Abstract
Background:
Considerable progress has been made in understanding genetic and environmental risk factors for multiple sclerosis (MS), yet we still cannot prevent MS.
Objectives:
To drive progress in primary and secondary prevention of MS by developing a comprehensive research agenda based on current knowledge.
Methods:
A global workshop convened people with lived experience, clinicians, researchers, and policymakers with expertise in MS, other chronic diseases, epidemiology, clinical trials, genomics, immunology, virology, behavioral science, and public health to develop pathways to advance the MS prevention agenda.
Results:
Regarding primary prevention, workshop participants recommended acting on known modifiable risk factors; identifying novel etiological factors and determining how they lead to disease; and building coalitions including organizations with shared interests. Regarding secondary prevention, workshop participants recommended identifying biomarkers relevant from initial immune dysregulation through to initial clinical presentation, linking biomarkers to long-term MS outcomes, developing cost-effective screening tools for MS and point-of-care testing, and developing prodromal criteria for MS that could be implemented clinically. Communication and evaluation frameworks, adoption of an implementation mind-set, and engagement of public health were highlighted as key supporting elements.
Conclusion:
This workshop sets the stage for developing a global prevention agenda for MS.
Introduction
In 2020, nearly 3 million people were living with multiple sclerosis (MS) worldwide. 1 Costs to affected individuals, their families, health systems, and society remain high. 2 Considerable progress has been made in understanding genetic and environmental risk factors for MS, and its pathobiology. The expanding array of disease-modifying therapies (DMTs) reflects highly effective global scientific collaboration. Yet, MS still cannot be prevented.
The Pathways to Cures Roadmap aims to coordinate global efforts to cure MS via three pathways. 3 The Stop pathway seeks to halt disease activity or injury to the central nervous system from the point of intervention onward. The Restore pathway seeks to repair the central nervous system and to develop strategies to maintain or improve function. The End pathway seeks to prevent new cases of MS. Notably, among the 2346 MS-related projects being funded by MS societies and government sources globally, only 197 were devoted to the End pathway, 4 accounting for 6% of funds awarded. In 2024, the updated Pathways to Cures Roadmap highlighted that the Stop pathway should target early detection and treatment, 3 a concept closely aligned with preventing new cases of MS.
Researchers in other immune-mediated diseases have moved ahead with clinical trials to prevent development of disease, or to delay presymptomatic disease from progressing to clinical manifestations.5,6 These and other advances such as the success of clinical trials in people with radiologically isolated syndrome (RIS)7,8 indicate that now is the time to intensify efforts to prevent MS, despite the challenges of designing appropriate studies.
In April 2025, MS Canada and MS Australia sponsored an international workshop to develop a global research agenda for preventing MS. Participants included people with lived experience; clinicians, researchers and policymakers with expertise in adult and pediatric MS along with expertise in other immune-mediated diseases, epidemiology, clinical trials, immunology, virology, genomics, artificial intelligence, biomarkers, behavioral science, and public health (Appendix 1). Herein, we present the discussions held and recommendations made during that workshop.
Developing a framework for prevention in MS
Workshop participants adopted consistent terminology to describe the evolution of MS from the susceptibility stage to clinical manifestations (Figure 1). At birth, individuals are susceptible to disease based on their genetics; interactions between genetics and exposures from the perinatal period onwards result in the biological onset of MS. How biological onset is defined or measured is a difficult question. It is unknown how soon after biological onset or if any clinical symptoms or signs would be observed or if changes on conventional magnetic resonance imaging would be observed, but immunological abnormalities may be identifiable, as with other immune-mediated diseases.9,10 Biological onset differs from causal sufficiency as (presumably) not all those who reach causal sufficiency will progress to biological onset of MS. Nor do some (yet undetermined number) with biological onset go on to manifest MS clinically or radiologically. Importantly, from biological onset onwards, affected individuals have the disease; meaning that potential interventions are targeting individuals with MS, not healthy people. After biological onset, a constellation of signs and symptoms may develop that do not include events typical of central nervous system demyelination although non-specific neurological and non-neurological symptoms may occur; this is the prodromal stage. Such prodromal stages are recognized in many diseases, including those that are immune-mediated and neurodegenerative.11,12 However, the length of the prodromal stage may vary substantially between individuals, and it may not be readily identifiable in everyone. Following the prodromal stage, individuals manifest neurological signs or symptoms in combinations that are typical of MS, such as transverse myelitis (which may cause lower limb numbness, weakness, bowel and bladder dysfunction). Individuals with RIS, who have imaging findings compatible with demyelinating disease in the absence of overt signs or symptoms of neurological disease, are challenging to place in this framework. Historically, these individuals were not classified as having MS, but some might be classified as being in the prodromal stage. This is particularly true if their imaging was done for symptoms such as headache or fatigue, which appear to be features of the MS prodrome; health care use is higher among individuals with RIS before their RIS diagnosis than in controls, although a “typical” MS prodrome has not been identified to date. 13 The 2024 revision of the diagnostic criteria now allows the diagnosis of MS in some individuals with RIS and possibly some individuals who would currently be classified as having prodromal MS; 14 the continued evolution of the diagnostic criteria will need to be carefully considered in the design of prevention trials and description of the course of MS.
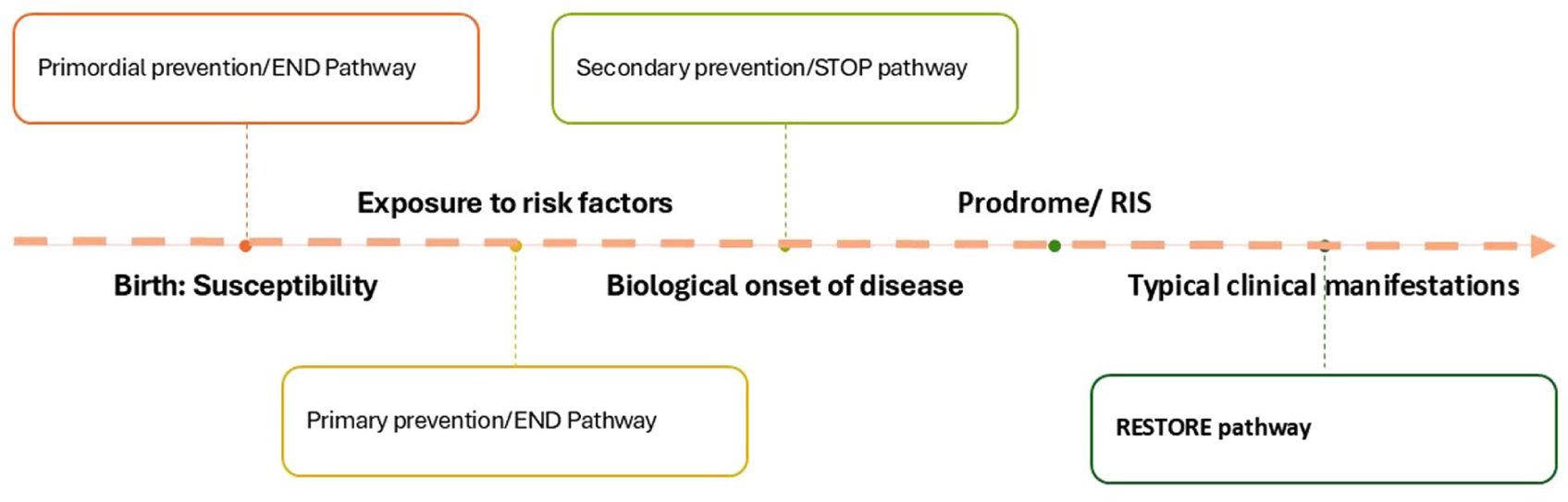
Schematic of disease evolution and timing of prevention interventions
Public health nomenclature characterizes prevention interventions relative to disease onset. Primordial and primary prevention efforts seek to inhibit the biological onset of the disease, so that no new cases of MS develop. Primordial prevention efforts prevent risk factors, such as obesity or infectious mononucleosis, from occurring. Primary prevention efforts target acquired risk factors to prevent disease, such as mitigating vitamin D insufficiency with vitamin D supplementation. Within the Pathways to Cures Roadmap, primordial and primary prevention correspond to the End pathway. 3 In contrast, secondary prevention efforts seek to identify disease in its earliest stages (biological onset prior to the prodrome or during the prodrome), so that interventions can prevent or delay further disease evolution; this corresponds to the Stop pathway.
Prevention of MS could be cost-effective, improving health while mitigating long-term health system and societal costs. Effective management, including DMT, is needed to improve the ability of persons living with MS to remain healthy, employed, and engaged in their communities. However, MS is costly to manage over a lifetime2,15 and some people are unable to access good MS treatment and care.
Risk factors for MS and primary prevention
Primary prevention efforts for MS will need to choose whom to target (Table 1); what risk factors to target; when in the individual’s life course to intervene; and how to intervene. In that context, the workshop briefly reviewed current knowledge of MS etiology. Genetically, MS risk is polygenic, involving over 230 common DNA variants, the majority outside of the genomic region encoding the human leukocyte antigen (HLA) system. 16 A recent multi-ancestry genome-wide association and co-localization study found that some of these variants are shared with other autoimmune diseases and major depressive disorder, while others appear to be MS-specific, affecting immune cells, inhibitory neurons, and glia. 17
Identifying individuals to participate in primary and secondary prevention trials.

These individuals have disease; thus, interventions at this stage are not targeting healthy volunteers, in contrast to trials identifying those at elevated risk of MS in whom biological onset of disease has yet to occur. **First-degree relatives (parents, children, siblings) in simplex families also have an increased risk of MS but not as high as in multiplex families or monozygotic twins; so they are not as high a priority population.
While multiple environmental factors have been examined in MS, only a few have consistent supporting epidemiological evidence, including Epstein-Barr virus (EBV), childhood and adolescent obesity, low sunlight exposure, vitamin D insufficiency/deficiency, and cigarette smoking. 18 Of these, EBV may offer the biggest preventive impact. 19 Most of these risk factors interact with genetic factors to amplify MS risk. 18 Much of the existing evidence regarding risk factors for MS has been drawn from populations of White European ancestry, raising concerns regarding the generalizability of this evidence to populations with differing ancestries and ethnicities. More recent studies in MS suggest these associations are similar in direction in MS populations who identify as Black or South Asian, 20 although inconsistencies have been reported regarding vitamin D insufficiency. 21
Workshop participants acknowledged gaps in our understanding of disease etiology that need to be addressed. First, other, undiscovered etiological factors almost certainly exist, and many people exposed to each (or multiple) identified risk factors will never develop MS. Second, we do not know which causal model 22 explains how exposure to environmental risk factors in a genetically susceptible individual causes MS. Do different environmental factors have differential effects across life stages? That is, is there a sensitive period when these exposures must occur, as suggested by migration studies, the greater importance of childhood/adolescent than adult obesity, and influence of age at exposure to EBV?23,24 Do risk factors accumulate such that the accumulation and duration of exposures is more important than their timing, or is a chain-of-risk model in which each exposure increases the risk of another exposure more appropriate? Third, the mechanisms by which these risk factors lead to biological onset of disease are incompletely understood, although progress is being made; for example, a growing literature links EBV to MS via molecular mimicry, immortalization of B cells, and immunomodulation among others.25 –29 Elucidating those mechanisms would offer the potential for preventive interventions that did not involve individual behavior change; this is important because behavior is hard to modify, and common pathways may underlie multiple risk factors. Fourth, what is an acceptable level of risk for an intervention aimed at preventing MS, does it vary according to the individual’s risk of MS and personal risk tolerance, and how do we balance intervention risks with uncertain benefits?
Considerable discussion focused on EBV given advances in understanding the mechanisms underlying the relationship between EBV and MS, and potential availability of an EBV vaccine. Key considerations regarding EBV-related interventions to prevent MS are discussed in recent reviews.30,31 Concerns specific to primary prevention of MS include the lack of sterilizing immunity provided by vaccination, 32 although this is not universally accepted as a concern, acceptable age of vaccination to ensure uptake and impact, and whether targeting infectious mononucleosis would have sufficient preventive effect. Targeting latent and lytic EBV infection via antivirals is an alternative strategy to vaccination, although potentially more relevant to secondary prevention; this is discussed elsewhere. 33
Workshop participants noted multiple potential facilitators that could address some of the challenges identified, as well as targets for early investment (Table 2). They reiterated the importance of acting on what we know now, potentially via targeting modifiable factors, building or joining coalitions to take collective action, building the economic argument for primary prevention, and engaging with public health (discussed later). Based on the component cause model, workshop participants emphasized the importance of a multipronged approach targeting multiple risk factors.
Considerations for primary and secondary prevention studies.
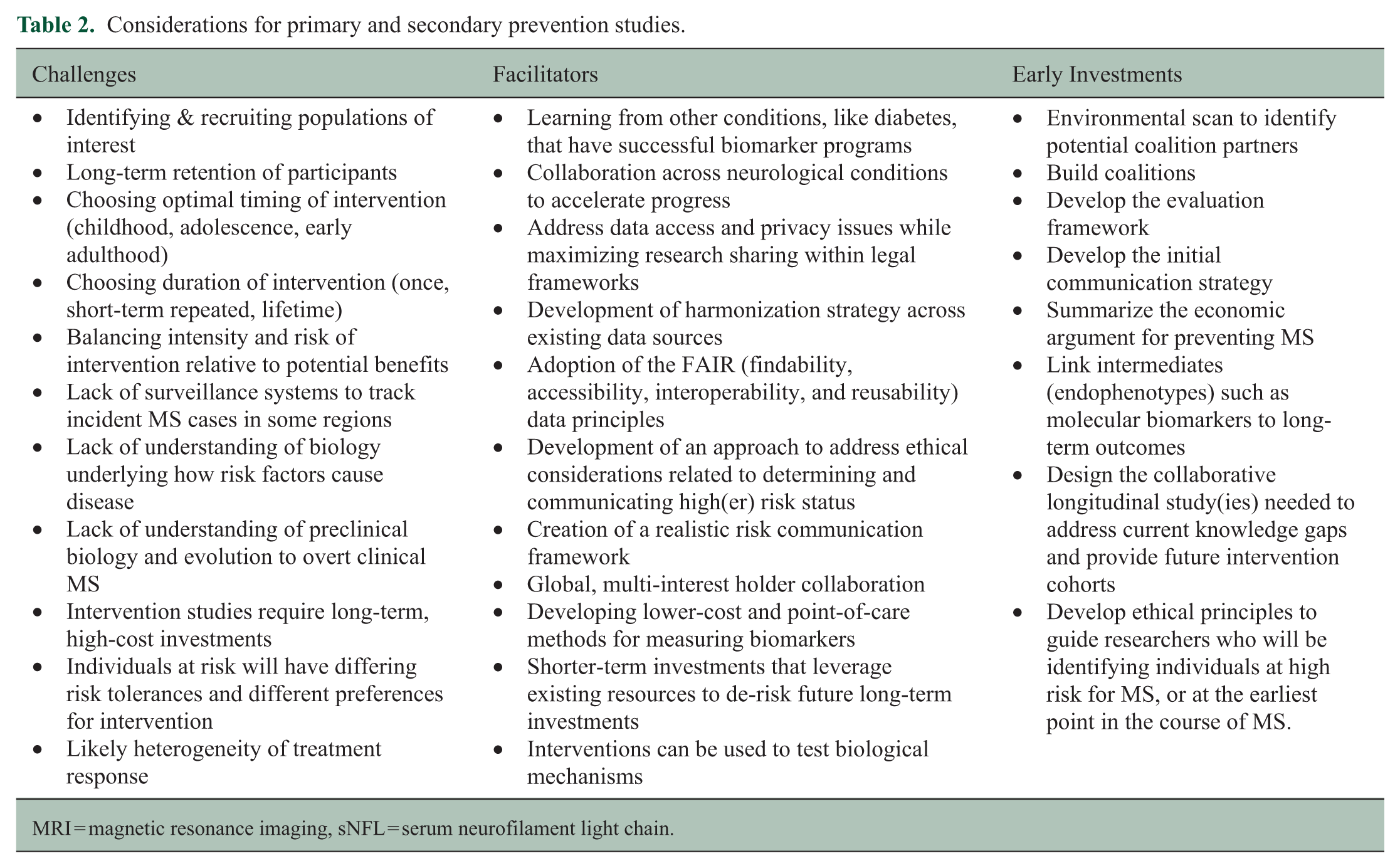
MRI = magnetic resonance imaging, sNFL = serum neurofilament light chain.
Secondary prevention
As with primary prevention efforts, secondary prevention efforts will need to identify: (i) whom to target, that is, how to identify individuals in whom biological onset of disease has occurred but presentation with first typical symptoms has not occurred (Table 1); (ii) what risk factors to target; (iii) when in the individual’s life course to intervene; and (iv) how to intervene. Existing evidence strongly supports the concept of a prodromal phase in MS, but contemporaneous recognition is difficult without an operational definition of the prodrome. Changes in health care use start 5 to 20 years before clinical presentation with typical symptoms in adult and pediatric populations.34,35 Cognition and school performance are lower before clinical presentation in people with MS than without MS.36 –38 Serum neurofilament light chain levels rise, up to 10 years before clinical presentation, indicating preceding neuroaxonal injury.39 –42 Further, a recent study reported that 10% of people with MS have a common autoantibody signature in serum that is present before clinical presentation and that appears to be highly specific for MS. 42 Other autoantibody biomarkers may be assessed in the future.43 –45
Workshop participants recommended continuing work to identify and validate biomarkers to support early detection of disease and intervention (Table 1). Biomarkers that detect disease at the point of initial immune dysregulation were viewed as particularly valuable. Studies linking intermediates or endophenotypes (biomarkers, prodromal symptoms) to subsequent confirmed MS, potentially decades later, are needed to support feasible, short-duration trials of interventions. A better understanding of the paths from prodromal symptoms to first demyelinating event and subsequent diagnosis is needed to understand opportunities for earlier diagnosis,46,47 as are validated prodromal criteria for MS. Tools for early detection of MS need to be available at point of care, and accessibility and cost must not be barriers to global use. 48 These recommendations were generally consistent with prior recommendations from a workshop supported by MS Canada aimed at advancing understanding of prodromal MS. 49
Intervention
The five-tier health impact pyramid describes how different types of interventions influence health (Figure 2). 50 The bottom tier has the greatest population-level impact and captures changes in social determinants of health such as poverty or pollution. Interventions in the second tier modify the environmental context so that healthy choices become the default. Interventions in the third tier happen only once or rarely but provide ongoing benefits; unlike the first two tiers, they are applied at the individual level. The fourth tier captures clinical interventions which require ongoing use, such as medications. The final tier captures health education interventions targeted at the individual; these are the least effective public health interventions. Generally, individual-level interventions are more intensive, requiring more resources per individual, but can be more targeted and more effective for the individual. Any intervention applied at the individual level requires thoughtful risk communication and must be acceptable to support high uptake; this means that assessing psychological outcomes of interventions being tested is critical. Population-level interventions are of lower intensity, but have broader reach, potentially achieving greater impact overall.
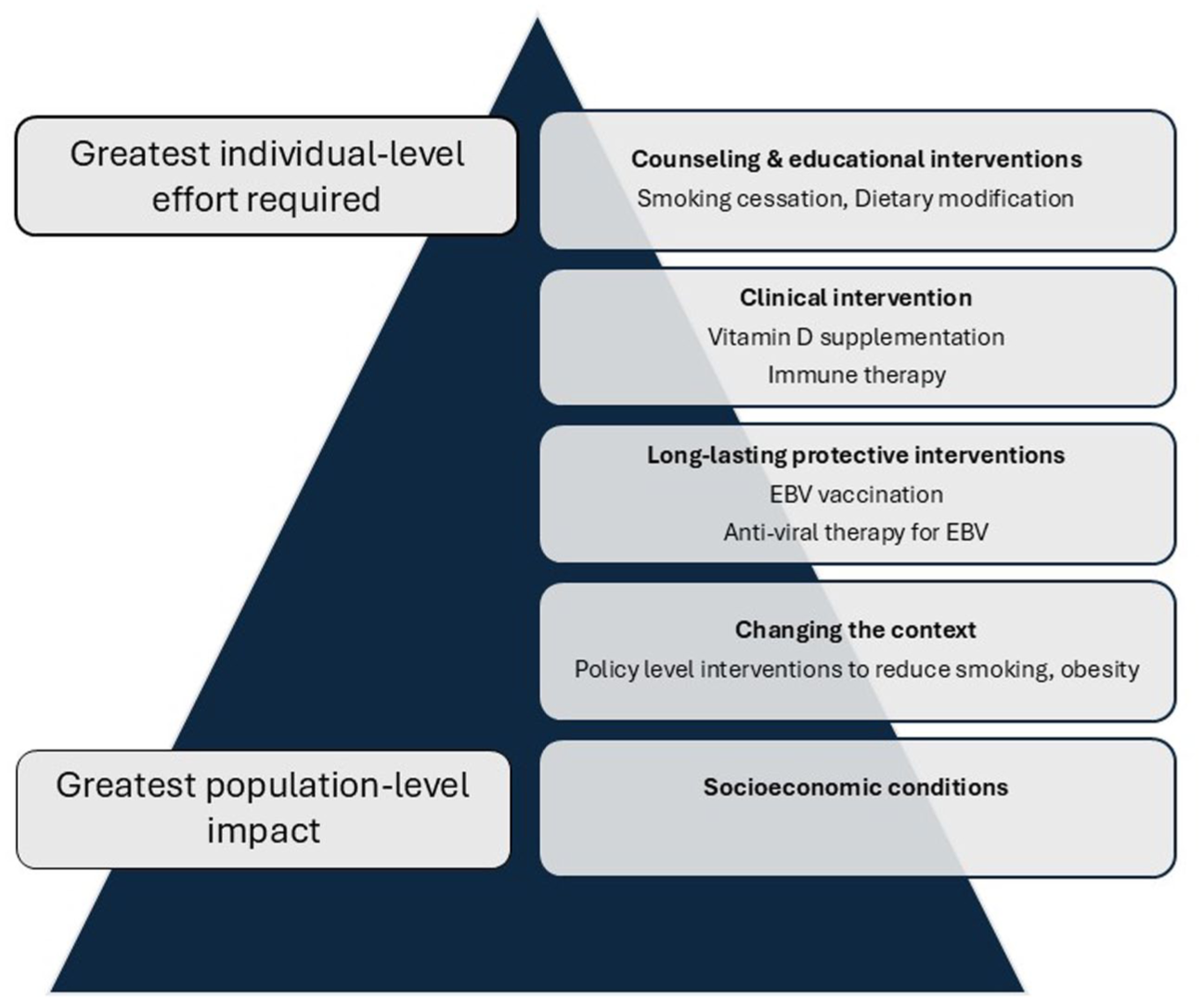
Health impact and intervention strategies for multiple sclerosis (adapted from (Frieden 2010)).
A workshop presentation provided insight into the application of interventions from three tiers of this pyramid for cancer prevention. 50 Smoking prevention efforts, for example, have used policy levers and legislation such as cigarette taxes and legislation banning indoor smoking in public places to modify environmental context at the population level. Vaccinations for human papilloma virus, which causes cervical cancer, are a protective intervention that requires administration of 1–3 injections in an individual’s lifetime. Dietary counseling to reduce obesity is an individual-level educational effort representing the final tier of the health pyramid. Workshop participants emphasized that any strategy for preventing MS will need to use interventions targeting a range of factors at different levels of the impact pyramid (Figure 2); at the population level, ensuring impact can be captured is crucial, while at the individual level, choice is important. The type of intervention may also vary at different time points in an individual’s lifespan.
Prevention efforts must adopt a global lens, recognizing that MS affects people worldwide 1 and that access to expertise, infrastructure, and involvement of people at risk of MS from many regions will be needed. The benefits of targeting risk factors for MS are likely to have more “global” effects in preventing other diseases. Implementation of efficacious interventions will need to be tailored to local context; in low-resource regions, this may require building capacity and working with communities to deliver the intervention. 51 Where multiple scientifically sound interventions may be available, we need to prioritize testing interventions that are cost-effective and can be implemented globally.
Measuring success
Workshop participants discussed the need for an evaluation framework to measure the success of prevention initiatives and identified several key elements. These included: (i) providing statements about the mission, current state, and desired future state; (ii) identifying all interest holders and their roles; (iii) measuring engagement (who, in what ways, how often); (iv) measuring the processes or tools created or implemented (such as a surveillance framework for measuring MS incidence); (v) measuring uptake of processes and tools (by whom, where); and (vi) measuring the impact of those processes broadly. Specifically, measuring these impacts should go beyond “no new MS cases.” For example, preventing infectious mononucleosis would reduce missed time from school and work among teens and young adults, reduce short-term health care costs related to diagnosis and management, and reduce long-term impacts related to EBV-associated syndromes and cancer. 52
The evaluation framework can be supported by requiring investigators to identify and measure outputs for each study outcome and by including economic evaluations in intervention studies that will support implementation by health systems. The benefits of developing a framework at the outset include: clearly identifying the gaps that need to be filled, identifying potential partners to form a coalition to support prevention by articulating who else may benefit from interventions, providing evidence of progress (such as improved awareness that MS could be prevented, number of coalition partners, uptake of interventions) to support engagement of additional partners and bolster the collective economic argument for prevention, and to identify what strategies and priorities show early signs of success or failure to allow course corrections.
Coalitions, engagement, and implementation
Building coalitions and collaborations is critical to preventing MS. Coalitions can pool knowledge, expertise, funding, and other resources from multiple sectors.53,54 Coalitions can more effectively build community awareness, facilitate communication across sectors, share the risks and responsibility of the plans, and benefit from more diverse perspectives and strategies, making them more effective at advocating for changes in health policy and developing sustainable interventions that benefit all interest holders. 54 Potential interest holders include people with lived experience of MS and their families, those at risk of MS who do not have a family member with MS, local and international MS organizations, and organizations targeting risk factors or conditions with overlapping risk factors that are relevant to MS (Table 3).
Examples of potential coalition partners.
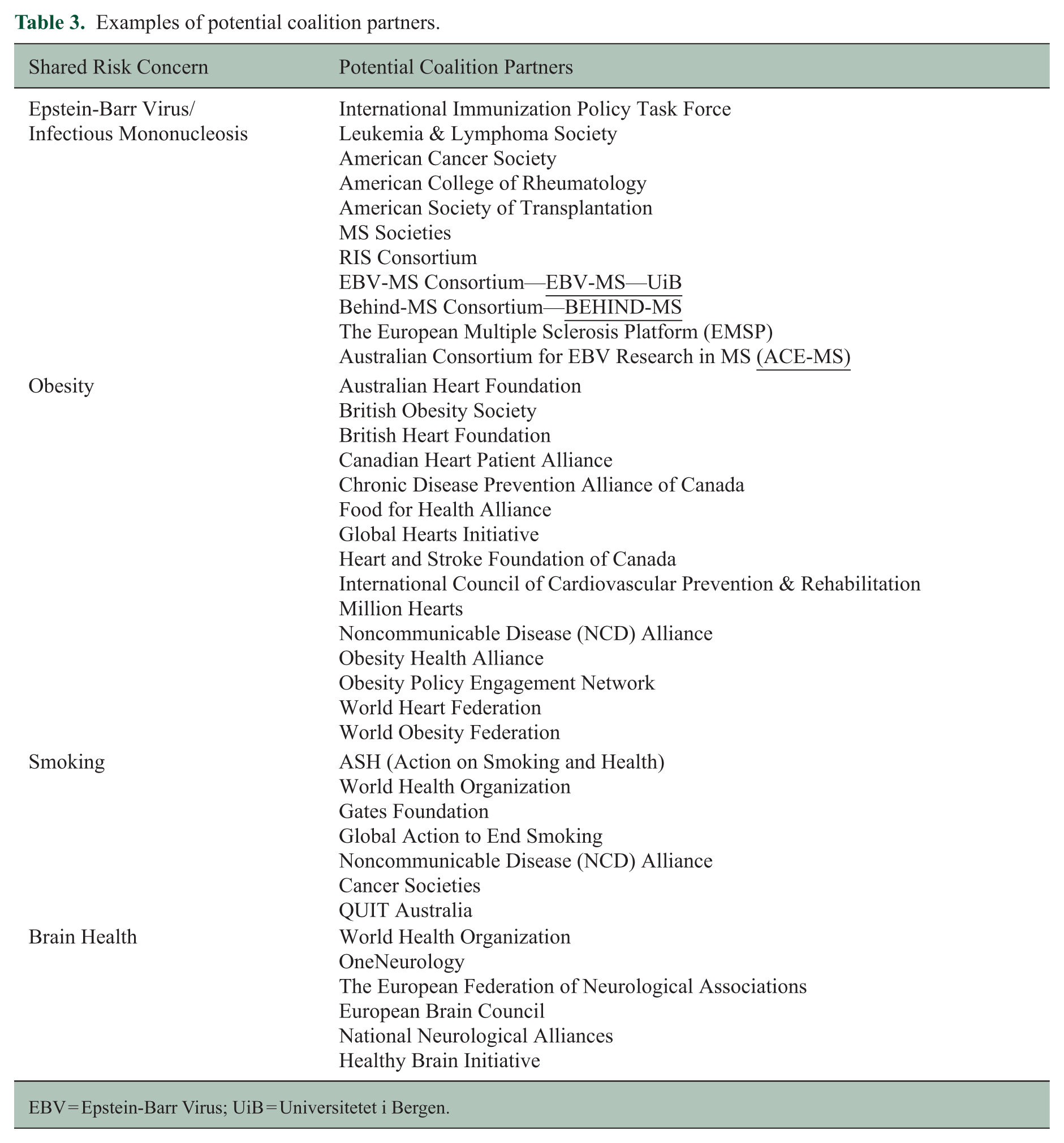
EBV = Epstein-Barr Virus; UiB = Universitetet i Bergen.
Workshop participants emphasized the importance of incorporating individual and community perspectives from across the world and placing them in the context of relevant behavioral change frameworks 55 to ensure development and adoption of effective preventive interventions. For example, to prevent MS with an EBV vaccine, a vaccine that prevents EBV must exist, be approved for use, be offered by the health system without barriers to access such as high cost, and be acceptable to the individual and their family. These challenges are exemplified by the varied uptake of the HPV vaccine for cervical cancer prevention, 56 and approval of the sole therapy (teplizumab) proven to delay onset of stage 3 (clinical) type 1 diabetes onset only in the United States. 57
Any coalition aimed at preventing MS must engage with public health agencies. Public health broadly focuses on health protection, disease prevention, and health promotion to improve population health outcomes. The core challenge when engaging public health agencies in efforts to prevent MS is that public health focuses on population health broadly, although engagement in disease-focused activities can occur; examples include the COVID-19 pandemic, AIDS, heart disease, diabetes, and obesity. A solid evaluation framework and evidence-based case for prevention will facilitate the engagement of public health and other coalition partners.
Communication and knowledge mobilization
Workshop participants emphasized the importance of a comprehensive communication and knowledge mobilization plan to support prevention efforts. The specific strategies employed will depend on the message (e.g. primary vs secondary prevention, intervention type) and the audience (e.g. primary care providers, MS care providers, the general population, family members of people with MS). Messaging must be consistent with language used by other global initiatives in the MS field, including the Pathways to Cures strategy, the diagnostic criteria for MS, and the disease course descriptors.
For primary prevention, workshop participants recommended fitting MS into existing campaigns about health behaviors (e.g. smoking, obesity) and providing information about addressing risk factors for MS (e.g. smoking, obesity, vitamin D insufficiency) when people engage with providers about their health, although the impact of this approach is hard to measure. For secondary prevention, messaging is more challenging because it involves earlier detection of disease, at a time when an individual may still feel well. When interventions are not available, detection of early disease may adversely affect emotional well-being, medicalize daily life, and increase the risk of stigma and discrimination related to insurance, as well as in employment and social settings. 58 However, individuals have the right to information about their health. With consent and support, an understanding of their prognosis may allow individuals to plan better for their future. 59 Messaging about when MS begins will need to shift gradually, a challenge which may be informed by the experience of type 1 diabetes when presymptomatic stages were incorporated into the disease framework. 9 Health systems must be prepared to handle increased awareness of MS and concerns of a larger number of individuals who may present requesting investigation of possible early MS, to ensure this does not adversely affect access to care and wait times, and to manage the related ethical issues mentioned earlier.
Future directions
Our understanding of etiological factors for MS and of the evolution of the disease preceding clinical presentation has advanced substantially but sustained efforts are needed to achieve the goal of preventing MS. MS Canada, MS Australia, and coalition partners will build on the recommendations from the workshop to develop a comprehensive prevention strategy, including building international coalitions that expand beyond those usually engaged in the field of MS, as well as communication and evaluation frameworks. The identified areas for investment need to be prioritized and funded, via future calls for applications. These efforts will need to align with the global Pathways to Cures strategic plan and other efforts to better describe, diagnose, and manage MS. With collective, coordinated action, the goal of preventing MS can be achieved.
Footnotes
Appendix 1
Data availability statement
No data sets were generated or analyzed.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RAM receives research funding from: CIHR, MS Canada, National Multiple Sclerosis Society, CMSC, the Arthritis Society, Pfizer Foundation, Public Health Agency of Canada, MMSF, CHRIM, Brain Canada and the US Department of Defense and is a co-investigator on studies receiving funding from Biogen Idec, Roche Canada. She holds the Multiple Sclerosis Clinical Research Chair (Dalhousie University).
RD has received honoraria for speaking and/or traveling from Biogen, Eisai, Merck, Roche, and Janssen. Advisory boards for Roche, Biogen, Sandoz, Sanofi, Novartis, and Merck in the past 2 years. Grant support from Biogen, Merck, and Celgene.
SEB has received consulting honoraria for Alumis Inc. and is a co-founder of Mate Bioservices.
MS received research support and consulting fees from Allergan, Biogen, BMS, Horizon, Merck-Serono, Novartis, Roche, Sanofi, and Teva and holds a patent on EBNA2 alleles in multiple sclerosis (IT1417523 EP2981625).
BVT has received honoraria for speaking from Biogen, Novartis, and Merck in the last 2 years.
BD has nothing to disclose.
PV has nothing to disclose.
JL has nothing to disclose.
JP has nothing to disclose.
BB has nothing to disclose.
CB has nothing to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The workshop was supported by MS Canada and MS Australia.
