Abstract
Background:
The cascade sign was reported in 2003 as pathognomonic of the parenchymal form of neuro-Behçet’s. At the time, diencephalo-mesencephalic lesions in neuromyelitis optica (NMO) were largely ignored. Only with the 2015 revision of NMO spectrum disorders diagnostic criteria, were localizations in these areas fully acknowledged.
Case report:
Here we report a case of AQP4-IgG+ NMOSD presenting with acute transverse myelitis, bilateral optic neuritis, with early brain magnetic resonance imaging (MRI) showing confluent diencephalo-mesencephalic lesions evoking the cascade sign (Figure 1, a-b).
Conclusion:
MRI presentation evoking the cascade sign might hinder the correct differential diagnosis and delay appropriate treatment in AQP4-IgG+ NMOSD.
Introduction
“Neuromyelitis optica spectrum disorder” (NMOSD) encompasses a range of inflammatory disorders of the central nervous system (CNS), primarily affecting the optic nerves, spinal cord, and brainstem. NMOSD is caused by IgG targeting AQP-4, a water channel in astrocytes with a pivotal role in maintaining the physiological functions of the blood–brain barrier (BBB) and the ependymal system. Anti-AQP-4 antibodies cause damage of the astrocytes, with secondary demyelination. 1 The typical magnetic resonance imaging (MRI) features are characterized by periependymal white matter lesions surrounding the ventricular system, especially located in the diencephalic area, the dorsal brainstem and around the lateral ventricles. 2
The cascade or waterfall sign is a neuroradiological finding considered pathognomonic of neuro Behçet’s disease (NBD), consisting of an extensive involvement of the meso-diencephalic junction, whose shape, in the coronal T2/FLAIR sequences, resembles a waterfall. The sign was described for the first time in 2003 3 and subsequently reported in more recent studies.4,5
Case report
A 42-year-old woman presented to the emergency room complaining of paresthesia, leg weakness and sphincter disturbances starting 2 days earlier. Paresthesia initially involved the left lower limb and subsequently extended to the ipsilateral hemiabdomen and the contralateral foot over the following 24 hours. Sensory symptoms were associated with bilateral leg weakness, constipation and urinary retention. Family history was negative for neurological disorders, her past medical history did not disclose similar episodes. She denied infections or fever in the last 4 weeks.
General examination revealed no mouth or genital aphthous ulcerations nor skin lesions (such as papule, pustule or erythema nodosus-like); eye examination did not disclose anterior segment abnormalities, such as uveitis. Neurological examination showed mild weakness of the left upper limb, marked weakness of the lower limbs, especially distally on the right, a sensory level at D4-D5 on the left and D10 on the right, bilateral Babinski sign, bilateral ankle clonus and urinary retention (post-void residual of 700 cc of urine). Routine blood tests were unremarkable, markers of systemic inflammation were negative. Brain MRI showed a T2-weighted hyperintense right meso-diencephalic lesion characterized by contrast enhancement with a patchy distribution, evocative of the “cascade sign,” associated with additional smaller lesions localized in the left meso-diencephalic area, the subcortical white matter bilaterally, the centrum semiovale, and the right superior cerebellar peduncle (Figure 1, a-b). Spinal MRI showed two large T2-hyperintense foci extending from medulla to the thoracic spinal cord (Figure 1, c-d). Cerebrospinal fluid (CSF) and serological findings are reported in Table 1. The main relevant results were positive anti-AQP4-IgG, allowing the diagnosis of AQP4-IgG+ NMOSD, along with negative HLA-B51. Treatment started the day after admission with high-dose intravenous methylprednisolone (1 g/day) for 5 days, without clinical improvement. The patient subsequently worsened, with progressive and marked weakness of the left upper limb.
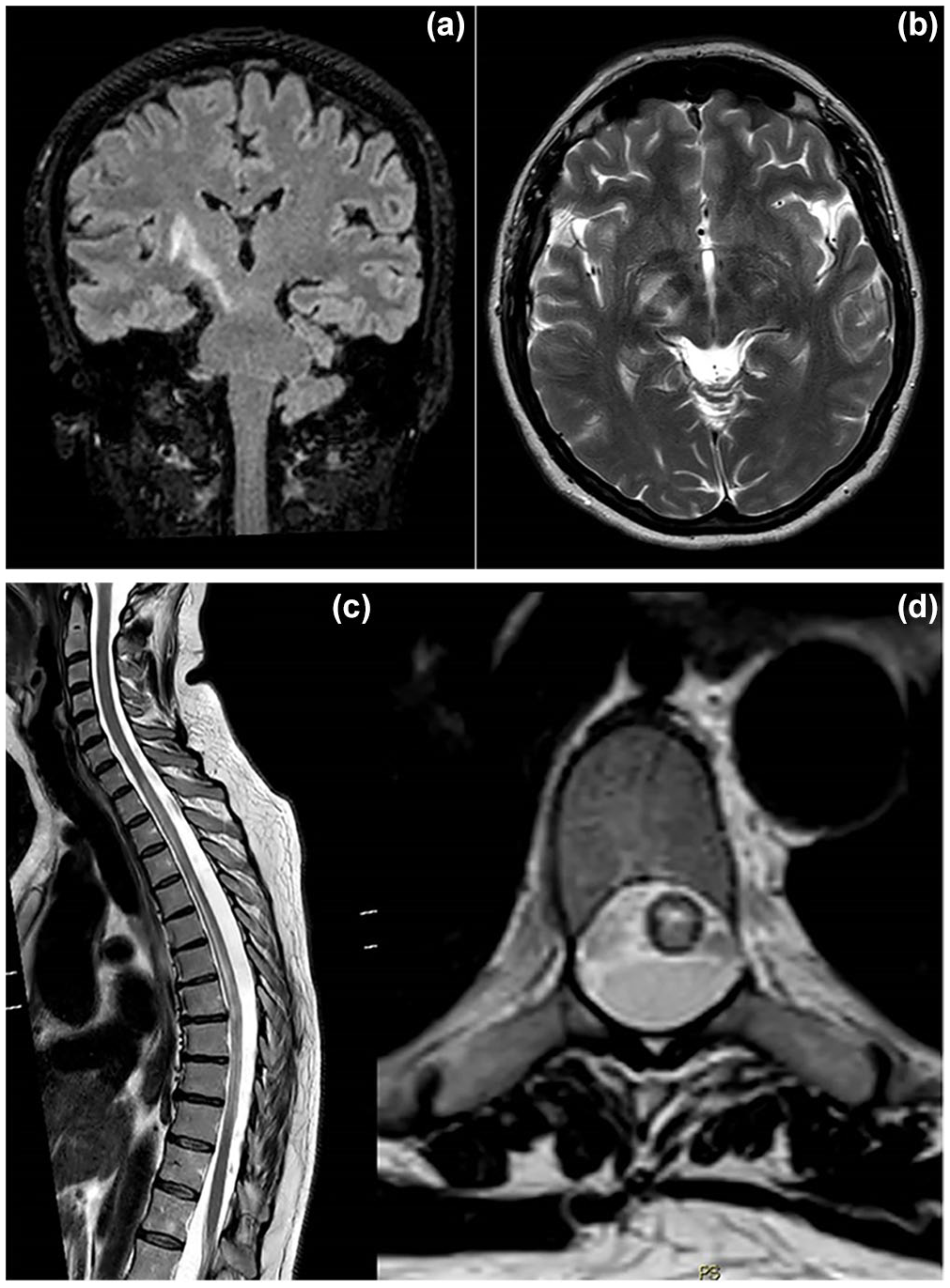
Brain MRI (a, b) shows a T2-weighted hyperintense right meso-diencephalic lesion and smaller lesions in the left meso-diencephalic area, centra semiovalia and in the right superior cerebellar peduncle. Spinal MRI (c, d) shows two large T2-hyperintense foci in the medulla abutting up to the thoracic segments.
Laboratory findings.
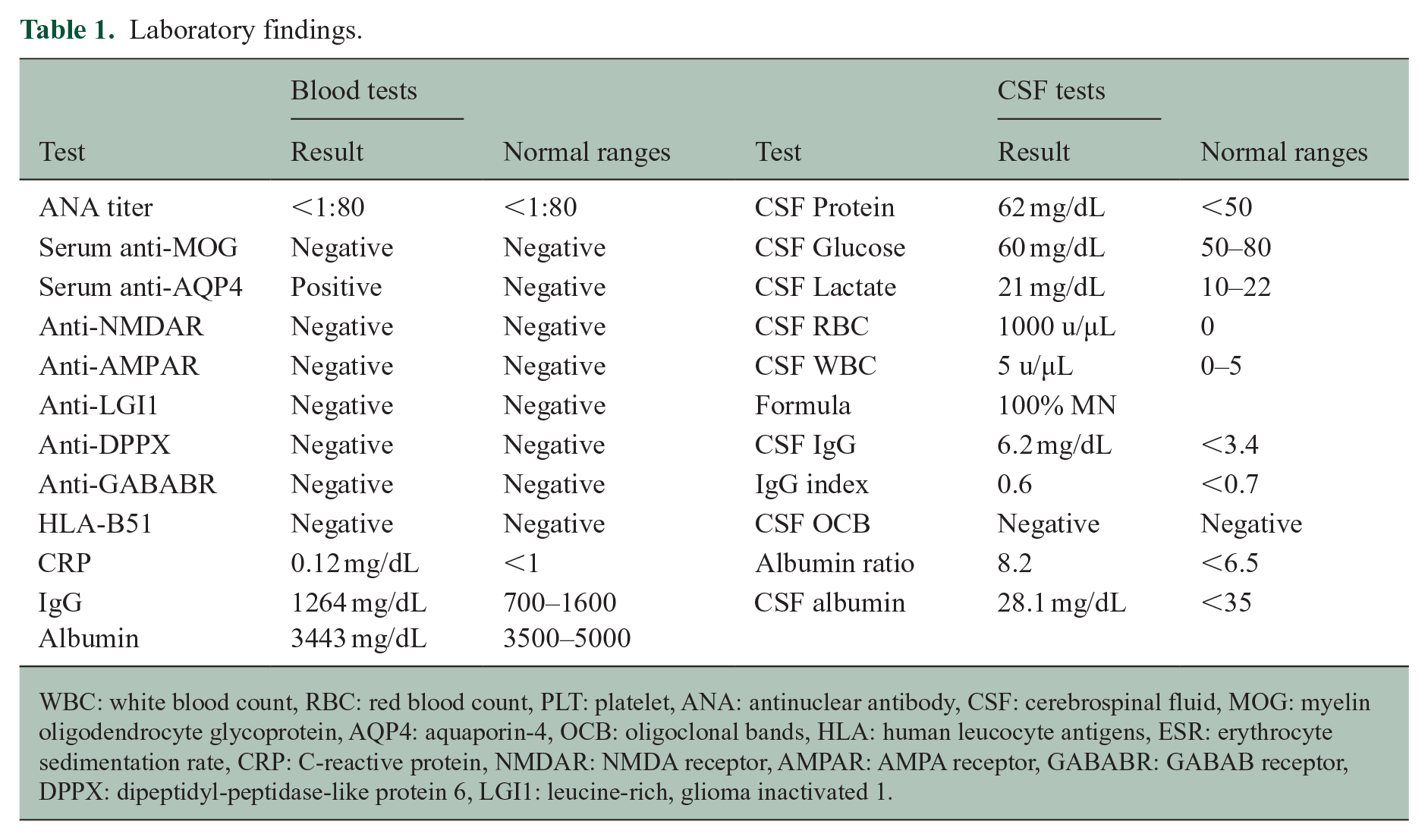
WBC: white blood count, RBC: red blood count, PLT: platelet, ANA: antinuclear antibody, CSF: cerebrospinal fluid, MOG: myelin oligodendrocyte glycoprotein, AQP4: aquaporin-4, OCB: oligoclonal bands, HLA: human leucocyte antigens, ESR: erythrocyte sedimentation rate, CRP: C-reactive protein, NMDAR: NMDA receptor, AMPAR: AMPA receptor, GABABR: GABAB receptor, DPPX: dipeptidyl-peptidase-like protein 6, LGI1: leucine-rich, glioma inactivated 1.
According to the diagnosis of NMOSD, plasmapheresis was started 1 week after admission, with clinical stabilization and no further progression of the disease.
Discussion
Thorough investigation of potential “better explanations” for hyperacute cases risking major disability is mandatory to devise the most appropriate treatment strategy.
Our case presented brain MRI features suggestive of NBD, a systemic inflammatory disease with neurological involvement in about 9% of cases. NBD can present acutely with febrile attacks and marked CSF abnormalities or as a chronic progressive form with cognitive impairment, ataxia and significant MRI abnormalities. 6 The absence of typical clinical (mouth and genital ulcers, skin lesions, uveitis) and laboratory neuro-Behçet’s findings and the extensive work-up, showing anti-AQP-4 IgG, allowed a timely diagnosis of AQP4-IgG+ NMOSD.
In this setting, the early detection at brain MRI of the “cascade sign” might have induced a quick diagnosis of NBD and treatment with an anti-TNF-alpha monoclonal antibody. This, in turn, might have caused a severe relapse of NMOSD, since treatment with anti-TNF-alpha monoclonal antibodies has been shown to be associated with an increased risk of inflammatory CNS events. 7 Awareness that the cascade sign might represent an early MRI finding in NMOSD, and prompt treatment with plasmapheresis followed by anti-CD20 treatment might help prevent early and severe disability due to the intensity, poor recovery and recurrence of the attacks, decreasing the risk of disability accrual. 8
NMOSD typically follows a relapsing-remitting course and usually presents with acute transverse myelitis or optic neuritis, symptoms common also in neuro-Behçet’s. 5 Diagnosis is based on the 2015 international consensus criteria, requiring at least one core clinical feature, detection of AQP4-IgG, and exclusion of other diagnoses. 9
The majority of patients affected by NMOSD show, paralleling relapses, an increase in MRI lesion load in areas with high AQP-4 protein expression. Typical locations of MRI brain lesions in NMOSD encompasses the diencephalon (surrounding the third ventricles and cerebral aqueduct), the midbrain (especially adjacent to the fourth ventricles) and along the corticospinal tracts (up to 44% of the cases in some cohorts), consistent with the high expression of the protein. 2 The disruption of the astrocyte cell membranes determines poorly marginated (“cloud-like”) enhancing lesions, while damage of the ependymal lining is associated with a typical periependymal enhancement. 10
The definition of “cascade” or “waterfall” sign, to our knowledge, has been used only in the parenchymal form of NBD. It is plausible that the shape of the lesion mimicking a cascade, in our case, is related to the distribution of the AQP-4 protein along the meso-diencephalic area, although typically the involvement of the meso-diencephalic area in NMOSD is more blurred (cloud-like), with ill-defined lesions extending from the posterior limb of the internal capsule to the cerebral peduncle of the pons, and along the pyramidal tract.
Conclusion
This case highlights an early MRI presentation of NMOSD, which, if overlooked, might hinder the correct differential diagnosis with other neuroinflammatory diseases and delay appropriate treatment. In the presence of the cascade sign, testing for AQP4-IgG should be considered to ensure timely management and appropriate treatment.
Footnotes
Data Availability Statement
The data that supports the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In the last 3 years (2022–2024), AL received personal compensation from Alexion, Amgen/Horizon, Biogen, Bristol Myers Squibb/Celgene, Janssen/Johnson & Johnson, Merck Serono, Novartis, Roche, Sanofi/Genzyme and Fondazione Italiana Sclerosi Multipla (FISM). Her institutions received research grants from Novartis, Roche, and Sanofi/Genzyme. FC has acted as a member of the Scientific Advisory Boards of Alexion. He has received speaker honoraria and travel support from Alexion, Merck and Novartis.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
