Abstract
Background:
This randomised controlled trial investigated the effect of a personalised digital lifestyle management application (‘levidex’) on inflammatory disease activity in newly diagnosed people with MS (pwMS), compared to a non-personalised application (‘dexilev’) that covered similar lifestyle-related content.
Methods:
Participants (N = 234) were recruited from July 2019 to April 2022 in 20 study centres in Germany and randomised to levidex (intervention group (IG), n = 115) or ‘dexilev’ (control group (CG), n = 119). Follow-up data was collected over 1-2 years. The combined primary endpoint (new T2 lesion and/or relapse) was analysed using Cox proportional hazards regression. Key secondary endpoints included self-reported quality of life and health behaviour.
Results:
There was no difference in the time to the first relapse and/or new T2 lesion between IG and CG (Hazard Ratio: 0.91; 95% confidence interval [CI]: [0.66, 1.27], p = 0.596). After 3 months, self-reported diet quality was higher in the IG (0.43; 95% CI: [0.14, 0.72], p = 0.0037). There was no difference in other secondary endpoints between IG and CG after 3 and 12 months.
Conclusions:
This study failed to meet its primary endpoint and usage of levidex did not differ from dexilev in its effects on inflammatory disease activity or behaviour change in this cohort of pwMS.
Keywords
Key messages
Compared to a non-personalised psychoeducational digital intervention, the CBT-based, personalised digital intervention levidex did not differ in its effects on inflammatory disease activity in terms of new T2 lesions and new relapses or health behaviour change within 1–2 years in this cohort of people with multiple sclerosis.
At the beginning of the study, quality of life, patient activation, physical activity, and diet quality were already moderate to high, which may have left little room for further improvements.
Further research should examine the effects of digital health interventions on people with multiple sclerosis who experience reduced quality of life, problematic health behaviours, or poor mental health.
Introduction
Multiple sclerosis (MS) is increasingly earlier diagnosed, especially based on elaborate magnetic resonance imaging (MRI) criteria recently emphasised by presentation of McDonald 2024 criteria. 1 Early disease-modifying therapy (DMT) directly after MS diagnosis is recommended. 2 However, many patients have limited disease activity early in the course and adherence to injectable or oral DMTs in the first 2 years may decline to 30%–50% among people with MS (pwMS). 3 The reasons are manifold and include uncertainty regarding benefits and side effects or access to care. Moreover, pwMS are often motivated to take an active role in managing their health and well-being. Internet sources often provide contradictory and poorly curated advice on lifestyle-related topics, 4 and existing care structures can rarely meet the complex information needs of pwMS.
Despite methodological limitations of clinical trial evidence, epidemiological research has shown that lifestyle factors have a major impact on mortality. 5 There is substantial evidence that exercise improves fitness, mobility, quality of life (QoL), and symptoms in MS, 6 and preliminary evidence has hinted at a possible impact of exercise on disease evolution. 7 Nutritional factors have been researched in MS for years, but the impact of any dietary pattern is not clear. 8 However, obesity and cardiovascular risk factors have a relevant negative impact on disease evolution. 9 Furthermore, a randomised controlled trial (RCT) has shown an impact of cognitive behavioural stress management training on inflammatory MRI activity. 10
While partly guided web-based lifestyle interventions based on behaviour change methodology have been developed, 11 no purely web-delivered lifestyle behaviour change programme is available in Germany. We chose a combined behaviour change programme called ‘levidex’ that provides information on health consequences of various health behaviours, aiming to motivate users to take an active role in managing MS from an early stage. This approach allows users to decide autonomously which aspects of their health behaviour they wish to change, supporting self-determined decision-making. We hypothesised that levidex can reduce inflammatory disease activity in early MS, as measured clinically by relapses or new T2 lesions. Therefore, the primary aim of the POWER@MS1 trial was to investigate whether levidex is more effective than the active control application ‘dexilev’ in reducing inflammatory disease activity in early MS.
Methods
The Consolidated Standards of Reporting Trials (CONSORT) 2010 checklist for the reporting of this study is provided as a supplemental file. Extensive study design details have been reported earlier. 12
Trial design
This was a rater-blinded, randomised controlled parallel group superiority trial of a 12 months digital lifestyle management application (levidex) compared to an active control digital application (dexilev). As the primary objective, we investigated whether levidex can reduce inflammatory disease activity in early MS, as measured clinically by relapses or new T2 lesions on MRI, to a greater extent than dexilev. As secondary objectives, we investigated whether levidex is superior to dexilev in its effects on QoL, physical activity, diet quality, psychological burden, patient autonomy, and empowerment. Participants were randomised to levidex or dexilev using an allocation ratio of 1:1 by block randomisation through a computer-generated system (see Randomisation).
Eligibility criteria
People aged between 18 and 65 years with a possible or confirmed relapsing-remitting MS for up to 12 months, MS-typical cerebrospinal fluid findings with detection of oligoclonal bands, and ⩾2 MS-typical lesions on T2-weighted images on cerebral MRI scans were eligible. From July 2019 to February 2021, only untreated pwMS with no planned DMT start within 3 months after inclusion were recruited. Due to a poor recruitment rate partially due to the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic, the inclusion criteria was broadened from March 2021 to March 2022, including pwMS who were already treated with a DMT.
Setting and locations
From July 2019 to March 2022, 20 study centres (academic hospitals, community clinics and private practices with an MS specialisation) in Germany were involved in eligibility screening and recruitment. All relevant data were collected in the study centres and radiological practises or online. Paper-based records were entered into the electronic data capture system secuTrial®.
Interventions
Intervention group (IG): levidex
Levidex offers 16 purely web-based modules without any trainer (see Appendix 1) and additional accompanying material (e.g. audio exercises, worksheets in pdf-format, embedded self-monitoring questionnaires), which can be accessed online via Internet-based devices (e.g. computer, smartphone, tablet) over 12 months. Levidex modules can be completed in about 30–45 minutes, depending on reading speed and participation in optional exercises. A detailed description of levidex and the development process has been published elsewhere. 13 To initiate health behaviour modifications among pwMS, levidex is based primarily on cognitive behavioural therapy (CBT) techniques and leverages a broad range of relevant behaviour change techniques (BCTs). It conveys relevant knowledge in the areas of MS treatment and health behaviour management and engages users in personalised ‘simulated dialogues’, where subsequent content is adjusted based on users’ individual selections of multiple pre-defined response options. To support adherence, an optional reminder system (emails and short text messages) has been incorporated. Usage of levidex was monitored biweekly and reacted on by a team member of the coordinating centre after 4 weeks of non-usage.
Active control group: dexilev
Participants randomised to the control group (CG) received 12 months of access to the active control programme dexilev. It is a web-based psychoeducational programme composed of 13 conversationally written modules (see Appendix 1). Each module provides guidance, practical tips, and suggestions to support active self-management, enhanced with visual aids. Module content was compiled and curated based on material available from the national MS society. Module completion in dexilev requires about 10–15 minutes. In contrast to levidex, dexilev does not personalise content, is not explicitly based on CBT techniques, and does not include additional material. Dexilev operates on the same technical platform as levidex and also includes a reminder system and usage monitoring, as applied in the IG.
Outcomes
Follow-up data were collected over 12–24 months. Outcomes and measurement time points are provided in Table 1 and the published study protocol. 12
Outcome measures and measurement time points adapted from Krause et al. 12
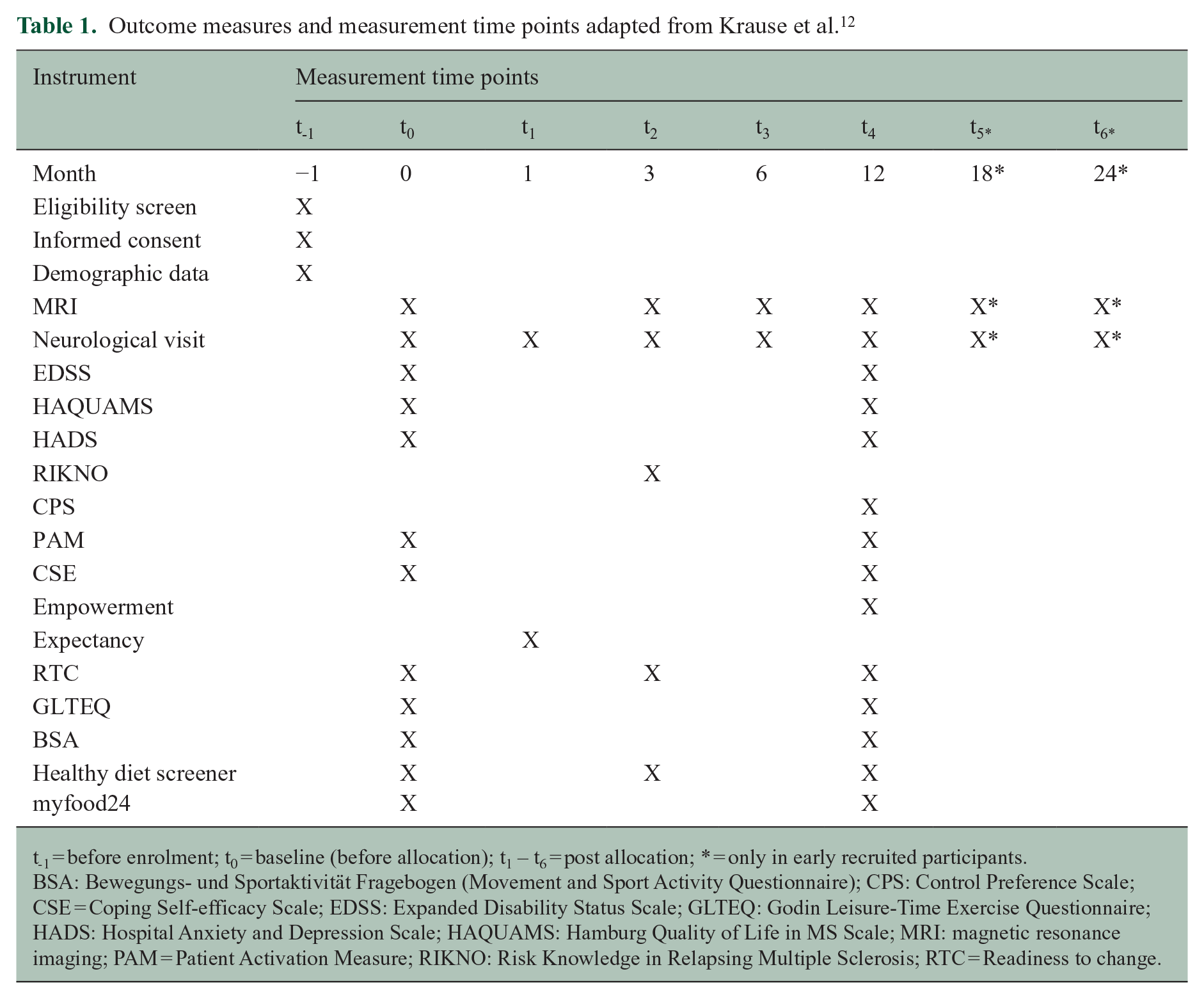
t-1 = before enrolment; t0 = baseline (before allocation); t1 – t6 = post allocation; * = only in early recruited participants.
BSA: Bewegungs- und Sportaktivität Fragebogen (Movement and Sport Activity Questionnaire); CPS: Control Preference Scale; CSE = Coping Self-efficacy Scale; EDSS: Expanded Disability Status Scale; GLTEQ: Godin Leisure-Time Exercise Questionnaire; HADS: Hospital Anxiety and Depression Scale; HAQUAMS: Hamburg Quality of Life in MS Scale; MRI: magnetic resonance imaging; PAM = Patient Activation Measure; RIKNO: Risk Knowledge in Relapsing Multiple Sclerosis; RTC = Readiness to change.
Primary outcome
The primary endpoint was inflammatory disease activity (new relapse or lesion on T2-weighted images on MRI scans). A protocol with recommended slice variation from 1 to 3 mm was applied for MRI assessments. Relapses were evaluated clinically during neurological follow-up visits. In case of events, the primary endpoint was formally reached, but participants remained in the study.
Secondary outcomes
Physical impairment was determined by neurologists in the study centres using the Expanded Disability Status Scale (EDSS). 14 All other secondary outcomes were obtained using paper- and web-based patient-reported outcome measures (PROMs). The key secondary endpoint, MS-related QoL, was measured with the Hamburg Quality of Life in MS Scale (HAQUAMS). 15 Moreover, the Hospital Anxiety and Depression Scale (HADS), 16 Risk Knowledge in Relapsing MS (RIKNO), 17 the Control Preference Scale (CPS), 18 the Patient Activation Measure (PAM), 19 the Coping Self-efficacy Scale (CSE), 20 Empowerment, 21 Expectancy, 22 the Godin Leisure-Time Exercise Questionnaire (GLTEQ), 23 the Movement and Sport Activity Questionnaire (BSA), 24 a healthy diet screener, 25 the 24-h dietary recall tool myfood24 26 and readiness to change (RTC), 27 were used.
Sample size
Initially, a sample size of 328 pwMS was calculated. 12 Based on a data export of event rates (new T2 lesion or relapse) from 135 pwMS included by 16 August 2021, a pre-defined blinded sample size recalculation was conducted. Two different definitions of a relapse (‘confirmed’ and the combination of ‘confirmed’ and ‘possible’) were reviewed, as the clinical assessment of relapse occurrence is not clear in all cases. The calculation of event rates for new lesions and confirmed relapses resulted in a required sample size of 250 participants (125 per group including 20% dropouts). Addressing confirmed and possible relapses resulted in a sample size of 216 participants (108 per group, including 20% dropouts). The conclusion was to target a sample size of 240 pwMS.
Randomisation
Concealed group assignment was undertaken externally through secuTrial®. Participants were randomised to the IG or CG in a 1:1 allocation ratio by block randomisation through a computer-generated system. Randomisation was initially stratified by centre with a block size of four patients each. Randomisation was additionally stratified by DMT status after the amendment of exclusion criteria. Access (login) details were provided by an unblinded study team member.
Blinding
This trial was investigator blinded and participating study centres were not provided with any information about group assignment. Blinding of participants was pursued, but only possible to a limited extent, as some pwMS and their neurologists realised the allocation during clinical visits.
Statistical methods
Details of statistical analyses were pre-specified in the published statistical analysis plan (see ClinicalTrials.gov, NCT03968172). The analysis was performed in R version 4.3.1. 28 The primary endpoint was evaluated using Cox proportional hazards regression that, in addition to treatment, included study centre as a factor. We did not account for age, disease duration or EDSS scores due to low variability based on the eligibility criteria. The treatment effect is reported as hazard ratio (HR) with 95% confidence interval (CI) and p value testing the null hypothesis H0: HR = 1. Kaplan–Meier curves were used to illustrate the treatment effect for both groups. A negative binomial regression was calculated for the annualised T2 lesion rate difference between IG and CG. Secondary endpoints were analysed using analysis of covariance (ANCOVA) models adjusted for baseline assessments and centre. Least square mean differences between IG and CG are reported with 95% CI and p values testing the null hypothesis of no intervention effect. Subgroup and moderator variable analyses were performed for early DMT, no DMT, programme adherence (defined as completion of at least 12 levidex modules (without booster modules) versus all 13 dexilev modules), low QoL (HAQUAMS score ⩾ 2), physical impairment (EDSS ⩾ 2) as well as moderate to severe symptoms of anxiety (HADS-A score 8–17) and depression (HADS-D score 8–16). Following the intention-to-treat principle, all pwMS were analysed in the group they were randomised to. Missing data were handled via multiple imputation using mice 29 and the baseline variable, sex and age as predictors. Early study discontinuations were treated as independent right censoring in the primary analysis.
Results
Participants and follow-up
Participants were enrolled from 9 July 2019 to 28 March 2022 and followed up until 5 April 2023. Numbers for screening, allocation, follow-up and data analysis are reported in Figure 1. Out of 234 randomised pwMS, 211 (90%) completed follow-up surveys. While the core study period was 12 months, early recruited participants were invited to remain in the study. Therefore, we included data of 28% (IG) and 29% (CG) until 24 months. In total, 234 pwMS were analysed by original assigned group (IG: n = 115, CG: n = 119; see Figure 1) with a mean follow-up of 16.93 months. One participant was excluded from primary outcome analyses due to missing MRI data after randomisation. Baseline characteristics (see Table 2) have been reported earlier and demonstrate a minor level of psychological and physical impairment as well as a high level of patient activation. 30
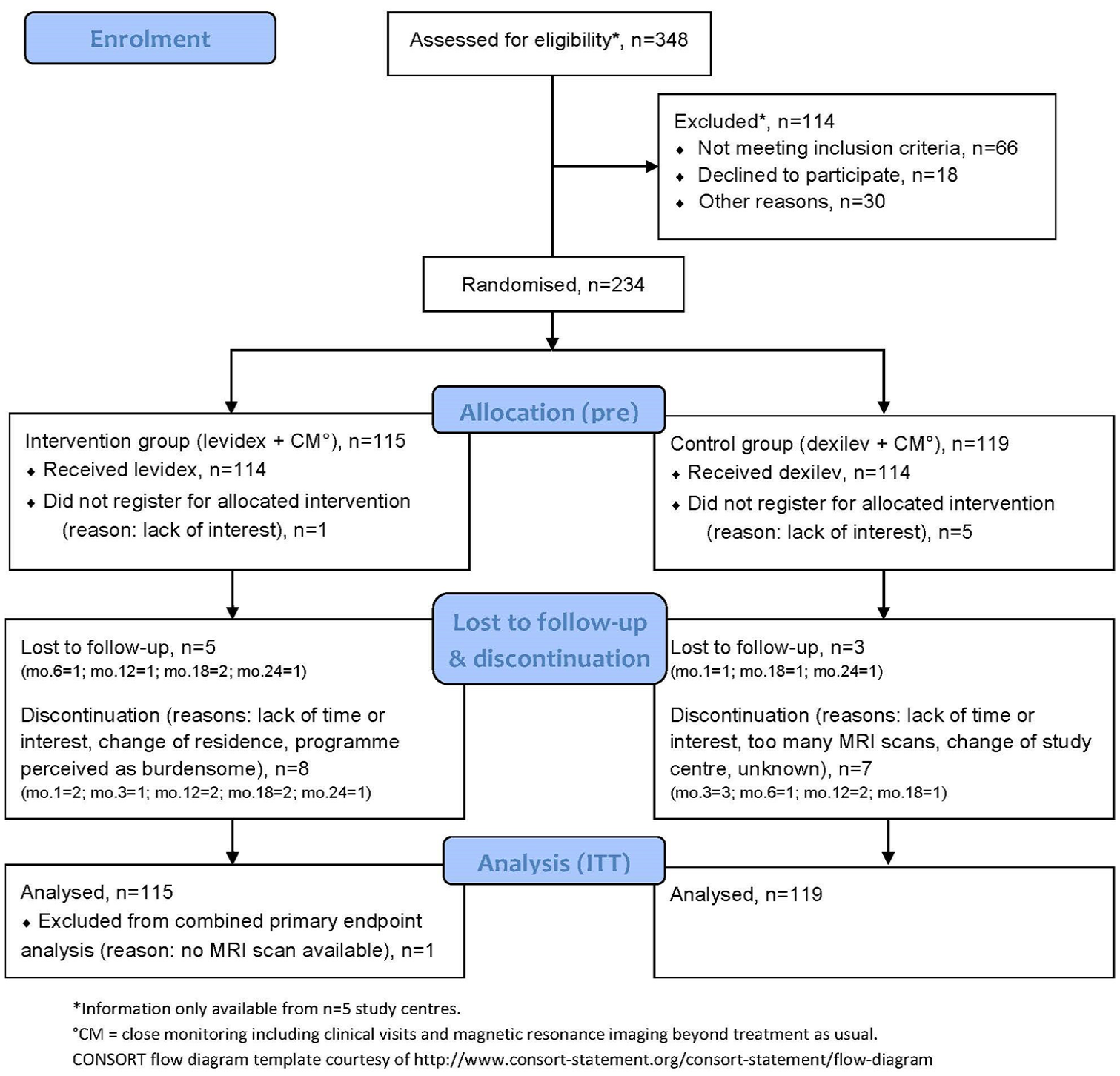
CONSORT participant flow diagram.
Baseline demographic characteristics.
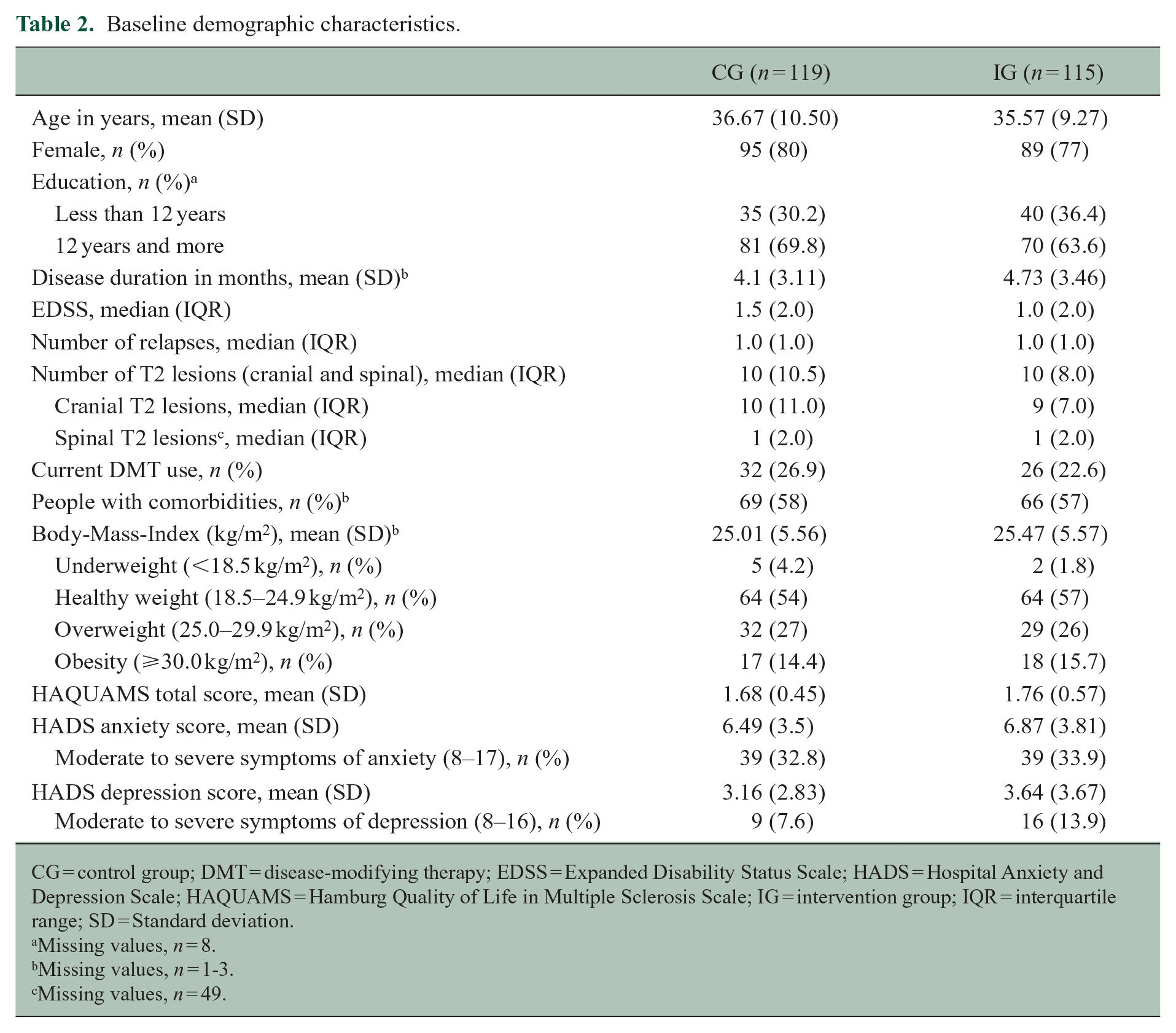
CG = control group; DMT = disease-modifying therapy; EDSS = Expanded Disability Status Scale; HADS = Hospital Anxiety and Depression Scale; HAQUAMS = Hamburg Quality of Life in Multiple Sclerosis Scale; IG = intervention group; IQR = interquartile range; SD = Standard deviation.
Missing values, n = 8.
Missing values, n = 1-3.
Missing values, n = 49.
Primary outcome
There was no difference between the IG and CG in the time to the first (possible) relapse and/or new T2 lesion (ITT analysis, HR 0.91 (95% CI: [0.66, 1.27]), p = 0.596); see Figure 2). There were 71 events (47 T2 lesions, 23 relapses, 1 both) in the IG and 70 events (41 T2 lesions, 28 relapses, 1 both) in the CG. Over the full length of follow-up, there was an average of 2.3 (SD 4.4) new T2 lesions in the IG and 2.2 (SD 4.4) new T2 lesions in the CG. There was no difference between the groups in the annualised T2 lesion rate. Incidence rate ratio was 0.8 (95% CI: [0.48, 1.33], p = 0.3725). Analysis of untreated pwMS (HR 1.08 (95% CI: [0.64, 1.82], p = 0.78) and highly adherent participants (HR 1.25 (95% CI: [0.82, 1.92], p = 0.298) showed no group differences. Detailed data on subgroup analyses of primary outcome components is provided in Appendix 2.

Kaplan–Meier curves for the combined primary endpoint (new T2 lesion or relapse) by treatment group.
Secondary outcomes
Four weeks post randomisation, compared to CG participants who had used dexilev, IG participants with access to levidex perceived the programme as more convincing (OR 2.80 (95% CI: [1.64, 4.76]), p < 0.001), had a higher expectation regarding a positive effect on their condition (OR 2.72 (95% CI: [1.57, 4.71]), p < 0.001) and were more likely to recommend the programme to a friend with similar problems (OR 4.70 (95% CI: [2.46, 8.97]), p < 0.001). Three months post randomisation, risk knowledge was higher in the CG (HR −0.52 (95% CI: [−0.93, −0.10]), p = 0.015). However, the difference of 4.2 versus 4.7 points out of 10 was marginal. Diet quality was higher in the IG three months post randomisation (HR 0.43 (95% CI: [0.14, 0.72]), p = 0.004). No effects on MS-specific QoL, disability, role preference, coping ability, symptoms of anxiety and depression, patient activation, physical activity, RTC or diet quality were observed 12 months post randomisation. Detailed statistical results for secondary outcomes are provided in Appendices 3–5.
Intervention and CG programme adherence
Out of 115 randomised IG participants, 114 (99%) logged on. The first six levidex modules were completed by 70%, up to 12 modules by 50% and all 16 modules by 42% of randomised participants. An average of 31 (SD 42.1) total days with activity were detected in the IG. Out of 119 randomised CG participants, 113 (95%) logged on. The CG showed a completion rate of 78% with an average of 10.5 (SD 4.1) total days with activity. Only 41% in the IG (11 missings) and 56% in the CG (13 missings) correctly detected their group allocation after 12 months. Regarding programme adherence, 58 (50%) participants in the IG and 93 (78%) participants in the CG demonstrated high use. The exploratory analysis of the combined primary endpoint showed a trend in favour of the IG (HR 0.59 (95% CI: [0.33, 1.09]), p = 0.09) and a high programme use (HR 0.60 (95% CI: [0.34, 1.05]), p = 0.07). There was no interaction between the treatment group and programme adherence (HR 1.85 (95% CI: [0.89, 3.84]), p = 0.101, see Appendix 2).
Ancillary analyses
Subgroup analyses on participants with lower QoL (HAQUAMS score ⩾ 2, HR 1.02 (95% CI: [0.54, 1.92]), p = 0.96), higher physical impairment (EDSS ⩾ 2, HR 0.9 (95% CI: [0.47, 1.72], p = 0.75) and moderate to severe symptoms of anxiety (HADS-A score 8–17, HR 0.97 (95%-CI: [0.54, 1.73], p = 0.91) or depression (HADS-D score 8–16, HR 0.25 (95% CI: [0.06, 1.05]), p = 0.06) showed no effect on the primary endpoint. When combining all cut-offs, no effect on the primary endpoint was detected (HR 1.06 (95% CI: [0.68, 1.64], p = 0.81).
Harms
No adverse events or harms (side effects, risks or complications) beyond psychological impact were expected and obtained during study conduct.
Discussion
This RCT assessed the impact of a purely web-based and combined health behaviour change intervention on inflammatory disease activity in MS. Disease activity of this early MS cohort, measured by number of new relapses and/or T2 lesions, aligns with data reported for another large early German MS cohort. 31 As the main finding, we could not demonstrate a slower evolution of clinical relapses or T2 lesions in the IG, compared to the CG. Moreover, the IG showed no sustained altered health behaviours, compared to the CG, as assessed by PROMs. However, after three months of using levidex, which is marked by exposure to the nutrition-related modules of the intervention, IG participants reported higher diet quality than those in the CG. The clinical relevance of the short-term improvement in diet quality (0.43 points on a scale of 0–10) is unclear and requires further investigation. Adherence to levidex (IG) corresponds to data reported in other trials investigating digital health applications.32,33 When examining participants who completed nearly all (⩾75%) or all modules (n = 151), no different pattern emerged compared to those who completed fewer than 75% of modules.
Behaviour change is a substantial challenge in life and health. Theory-based interventions using appropriate strategies and behavioural targets are needed, 34 but the required number and combination of active ingredients in interventions targeting health behaviour is a matter of ongoing research. 35 In MS, most work in the area of behaviour change has focussed on neuropsychiatric domains of the disease with considerable evidence for CBT-based approaches in face-to-face, group-based or even digital interventions. 36 Efforts to enable behaviour change have also been undertaken in physical activity and exercise, indicating short-term benefits of up to 6 months based on social cognitive theory approaches. 37 Motl et al. 11 could recently show altered physical activity behaviour with a predominantly web-based behaviour change intervention based on this approach. Patient empowerment strategies represent an additional approach and studies focusing behaviour change in chronic diseases have been reviewed in 2021. 38 However, most of the concepts, even if Internet-delivered, made use of some personalised approach and the evidence of purely computerised interventions for MS management is limited. 39 Although levidex enables some degree of individualisation while it is completely software-based, the amount of personalisation is limited. Feedback from process evaluation interviews with participants (analysis ongoing) however indicates a high appreciation of the tool and higher behaviour change than documented in our secondary outcome measures. Therefore, the study indicates that responsive instruments for behaviour change in domains as physical activity and diet need further development. However, studies with higher individualisation offering an array of support options up to longitudinal personal coaching are warranted.
While there was no effect on MS-related QoL in this cohort, levidex has shown clinically relevant improvements in MS-related QoL in a sample of 421 pwMS who were diagnosed for at least 1 year and participated in a purely web-based RCT. 40 Differing from this study sample, the earlier trial included pwMS with lower QoL as well as later stages of MS, which might explain the significant effect on QoL observed in the earlier study. This work led to the inclusion of levidex in the preliminary directory of reimbursable digital health applications (DiGA) which is part of the German eHealth infrastructure. A further web-based RCT including all forms and stages of MS is currently conducted to evaluate the benefits for QoL.
Limitations
A personal coach might have enhanced behaviour change and sustainability at least in a subgroup of participants. However, this remains speculative, given that meta-analyses have shown that personal behavioural interventions do not always outperform digital interventions, even though they are presumably more personalised and adaptive. 41 The multiple behaviour change intervention may have had differential physiological effects based on the chosen domain of behaviour change. This heterogeneity may have diluted treatment effects. There is furthermore heterogeneity in MRI assessments (e.g. protocol applied) and randomisation may only partly have compensated this limitation. In addition, participants showed a high MS-related QoL and patient activation already at baseline, along with low levels of depression and anxiety, making any further improvements difficult. While self-reported moderate to severe symptoms of anxiety were present in one-third of participants, the close monitoring with frequent clinical visits and MRI scans above German standard care in both treatment groups might itself have had a positive impact (e.g. lower psychological stress due to an increased sense of security), which might have led to an underestimation of the benefits of the intervention. 39 Furthermore, the use of an active psychoeducational control programme (dexilev), based on credible material from the German MS society, might have had effects on outcomes that were similar to the IG programme (levidex). Indeed, in the absence of a no-treatment or care-as-usual CG, it is possible that both active conditions were similarly effective (or ineffective), which is why no systematic post-treatment between-group differences were observed. For the assessment of health behaviour, only PROMs were used while perhaps objective outcomes represent more accurate and valid measures.
Further research should particularly focus on individuals with low QoL and problematic health behaviours. In addition, objective outcome measures, such as accelerometry, might help detecting subtle behaviour changes more accurately.
Conclusion
This pragmatic RCT did not meet its ambitious primary endpoint. Additional personal support might induce more pronounced behaviour change and possibly have a greater impact on MS disease activity, but such potential advantages remain to be demonstrated, and they must be weighed against the higher costs and limited scalability of person-delivered treatments, compared to fully automated digital interventions. Nevertheless, the study shows that close clinical and MRI follow-up in the first years after diagnosis and extensive lifestyle information is appreciated and adhered to by people recently diagnosed with MS.
Supplemental Material
sj-docx-1-msj-10.1177_13524585251356410 – Supplemental material for Digital lifestyle management application (levidex) for empowerment and health behaviour change in early multiple sclerosis – Results of the randomised controlled POWER@MS1 trial
Supplemental material, sj-docx-1-msj-10.1177_13524585251356410 for Digital lifestyle management application (levidex) for empowerment and health behaviour change in early multiple sclerosis – Results of the randomised controlled POWER@MS1 trial by Nicole Krause, Carlotta Derad, Barbara von Glasenapp, Karin Riemann-Lorenz, Björn Meyer, Thomas Asendorf, Herbert Temmes, Markus van de Loo, Stefan M Gold, Charlotte Schubert, Klarissa H Stürner, Clemens Warnke, Stephan Schmidt, Tim Friede and Christoph Heesen in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The authors want to thank all participating pwMS as well as all neurologists and study nurses in the participating study centres and radiologists involved in the study for their support. They thank Ina Schröder (study nurse in Kiel), Monika Höveler (study nurse in Cologne), Monika Schmitz (study nurse in Bonn) and Veronika Grundner (study nurse in Munich) for their support in recruitment and study conduct.
Protocol
The study protocol was published 12 and planned statistical analyses were uploaded as part of the trial registration (NCT03968172) before data export and analyses.
Monitoring
External independent data monitoring was performed by the contract research organisation CTC North GmbH & Co. KG throughout the study duration. This included onsite visits at the coordinating centre as well as remote data checks in secuTrial® for all POWER@MS1 study centres.
Author contributions
CH is the principal investigator and led the planning and conduct of the full study with support from NK, CD, BvG, TA, KRL, HT, SMG and TF. NK and CH wrote the first draft of the paper. CD, TA and TF conducted the statistical analyses and specifically revised these sections of the paper. MvdL contributed to the study as a pwMS expert. CS, KHS, CW, SS and the POWER@MS1 study group were involved in recruitment and study conduct in the study centres. All authors conceived the study, revised the manuscript for relevant scientific content and approved the final version.
Data availability statement
All relevant data are within the manuscript and its supplemental material.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: TF reports consultancies including data monitoring committees for Actimed, Apellis, Aslan, Bayer, BiosenseWebster, BMS, CSL Behring, Daiichi Sankyo, Enanta, Fresenius Kabi, Galapagos, IQVIA, Immunic, KyowaKirin, LivaNova, Minoryx, Novartis, PINK gegen Brustkrebs, PPD, RECARDIO, Recordati, Relaxera, Roche, Servier, Viatris, VICO Therapeutics and Vifor. CW has received institutional support from Novartis, Alexion, Sanofi Genzyme, Biogen, Merck, Janssen, Bayer and Roche. SMG reports grants from German Ministry of Education and Research, German Ministry of Health, German Research Foundation, National MS Society (USA), and Wellcome Trust and personal fees from Hexal, Tegus, and Angelini. CH has received research funding support from BMS, Merck, Novartis, Sanofi. He has received speaker honoraria from Novartis and Roche. CS reports speaker honoraria from Alexion, Argenx, Desitin and TAD and honoraria for advice for Alexion and Argenx. PO has received research support, lecture fees and travel expenses from Alexion, Anavex Life Sciences, Bayer Health Care, Bial, Biogen, Immunic Therapeutics, Janssen, Merck Serono, Novartis, Pfizer, Roche, Sanofi Genzyme and TEVA. BM is an employee of GAIA AG, the company that developed, owns, and operates levidex, the digital health application investigated in this trial. AB receives funding from the Innovationsausschuss of the German Federal Joint Committee (G-BA; grant 01VSF23040) and from the German Federal Ministry of Education and Research (BMBF; grant 01ZZ2102B). He has received consulting and/or speaker fees from Alexion, Argenx, Biogen, CSL Behring, Horizon/Amgen, Merck, Neuraxpharm, Novartis, Roche and Sandoz/Hexal, and his institution has received compensation for clinical trials from Alexion, Biogen, Merck, Novartis, Roche, and Sanofi Genzyme; all outside the present work. JK received personal compensation for consulting as well as speaker honoraria from Bayer, Biogen, Bristol Myers Squibb, CSL Behring, Grifols, Janssen, Merck Serono, Novartis, Roche, Sanofi Genzyme, Takeda and Teva. KG has received research support, lecture fees and travel expenses from Abbvie, Allergan, Almirall, Bayer, Biogen, Celgene, Desitin, Eisai, Sanofi Genzyme, Hexal, Ipsen, Merck, Novartis, Octapharma, Roche and Teva. KHS has received lecture fees, honoraria for advice and travel expenses from Merck, Roche, Sanofi Genzyme and ArgenX. SS received personal compensations for serving on scientific advisory boards for Hoffmann-La Roche, Novartis, Merck Serono, Bayer Vital, Biogen, Genzyme, and Teva and received travel funding and/or speaker honoraria from Hoffmann-La Roche, Novartis, Merck Serono, Bayer Vital, Biogen, Genzyme, and Teva. All others have nothing to declare.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Public funding for this project was provided by the Innovationsfonds, Innovationsausschuss beim Gemeinsamen Bundesausschuss, Wegelystraße 8, 10623 Berlin, Germany (01VSF17015) without any involvement of the funding body in study related aspects. The commercial version of the digital health application levidex was developed prior to the commencement of this project; its development was not funded by the German Innovation Fund. The programme version examined in this trial differed in some aspects from the commercial version of levidex: (a) additional content was added on the topic of treatment-related shared-decision-making, and (b) annotated scientific references within the programme were reviewed and updated and added by the study team. These modifications are not part of the commercially available version of levidex.
Ethical considerations
The study has been approved by the Ethics Committee of the Hamburg Chamber of Physicians (PV6015) and the ethics committees of participating study centres.
Consent to participate
Informed consent was obtained by neurologists in participating MS centres. A copy of the signed informed consent was sent to the coordinating study centre by fax. Participants were allowed to withdraw their consent at any time. There was no financial compensation for participation. After reaching the primary endpoint, participants were asked to remain in the study. Only the study team (investigators) and Alexander Stahmann (medical information scientist at the German MS Registry) had access to the final trial dataset.
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
