Abstract
Background:
While disability progression in multiple sclerosis (MS) is age-dependent, the exact timing of neuroaxonal degeneration throughout the patient lifespan remains unclear.
Objectives:
To investigate the influence of age on retinal neurodegeneration after the first unilateral episode of optic neuritis (ON).
Methods:
We measured peripapillary retinal nerve fibre layer (pRNFL) and macular ganglion cell inner plexiform layer (mGCIPL) absolute thickness and intereye differences (IEDs) in 61 MS patients with a single unilateral ON 6–24 months prior (MS-ON) and 61 MS patients without ON history (MS-NON). Participants were 1:1 matched based on age, sex, and Expanded Disability Status Scale (EDSS). Statistical significance was evaluated using mixed linear effects models.
Results:
Age and ON status of eyes significantly interacted in predicting absolute pRNFL and mGCIPL thickness (pRNFL:
Discussion:
Our data show evidence of increased relapse-associated neuroaxonal damage in older individuals. This suggests less neuronal resilience following attacks and implicates age-effects beyond insidious progression. The growing population of older people with MS (pwMS) may be at increased risk of neurodegeneration and permanent disability worsening after acute MS attacks. Longitudinal studies should confirm this.
Introduction
Multiple sclerosis (MS) is an autoimmune disease of the CNS characterised by demyelination and neuroaxonal degeneration. 1 While autoimmune-mediated demyelination associated with acute relapses is effectively targeted by disease-modifying therapies (DMTs), our understanding of the causes and timing of neurodegeneration in MS remains limited.1,2 Disability progression in MS is age-dependent. 3 While younger people with MS (pwMS) are more prone to experiencing relapse-associated worsening (RAW), aging is typically accompanied by a gradual shift towards progression independent of relapse (PIRA). 4 There is additional evidence that older patients with MS may exhibit incomplete recovery from relapses. 5 However, the direct contribution of age to neurodegeneration after an acute attack remains unclear. Whether age-associated disability in MS is solely the result of cumulative injury or whether disability depends on the timing of injury, is currently unknown.
The afferent visual system provides a valuable model in MS research by uniquely enabling pathway-specific longitudinal assessment of neurodegeneration, while also being a frequent site of inflammatory activity.6,7 Damage to retinal ganglion cells and their axons resulting from optic neuritis (ON) is linked to a decrease in macular ganglion cell inner plexiform layer (mGCIPL) and peripapillary retinal nerve fibre layer (pRNFL) thickness as measured by optical coherence tomography (OCT). 7 OCT uses coherent light to produce high-resolution, non-invasive, cross-sectional images of the retina. Besides a reduction in absolute pRNFL and mGCIPL thickness, intereye differences (IEDs) of pRNFL and mGCIPL can be used as indicators of previous ON attacks and to quantify the severity of ON-related damage.8–10
Regardless of the overall rate of neural decline, it is unclear how individual attacks contribute to neurodegeneration over the course of MS. An increasing vulnerability of neurons towards stressors or a chronic increase in neuronal stressor load might promote greater attack-related neurodegeneration in aging pwMS. 11 This study aimed to understand, whether the neuroaxonal loss resulting from a single MS attack is age-dependent. We hypothesised that higher age at ON is associated with greater subsequent retinal atrophy.
Methods
Participants
This cross-sectional analysis comprised 122 pwMS: Sixty-one pwMS with previous ON (MS-ON) and 61 pwMS without ON history (MS-NON) (Figure 1). Participants of both groups were derived from large observational cohorts at UCSF (UCSF EPIC study,
12
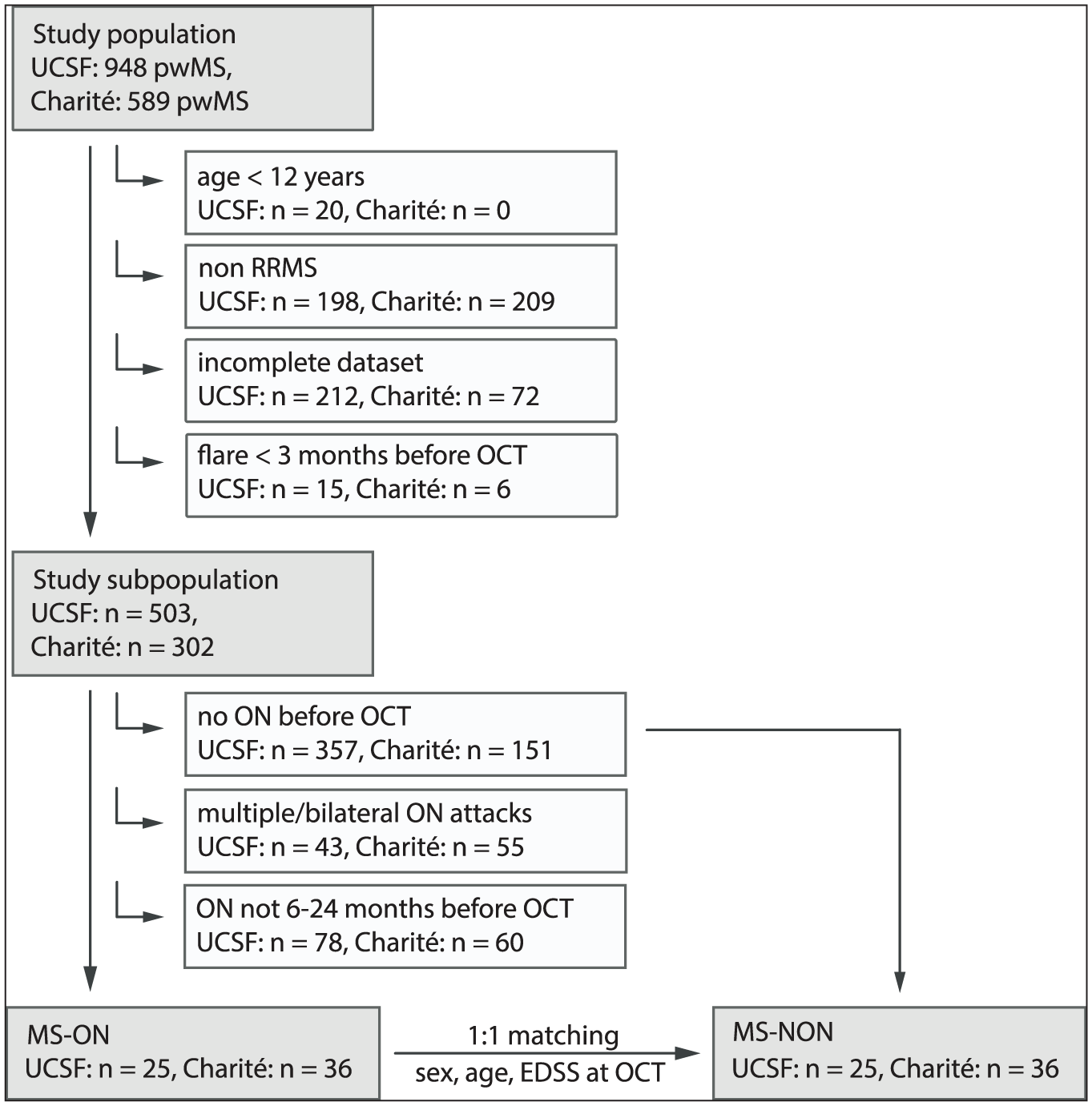
Cohort selection flow chart.
The study was approved by the local ethics committees in San Francisco and Berlin (Charité Universitätsmedizin Berlin: EA1/362/20, EA1/163/12, EA1/182/10) and performed in line with the current version of the Declaration of Helsinki. All participants gave written informed consent before study inclusion.
OCT acquisition and measurements
Experienced personnel performed OCT acquisition, quality control, and image data reading at UCSF (DC, SCM) and Charité (CB, GK). Images of macular and peripapillary retinal layers were acquired with spectral domain OCT devices (Spectralis, Heidelberg Engineering, Heidelberg, Germany). Pupil dilation was not performed. Quality control was executed in compliance with OSCAR-IB criteria, 16 and OCT data were reported according to the APOSTEL guidelines. 17 Semi-automatic layer segmentation was performed using the OCT manufacturer’s software (eye explorer, Heidelberg Engineering; versions 1.10.12.0 (UCSF) and 1.10.2.0 (Charité)). pRNFL thickness was quantified from 12° ring scans around the optic nerve head with activated eye tracker. mGCIPL thickness was calculated as average thickness from macular volume scans by placing 3.45 mm diameter cylinders around the fovea.
Visual function
Visual function was measured for each eye separately. High-contrast visual acuity (HCVA) was acquired using retro-illuminated Early Treatment in Diabetic Retinopathy (ETDRS) charts at 4 m distance. Decimal acuity (Charité) or letter acuity (UCSF) scores were converted to the logarithm of the Minimum Angle of Resolution (logMAR). Low-contrast visual acuity (LCVA) was tested with Sloan 2.5% contrast charts, with the letter acuity (range 0–60) as the outcome of interest. Correction was either carried out using optimal optical (Charité) or habitual correction (UCSF).
Statistical analysis
All parameters are reported as mean ± SD unless specified otherwise. The relationship between age and pRNFL/mGCIPL thickness was examined using linear mixed-effects models including an interaction term between ON status of eyes and age at OCT, as well as subject-specific intercepts. Contralateral, ON-unaffected eyes of MS-ON participants (CON eyes) and eyes from MS-NON participants (NON eyes) were combined to increase power. Age at OCT was centred around the mean to allow interpretation of ON effects at mean age. Estimated marginal means were derived from these models to predict pRNFL/mGCIPL mean thickness differences between ON-affected and ON-unaffected eyes at ages 20, 30, 40, and 50 years. Effect size of interaction terms was further assessed by obtaining marginal ΔR² and standardised coefficients. Marginal ΔR² was calculated as the difference in marginal R² between models with and without the interaction term. The association between age and pRNFL/mGCIPL IEDs per MS-ON participant was analysed with linear regression models. IEDs were calculated as the absolute value of the difference in pRNFL/mGCIPL thickness between both eyes. Percentage IEDs were obtained by normalising absolute IEDs to the eye with greater pRNFL/mGCIPL thickness. Furthermore, the MS-ON group was split based on median age (35 years). The difference between pRNFL/mGCIPL IEDs in these two subgroups was analysed with the Mann–Whitney U-test to allow for non-parametric testing of non-normally distributed data. Normality of linear model residuals was assessed graphically with density curves. Given the collinearity of the structural measures and the limited number of comparisons, we did not apply formal multiple-comparison corrections. The impact of centre, sex, disease duration, DMT treatment (yes/no), steroid treatment (yes/no, only for IED comparisons within the MS-ON group) and time between ON and OCT (only for IED comparisons within the MS-ON group) as additional predictor variables was investigated by comparing hierarchically nested models. Goodness of fit was assessed through likelihood ratio testing, implemented via the
Results
Patient population
Sixty-one participants with RRMS were included in both the MS-ON and MS-NON group (UCSF:
Cohort description.
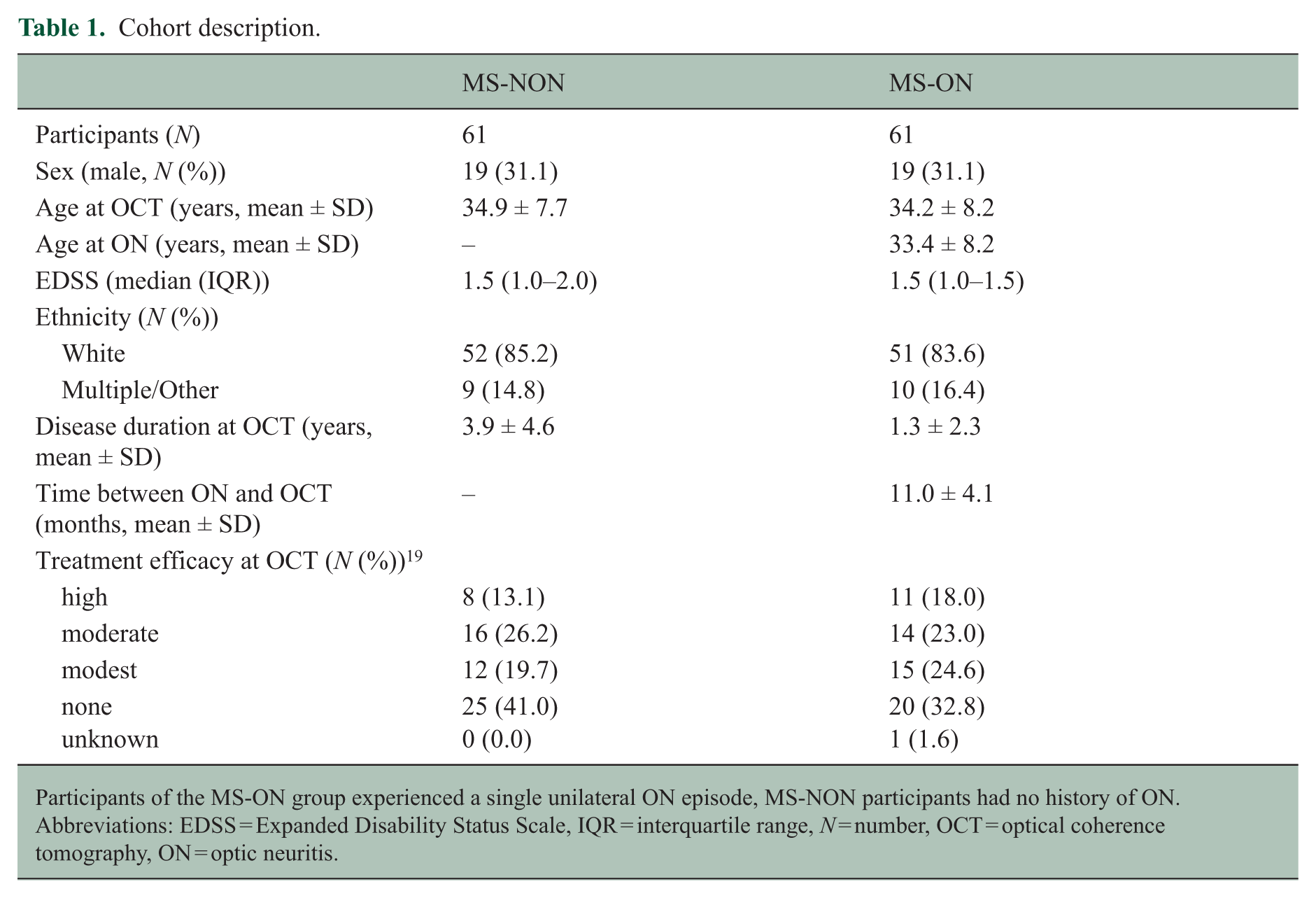
Participants of the MS-ON group experienced a single unilateral ON episode, MS-NON participants had no history of ON. Abbreviations: EDSS = Expanded Disability Status Scale, IQR = interquartile range,
ON leads to retinal atrophy
pRNFL/mGCIPL thickness was significantly lower in ON eyes compared to CON and NON eyes (ON eyes: pRNFL (median (IQR)): 92.00 (81.65–99.50) µm, mGCIPL (median (IQR)): 77.02 (69.53–83.55) µm; CON eyes: pRNFL (median (IQR)): 103.00 (94.17–108.27) µm, mGCIPL (median (IQR)): 87.72 (83.44–89.90) µm; NON eyes: pRNFL (median (IQR)): 100.83 (94.73–105.00) µm, mGCIPL (median (IQR)): 88.11 (82.81–93.76) µm;
Description of OCT-derived outcome parameters in ON-affected and -unaffected eyes/participants.
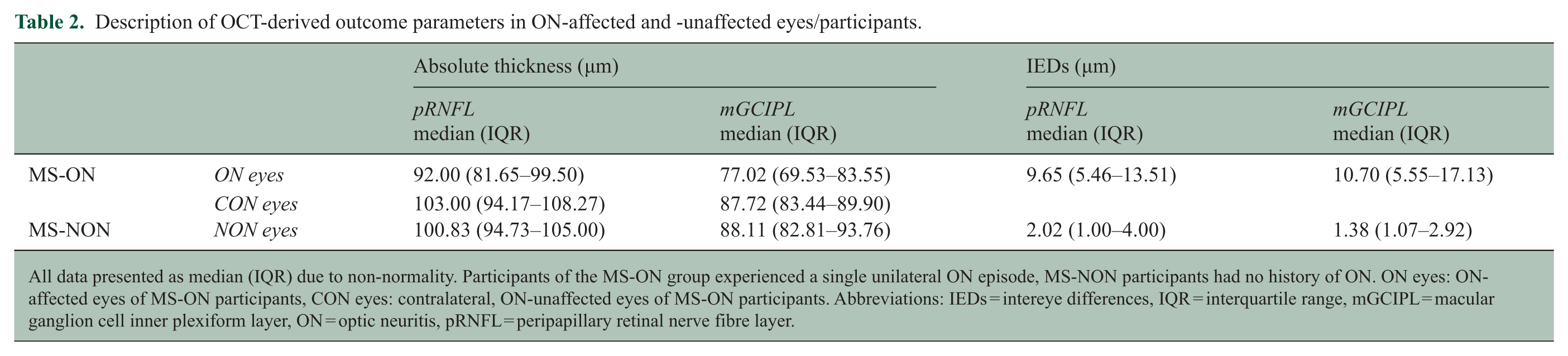
All data presented as median (IQR) due to non-normality. Participants of the MS-ON group experienced a single unilateral ON episode, MS-NON participants had no history of ON. ON eyes: ON-affected eyes of MS-ON participants, CON eyes: contralateral, ON-unaffected eyes of MS-ON participants. Abbreviations: IEDs = intereye differences, IQR = interquartile range, mGCIPL = macular ganglion cell inner plexiform layer, ON = optic neuritis, pRNFL = peripapillary retinal nerve fibre layer.
Retinal atrophy after ON is associated with age
As expected, older age was associated with lower pRNFL/mGCIPL thickness in ON-affected and -unaffected eyes (ON eyes: pRNFL:
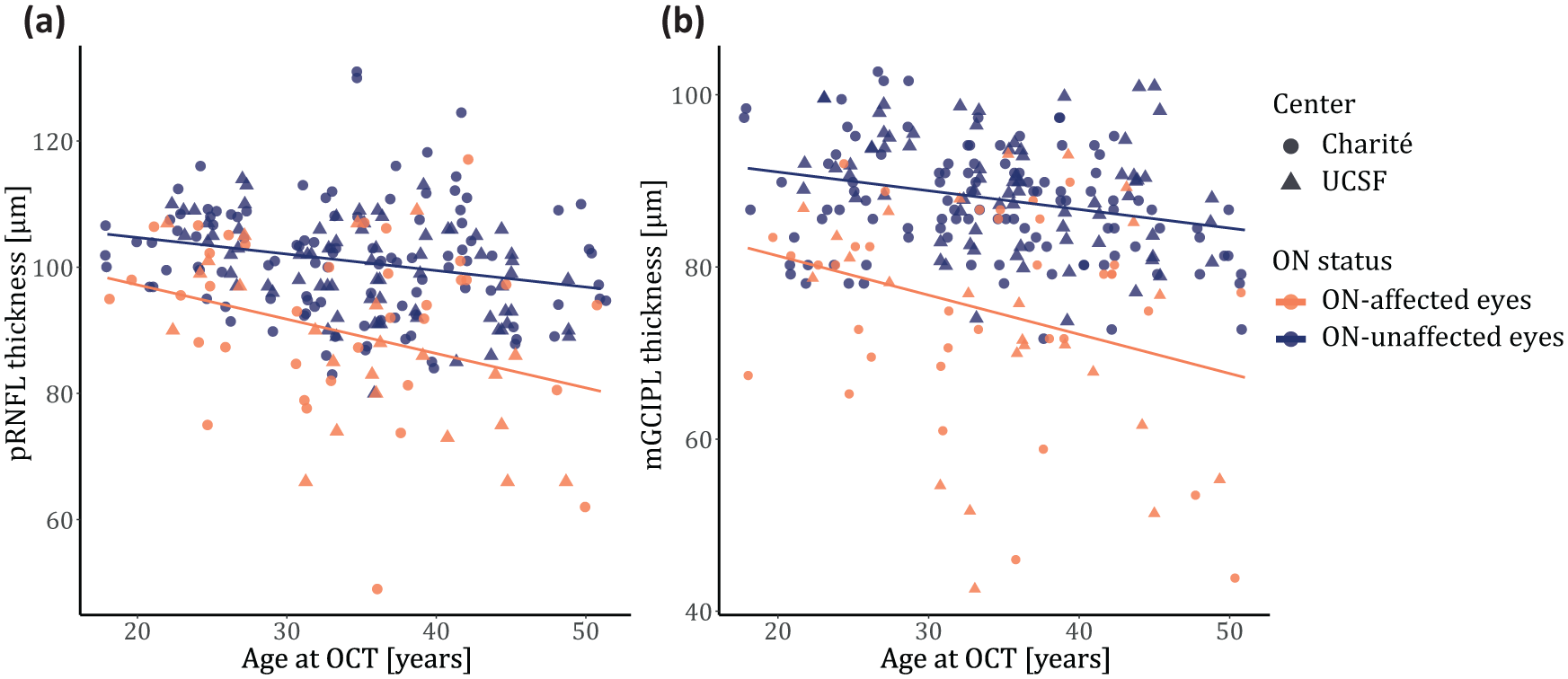
Age dependency of pRNFL/mGCIPL thickness in ON-affected (orange) and -unaffected eyes (blue).
Linear mixed-effects modelling of pRNFL/mGCIPL thickness (µm) in ON-affected and -unaffected eyes, depending on age.
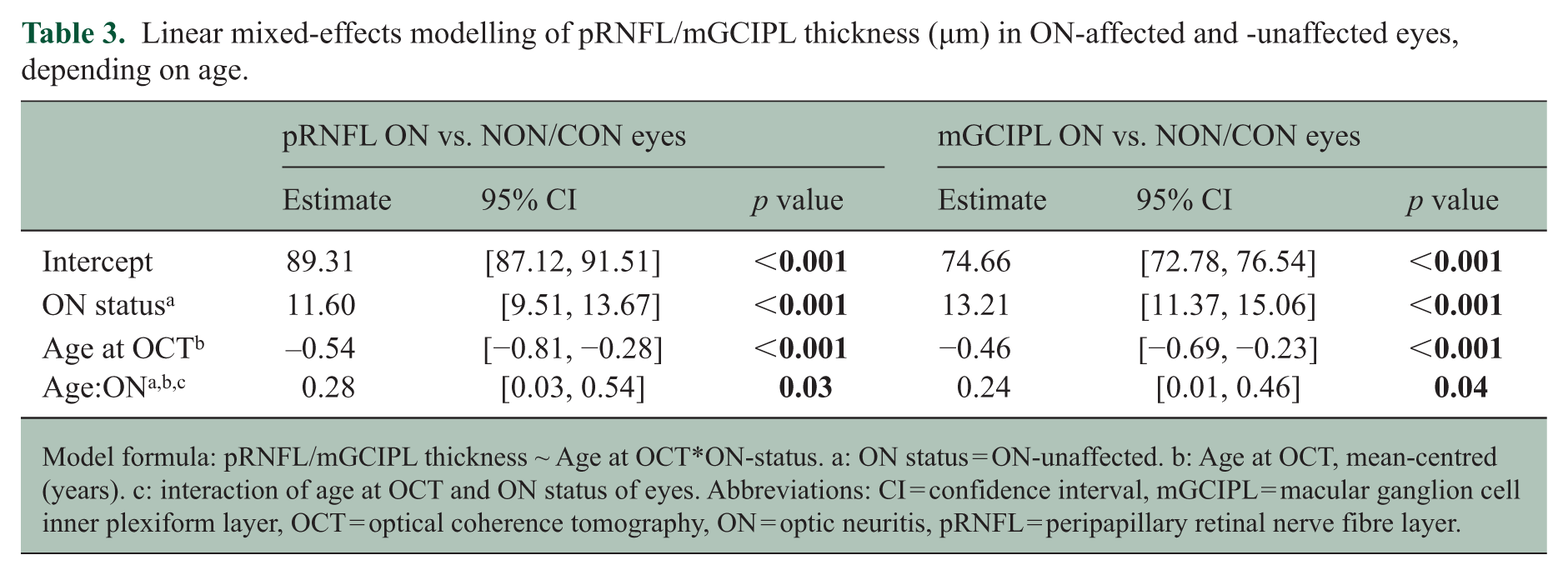
Model formula: pRNFL/mGCIPL thickness ~ Age at OCT*ON-status. a: ON status = ON-unaffected. b: Age at OCT, mean-centred (years). c: interaction of age at OCT and ON status of eyes. Abbreviations: CI = confidence interval, mGCIPL = macular ganglion cell inner plexiform layer, OCT = optical coherence tomography, ON = optic neuritis, pRNFL = peripapillary retinal nerve fibre layer.
Estimated marginal mean differences in pRNFL were predicted to increase from −7.49 µm (95% CI = [−11.72, −3.27]) at 20 years to −15.97 µm (95% CI = [−20.56, −11.39]) at 50 years of age (Table 4). For mGCIPL, the predicted mean difference between ON-affected and -unaffected eyes increased from −9.74 µm (95% CI = [−13.5, −5.99]) at 20 years to −16.89 µm (95% CI = [−20.96, −12.82]) at 50 years of age (Table 5).
Changes in estimated marginal mean pRNFL thickness differences between ON-affected and -unaffected eyes from 20 to 50 years of age.
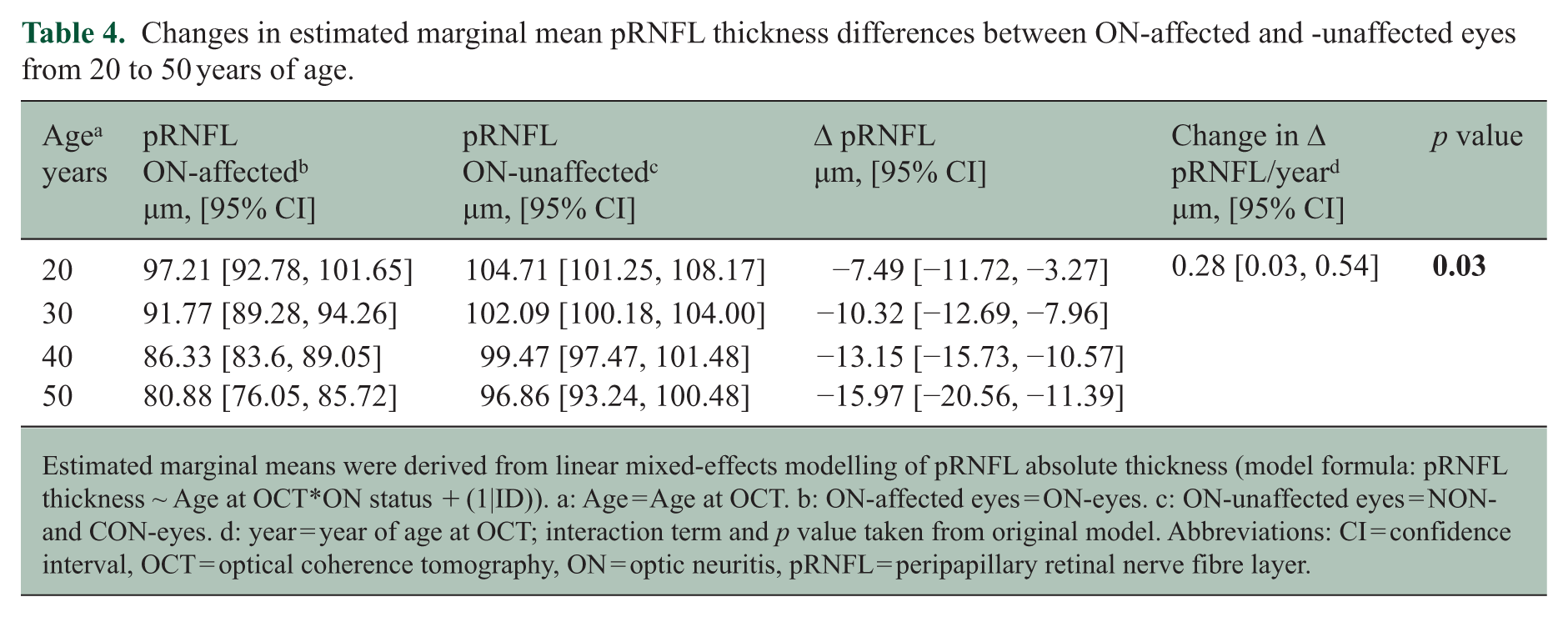
Estimated marginal means were derived from linear mixed-effects modelling of pRNFL absolute thickness (model formula: pRNFL thickness ~ Age at OCT*ON status + (1|ID)). a: Age = Age at OCT. b: ON-affected eyes = ON-eyes. c: ON-unaffected eyes = NON- and CON-eyes. d: year = year of age at OCT; interaction term and
Changes in estimated marginal mean mGCIPL thickness differences between ON-affected and -unaffected eyes from 20 to 50 years of age.
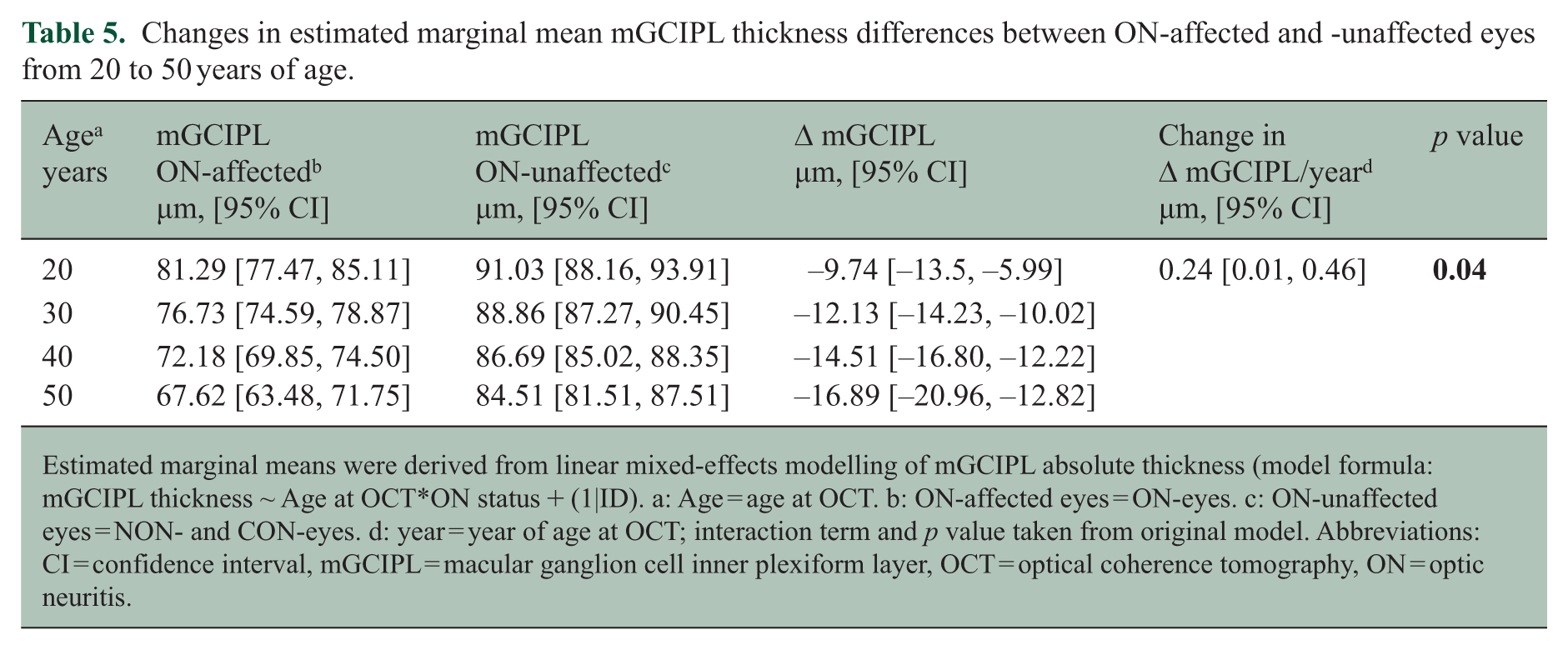
Estimated marginal means were derived from linear mixed-effects modelling of mGCIPL absolute thickness (model formula: mGCIPL thickness ~ Age at OCT*ON status + (1|ID). a: Age = age at OCT. b: ON-affected eyes = ON-eyes. c: ON-unaffected eyes = NON- and CON-eyes. d: year = year of age at OCT; interaction term and
Consistently, there was a numerical increase in pRNFL and mGCIPL IEDs of MS-ON participants with greater age, not meeting the predefined threshold of statistical significance (pRNFL:
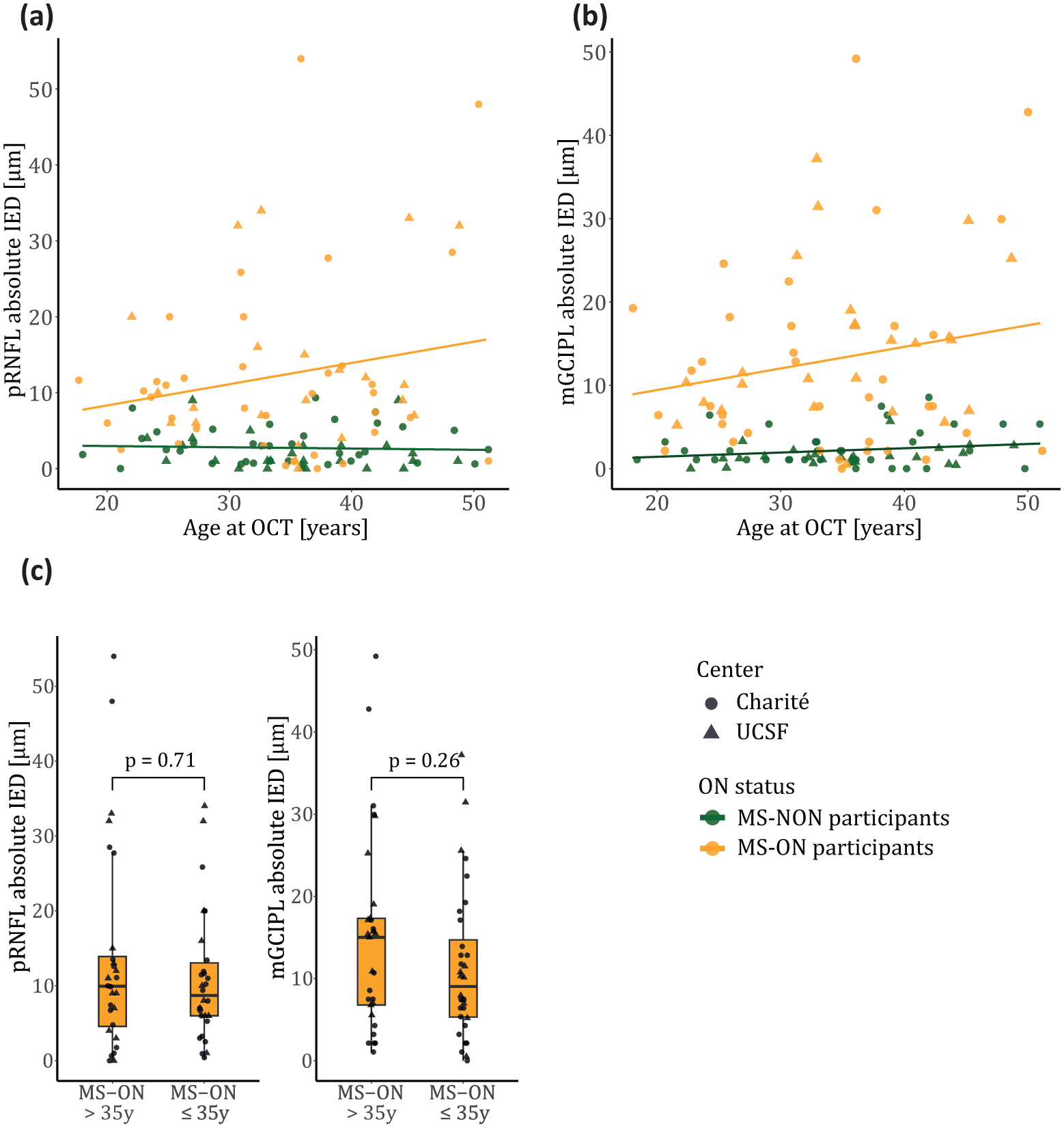
Age dependency of pRNFL/mGCIPL IEDs in MS-ON and MS-NON participants. Participants of the MS-ON group (yellow) experienced a single unilateral ON episode, MS-NON participants (green) had no history of ON.
Age (in IED models) or the interaction of age and ON status (in absolute thickness modelling) presented the best predictors of OCT-derived outcome parameters in all statistical models, when adding additional predictor variables (Supplemental Tables 1 and 2). In pRNFL/mGCIPL thickness models, age and ON status remained the only independent variables with statistical significance. Model estimates remained relatively stable (pRNFL: Age:ON interaction term = 0.28–0.30, mGCIPL: Age:ON interaction term = 0.23–0.24; pRNFL absolute IED: Age at OCT = 0.28–0.34, mGCIPL absolute IED: Age at OCT = 0.23–0.26). In addition, the studied outcome parameters were not significantly influenced by differing treatment efficacies (data not shown).
After outlier exclusion, the significant interaction of age and ON status persisted in modulating pRNFL thickness (Age:
Visual recovery after ON depends on age
HCVA data were available for 41 ON eyes (UCSF:
Description of visual acuity parameters in ON-affected and -unaffected eyes by centre.
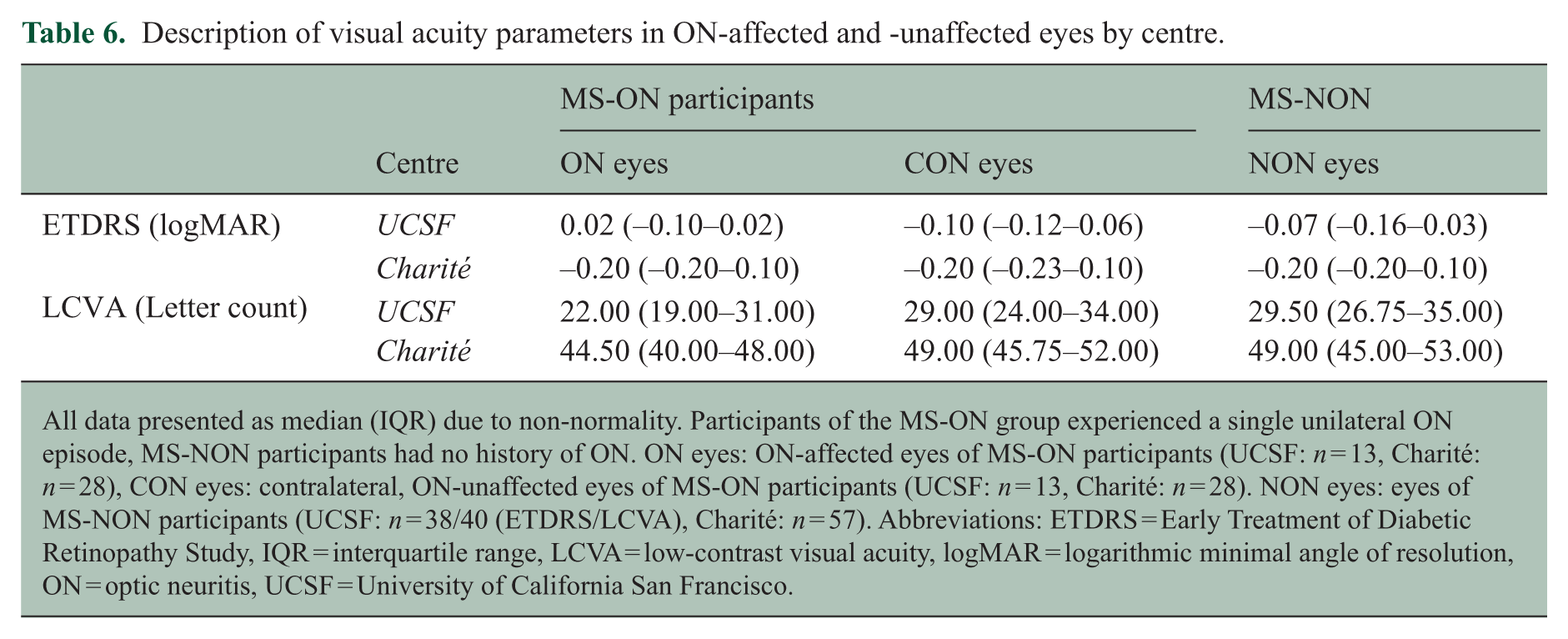
All data presented as median (IQR) due to non-normality. Participants of the MS-ON group experienced a single unilateral ON episode, MS-NON participants had no history of ON. ON eyes: ON-affected eyes of MS-ON participants (UCSF:
Linear mixed-effects modelling of HCVA (ETDRS, logMAR) in ON-affected and -unaffected eyes, depending on age.
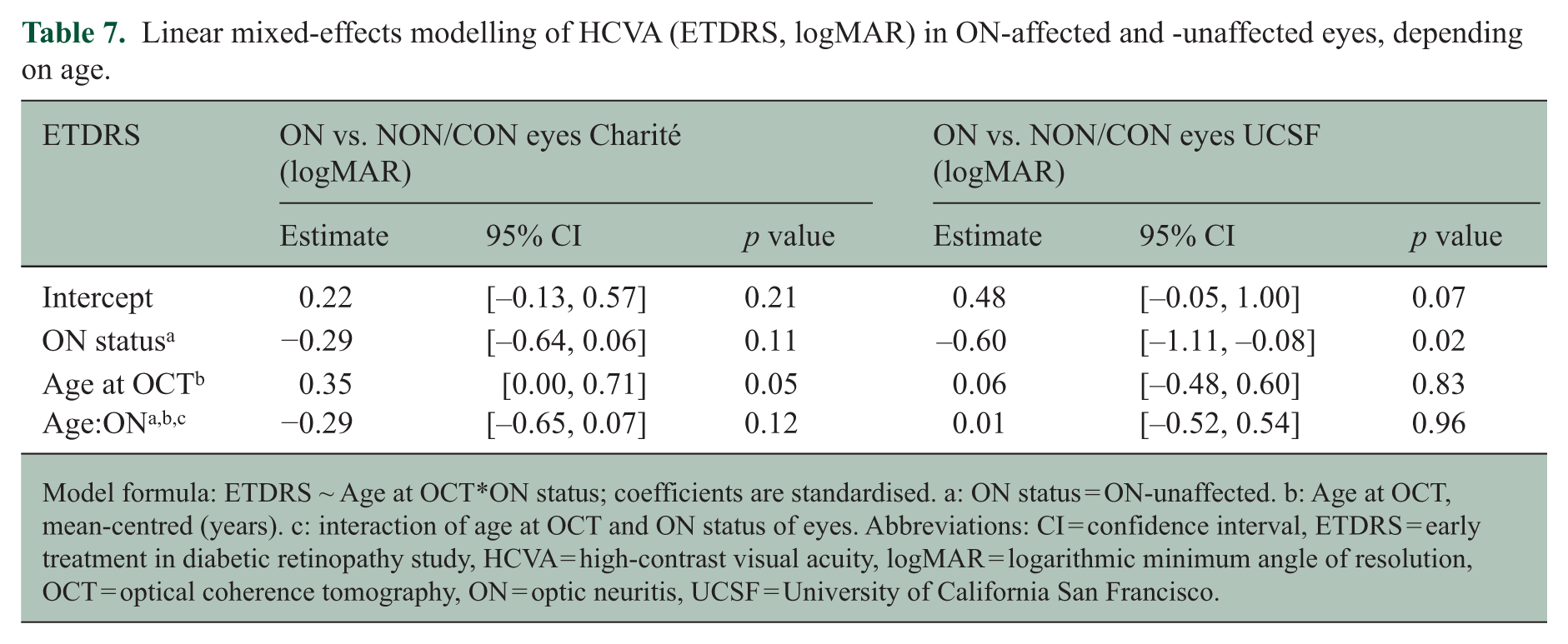
Model formula: ETDRS ~ Age at OCT*ON status; coefficients are standardised. a: ON status = ON-unaffected. b: Age at OCT, mean-centred (years). c: interaction of age at OCT and ON status of eyes. Abbreviations: CI = confidence interval, ETDRS = early treatment in diabetic retinopathy study, HCVA = high-contrast visual acuity, logMAR = logarithmic minimum angle of resolution, OCT = optical coherence tomography, ON = optic neuritis, UCSF = University of California San Francisco.
Linear mixed-effects modelling of LCVA (letter count) in ON-affected and -unaffected eyes, depending on age.
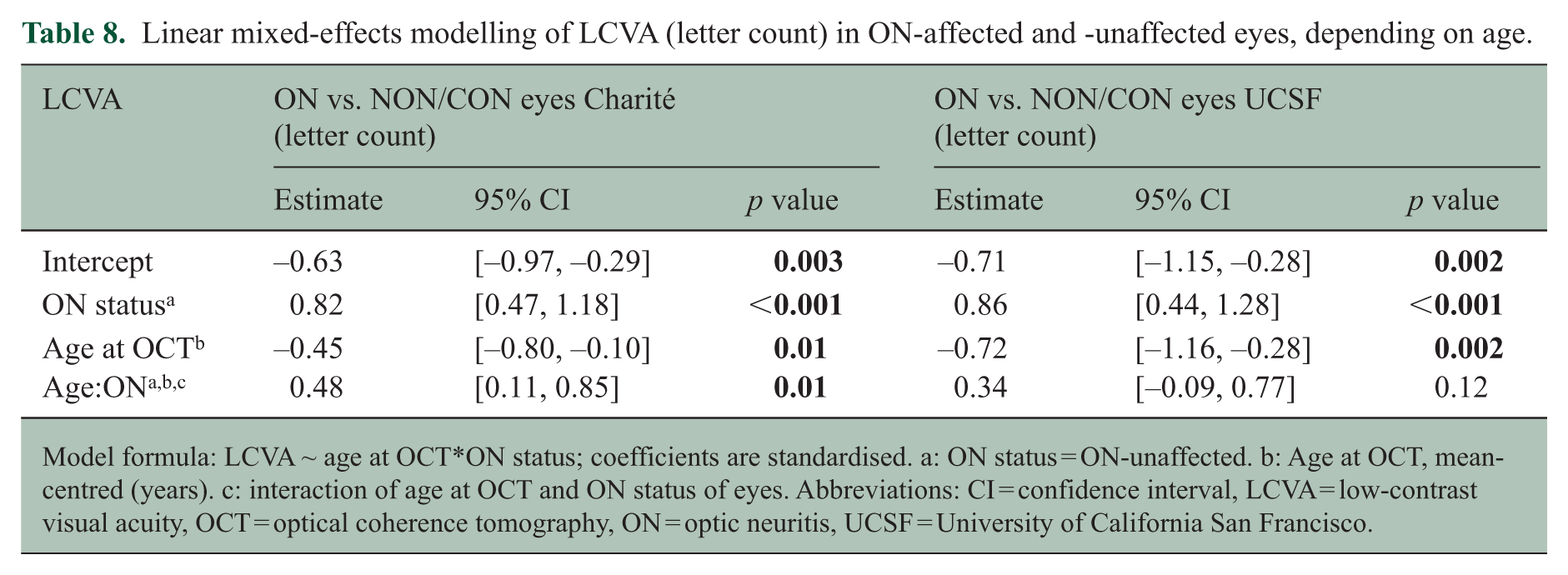
Model formula: LCVA ~ age at OCT*ON status; coefficients are standardised. a: ON status = ON-unaffected. b: Age at OCT, mean-centred (years). c: interaction of age at OCT and ON status of eyes. Abbreviations: CI = confidence interval, LCVA = low-contrast visual acuity, OCT = optical coherence tomography, ON = optic neuritis, UCSF = University of California San Francisco.
Discussion
While neurodegeneration is recognised as the cause of chronic disability in MS, drivers of age-related disability accrual are only poorly understood. 3 We aimed to analyse the relationship between age and ON-related retinal atrophy. Previous studies have suggested greater overall neurodegeneration rates in younger pwMS,20–22 but age differences in the contribution of individual attacks to the neurodegenerative load were, to our knowledge, previously unknown. A better understanding of age-related differences in vulnerability towards relapse-associated damage could help inform ongoing discussions regarding discontinuation of treatment in older pwMS. 35
Our data suggest that retinal neuroaxonal loss during acute events is age-dependent. This was reflected in the interaction of age and ON status of eyes in linear mixed-effects models of pRNFL and mGCIPL thickness, adding to the known age-dependent neuroaxonal loss in MS, which is also observed in the retina.23,24 Older age was also associated with a tendency towards increased pRNFL/mGCIPL IEDs after ON. Moreover, the interaction between age and ON status also influenced LCVA 6–24 months after ON. This suggests that older age at ON is associated with impaired functional recovery. Multiple other studies have previously demonstrated an association of age with declining functional recovery from MS relapses.5,23–26 However, the biological basis of this has not been determined. Our work suggests that early neuroaxonal loss is a likely significant contributor to this failure to recover functionally.
A previous study reported no predictive effect of age on HCVA 12 months after a first episode of ON in pwMS, while pRNFL thickness at 12 months was significantly associated with age at baseline in univariate regression analysis. 27 Due to the lack of a control group of pwMS without ON, this study could not separate the attack-specific contribution of age from the results of natural and MS-related aging. 27
Major strengths of our study include the close 1:1 matching of an MS-NON group to control for age- and attack-unrelated neurodegeneration, as well as a focus on patients after their first clinical ON attack. Since ON events often occur early in the disease, most participants were DMT-naïve at the ON event, mitigating possible effects of differing treatment efficacies. Older and younger MS-ON participants did not significantly differ with respect to EDSS and disease duration. This argues against the assumption that ON activity in older participants could be indicative of a more severe MS course.
Nevertheless, limitations have to be considered: The lack of baseline OCT data before ON events and the relatively narrow age span present limiting factors of our study. Owing to the cross-sectional design, it cannot be excluded that retinal asymmetry or thinning of retinal layers may be due to factors unrelated to age and retinal inflammation. As ON frequently presents the heralding event in MS, obtaining baseline data remains a challenge. However, the fact that IEDs remained low in MS-NON participants throughout the studied age span supports the underlying assumption that the increase in IEDs observed in MS-ON participants can be substantially attributed to the prior ON episode. ON-unaffected eyes of MS-ON participants can thus serve as baseline surrogates for the affected eyes. Considering that only 26% of participants were older than 40 years, the reported effects might be stronger when studied in cohorts with larger age ranges, although recruitment of older participants is limited by lower attack frequencies28,29 and the requirement of no prior ON attack. The average difference in pRNFL/mGCIPL thickness of 0.24–0.28 µm per year of age at ON estimated by our models however amounts to an average difference of ~2–3 µm per decade of age. This difference could already be considered clinically meaningful: previously published cross-sectional data estimated an average reduction in pRNFL thickness of 2.2–2.9 µm to be associated with a 1-line change in HCVA testing.30,31 In addition, our study suggests impairment of LCVA recovery in older patients with ON, further underlining the clinical implications of our structural findings.
The outlier-dependency of the mGCIPL data and the marginal statistical significance in IED analyses could be explained by the relatively small sample size. Generally, our variables of interest only explained small parts of the overall variance of our outcome data. Differences in attack severity might explain some of this additional data variability. Data on visual acuity at nadir was unfortunately not available to be included as a measure of attack severity. Given that older pwMS typically show decreased adaptive immune activity, it is plausible that relapse-related inflammation is less severe in this group. 32 Differences in the time between ON and OCT could also have added to the data variability. Furthermore, overall disability of both MS-ON and MS-NON participants was relatively low. While this might be due to the short disease duration associated with first-time ON attacks, we cannot exclude that our study population might not be reflective of all disease courses.
Larger, prospectively gathered datasets from multiple centres and in more diverse populations are needed to replicate and expand upon these findings. The inclusion of additional measures of neurodegeneration is crucial, since age-related neurodegeneration has been suggested to be region- and assessment-dependent. 20 Attack definitions may however be more challenging to standardise when studying other areas of the CNS, for example, with magnetic resonance imaging (MRI).
Preclinical studies should further elucidate possible mechanisms behind age-related changes in neuroaxonal loss, such as impaired remyelination and changes in neuronal vulnerability. The degree of demyelination of the optic nerve has been shown to correspond with retinal neurodegeneration and remyelinating treatment has been associated with increased neuroaxonal stability.33,34 Since impaired remyelination is a known pathophysiological feature associated with aging in MS, age-related remyelination failure and increased neuroaxonal loss might be linked. 32 In addition, neuronal senescence might reduce the intrinsic capacity for self-protection. Unravelling the contribution of these complex mechanisms to age-associated neuronal loss could provide essential insights for future therapeutic strategies to prevent neurodegeneration and disability progression in MS.
Despite the relatively young age of our participants, our data suggest an association between age at ON and subsequent retinal neurodegeneration. In the clinical setting, an enhanced understanding of age-effects in MS attacks could aid both as a prognostic factor and help guide therapeutic decision-making. Although less frequent, individual MS attacks might have more detrimental effects in older patients. This conclusion is also relevant in the context of emerging discussions regarding discontinuation of treatment in aging pwMS. 35
Supplemental Material
sj-docx-1-msj-10.1177_13524585251374507 – Supplemental material for Investigating the impact of age on neurodegeneration after optic neuritis in multiple sclerosis using optical coherence tomography
Supplemental material, sj-docx-1-msj-10.1177_13524585251374507 for Investigating the impact of age on neurodegeneration after optic neuritis in multiple sclerosis using optical coherence tomography by Henriette KM Reinsberg, Frederike C Oertel, Daniel Casillas, Gelsomina Kaufhold, Shivany Condor Montes, Charlotte Bereuter, Makenna C Chapman, Pia S Sperber, Bruce A Cree, Judith Bellmann-Strobl, Stephen L Hauser, Tanja Schmitz-Hübsch, Christian Cordano, Hanna G Zimmermann, Ahmed Abdelhak, Friedemann Paul and Ari J Green in Multiple Sclerosis Journal
Footnotes
Acknowledgements
University of California, San Francisco MS-EPIC Team: Refujia Gomez, Meagan Harms, Tiffany Cooper, Anna Sindalovsky, Stacy Callier, Harkee Halait, Adam Santaniello, Adam Renschen, Simone Sacco MD, Nico Papinutto PhD, Ariele Greenfield MD, Joanne Guo MD, Sasha Gupta MD, Joseph Sabatino MD PhD, Richard Cuneo MD, Jill Hollenbach PhD, Jeffrey Gelfand MD MAS, Riley Bove MD MSc, Samuel Pleasure MD PhD, Michael Wilson MD, Roland G Henry PhD, Sergio Baranzini PhD, Jorge R Oksenberg PhD, Stephen L. Hauser MD.
HKMR thanks the DAAD PROMOS program and the German Academic Scholarship Foundation for fellowship support
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: F.C. Oertel serves as a board member of IMSVISUAL, received research grants by the Hertie foundation, Novartis, Deutsche Forschungsgemeinschaft and the Deutsche Gesellschaft für Neurologie, received speaker honoraria by UCB and fellowship support by NMSS and AAN.; B.A.C. Cree has received personal compensation for consulting from Alexion, Autobahn, Avotres, Biogen, Boston Pharma, EMD Serono, Hexal/Sandoz, Immunic AG, Kyverna, Neuron23, Novartis, Sanofi, and TG Therapeutics and received research support from Genentech and Kyverna.; J.Bellmann-Strobl received institutional research support from NEMOS e.V., Alexion and Bayer AG, personal compensation from Alexion, speaking honoraria and travel grants from Bayer Healthcare, Horizon/Amgen, Novartis and Sanofi-Aventis/Genzyme, in addition received compensation for serving on a scientific advisory board of Alexion, Roche and Merck, all unrelated to the presented work.; S.L. Hauser currently serves on the scientific advisory board of Accure, Alector, Annexon, Hinge Therapeutics. He has previously consulted for BD, Moderna, NGM Bio, Nurix Therapeutics, Pheno Therapeutics and previously served on the Board of Directors of Neurona. Dr. Hauser also has received travel reimbursement and writing support from F. Hoffmann-La Roche and Novartis AG for anti-CD20-therapy-related meetings and presentations. He has received grants by NIH/NINDS (R35NS111644), NMSS (SI-2001-35701) and Valhalla Foundation.; T. Schmitz-Hübsch received speaking honoraria from AbbVie, Bayer, Roche and research grants from bms/Celgene, Roche. C. Cordano reports funding from the Italian Multiple Sclerosis Foundation (FISM) Rita-Levi Montalcini Grant code 2019/BC/002.; H.G. Zimmermann reports grants from Novartis, unrelated to this study.; A.Abdelhak received research grants from the Department of Defence, the German Multiple Sclerosis Society, and the Weill Institute for Neurosciences, he received an advisory fee from OctaveBio and speaking honoraria from Roche, all unrelated to this study.; F.Paul received research support, grants and personal fees by Deutsche Forschungsgemeinschaft, Bundesministerium für Bildung und Forschung, Einstein Foundation, Guthy Jackson Charitable Foundation, EU FP7 Framework Programme, Biogen, Sanofi Genzyme, Merck Serono, Novartis, Bayer, Roche, Alexion, Horizon, Neuraxpharm, Almirall, GlaxoSmithKline, Hexal, Viela Bio, UCB, Mitsubishi Tanabe and Celgene. He is an advisory board member of Celgene, Roche, UCB and Merck, academic editor at PLos One and associate editor of Neurology® Neuroimmunology & Neuroinflammation.; A. J. Green reports research support, grants and personal fees from NINDS (R01NS105741, R01AG062562, R01AG038791), NMSS (RG-1707-28564), SBIR, All May See, Rachleff Family, Westridge Foundation, JAMA Neurology, Anonymous philanthropy, Roche, Pipeline Pharmaceuticals, the National MS Society, UCSF Innovation Programme for Remyelination and Repair: IP/Patents Bazodoxefine and other miscellaneous outside the submitted work. In addition, Dr. Green has a patent Small Molecule drug for Remyelination pending and has worked on testing off label compounds for remyelination. The remaining authors have no conflicts to disclose.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Considerations
The study was approved by the local ethics committees in San Francisco and Berlin (Charité Universitätsmedizin Berlin: EA1/362/20, EA1/163/12, EA1/182/10) and performed in line with the current version of the Declaration of Helsinki.
Consent to Participate
All participants gave written informed consent before study inclusion.
Consent for Publication
Not applicable.
Data Availability
Anonymised data used for this study are available from the corresponding author upon reasonable request.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
