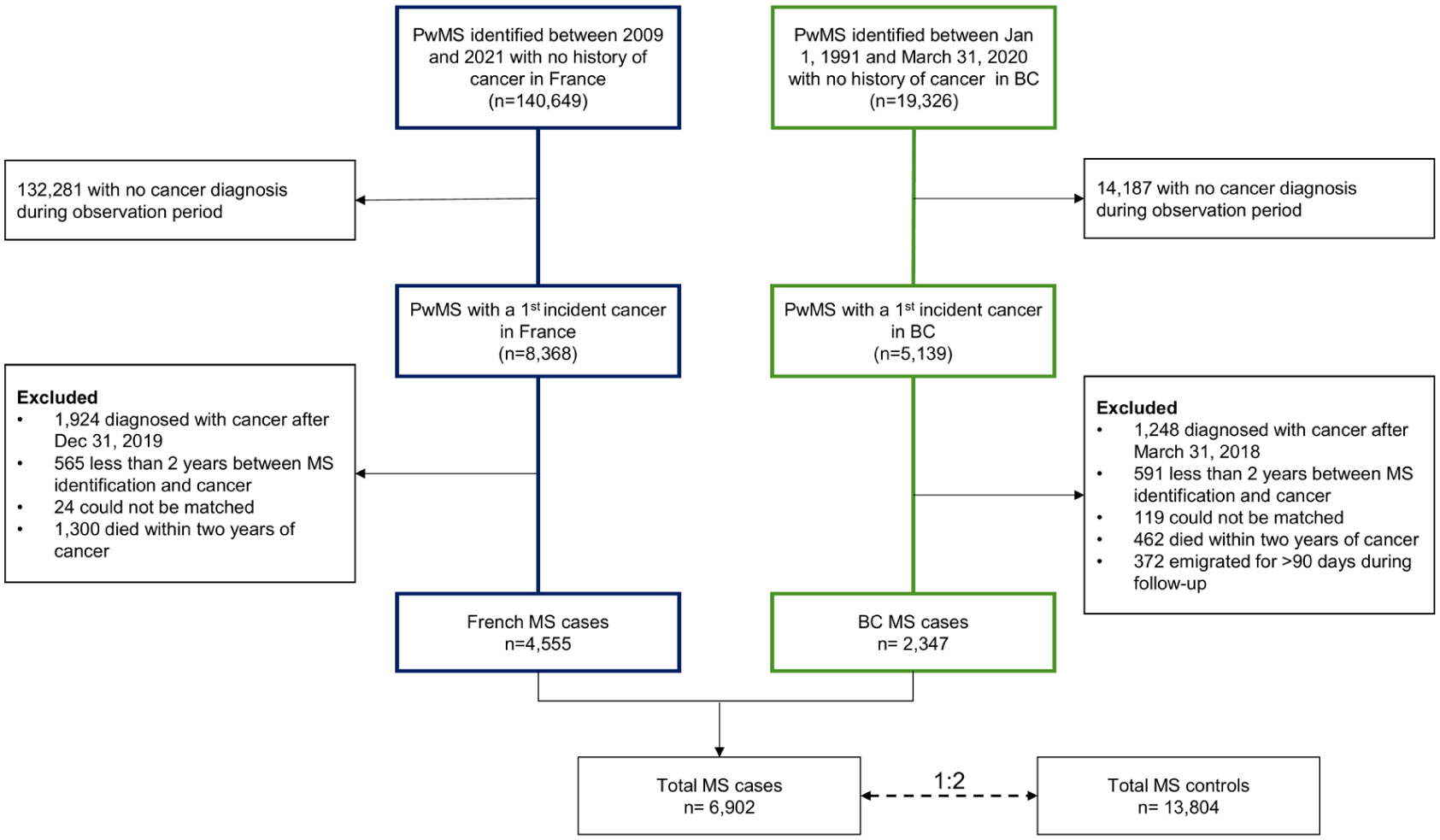
Population flowchart.
Characteristics of patients living with multiple sclerosis with (cases) and without (controls) a cancer diagnosis at the index date (n = 20,706).
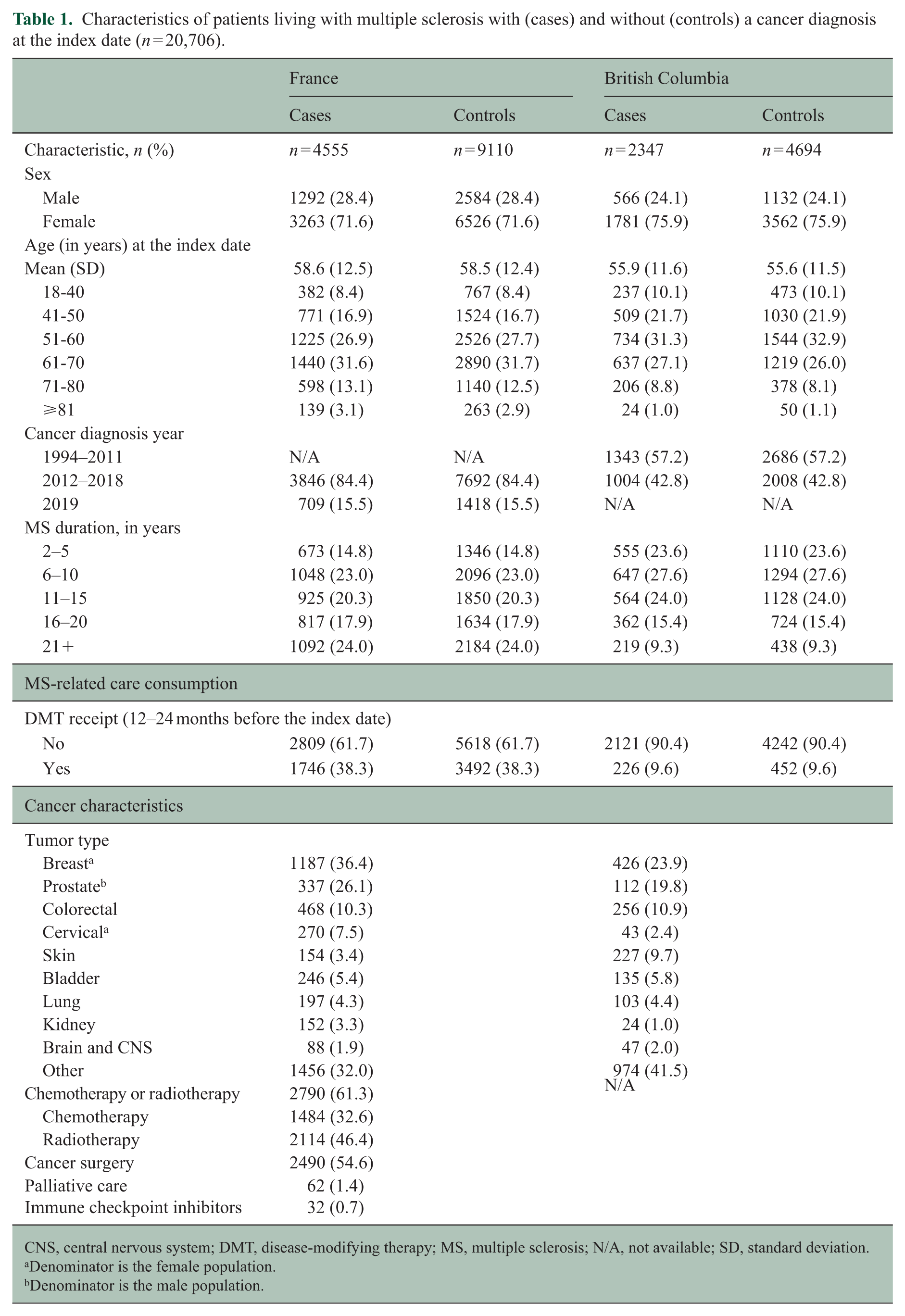
CNS, central nervous system; DMT, disease-modifying therapy; MS, multiple sclerosis; N/A, not available; SD, standard deviation.
Denominator is the female population.
Denominator is the male population.
Neurologist visits
Overall, the percentage of pwMS who saw a neurologist was higher among cases than controls (France 75.1% vs. 72.7%; BC 72.0% vs. 68.7%) with an overall decrease in both groups over time (Table 2). Before and after cancer, the neurologist visit rate was slightly higher for cases than controls, but the post-cancer IRR was only modestly significant (IRR; pre-cancer = 1.08 [1.04–1.13], p = 0.001; post-cancer = 1.04 [1.000–1.09], p = 0.05) (Table 3). When comparing pre- and post-cancer IRRs, they did not differ (SR = 0.96 [0.91–1.005], p = 0.08). Results were similar across regions and age groups (Figure 3 and Supplemental Figure 2). In addition, post- vs. pre-SR were comparable across time periods (2018–2019 = 0.97 [0.88–1.07]; 2012–2017 = 0.94 [0.89–1.00], Figure 4, Supplemental Table 5).
Yearly multiple sclerosis-related care consumption.
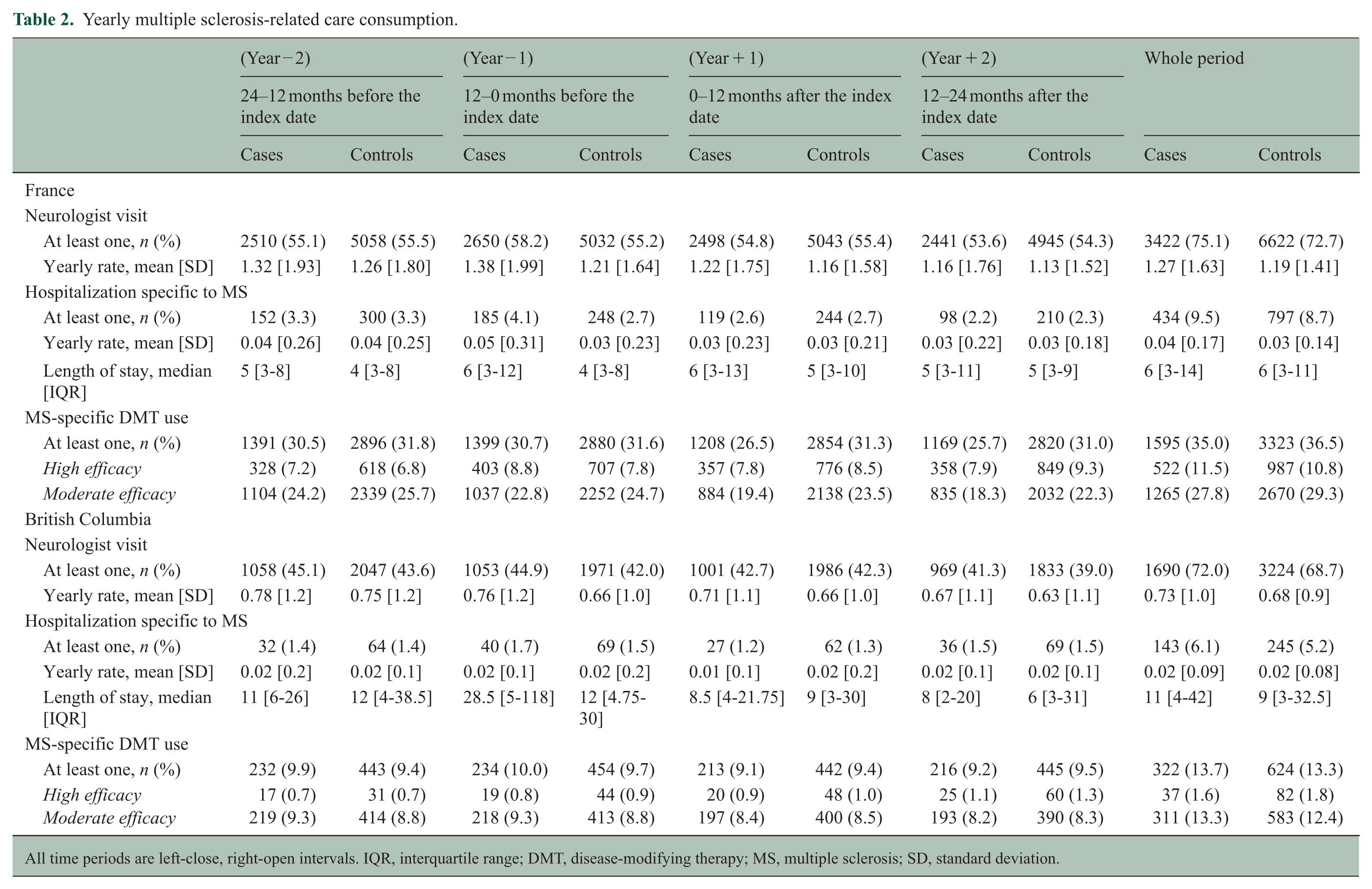
All time periods are left-close, right-open intervals. IQR, interquartile range; DMT, disease-modifying therapy; MS, multiple sclerosis; SD, standard deviation.
Mixed-effect model estimates to assess the impact of the cancer diagnosis on multiple sclerosis-related care by region and pooled.
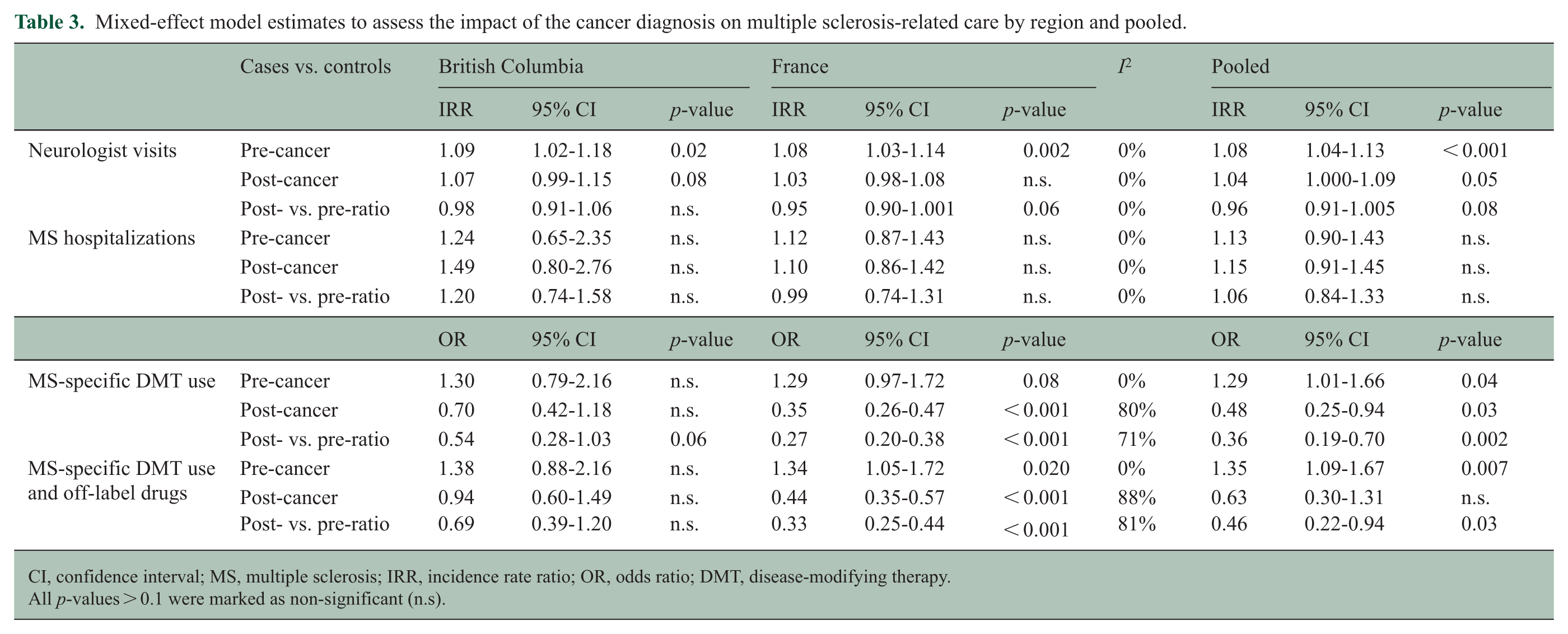
CI, confidence interval; MS, multiple sclerosis; IRR, incidence rate ratio; OR, odds ratio; DMT, disease-modifying therapy.
All p-values > 0.1 were marked as non-significant (n.s).
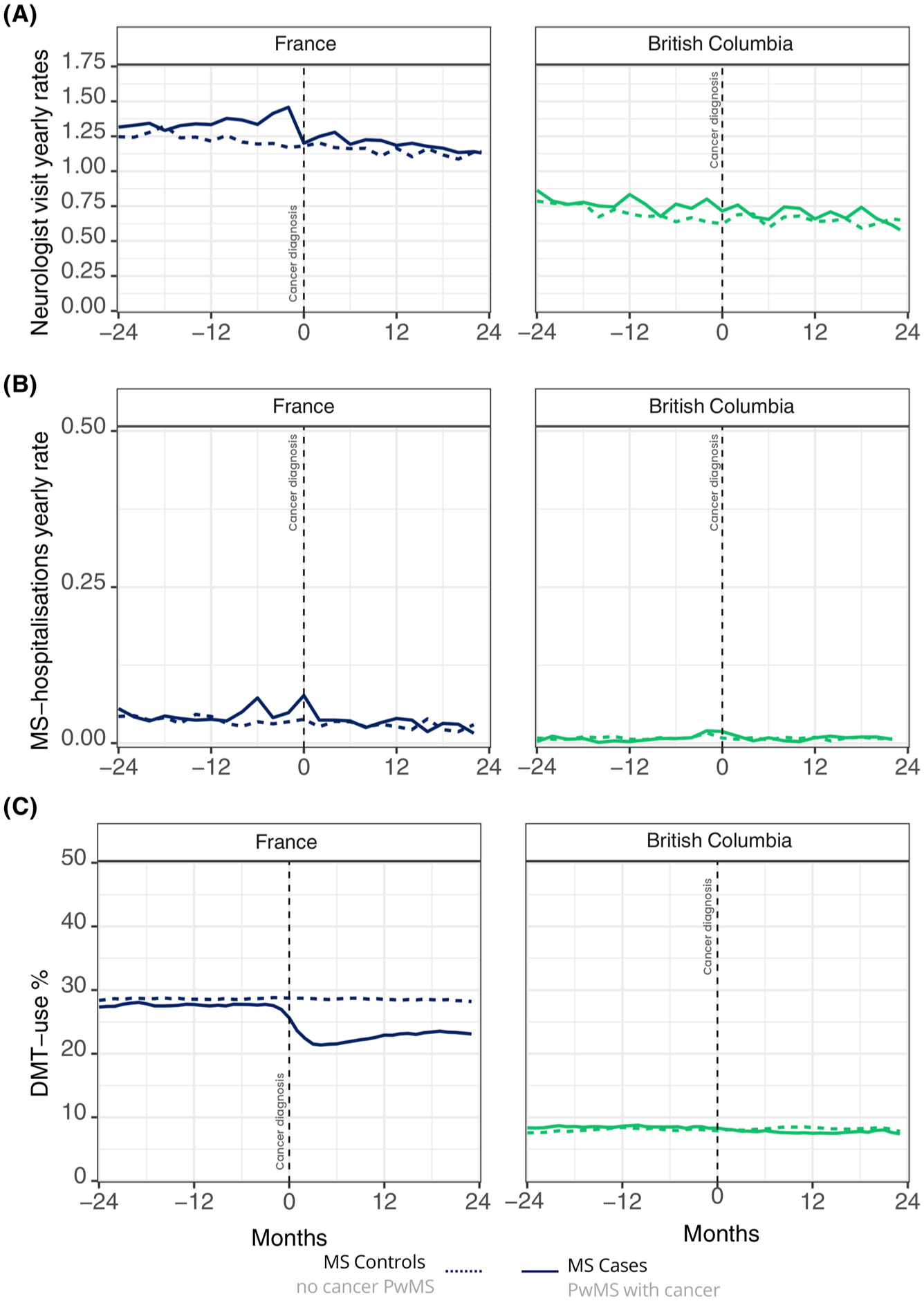
MS-related outcomes over time, cases vs. controls. (a) Yearly neurologist visit rates. (b) Yearly MS hospitalization rates. (c) Percentages of patients on MS-specific DMT.
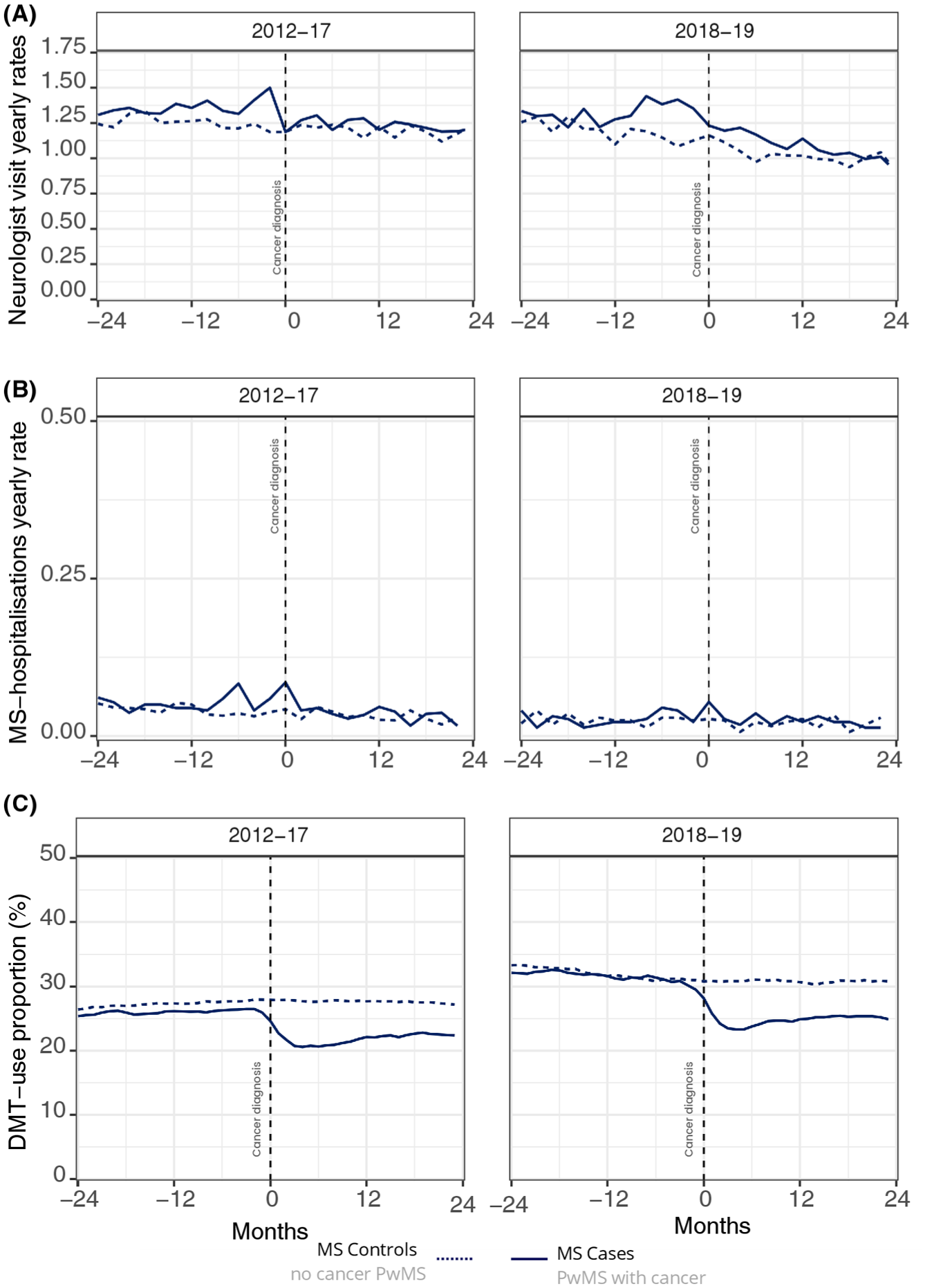
MS-related outcomes over time, cases vs. controls, according to time period in France (n = 13,665). (a) Yearly neurologist visit rates. (b) Yearly MS hospitalization rates. (c) Percentages of patients on MS-specific DMT.
By DMT use
In the French cohort, those receiving DMT had a more pronounced neurological follow-up overall. Over the study period, >90% of PwMS in the high- and moderate-efficacy DMT groups saw a neurologist but only 62% in the untreated group (Supplemental Table 6). Those using a high-efficacy DMT were younger, had the highest mean annual neurologist visit rate (Supplemental Table 6), and were more frequently on chemotherapy (Supplemental Table 7). Among those receiving high- and moderate-efficacy DMT, cancer did not have any impact on neurologist visit rates (SR = 0.98 [0.83–1.16] and 0.98 [0.93–1.03], respectively). Conversely, among those not receiving DMT, the neurologist visit rate was higher among cases than controls only before cancer diagnosis (IRR, pre-cancer = 1.13 [1.04–1.22]; post-cancer = 1.03 [0.95–1.11]) which resulted in a significant post- vs. pre-cancer SR = 0.91 [0.84–0.99].
MS hospitalizations
During the study period, there were few MS hospitalizations in France (cases = 434, 9.5%; controls = 797, 8.7%) and BC (cases = 143, 5.9%; controls = 245, 5.1%, Table 2). In both regions, the pre- and post-cancer MS hospitalization rates did not differ between cases and controls (IRR, pre-cancer = 1.13 [0.90–1.43]; post-cancer = 1.15 [0.91–1.45]) (Table 3). The pre- and post-cancer IRRs also did not differ, showing no impact of cancer on MS hospitalization rates (SR = 1.06 [0.84–1.33]). In the French cohort, similar results showing no difference were found when stratifying by DMT use (Supplemental Figure 3). Those findings were consistent across time periods, showing no difference (2018–2019 = 1.21 [0.62–2.33]; 2012–2017 = 0.92 [0.67–1.28], Figure 4, Supplemental Table 5).
DMT use
Over the study period, fewer individuals received MS-specific DMT in BC than in France (13.4% vs. 36.0%), especially high-efficacy DMT (1.7% vs. 11.0%). After cancer, fewer cases were on DMT than controls (post-cancer OR = 0.48 [0.25–0.94]), specifically in the French cohort (post-cancer OR = 0.35 [0.26–0.47]) (Table 3). The comparison of the pre- and post-cancer estimates showed that cancer had a negative effect on DMT use (SR = 0.36 [0.19–0.70]). This effect, albeit less strong, remained when including off-label drugs (SR = 0.46 [0.22–0.94]). This decrease in DMT use was observed across all age groups (Supplemental Figure 4) as well as across all time periods (2018–2019 = 0.16 [0.08–0.34]; 2012–2017 = 0.33 [0.22–0.47], Figure 4, Supplemental Table 5). In the French cohort, post hoc analyses by selected individual DMTs, including S1P-modulators and anti-CD20, are shown in Supplemental Figure 5.31,32
By cancer treatment
In the French cohort, cases who received chemotherapy were on average younger and more likely to be on DMT (Supplemental Table 8). In all cancer treatment groups, the percentage of DMT users decreased after the index date; however, the extent and statistical significance varied across groups (Figure 5). The chemotherapy group experienced the most significant decrease in DMT use (SR = 0.24 [0.14–0.40]). DMT use also decreased in the radiotherapy alone (SR = 0.36 [0.19–0.67]) and surgical intervention-only groups (SR = 0.38 [0.18–0.78]). Findings remain virtually unchanged in the post hoc descriptive analysis excluding off-label/non-specific DMTs (including rituximab) from the analysis (Supplemental Figure 6).
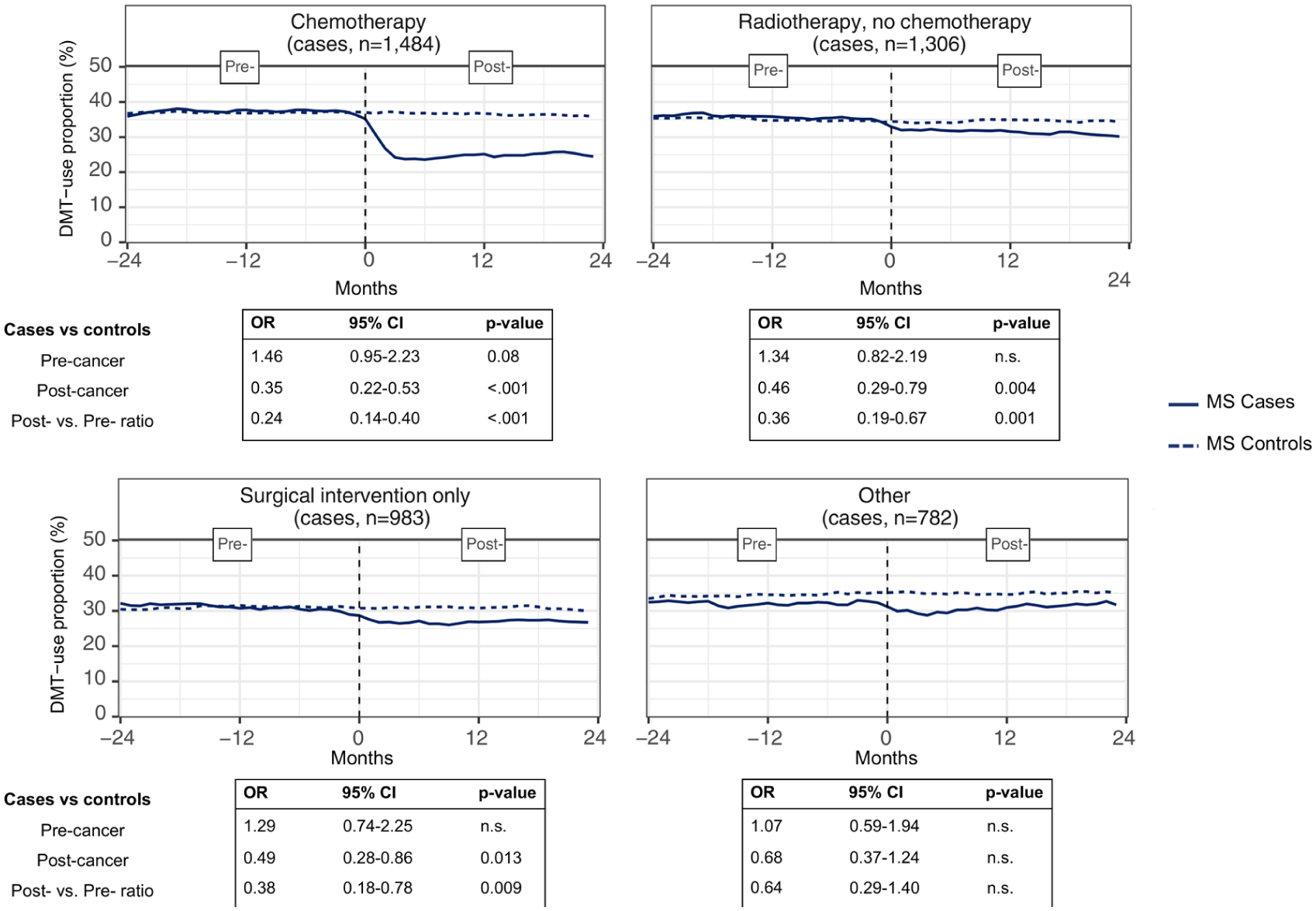
Percentage of patients using MS-specific DMT and off-label drugs: mixed-effect model results according to the cancer treatment type, cases vs. controls, France.
Discussion
We assessed the impact of cancer on MS-related care and DMT use in a large multinational population-based cohort of matched pwMS with and without cancer. Overall, cancer did not have a major effect on neurological care and MS hospitalizations in the 2 years after the diagnosis, independently of age, sex, residence, and MS duration. These findings remained consistent in pwMS across all MS treatment groups as well as across time periods.
Conversely, DMT use decreased sharply after the cancer diagnosis, overall and across all cancer treatment groups, but especially in individuals receiving chemotherapy. This decrease was also observed in those diagnosed with cancer after 2018, reflecting current MS practices. Cancer did not affect the rates of neurologist visits and MS hospitalizations, possibly suggesting a decision to withhold DMT and not patient-led care withdrawal. This decision could be due to multiple factors: oncology-driven treatment prioritization, contraindications imposed by oncologists, or temporary clinical instability. It could also be neurologist or patient-driven due to concerns about the role of DMT in cancer incidence or severity. The fact that chemotherapy can have a beneficial and long-lasting immunomodulatory effect on the course of MS could justify temporary DMT cessation.15,22,24 Furthermore, some DMT–chemotherapy combinations may be contraindicated due to cumulative risks, most notably infectious ones. However, radiotherapy and surgery do not act as MS treatment; therefore, the decrease in DMT use in these treatment groups is more challenging to interpret. In addition, the adverse effect of cancers on DMT use seemed larger in the French cohort.
A key challenge was ensuring comparability between pwMS with cancer (cases) and matched cancer-free pwMS (controls), both alive during the study period. The case population was highly specific: ill enough to develop cancer yet healthy enough to survive ⩾2 years post-diagnosis. To maximize compatibility and ensure similar levels of health and health-related behaviors, individuals were matched on criteria as stringent as possible. In addition, pre-cancer MS-related healthcare utilization indicators were broadly similar, suggesting a high degree of comparability between cases and controls.
Several differences emerged between the French and BC cohorts. First, at cancer diagnosis, BC patients were younger (55.9 vs. 58.6 years), likely reflecting differences in the cancer identification algorithm (cancer identification through physicians’ visits in BC which may identify earlier or less severe tumors), cancer type distribution, and study periods (France: 2011–2021; BC: 1992–2020). Indeed, the study period in BC included a time of elevated MS mortality, resulting in a younger overall population. Second, for all three outcomes, MS-related healthcare consumption was higher in France than in BC, especially concerning DMT use, for both cases and controls. This could be explained by differences in care practice and use of healthcare services between these regions. Regardless of these differences, the impact of cancer on MS-related care consumption was similar in both cohorts.
Although population-based medico-administrative databases allow studying large cohorts and diminishing the selection bias, they can be limited in terms of available information, such as lifestyle-related factors and clinical data (e.g. disease progression or activity [relapses]). In addition, cancer severity or stage at diagnosis was not available and could be an important factor regarding treatment choices, specifically regarding discontinuation, as some DMTs are contraindicated in the case of progressive cancer.33,34 Finally, DMT exposure windows, based on filled prescriptions, may introduce minor misclassification. Regional differences also existed; for example, information on cancer treatment types was available only in France, whereas diagnoses (ICD codes) related to physician visits were available only in BC. The modest use of high-efficacy DMTs likely reflects an older, less inflammatory population, the study covering multiple treatment paradigms, and estimates align with recent population-based studies.4,35
To conclude, this study offers valuable insights into how the diagnosis of cancer affects MS neurological care and treatment decisions, yielding reassuring and consistent results across two regions on separate continents. We used population-based data that allowed the inclusion of virtually all pwMS with a cancer diagnosis in France and BC between 1994 and 2019, resulting in a large, population-based cohort. It also allowed addressing the same questions in two regions with different MS care practices over a long time period. Our time-period–stratified analysis, particularly for the 2018–2019 cancer diagnosis group followed up until 2021 in France, demonstrated that our observations remained consistent over time, thereby reinforcing the relevance of these findings within the context of contemporary MS management practices. As the MS population grows and ages, this issue is becoming increasingly relevant. Further research is needed on care coordination to develop unified, evidence-based guidelines for managing MS in the context of cancer. 23
Supplemental Material
sj-docx-1-msj-10.1177_13524585261437966 – Supplemental material for Impact of cancer on multiple sclerosis–related healthcare and disease-modifying drug use: A multinational cohort study
Supplemental material, sj-docx-1-msj-10.1177_13524585261437966 for Impact of cancer on multiple sclerosis–related healthcare and disease-modifying drug use: A multinational cohort study by Chloe Pierret, Geraldine Androdias, Christine Lebrun-Frenay, Sandra Vukusic, Myriam Maumy, Huah Shin Ng, Feng Zhu, Helen Tremlett and Emmanuelle Leray in Multiple Sclerosis Journal
Footnotes
Acknowledgements
We would like to acknowledge the members of the team who facilitated gaining funding for the wider program of work related to MS (Dr Karim, Dr Wijnands, Dr Marrie, Dr Maxwell, Dr Zhao, and Dr Yusuf).
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.P. has nothing to disclose. G.A. has received personal compensation for consulting, serving on a scientific advisory board, speaking, or other activities with Biogen, Janssen, Merck, Novartis, Roche, and Sanofi-Genzyme. C.L.-F. has research support in the last 3 years from the EDMUS Foundation. S.V. has received non-personal consulting and lecturing fees, travel grants, and unconditional research support from Biogen, Janssen, Merck, Novartis, Roche, Sandoz, and Sanofi. M.M. has nothing to disclose. H.S.N. has received research support from the Cancer Council South Australia’s Early Career Research Fellowship. F.Z. has nothing to disclose. H.T. has received research support in the last 3 years from the Canada Research Chair Program, National MS Society, Canadian Institutes of Health Research, Canada Foundation for Innovation, MS Canada, and the EDMUS Foundation. Advisor for Canada’s Drug Agency (2024–2025, payment declined). E.L. consulting and lecture fees or travel grants from Alexion, Biogen, Merck, Novartis, Roche, and Sanofi are not related to the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by the French Foundation for Research in Multiple Sclerosis (ARSEP) mobility grant, the French Public Health Doctoral Network (RDSP) mobility grant, the ARSEP-EDMUS Foundation, the Clinical Neuroscience Institute of Rennes (INCR), and the National MS Society and MS Canada (RG5063A4/1/RFA-2103-37392; EGID: P002/903124; PI: H.T.). None of the study funders had a role in the methods, analyses, writing of the manuscript, or the decision to submit it for publication.
Ethical Considerations
France: Data access for the study was approved according to the current French legislation (Articles R.1461–11 to R.1461-17 of the French Public Health Code and Data Protection Authority decision CNIL-2016-316), and the study was declared to the EHESP SNDS registry, equivalent to an Institutional Review Board approval. British Columbia: This study was approved by the University of British Columbia’s Clinical Research Ethics Board. Access to data provided by the Data Steward(s) is subject to approval but can be requested for research projects through the Data Steward(s) or their designated service providers.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
ORCID iDs
Data Availability
A request for data access may be made, but it would require the previous approval of the French regulatory authorities and/or Canadian Data Stewards from PopulationData BC. France: The SNDS database is the property of the French health insurance system. British Columbia: Access to data provided by the Data Stewards is subject to approval but can be requested for research projects through the Data Stewards or their designated service providers. The following datasets were used in this study: PharmaNet, Discharge Abstract Database, Medical Services Plan, BC Vital Events, and Statistics and Census Geodata. You can find further information regarding these data sets by visiting the PopData project webpage at ![]() . All inferences, opinions, and conclusions drawn in this publication are those of the authors and do not reflect the opinions or policies of the Data Stewards.
. All inferences, opinions, and conclusions drawn in this publication are those of the authors and do not reflect the opinions or policies of the Data Stewards.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
