Abstract
Background:
The effects of smoking and vascular risk factors (VRF) on relapse activity in myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) are not well understood. We aimed to investigate these.
Methods:
MOGAD patients in the Oxford National Neuromyelitis Optica (NMO) Service with known smoking or VRF (hypertension, hypercholesterolaemia, type 2 diabetes, elevated body mass index) status were considered for inclusion. Outcomes were time-to-relapsing course and relapse rate. Cox and negative binomial regression were utilised.
Results:
We included 172 patients. Current smokers had a 2.2 times higher relapse rate than never smokers (adjusted rate ratio 2.23 [95% CI = 1.35–3.68], p = 0.0017). Relapse rate was similar between past and never smokers (adjusted rate ratio 0.81 [0.51–1.29], p = 0.38). For time-to-relapsing course, multivariable analysis did not identify smoking as associated although univariable analysis showed a consistent trend with shorter time-to-relapse in current than never smokers (hazard ratio 1.76 [0.97–3.22], p = 0.06). There was no difference in either outcome by VRF status.
Conclusion:
Relapse rates were doubled in current than never smokers, whereas these were similar between past and never smokers. VRF did not associate with relapse risk. Smoking cessation should be encouraged in current smokers with MOGAD as past smokers were not observed with this poorer outcome.
Keywords
Introduction
Myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) is a recently defined inflammatory disease of the central nervous system. 1 Following the onset attack, between 30% and 50% of patients may develop a relapsing disease course.2,3 Disability accrual after onset in MOGAD is relapse driven, in contrast to multiple sclerosis in which relapse-associated and relapse-independent disability progression are observed. 4 Although MOGAD attacks are often less disabling compared to aquaporin-4 antibody-positive neuromyelitis optica spectrum disorder, one MOGAD cohort reported 59% with persistent disability at last follow-up and associated with number of attacks and worse recovery after onset attack. 5 There are currently no randomised-controlled trial evidence to guide immunosuppressive treatment in MOGAD, and available treatments are empirically used to prevent future relapse. Observational studies support the steroid-responsive nature of MOGAD in relapse prevention.6,7
Smoking and other vascular risk factors (VRF) may co-exist in patients with MOGAD. The impact these factors have on disability recovery following the onset attack of MOGAD has been investigated, with current smoking status associated with poorer recovery.8,9
It is not clear whether smoking and VRF affect relapse risk in MOGAD. One study had investigated relapse risk and smoking in 70 MOGAD patients with disease durations between 4 and 5 years and did not identify a significant difference between ever- and non-smokers. 9 To better understand the effect of these modifiable factors on disease activity, we investigated the association of smoking and VRF status with relapse risk through a retrospective analysis of a prospectively recruited single-centre observational cohort.
Methods
Study design and participants
Patients reviewed through the Oxford Neuromyelitis Optica Highly Specialised Service and who consented to participate in the Demyelinating Research Tissue Bank (approved by Oxford C Research Ethics Committee, reference 21/SC/0353) were considered for inclusion. In the Demyelinating Research Tissue Bank, participants with CNS demyelinating disease such as MOGAD are recruited into a prospective observational cohort with clinico-biological data collected at the time of each regular clinic visit, and clinical data retrospectively ascertained prior to entry. Clinical data collected include demographics, attack and relapse characteristics, and acute and chronic immunosuppressive treatments used. Data cut-off was December 2023. Through this consultant-led service, all patients are reviewed by at least one of two neurology consultants (M.I.L., J.P.) with expertise in NMOSD and MOGAD. Inclusion criteria were fulfilment of MOGAD diagnostic criteria,1 age at onset ⩾ 16 years, at least 1 year disease duration (i.e. duration between onset attack/first symptom and last follow-up), and available data on early treatment, relapses, and either smoking or non-smoking VRF status.
Exposures and outcomes
Cigarette smoking (referred to as ‘smoking’ through this paper) and VRF status were collected through patient-filled questionnaires done on service entry (at a median 0.8 years [IQR 0.4–2.5 years] after onset attack) and through clinical height and weight measurements. Demographic, attack and treatment data were collected prospectively during clinical follow-up, or retrospectively ascertained if prior to service entry.
Smoking status was classified as never, past or current smoker. For non-smoking VRF, presence or absence of previously diagnosed hypertension, hypercholesterolaemia and type 2 diabetes mellitus were collected. Elevated body mass index (BMI) was defined as BMI > 25 kg/m2. At least one non-smoking VRF was considered present if any of hypertension, hypercholesterolaemia, type 2 diabetes or elevated BMI was present. Immunosuppressive treatment (IST) was defined as oral prednisolone, oral steroid-sparing agents (such as azathioprine, mycophenolate, and methotrexate), maintenance intravenous immunoglobulin, monoclonal antibody therapies (such as rituximab, tocilizumab), cyclophosphamide, or multiple sclerosis disease-modifying therapies.
Outcomes were time-to-relapsing course and annualised relapse rate, respectively. A relapse was defined as a new clinical attack from MOGAD more than 30 days from the prior attack.
Statistical analysis
Summary statistics, Mann–Whitney U test and Fisher’s exact test were used to describe and compare baseline characteristics.
Kaplan–Meier curves and Cox regression were used to investigate time-to-relapsing course as the outcome. Baseline was set as time of onset attack. Kaplan–Meier curves and univariable Cox regression were used to assess the association of smoking status and non-smoking VRF status (encoded as binary variables for presence of at least one non-smoking VRF, or presence of each non-smoking VRF, respectively) with outcome. Covariates included in the multivariable Cox regression model were age at onset (years; modelled linearly), sex, onset attack phenotype, acute steroid use after onset attack and IST use modelled as a time-varying covariate. Patients without event occurrence were censored at last follow-up. Global Schoenfeld test was used to assess the proportional hazard assumption.
Relapse rate over the follow-up period (from onset attack to last follow-up) was assessed by negative binomial regression. Negative binomial regression was used as overdispersion was present (dispersion ratio > 1 for fitted Poisson models [range 2.0–3.0]). Relapse occurring within the follow-up period were counted; onset attack was not included in this count. Univariable regression was used to assess smoking status and non-smoking VRF status, with an offset term of log of the follow-up duration included to account for varying follow-up times of each patient. For multivariable analysis, a person-period dataset was generated in which each patient’s total follow-up was split at IST start and stop dates to account for time-varying IST use. Covariates included in the multivariable model were smoking status, non-smoking VRF status (binary variable based on presence of ⩾ 1 non-smoking VRF), age at onset (years; modelled linearly), sex, onset attack phenotype, acute steroid use after onset attack and IST use. An offset term of log of the duration of each interval was included. Robust standard errors were calculated by clustering based on patient ID.
In sensitivity analyses, we modelled either smoking or VRF differently in our multivariable models: i) smoking as a binary variable based on current smoking status (i.e. current smoker, or past/never smoker), and ii) non-smoking VRF as a numeric variable representing the number of non-smoking VRF the patient has and modelled linearly.
A two-tailed p < 0.05 was considered as significant. Statistical analyses were conducted in the R Statistical Environment (v4.4.1) with tidyverse, survival and MASS packages.10 –13
Results
Cohort characteristics
We included 172 patients with MOGAD who fulfilled our inclusion criteria (Figure 1). Median age at disease onset was 35.6 years (interquartile range [IQR] 28.3–49.0; Table 1) and most common onset phenotypes were isolated optic neuritis (60.5%) followed by isolated myelitis (19.2%). There were 67 (39%) patients who had at least one non-smoking VRF, the most common of which was elevated BMI (27.9%). Disease duration at last follow-up was a median 4.1 years (IQR 2.4–7.7). Among 148 patients with available smoking status data, over half were never smokers (78/148 [52.7%]), followed by 46 (31.1%) past smokers, and 24 (16.2%) current smokers.
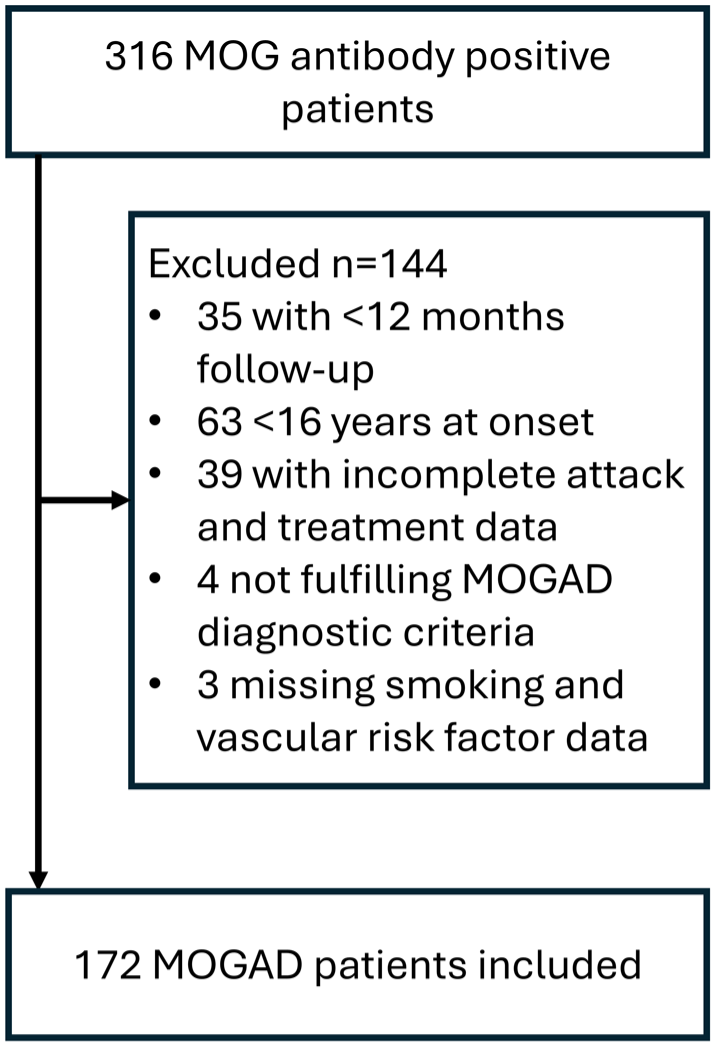
Patient inclusion flow chart.
Cohort characteristics.
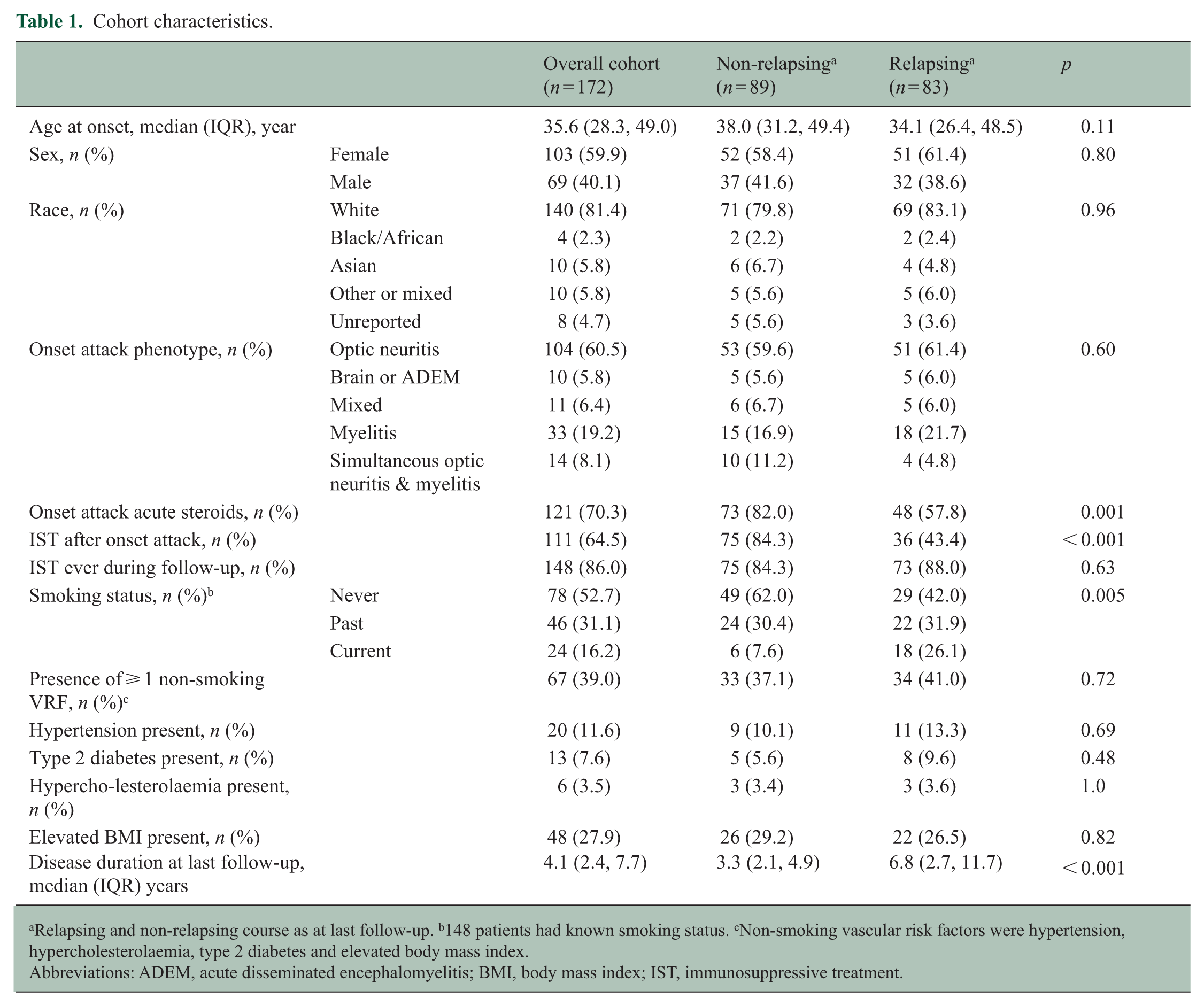
Relapsing and non-relapsing course as at last follow-up. b148 patients had known smoking status. cNon-smoking vascular risk factors were hypertension, hypercholesterolaemia, type 2 diabetes and elevated body mass index.
Abbreviations: ADEM, acute disseminated encephalomyelitis; BMI, body mass index; IST, immunosuppressive treatment.
Across a median 4.1 years disease duration, 83 (48.3%) developed a relapsing course. Of the 83 who relapsed, median time to first relapse (from disease onset) was 0.9 years (IQR 0.4–3.6). There was a higher proportion of current smokers among relapsers than non-relapsers (18/83 [26.1%] vs 6/89 [7.6%], p = 0.005), whereas the proportion of those with at least one non-smoking VRF was similar (34/83 [41.0%] vs 33/89 [37.1%], p = 0.72).
By smoking status, 58/78 (74.4%) never smokers, 29/46 (63%) past smokers and 12/24 (50%) current smokers were treated with IST after their onset attack (p = 0.068) and 72/78 (92.3%) never smokers, 38/46 (82.6%) past smokers and 19/24 (79.2%) current smokers were commenced on any IST across their follow-up period (p = 0.13). Comparing those with and without at least one non-smoking VRF, 69/105 (65.7%) without VRF and 42/67 (62.7%) with at least one VRF were on IST after onset attack (p = 0.81). Whereas, 89/105 (84.8%) without VRF and 59/67 (88.1%) with at least one VRF were on an IST at some point during their follow-up (p = 0.70). The most common IST used during follow-up were oral prednisolone (in 145/172 [84.3%] patients), azathioprine (32/172 [18.6%]), mycophenolate (24/172 [14.0%]) and methotrexate (7/172 [4.1%]).
Time-to-relapsing course
Kaplan–Meier curves suggested earlier time-to-relapsing course for current smokers, but with overlapping confidence intervals with past and never smokers (Figure 2(a)). In univariable Cox regression analyses, current smokers had an elevated hazard for a relapsing course relative to never smokers but this did not reach statistical significance (hazard ratio [HR] 1.76 [95% confidence interval = 0.97–3.22], p = 0.06; Table 2). Relapse risk was not significantly different between past and never smokers. There was also no difference between those with and without non-smoking VRF (Figure 2(b) and Table 2).
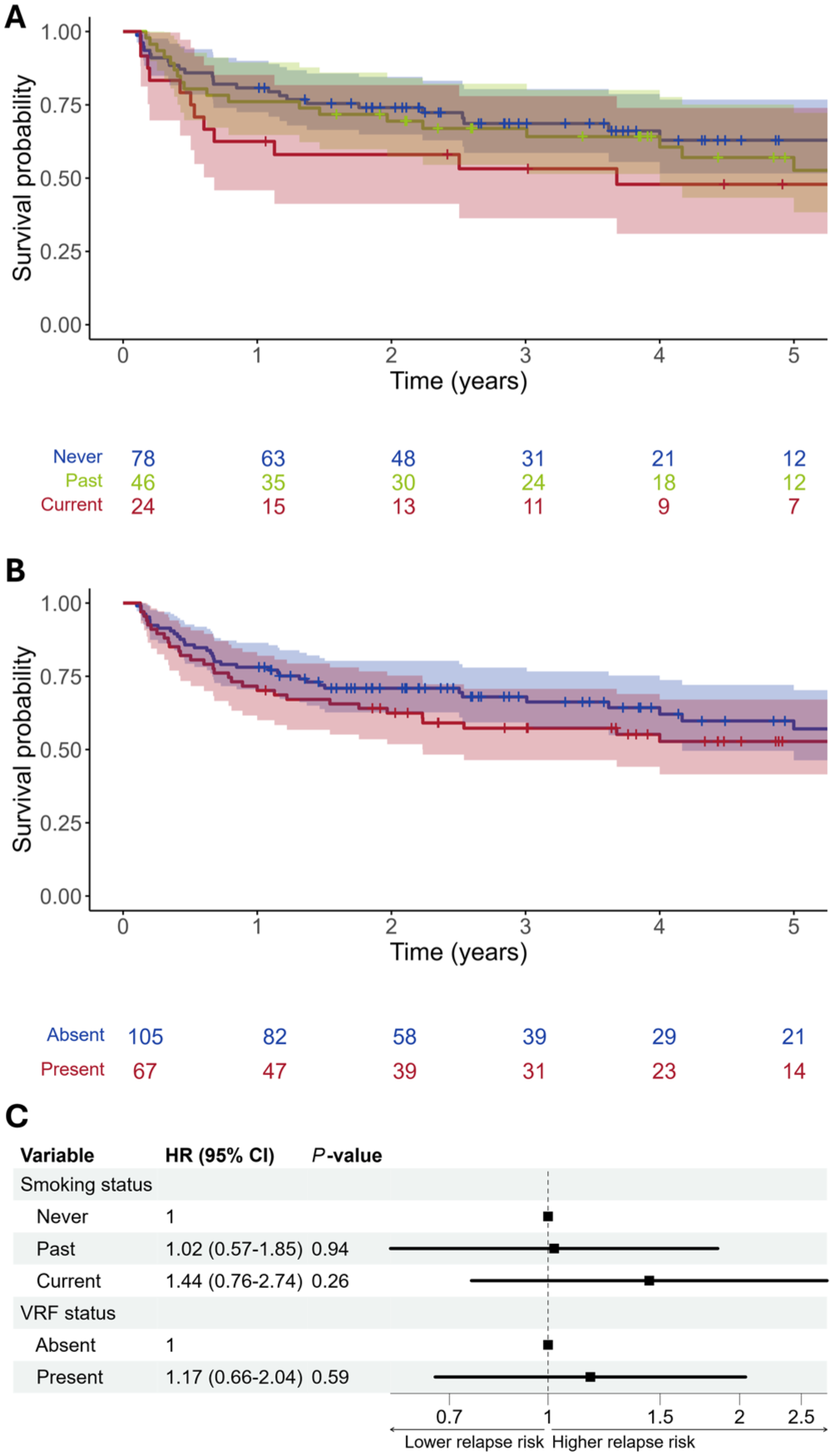
Time-to-relapsing course and smoking and vascular risk factor status.
Univariable Cox regression analyses for time-to-relapsing course.
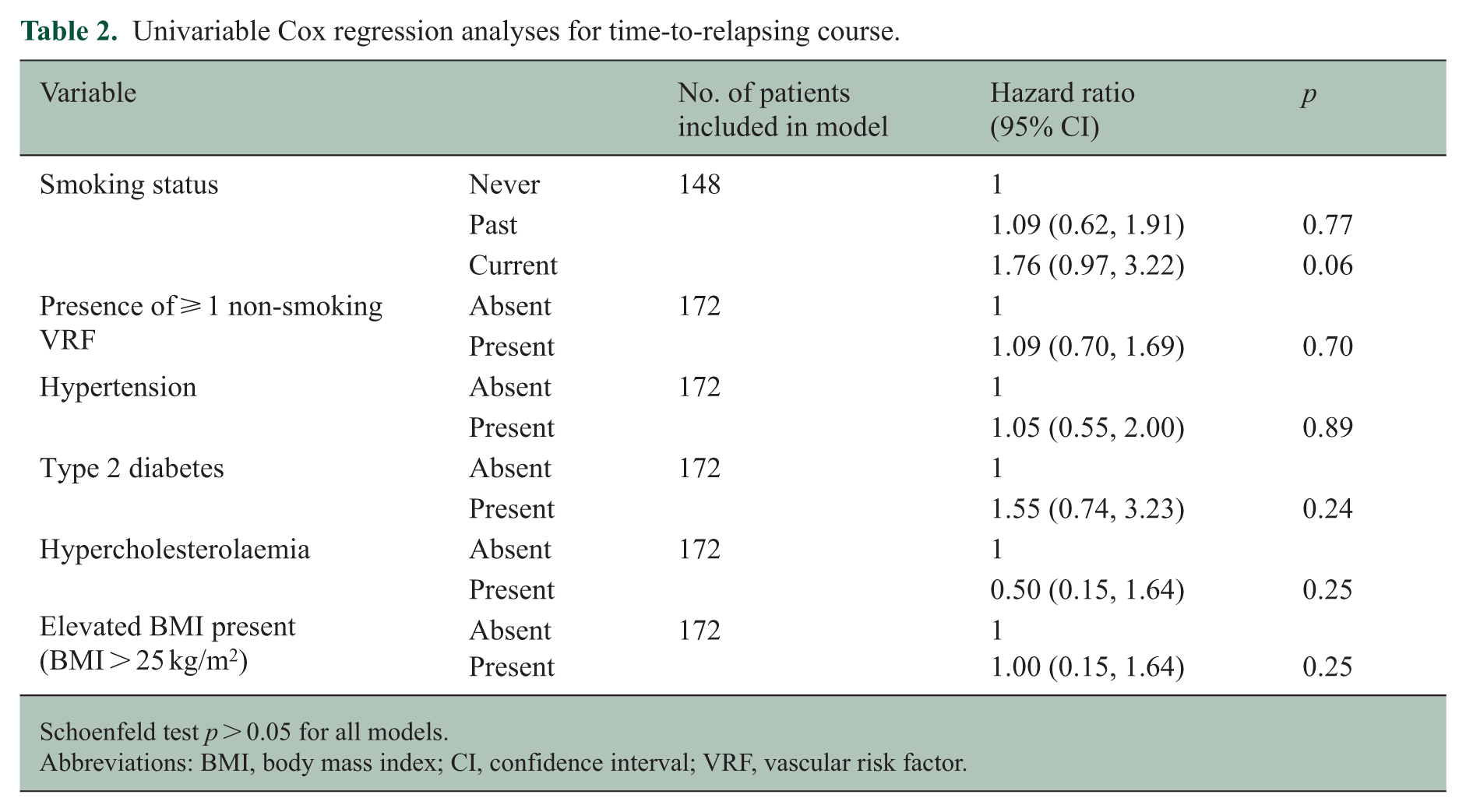
Schoenfeld test p > 0.05 for all models.
Abbreviations: BMI, body mass index; CI, confidence interval; VRF, vascular risk factor.
There were 148 patients with both smoking and VRF status available and we included them in our multivariable Cox regression model. Multivariable analysis showed no significant association of smoking or VRF status on risk for relapsing course (Figure 2(c) and Table 3). Sensitivity analyses showed unchanged results, with no significant associations between smoking and non-smoking VRF and outcome (Supplemental Tables 3 and 4).
Multivariable Cox regression results for time-to-relapsing course.
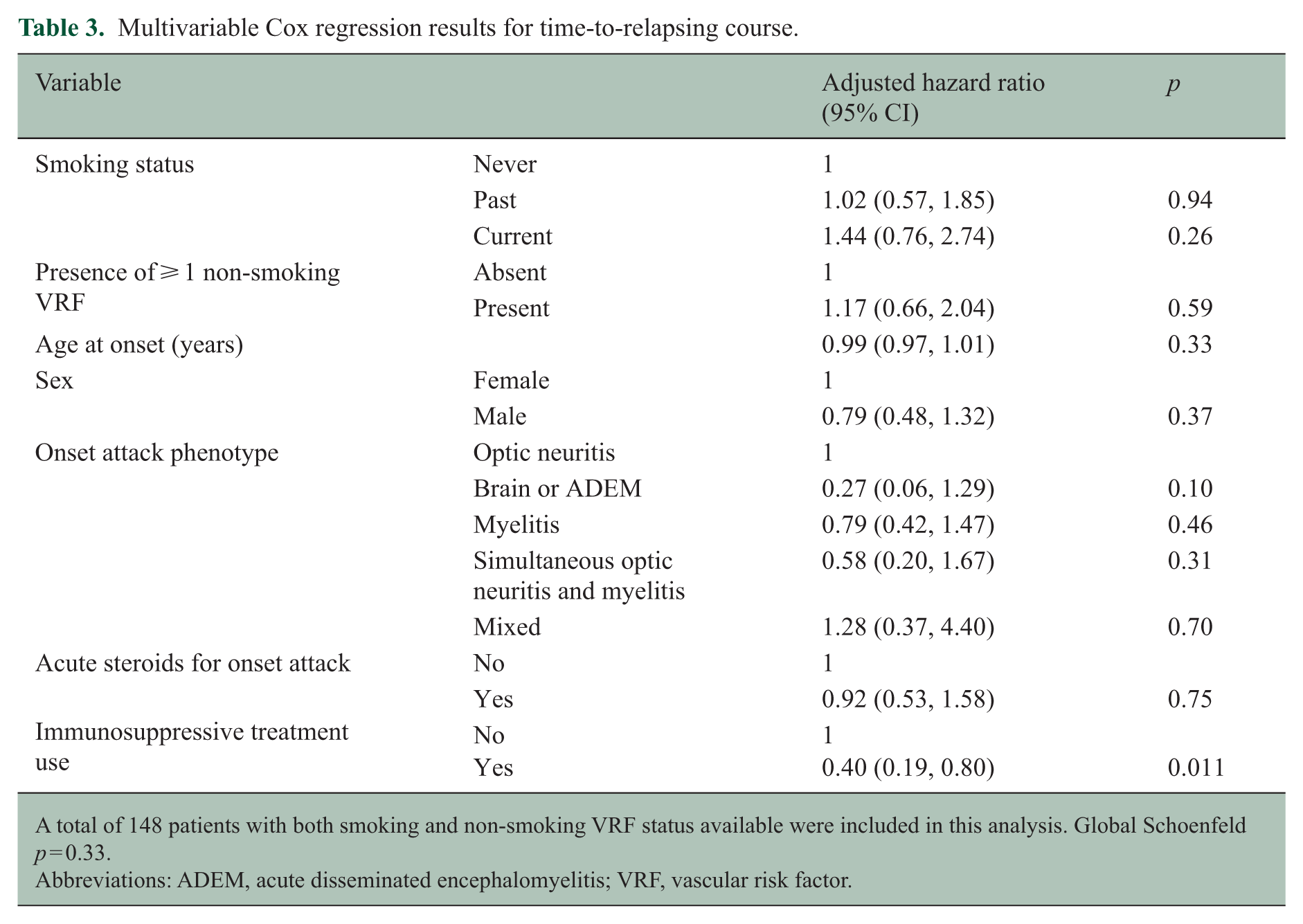
A total of 148 patients with both smoking and non-smoking VRF status available were included in this analysis. Global Schoenfeld p = 0.33.
Abbreviations: ADEM, acute disseminated encephalomyelitis; VRF, vascular risk factor.
Annualised relapse rate
By smoking status, crude annualised relapse rate was highest in current smokers compared to never and past smokers (Figure 3(a); annualised relapse rate for current smokers = 0.25, past smokers = 0.17 and never smokers = 0.18). Univariable negative binomial regression revealed a doubling of the relapse rate in current smokers compared to never smokers (rate ratio 2.10 [95% confidence interval 1.20–3.68], p = 0.01; Table 4) whereas relapse rate was not significantly different between past and never smokers.
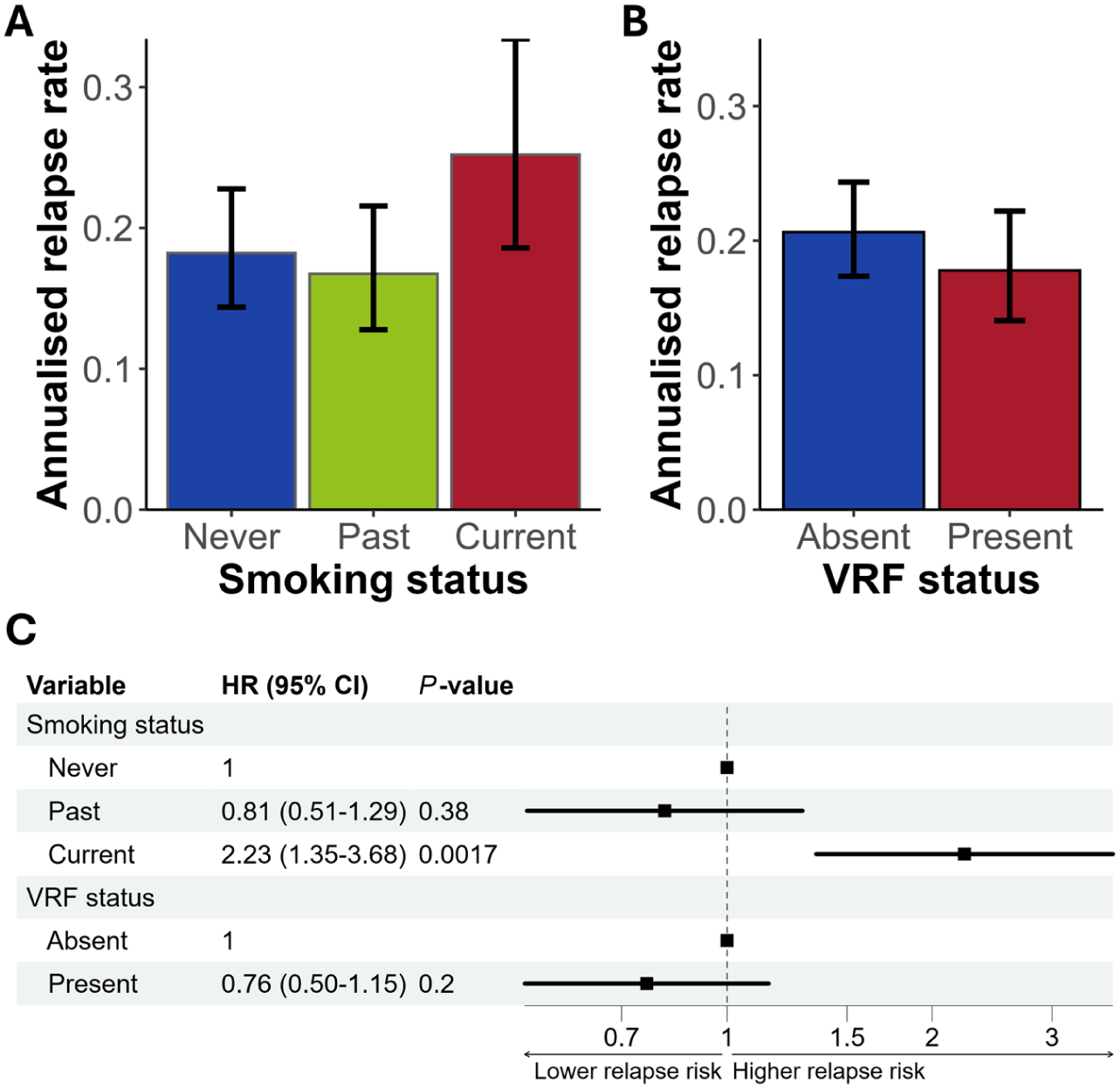
Annualised relapse rate and smoking and vascular risk factor status. Crude annualised relapse rates were calculated by smoking status (a; categories of never [ref], past and current smoking) and non-smoking vascular risk factor (b; binary variable for the presence of ⩾ 1 VRF) status, respectively. Forest plot for multivariable negative binomial regression results for smoking and non-smoking VRF status (c), adjusted for covariates of age at onset (modelled linearly), sex, onset attack phenotype, acute steroid use following onset attack, and immunosuppressive treatment use, with offset term included of log of the interval duration. CI, confidence interval; VRF, vascular risk factor.
Univariable negative binomial regression analyses for relapse rate.
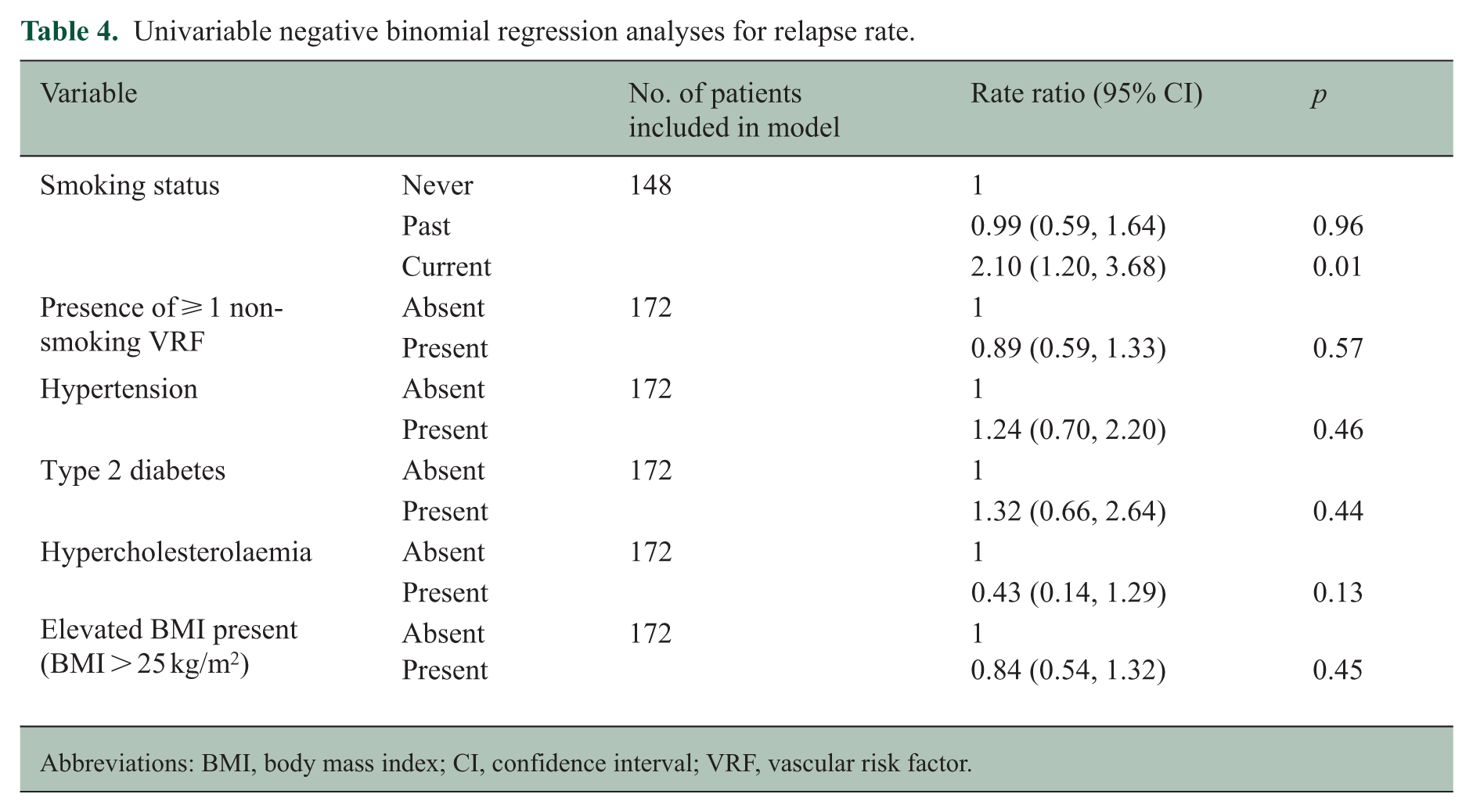
Abbreviations: BMI, body mass index; CI, confidence interval; VRF, vascular risk factor.
Crude annualised relapse rate was similar between those with and without any non-smoking VRF (Figure 3(b); annualised relapse rate for those with non-smoking VRF = 0.18, and without non-smoking VRF = 0.21). Univariable analyses showed no significant association with VRF status or presence of individual non-smoking VRF on relapse rate (Table 4).
Multivariable negative binomial regression, of data from 148 patients with both smoking and VRF status available, was consistent with our univariable regression results (Figure 3(c) and Table 5). Current smokers had a 2.2 times higher annualised relapse rate compared to never smokers (adjusted rate ratio 2.23 [95% CI 1.35–3.68], p = 0.0017), with no difference between past and never smokers. Presence of at least one non-smoking VRF was not significantly associated with relapse rate. In sensitivity analyses, current smoking status remained significantly associated with elevated relapse rate in both models (Supplemental Tables 5 and 6).
Multivariable negative binomial regression results for relapse rate.
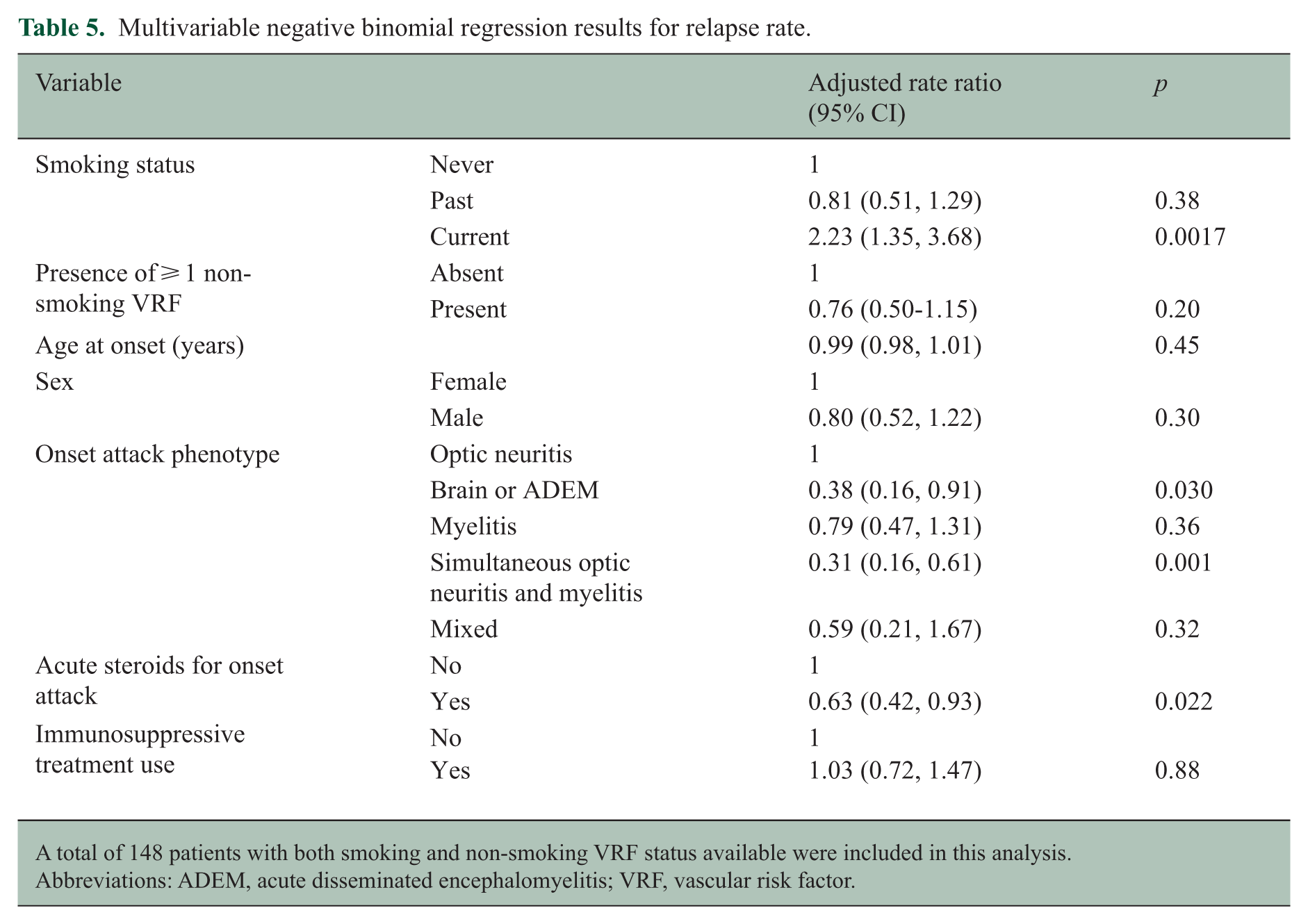
A total of 148 patients with both smoking and non-smoking VRF status available were included in this analysis.
Abbreviations: ADEM, acute disseminated encephalomyelitis; VRF, vascular risk factor.
Discussion
In our retrospective observational study of a single specialist service cohort, we found that patients with MOGAD who were current smokers had a greater relapsing propensity over time than never smokers, whereas relapse rates were not significantly different between past and never smokers. Non-smoking VRF were not observed to associate with relapse risk. In our multivariable analysis for time-to-relapsing course, neither smoking nor VRF status were associated with an increased hazard, although univariable analysis suggested elevated risk in current smokers. Overall, these findings provide additional impetus for smoking cessation as a part of MOGAD management.
To date, there have been few studies on smoking and VRF comorbidities and their interactions with MOGAD. Several studies from our group had examined the effects of smoking on recovery from disability following an attack. One study with 70 MOGAD patients observed a higher percentage of ever smokers had poor recovery compared to never smokers, but this did not reach statistical significance likely due to being underpowered. 9 In a subsequent study which included 236 MOGAD patients and classified smoking status as current, past or never, current smokers had a significantly lower chance of a good recovery after the onset attack compared to never smokers. 8 There is also evidence of smoking effects on radiological activity. The majority of T2 hyperintense lesions seen on neuraxial magnetic resonance imaging in MOGAD during an attack have been observed to resolve on interval imaging done several months later. 14 However, current and past smokers had higher risk for lesion persistence compared to never smokers. 15
We add to the data indicating detrimental effects of active smoking on MOGAD by showing that current smokers have an elevated relapse rate over time compared to never smokers. There was no significant difference between past and never smokers, suggesting smoking cessation results in the resumption of a baseline relapse propensity for MOGAD. A previous study did not identify a difference in relapse rates between ever and never smokers. 9 The combination of current and past smokers together as ‘ever smokers’, and their sample size which was under half of this current study’s, likely limited their ability to detect effects of smoking on relapse activity. In our current study, we observed a significant difference between current and never smokers in relapse rates but not in time-to-relapse which only measure one outcome rather than the effects on relapses over time and could be due to underpowering. However, the direction of the point estimate from our Cox regression model was consistent with the results from our analysis on relapse rates. Considering the data on smoking from our study and past studies examining other facets of disease activity, there is consistent evidence for harm from active smoking in MOGAD. All these data support smoking cessation for its benefits in MOGAD.
The biological mechanisms by which smoking leads to increased relapse activity or impaired disability recovery in MOGAD are unclear. Smoking has been associated with increased levels of pro-inflammatory cytokines in the circulation. Systemic inflammatory markers which have been found to be elevated in current smokers include serum interleukin-6 and C-reactive protein.16,17 Of interest is the suggested effectiveness of anti-interleukin-6 receptor monoclonal antibodies in the prevention of relapse in MOGAD as reported in case series and small observational studies.18 –21 Given these, interleukin-6 receptor signalling may be a pathway by which smoking modulates MOGAD disease activity, but this hypothesis needs to be tested. A recent study of the immunological effects of smoking showed that innate immune modulations are reversed with smoking cessation, whereas adaptive immune effects can persist even after smoking cessation due to epigenetic modifications. 22 In the context of our findings, reversal of innate immune changes due to current smoking may explain similar relapse rates between past and never smokers. There is also evidence for increased disease activity associated with smoking in other neurological and non-neurological autoimmune diseases, such as multiple sclerosis23 –25 and rheumatoid arthritis,26,27 suggesting possible shared pathways for perturbations relating to smoking.
The study of VRF on MOGAD has so far been limited. One study showed no significant association between the presence of VRF and degree of recovery following the onset attack. 8 A second study observed that MOGAD patients who had at least one cardiovascular comorbidity or risk factor were significantly older than those without any comorbidity, but with no significant difference in baseline disability. 28 In our study, we examined VRF and relapse activity and did not identify any association. One interpretation of our results is that the non-smoking VRF which we studied do not influence relapse activity. However, the number of patients with individual VRF were relatively small, with the most common non-smoking VRF present in our cohort being elevated BMI at 27.9%. Power was likely to have been limited to be able to detect any effects from individual VRF. Larger future cohorts with available comorbidity data will help to clarify any potential effects of non-smoking VRF on MOGAD disease activity.
Our study is one of few to investigate smoking and VRF and their effects on MOGAD. A key strength of our study is its cohort from a single specialist service, through which all patients are assessed by at least one of two neurologists with expertise in MOGAD. This minimises data heterogeneity and ensures inclusion of well phenotyped patients. In the United Kingdom, most patients with MOGAD are referred to and seen through one of two highly specialised services, and so our findings are likely applicable to a general MOGAD cohort. Although, there may be more patients with a relapsing course due to selection bias in the overall cohort from our specialised service. Since the prior study from our group, we have increased the granularity of smoking status by including current and past smoker categories. There are several limitations in the current study. Our service’s setup as a nationally commissioned service for MOGAD may help to mitigate against biases which observational studies are at risk of, such as information bias, although these may still be present. Smoking status was self-reported and not defined by a given smoking quantity (e.g. by pack-years), which may introduce heterogeneity within smoking groups. We had a small group of current smokers and we did not have available quantification of smoking history in pack years, which would be useful in exploration of the subgroup of current smokers in particular. However, the lack of difference between past and never smokers would suggest the level of smoking, at least once stopped, may not be important. We also had limited numbers of patients with each non-smoking VRF examined, which limited our ability to investigate the effect of each individually. Comorbidity status was determined by questionnaire or initial clinical height/weight assessment, rather than biochemically such as through lipid profiles or HbA1c. Self-reported comorbidity status likely represents a known prior diagnosis, although we did not have data on their control or management status. There is a possibility for misreporting of comorbidities by patients. The smoking and VRF data used in this study were collected at one timepoint following disease onset and then clinic entry, and we did not have start/stop dates to be able to account for changes to smoking status during follow-up. Although, we expect time-varying probabilities for smoking status change to be low, future studies with more granular exposure data will be important to confirm our findings. Despite adjustment for several variables in our multivariable models, there may be ongoing confounding from unmeasured variables which may affect our results. Our study was also not designed to investigate causal effects of treatment. Further work both in basic and clinical sciences is required to establish if there is a causal relationship between current smoking and elevated relapse risk.
In conclusion, active smoking was associated with an increased relapse rate of patients with MOGAD compared to non-smokers, whereas past smokers had similar relapse rate to non-smokers. Our results suggest a detrimental effect of current smoking on MOGAD disease activity and highlight the potential benefits of smoking cessation in MOGAD. Future studies with larger cohorts and more granular data on smoking and comorbidities will be useful to further understand the interactions between these and MOGAD, and inform how we should best manage them.
Supplemental Material
sj-pdf-1-msj-10.1177_13524585261429292 – Supplemental material for Myelin oligodendrocyte glycoprotein antibody-associated disease and relapse risk: Effects of smoking and vascular risk factors
Supplemental material, sj-pdf-1-msj-10.1177_13524585261429292 for Myelin oligodendrocyte glycoprotein antibody-associated disease and relapse risk: Effects of smoking and vascular risk factors by Wei Zhen Yeh, Fiona Chan, Anna Francis, Helmut Butzkueven, Ruth Geraldes, Maria Isabel Leite and Jacqueline Palace in Multiple Sclerosis Journal
Footnotes
Acknowledgements
We acknowledge Ms Madelina Miron and Ms Yvonne Sharawakanda, NMO specialist nurses, who have input data into the Oxford NMO database. W.Z.Y. was supported by an ECTRIMS Research Fellowship and acknowledges the ECTRIMS Research Fellowship Programme for its support. He was also supported by the Robert and Elizabeth Albert Travel Grant from the RACP Foundation, Mr Robert O. Albert AO and Mrs Elizabeth Albert. He is currently supported by an MS Australia Postdoctoral Fellowship (24-PDF-0166) and acknowledges MS Australia and MSWA for their support. The authors thank the highly specialised services NHS England for funding the Neuromyelitis Optica Service in Oxford.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: W.Z.Y. is currently supported by an MS Australia Postdoctoral Fellowship and was supported by an ECTRIMS Research Fellowship and the Robert and Elizabeth Albert Travel Grant from the RACP Foundation, Mr Robert O. Albert AO and Mrs Elizabeth Albert; and has received travel support from Biogen, UCB and the Guthy-Jackson Charitable Foundation outside of the submitted work. F.C. was awarded an ECTRIMS fellowship in 2023–2024. A.F. received a travel grant from Alexion in 2023. H.B.’s institution (Monash University) received compensation for consulting, talks, and advisory/steering board activities from Alfred Health, Biogen, Genzyme, Merck, Novartis; research support from Biogen, Merck, Multiple Sclerosis Australia, National Health and Medical Research (Australia), Novartis, the Oxford Health Policy Forum, the Pennycook Foundation, Roche. R.G. has received support for scientific meetings and courses and honoraria for advisory work from Bayer, Biogen, Merck, Novartis, Jasen, MIAC and UCB. M.I.L. is funded by the NHS (Myasthenia and Related Disorders Service and National Specialised Commissioning Group for Neuromyelitis Optica, UK) and by the University of Oxford, UK. She has been awarded research grants from UK associations for patients with myasthenia and with muscular disorders (Myaware and Muscular Dystrophy UK, respectively) and the University of Oxford. She has received speaker honoraria or travel grants from Biogen, Novartis, UCB Pharma and the Guthy-Jackson Charitable Foundation. She serves on scientific or educational advisory boards for Argenx, UCB Pharma and Viela Bio/Horizon Therapeutics/Amgen. J.P. has received support for scientific meetings and honorariums for advisory work From Merck Serono, Novartis, Chugai, Alexion, Roche, Medimmune, Amgen, Vitaccess, UCB, Mitsubishi, Amplo, Janssen; Grants from Alexion, Argenx, Clene, Roche, Medimmune, Amplo biotechnology; Patent ref P37347WO and licence agreement Numares multimarker MS diagnostics Shares in AstraZenica. Her group has been awarded an ECTRIMS fellowship and a Sumaira Foundation grant. A Charcot fellow worked in Oxford 2019–2021. She acknowledges partial funding to the trust by Highly specialised services NHS England. She is on the medical advisory boards of the Sumaira Foundation and MOG project charities, is a member of the Guthy Jackon Foundation Charity and is on the Board of the European Charcot Foundation and a member of MAGNIMS and the UK NHSE IVIG Committee and chairman of the NHSE neuroimmunology patient pathway and ECTRIMS Council member on the educational committee since June 2023. Currently, she is on the ABN advisory groups for MS and neuroinflammation and recently on neuromuscular diseases advisory group.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: W.Z.Y. was supported by an ECTRIMS Research Fellowship and acknowledges the ECTRIMS Research Fellowship Programme for its support. He was also supported by the Robert and Elizabeth Albert Travel Grant from the RACP Foundation, Mr Robert O. Albert AO and Mrs Elizabeth Albert. He is currently supported by an MS Australia Postdoctoral Fellowship (24-PDF-0166) and acknowledges MS Australia and MSWA for their support. The authors thank the highly specialised services NHS England for funding the Neuromyelitis Optica Service in Oxford.
Ethical Considerations
This study was approved by the Oxford C Research Ethics Committee, reference 21/SC/0353.
Consent to Participate
Written consent was obtained from included participants.
Consent for Publication
Not applicable.
Data Availability Statement
The anonymised grouped data that support the findings of this study are available from the corresponding author to qualified noncommercial investigators, on reasonable request and in accordance with consent and ethics requirements.
Supplemental Material
Supplemental material for this article is available online.
