Abstract
Description of a patient with multiple sclerosis (MS) who underwent immunotherapy with ocrelizumab and suffered a severe course of tick-borne encephalitis (TBE): A 33-year-old man presented with acute cerebellitis with tonsillar herniation. The initial suspected diagnosis of TBE was confirmed after a significant diagnostic delay, likely caused by negative serological testing due to B-cell depletion from ocrelizumab treatment for underlying MS. TBE diagnosis was made using polymerase chain reaction (PCR) and oligo-hybrid capture metagenomic next-generation sequencing (mNGS) of cerebral spinal fluid and brain biopsy samples which yielded a near-full length TBE Virus (TBEV) genome.
Case report
In 12/2020, a 29-year-old man was diagnosed with relapsing multiple sclerosis (MS). After a second relapse, therapy with ocrelizumab was initiated (07/2022; last administration in 02/2024), which has been tolerated without complications and led to a stable disease course.
In 06/2024, the patient presented to a peripheral hospital with fever, abdominal pain, nausea, and vomiting that had persisted for a week. No internal source of infection was identified, treatment with ceftriaxone was initiated. The patient’s condition worsened, and he was referred to our emergency department. Initial cerebral computer tomography (CT) scan was unremarkable (Figure 1(a)–(c)), while cerebral spinal fluid (CSF) contained 80 cells/μL, elevated protein levels (106 mg/dL). Infectious encephalitis was suspected, and ampicillin/acyclovir was added to ceftriaxone. However, the patient deteriorated as he did not follow instructions, had untargeted pain aversion in all extremities, a skew deviation, gaze deviation and spontaneous nystagmus to the right and up-beat nystagmus. Follow-up CT scan demonstrated cerebellar swelling, incipient gyrations, and narrowing of the fourth ventricle and basal cisterns. On the intensive care unit, protective intubation was performed and analgosedation was initiated. Due to cerebellitis with increasing swelling and deep cerebellar tonsils (Figure 1(d)–(f)), a suboccipital decompressive craniectomy was performed and an extra-ventricular drainage was placed (Figure 1(g)). Antibiotics were switched to meropenem and voriconazole was added. For initial microbiologic workup, blood cultures and CSF cultures were done and Multiplex-PCR (BIOFIRE® FILMARRAY® Meningitis/Encephalitis Panel, Biomérieux) targeting the 14 most common bacterial and viral pathogens (excluding TBEV), that cause central nervous system infections (and cryptococcus neoformans) and HSV, VZV single PCRs were undertaken from CSF. Serologic tests for TBEV were performed from serum and CSF (IgG, IgM using ELISA). All these tests were found negative. Two days after the first surgery acute lower incarceration with dilated, light-stiff and rounded pupils occurred. After immediate conservative intracranial pressure therapy, a partial right cerebellar resection and suboccipital re-craniectomy were performed (Figure 1(h) and (i)). Ganciclovir, sulfadiazine/pyrithiamine, and doxycycline were added to target a broader spectrum of pathogens. A broad spectrum of bacterial, fungal parasitic and viral pathogens was ruled out by diagnostic assays. Autoantibodies associated with paraneoplastic or autoimmune encephalitis were found negative. Follow-up imaging showed acute ischemia in the right posterior area with demarcation in the right thalamus, mesencephalon, pons and partially in the cerebellum as well as new T2-FLAIR hyperintensities in the right thalamus, temporobasal/mesial area and around the temporal horns (Figure 1(j)–(l)). In the case of a possible concomitant vasculitis process, methylprednisolone was given. Intravenous immunoglobulins were administered to compensate for hypogammaglobulinemia. A lymphocytic inflammatory reaction and pronounced histiocytic and monocytic activation with destruction of the Purkinje cells were detectable in the neuropathological examination (Figure 1(m)–(s)). Finally, CSF and brain biopsy samples were investigated by an experimental oligo-hybrid capture based metagenomic NGS protocol using the Twist Comprehensive Viral Research Panel (Twist Biosciences,
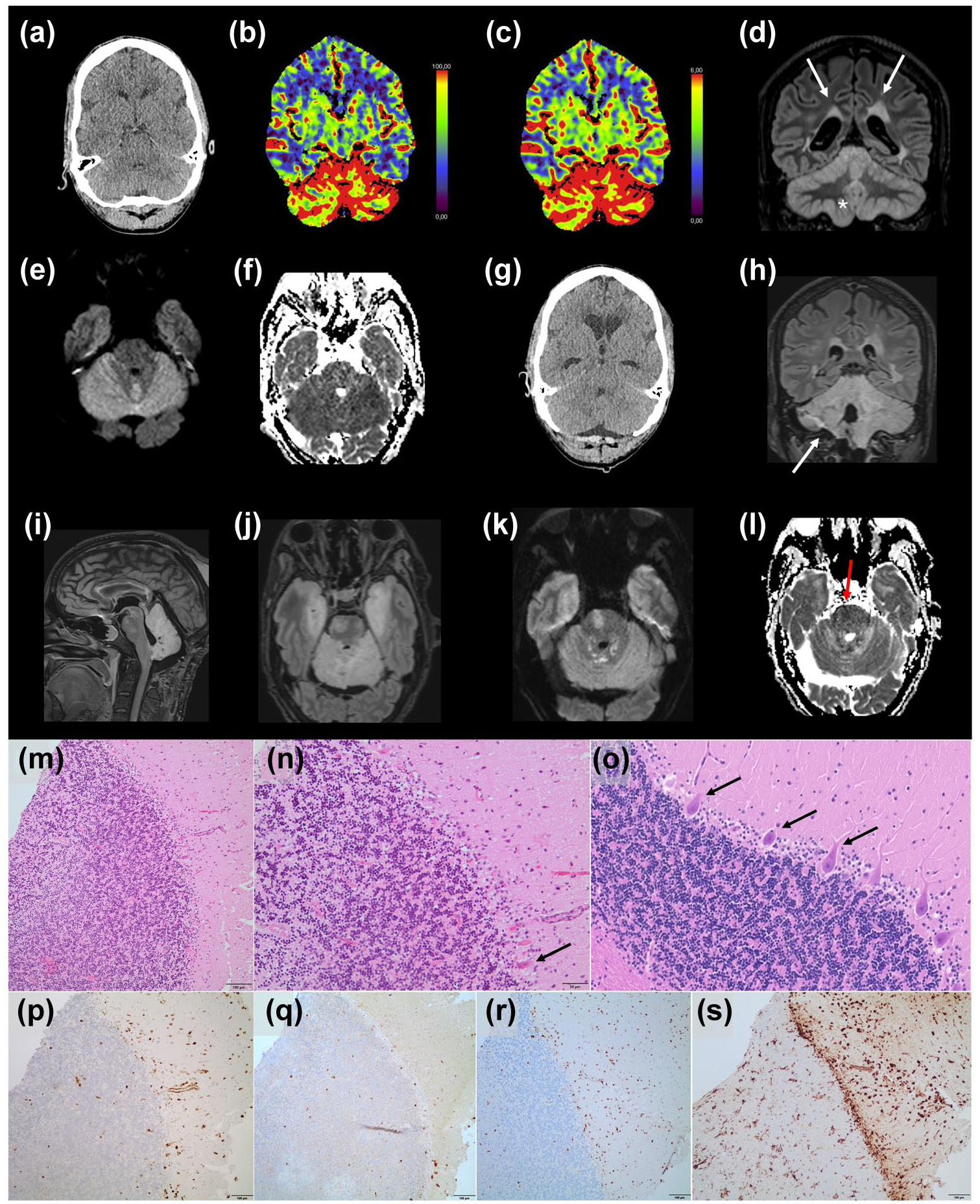
Neuroimaging and histology of cerebellar biopsy in a case of cerebellitis caused by tick-borne encephalitis flavivirus. Initial head CT was unremarkable (a). Follow-up CT showing cerebellar hyperperfusion with elevated CBF (b) and CBV (c). Subsequent MRI demonstrating hyperintense cerebellar edema predominantly affecting the cortex with herniation of the right cerebellar tonsil (d: T2-FLAIR coronal, asterisk). DWI shows corresponding hyperintensities (e: DWI-b1000) and slightly hypointense signal in the ADC map (f), in the sense of probable cytotoxic components. Furthermore, pre-existing confluent MS lesions were seen particularly in the periventricular zone (arrows in d). Follow-up scans after suboccipital trepanation (g: CT; h-i: T2-FLAIR coronal and sagittal) and resection of the right cerebellar tonsil (h, arrow) due to severe brain swelling. Transversal T2-FLAIR (j) showed new edema of both mesial temporal lobes which were rated vasogenic due to T2-shine-through phenomenon (k: DWI-b1000, hyperintense; l: ADC map, hyperintense). Furthermore, new cytotoxic edema (l; ADC, hypointense; arrow) was noted in the paramedian pons on the right and within the superior cerebellar vermis, consistent with acute ischemic small-vessel (probably para-infectious) stroke. Cerebellar cortex with mild infiltration by lymphocytes and macrophages as well severe reduction of Purkinje cells (arrow) and proliferation of Bergmann-Glia (H&E) (m and n (zoom from m)). For comparison a normal cerebellar Cortex (o). Pan-leukocyte marker (CD45) shows infiltrates diffuse in the cerebellar cortex and perivascular (p). Only a few CD45-positive cells were identified as T-cells (CD3), no B-cells (q). The majority of CD45-positive cells are histiocytes/macrophages (CD68) (r). Characteristic marked microglial activation along the Purkinje cell layer and to a lesser extent in the granular cell layer and the molecular layer (Cr3/43) (s).
During the disease progression, the patient developed a refractory non-convulsive status epilepticus, which could be resolved after treatment with levetiracetam, lacosamide, clobazam, and burst suppression anesthesia. The patient remained comatose; on discharge, the pupils were sluggishly reactive to light on both sides. In 08/2024, the patient died in a rehabilitation clinic.
Discussion
B-cell depletion therapy is increasingly being used to treat persons with MS (PwMS), as it is a safe and effective therapy. 2 However, the antibody response to pathogens is impaired leading to increased susceptibility to and severity of viral infections but also impacting diagnostic workup. The standard diagnostic algorithm for TBE is based on serological testing, since in the second phase of TBEV infection when CNS symptoms manifest, TBEV nucleic acids are most often undetectable in CSF. 3 B-cell-depleted patients can present with delayed, incomplete or even missing seroconversion to viral infections, in such cases diagnostic workup should involve PCR and/or mNGS. Since TBEV is not included in the most widely used multiplex-PCR CSF assays and commercial PCR systems for TBEV are not widely available, the diagnostic laboratory should be contacted to initiate these tests in such cases. As viral RNA levels decline after the initial phase and are further reduced by the onset of neurological symptoms, PCR testing frequently results in false-negative findings. In this case hybrid-capture mNGS proved to be a sensitive diagnostic tool.
In contrast to the healthy population, PwMS and immunotherapy are at increased risk for severe TBE courses. We advocate that PwMS in TBE risk areas should be recommended to use TBE vaccination. TBE vaccination is safe in PwMS, as no increase in the annual relapse rate nor a worsening in the Expanded Disability Status Scale was observed.4,5 However, as seen with other vaccines, reduced vaccine response rates are assumed in patients that are vaccinated already undergoing sphingosine-1-phosphate receptor modulator or B-cell depletion therapy.4,6 Immunization should be performed following the current consensus recommendations for PwMS. 7
According to anecdotal human TBE cases in Southern Germany and one mice study, the TBEV strain MucAr HB171/11 is assumed to have reduced neuroinvasiveness and neurovirulence compared to other TBE strains.1,8 However, in the present case a strong immunoreaction toward the Purkinje cells was observed, similar to previous reported fatal TBE cases,9,10 and TBE viral RNA could be recovered from cerebellar tissue. In mice, an increase in MucAr HB171/11 RNA load in CNS tissue was evident 10 days after infection, but significantly earlier in the comparison TBE strain. 8 We hypothesize that in immunocompetent hosts, a comprehensive immune reaction including early antibody formation, likely occurs before the CNS invasion of MucAr HB171/11 leading to a less severe disease course. In the present case, therapy with ocrelizumab and impaired antibody response may have been an aggravating factor for the fulminant course of TBE.
Supplemental Material
sj-docx-1-msj-10.1177_13524585251369425 – Supplemental material for Severe course of tick-borne encephalitis (TBE) in a patient with relapsing multiple sclerosis (MS) treated with ocrelizumab
Supplemental material, sj-docx-1-msj-10.1177_13524585251369425 for Severe course of tick-borne encephalitis (TBE) in a patient with relapsing multiple sclerosis (MS) treated with ocrelizumab by Jonathan A Gernert, Matthias Klein, Florian Schöberl, Katharina J Müller, Moritz L Schmidbauer, Robert Forbrig, Sigrun Roeber, Jochen Herms, Nicole Terpolilli, Milena Milakovic Obradovic, Alexander Graf, Christopher Dächert, Oliver T Keppler, Maximilian Münchhoff and Konstantinos Dimitriadis in Multiple Sclerosis Journal
Footnotes
Data Availability Statement
The data that support the findings of this study are available upon reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research presented here was, in part, funded by the Bay-VOC research initiative of the Free State of Bavaria, Germany (M.M., O.T.K.).
Ethical Approval
Not applicable.
Consent for Publication
The authorized guardian of the patient has given written consent for publication in an academic journal.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
