Abstract
Background:
Family planning is an important aspect of multiple sclerosis (MS), and neuromyelitis optica spectrum disorder (NMOSD) management. Knowledge gaps remain, including optimal perinatal management strategies, and fetal risks associated with disease-modifying therapy (DMT) exposure.
Objective:
To describe perinatal DMT use, together with pregnancy and neonatal outcomes prospectively recorded in the International MSBase Pregnancy and Women’s Health Registry.
Methods:
We report summary statistics for data collected between May 2020 and August 2024.
Results:
A total of 1887 relapsing-remitting MS (RRMS), 12 primary-progressive MS (PPMS), 2 radiologically isolated syndrome (RIS) and 21 NMOSD completed pregnancies were recorded, including 1644 (85.5%) live births, 208 (10.8%) miscarriages, and 6 (0.3%) neonatal deaths. Most women had unassisted (53.8%) or assisted (7.4%) vaginal births. Seventy five percent of pregnancies had DMT exposures within 6 months preconception; 19% of NMOSD, and 62% of MS pregnancies were DMT-exposed during gestation; 18.1% of pregnancies reported in-pregnancy monoclonal antibody DMT exposure. No overt safety signals were seen.
Conclusion:
This first report from the newly launched MSBase pregnancy registry, establishes an increasing number of pregnancies being conceived on monoclonal antibody therapies. Although no safety signals were observed, it is important to continue monitoring for safety signals in real-world databases as the use of highly effective therapies continues to increase perinatally.
Keywords
Introduction
Women are disproportionately diagnosed with multiple sclerosis (MS) and related disorders. Women with MS outnumber men by three-fold, 1 whereas women with neuromyelitis optica spectrum disorder (NMOSD) outnumber men by nine-fold. 2 Commonly diagnosed in their third to fifth decades of life, most women will use disease-modifying therapies (DMTs) for much of their lives. Our understanding of the impact of these conditions, and the use of DMT on women’s health, including fecundity, fertility, 3 and pregnancy,2,4 together with reproductive aging and menopause3,5 is evolving.
The evidence base for the safe and effective use of DMTs perinatally in women with MS is growing, increasing the confidence of neurologists to recommend and prescribe these medications during pregnancy planning and, for some DMTs, in pregnancy. Data from large registries including MSBase,6,7 the Canadian, 8 Danish, 9 French, 10 German,11,12 Italian, 13 and American 14 pregnancy registries, as well as purpose-designed studies 15 provide critical real-world insights that, in addition to post-marketing pharma-led studies, inform clinical guidelines on the use of these DMT through pregnancy and postpartum.4,16 The evidence base around NMOSD and NMOSD DMT use during pregnancy still remains limited, as 2000 exposed pregnancies are ideally required for each DMT to accurately define risks, including rare birth defects.2,17–19
While the international MS outcomes registry MSBase 20 has prospectively collected pregnancy-related information since 2004, detailed pregnancy and neonatal outcomes data were not recorded. With the growing number of DMT available to treat MS and NMOSD, the need to capture more detailed data is evident. In response, MSBase launched a prospective Pregnancy, Neonatal Outcomes and Women’s Health Registry 21 in 2020. The purpose of this registry is to address the impact of MS and related disorders, together with the impact DMTs on women’s health throughout the lifespan. 21 In this first update from our women’s health registry since its launch, we specifically report on pregnancy and birth outcomes in the context of an expanding number of DMT available for the management of MS and NMOSD through pregnancy.
Methods
The MSBase Pregnancy, Neonatal Outcomes and Women’s Health Registry
The MSBase Pregnancy, Neonatal Outcomes and Women’s Health Registry began collecting detailed maternal and neonatal health outcomes on 10 May 2020. 21 New data fields include assisted reproductive technology method, pregnancy outcome, obstetric/maternal complications, delivery method, reason for termination/miscarriage, congenital abnormality, birthweight, sex, and breastfeeding data.
Data are collected from 45 countries spanning the northern parts of Africa, North and South America, Asia, Australasia, Europe, and the Middle East.
Study ethics
The Registry is integrated within the MSBase Registry (registered within the WHO International Clinical Trials Registry Platform ID ACTRN12605000455662). The MSBase Registry has ethics approvals or exemptions granted by each participating site’s human research ethics committee. All participants provide informed consent for their de-identified clinical data to be shared with researchers.
Data acquisition and study population
Data were collected during routine outpatient neurology visits and recorded by neurologists or nurses in real-time and therefore reflect real-world clinical care. Data are collected from women of childbearing age with relapse-onset MS (ROMS), progressive-onset MS (POMS), radiologically isolated syndrome (RIS) and NMOSD (both probable and definite).
Definitions
Any woman of
Statistical methods
We report summary statistics for demographic, clinical, treatment, pregnancy, birth, and neonatal outcomes data collected to-date. All variables were assessed for normality using the Shapiro-Wilk normality test, and reported as means and standard deviations (SD) or as medians and interquartile ranges (IQRs) as appropriate. Categorical data are reported as numbers and percentages.
Data availability statement
MSBase is a data processor, warehousing data from individual principal investigators who agree to share their datasets on a project-by-project basis. Data access to external parties can be granted at the sole discretion of each MSBase Principal Investigator (the data controllers), who will need to be approached individually for permission. To make a request for data access, please contact the corresponding author for instructions.
Results
Patient and pregnancy characteristics
The MSBase Pregnancy, Neonatal Outcomes and Women’s Health Registry 21 actively followed 20,850 women of childbearing age between 10 May 2020 and 1 August 2024. Of these 20,850 women, 20,539 had a diagnosis of MS, 40 had a diagnosis of radiologically isolated syndrome (RIS), and 271 had a diagnosis of NMOSD. These women had an average age of 37.3 years (SD 7.1 years) at data extract, with 2411 women now aged between 45 and 49 years. The median follow-up duration for this cohort in the MSBase Registry was 5.25 years (IQR: 1.9, 9.9). The median disease duration at data extract was 8.5 years (IQR: 4.3, 14.0) for women with MS, and 7.2 years (IQR: 2.9, 11.3) for women with NMOSD.
We recorded 9541 pregnancies for 5806 (27.8%) of the women followed during the observation period (see Figure 1). Of these, 7024 pregnancies for 4299 women were completed prior to our registry launch, and 2517 (26.4%) pregnancies for 2146 women were captured from 10 May 2020 onwards. The contribution by country to the registry is displayed in Figure 2.
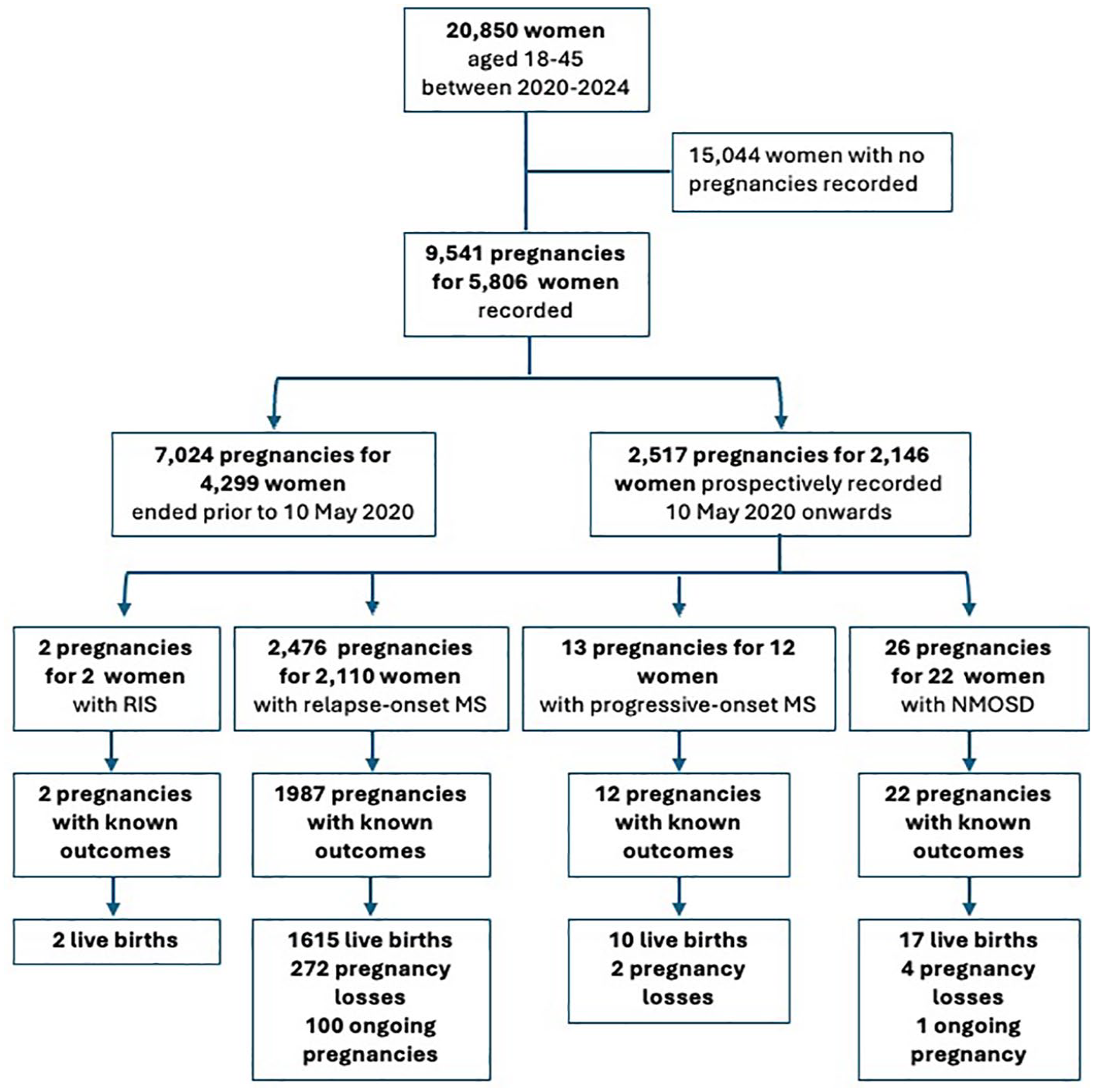
Consort flow diagram of study inclusion showing the number of pregnancies prospectively recorded, stratified by disease phenotype: radiologically isolated syndrome (RIS), relapse-onset MS (including secondary progressive MS), progressive-onset MS, and neuromyelitis optica spectrum disorder (NMOSD).
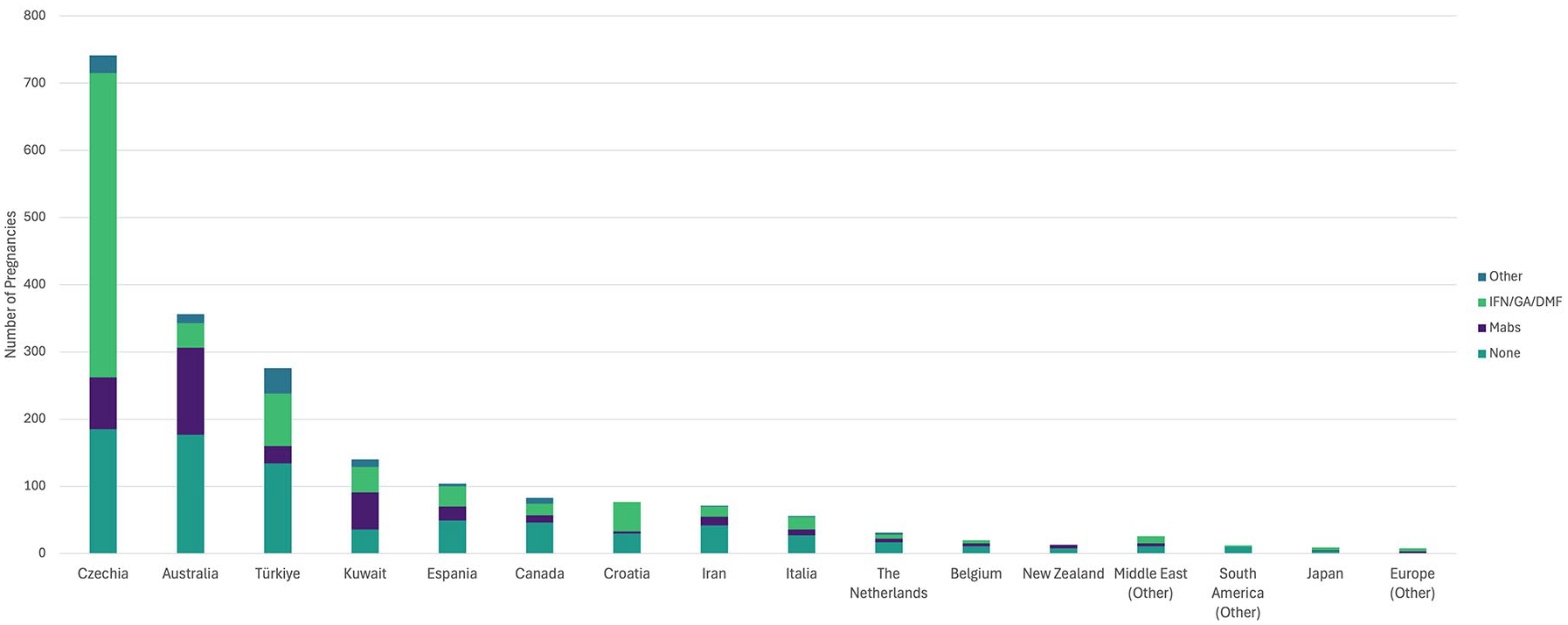
Number of pregnancies prospectively recorded by country, or region if fewer than 10 captured. This figure further displays the differences in regional peripartum management, showing the number of disease-modifying therapy (DMT) exposures during pregnancy, categorized by DMT type: none; interferons, glatiramer acetate or dimethyl fumarate (IFN/GA/DMF); monoclonal antibodies (Mabs, including: natalizumab, ocrelizumab, ofatumumab, rituximab, alemtuzumab); and all other therapies.
Of the 2146 women who had pregnancies recorded in the newly launched Registry; 640 women had pregnancies both prior to and after Registry launch, and 1,507 women had pregnancies only after pregnancy registry launch. A total of 1820 (84.8%) women had a single pregnancy, 285 (13.3%) had two pregnancies, and 41 (1.9%) women reported three or more pregnancies during the observation period. The mean maternal age at the last menstrual period (LMP) was 32.3 years (SD 4.7 years), with a median expanded disability status scale (EDSS) score of 1.5 (IQR: 1, 2) within 12 months of the LMP. The median follow-up post-pregnancy was 1.7 years (IQR: 0.8, 2.6 years; range: 0.1–4.2 years). Detailed, per-phenotype, patient characteristics at each pregnancy are summarized in Table 1.
Patient characteristics at each prospectively recorded pregnancy with known outcomes.
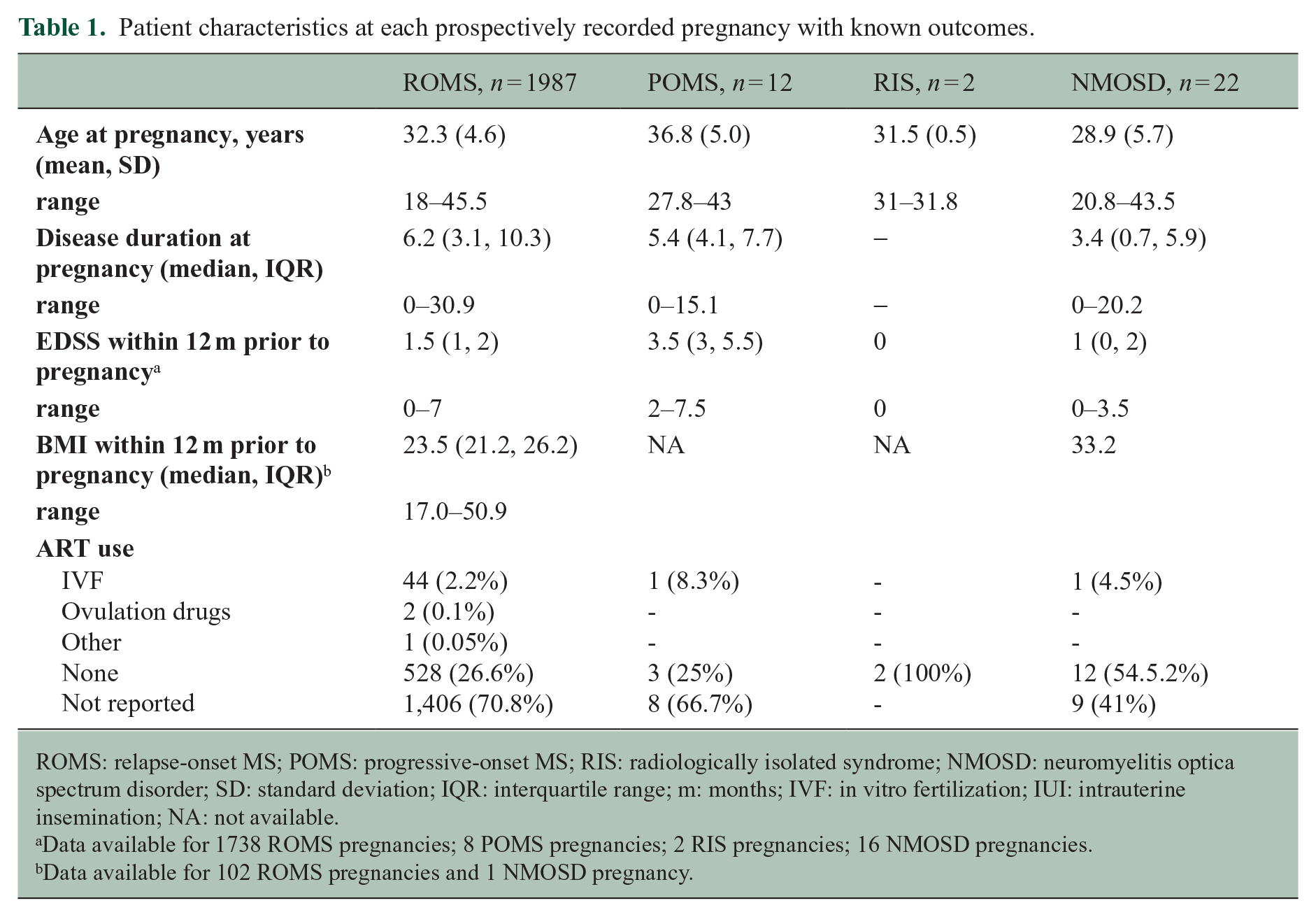
ROMS: relapse-onset MS; POMS: progressive-onset MS; RIS: radiologically isolated syndrome; NMOSD: neuromyelitis optica spectrum disorder; SD: standard deviation; IQR: interquartile range; m: months; IVF: in vitro fertilization; IUI: intrauterine insemination; NA: not available.
Data available for 1738 ROMS pregnancies; 8 POMS pregnancies; 2 RIS pregnancies; 16 NMOSD pregnancies.
Data available for 102 ROMS pregnancies and 1 NMOSD pregnancy.
We had limited ascertainment of several demographic factors for the 2,146 women with prospectively reported pregnancies. Educational status was available for 684 (31.9%) of women. Of these 57 (8.3%) had completed elementary school, 194 (28.4%) had completed high school, and 433 (63.3%) had completed a tertiary degree. Employment status was available for 660 (30.7%) of women. Of these women, 441 (66.8%) were employed, 45 (6.8%) were unemployed, 83 (12.6%) performed domestic duties, 90 (13.6%) were students, and 1 (0.15%) reported being on a disability support pension. Smoking status was only available for 99 (4.6%) of women, of which 15 (15.1%) were current smokers, 17 (17.2%) were past smokers, and 67 (67.7%) were never smokers. We recorded 592 pregnancies with known outcomes for which assisted reproductive therapy (ART) data were available. Of these, 543 (91.7%) did not use ART; 46 conceived using in vitro fertilization (IVF), two used ovulation drugs and one used other methods.
Pregnancy outcomes
As of 1 August 2024, 1922 (76.4%) of pregnancies were completed, 101 (4%) pregnancies were ongoing. 494 (19.6%) pregnancies did not have outcome data available beyond the date of the last menstrual period and were considered lost to follow-up. Of reported pregnancy outcomes 1464 (76.2%) of pregnancies resulted in full-term births, 180 (9.4%) were preterm births, totaling 1644 (85.5%) live births. 208 (13.7%) of pregnancies resulted in miscarriage, and 55 (2.9%) in termination. Six (0.3%) neonatal deaths and 9 (0.5%) ectopic pregnancies were reported. Of the total 1644 live births reported in our registry, birthweights were available for 413 term babies with a median weight of 3260 g (IQR: 3000, 3590). 918 (55.8%) babies were breastfed of which 150 (16.3%) reported exclusive breastfeeding for a median 3 months (IQR: 1, 5 months; range: 0–6 months). For detailed pregnancy outcomes by disease phenotype, see Table 2. Most pregnancies were singletons, with only 35 twin pregnancies reported. Of the twin pregnancies, 21 were born at term, nine preterm, four miscarriages, and one elective termination were also reported.
Pregnancy and neonatal outcomes by disease phenotype for pregnancies with known outcomes.
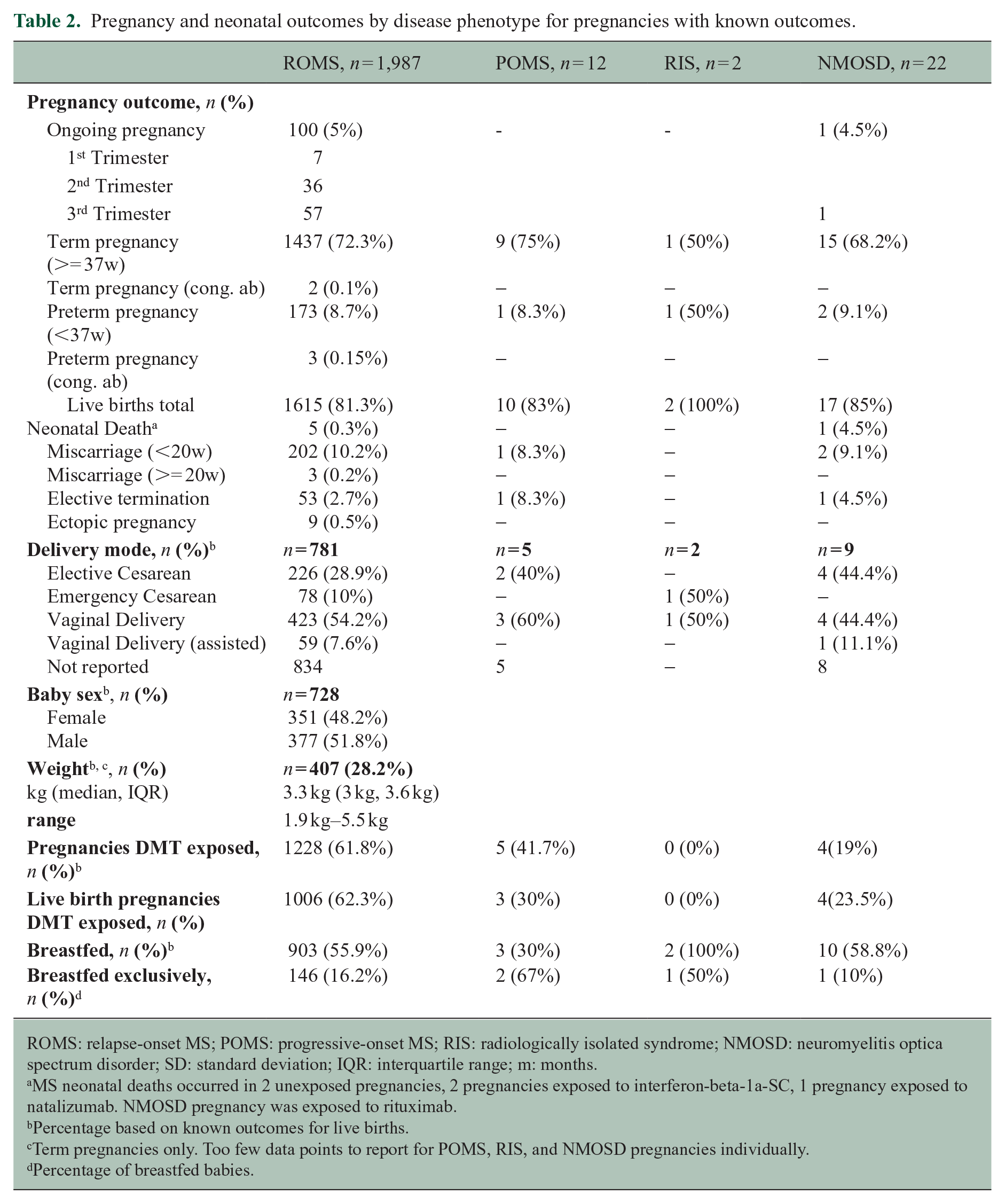
ROMS: relapse-onset MS; POMS: progressive-onset MS; RIS: radiologically isolated syndrome; NMOSD: neuromyelitis optica spectrum disorder; SD: standard deviation; IQR: interquartile range; m: months.
MS neonatal deaths occurred in 2 unexposed pregnancies, 2 pregnancies exposed to interferon-beta-1a-SC, 1 pregnancy exposed to natalizumab. NMOSD pregnancy was exposed to rituximab.
Percentage based on known outcomes for live births.
Term pregnancies only. Too few data points to report for POMS, RIS, and NMOSD pregnancies individually.
Percentage of breastfed babies.
The majority of women had unassisted (54.1%) or assisted (7.5%) vaginal births. Elective cesarean sections comprised 29.1% of delivery methods, most often performed in Iran (57.1%), Turkey (50.9%), Italy (46.3%), and Australia (37%). Birth complication data were reported for 614 (31.9%) completed pregnancies. Of these 535 (87.1%) reported no complications. The most common complications were gestational diabetes (n = 16) and pre-eclampsia or pregnancy-induced hypertension (n = 10). Antepartum hemorrhage was reported in five women. Additional complications reported included: growth restriction, large for gestational age, low birth weight, oligohydramnios, polyhydramnios, premature membrane rupture, placenta previa, placental abruption, and others.
Disease-modifying therapy exposures
In the 6 months prior to conception, 506 (25%) pregnancies with known outcomes had no DMT exposure and 1517 (75%) were DMT-exposed, of which 282 (18.6%) discontinued within the 6 months prior to conception with a median 31 days washout (IQR: 2 day, 97 days). Disease-modifying therapy (DMT) exposure in-pregnancy was reported for 1228 (61.8%) pregnancies in women with ROMS, 5 (41.7%) pregnancies in women with POMS, and 4 (19%) NMOSD pregnancies (Table 2). DMT exposure trends varied by country (Figure 2). The interferons, glatiramer acetate, and dimethyl fumarate were most commonly used in pregnancy with 37.8% of pregnancies exposed to these drugs. In-pregnancy monoclonal antibody infusions were recorded in 18.1% of pregnancies.
Of ROMS pregnancies, 1006 (62.3%) of live birth (term and preterm) pregnancies received DMT during pregnancy. The median length of DMT exposure in pregnancy was 42 days (IQR: 25, 218; range: 1–290). Figure 3 summarizes the DMT exposure trends in women with ROMS who had live births from the 6 months prior to conception through to 3 months postpartum. The per-DMT exposure durations for this cohort are summarized in Table 3. Postpartum, 811 (50.2%) ROMS women with live births resumed DMT use within 3 months (Figure 3, Table 3). Whereas, 183 (68%) women reporting miscarriages, terminations and other adverse pregnancy outcomes resumed DMT within 3 months of pregnancy completion.
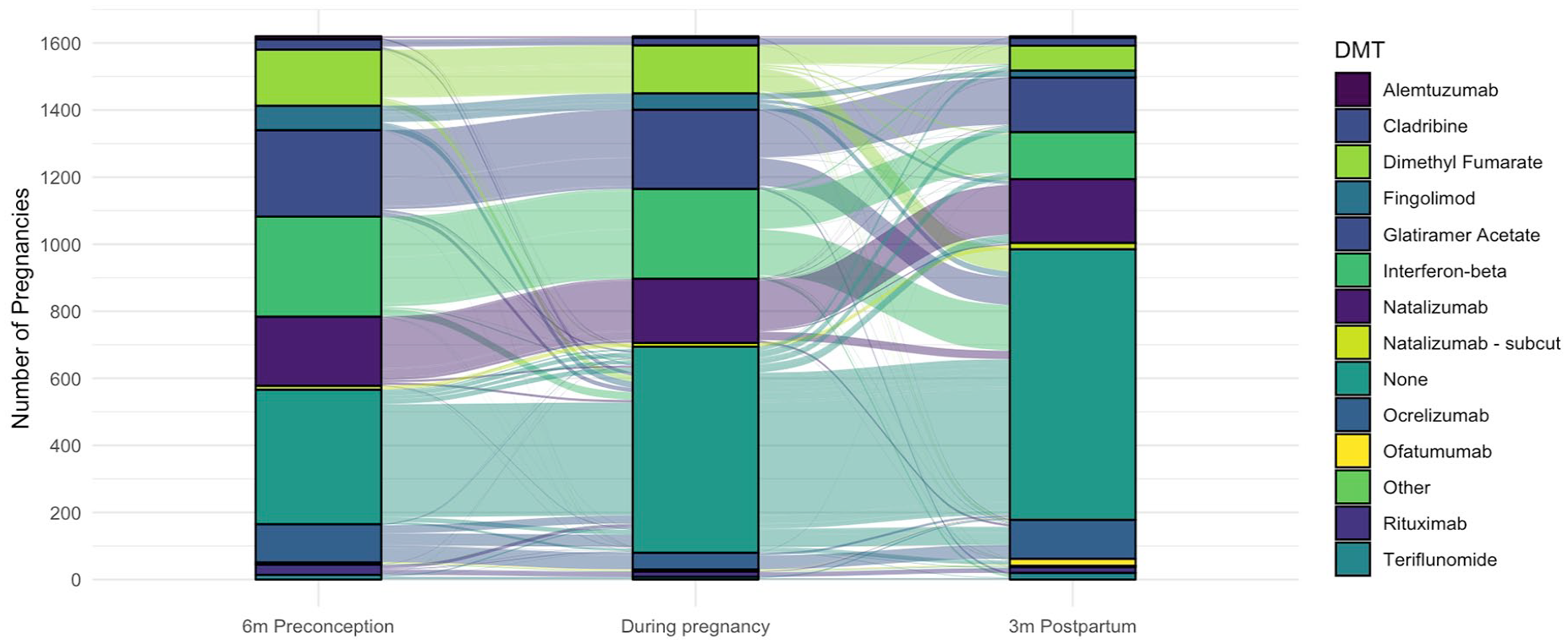
Frequencies and patterns of disease-modifying therapy (DMT) exposure 6 months preconception, during pregnancy (for any duration) and in the first 3 months postpartum in women with relapse-onset MS. DMTs with fewer than five exposures grouped together in the “other” category.
Disease-modifying therapy use associated with 1615 live births to women with relapse-onset MS.
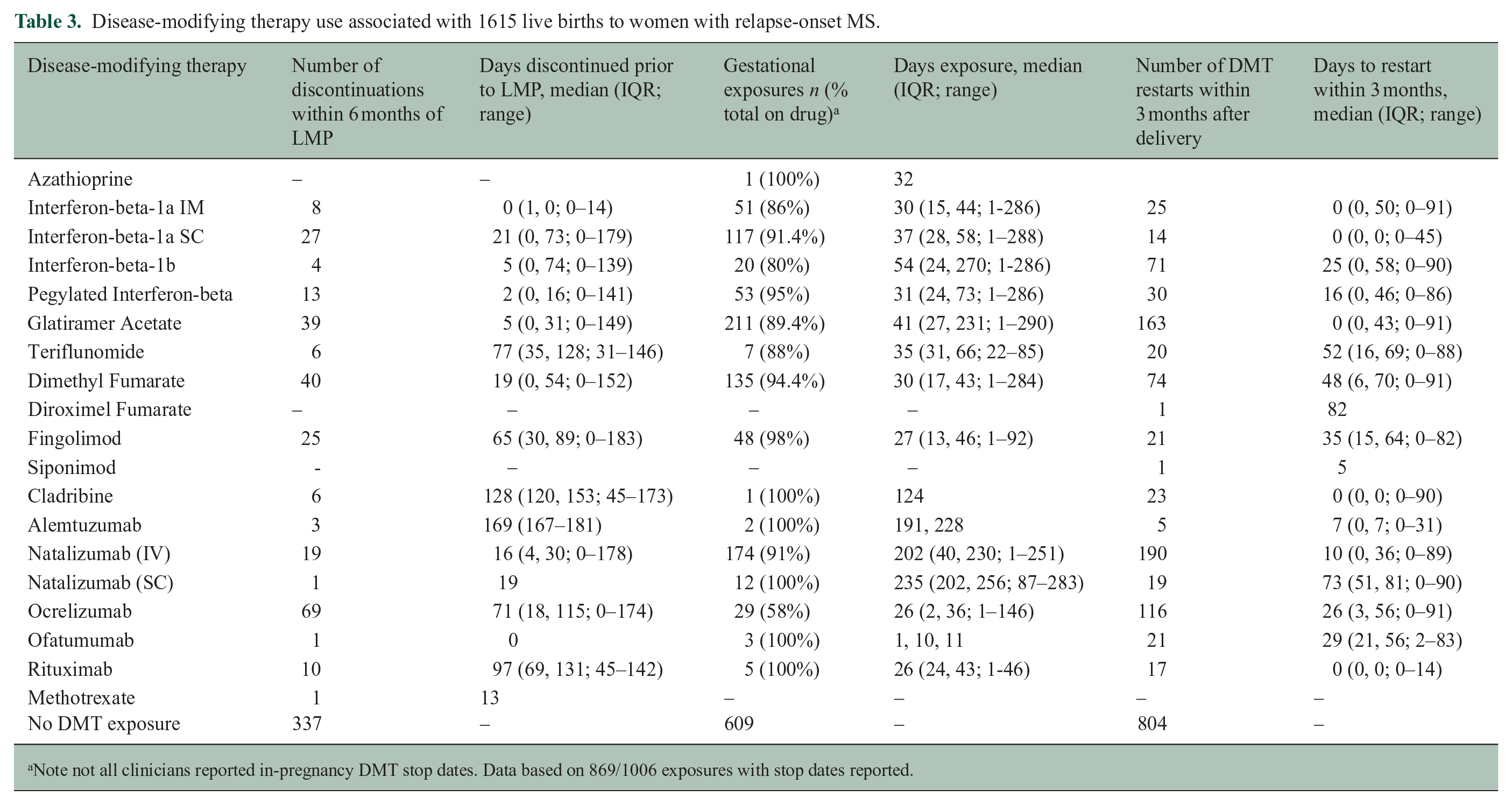
Note not all clinicians reported in-pregnancy DMT stop dates. Data based on 869/1006 exposures with stop dates reported.
In the NMOSD cohort, six rituximab exposures were recorded in the 6 months prior to conception. Of these, two ceased therapy prior to conception with a washout of 2 and 4 months, respectively. Four rituximab exposures occurred during pregnancy, median gestation at DMT exposure was 26 days (IQR: 24, 43). Of these six pregnancies, five resulted in healthy, term deliveries, and one in neonatal death. One woman was exposed to eculizumab 2.5 months prior to conception; this pregnancy was aborted. No women with NMOSD restarted DMT within 3 months postpartum; however, five women resumed rituximab a median 271 days (IQR: 219, 287) after the completion of their pregnancies. We did not observe any perinatal exposures to satralizumab.
Of 918 breastfed babies, 296 (32.2%) were DMT-exposed during breastfeeding in the first 3 months postpartum. All breastfeeding mothers who resumed DMT within the first 3 months postpartum had ROMS. The median time to DMT reinitiation during this period was 2 days (IQR: 0, 43; range: 0–91). The DMTs resumed during breastfeeding included glatiramer acetate (n = 78), interferon-beta preparations (n = 62), natalizumab infusions (n = 60), ocrelizumab (n = 20), pegylated-interferon-beta (n = 17), dimethyl fumarate (n = 14), rituximab (n = 12), natalizumab subcutaneous injections (n = 10), ofatumumab (n = 7) and others (n = 16).
Congenital abnormalities
In total, five pregnancies were reported with congenital abnormalities, three preterm pregnancies and two term pregnancies. Of the three preterm pregnancies, one had been exposed to glatiramer acetate for 38 days, another received rituximab at 43 days gestation, the third was not DMT-exposed. No specifics of the congenital abnormalities were reported for this group. One term pregnancy reporting a chromosomal abnormality had been exposed to dimethyl fumarate for 37 days. This mother had a history of prior pregnancies, one of which had also resulted in the birth of a child with congenital abnormalities. The other term pregnancy with congenital abnormality was not DMT-exposed.
Discussion
Family planning and pregnancy decisions are key considerations in the management of women with MS and NMOSD. In the era of highly effective disease-modifying therapies knowledge gaps remain, including best management strategies through pregnancy and whether there are fetal risks associated with DMT exposure in utero.
In this first report from the newly launched prospective MSBase Pregnancy, Neonatal Outcomes and Women’s Health Registry, we report 101 ongoing pregnancies, and pregnancy outcomes for 1922 completed pregnancies, including 21 completed pregnancies for women with NMOSD.
Over 85% of pregnancies with known outcomes resulted in live births, in line with other recently reported studies. 22 In our registry, 9% of births were preterm births, 10% of pregnancies were miscarried, reassuringly in line with data from German MS Pregnancy Registry, 23 and in line with global rates in the general population as per the World Health Organization.24,25
Interestingly, 75% of pregnancies were DMT exposed in the 6 months prior to conception, decreasing to 62% of pregnancies during gestation, an overall lower percentage of DMT use than in the German pregnancy registry that reported 76% exposure at pregnancy. 23 Ours may be a slight underestimate as we measured the date of last infusion for monoclonal antibodies, but did not estimate the duration of effect of these drugs. We know, for example that infusional anti-CD20 monoclonal antibodies have a durable effect, protecting against disease activity for many months into pregnancy and potentially beyond. 6 The recent study reporting pregnancy outcomes from the Roche pharmacovigilance study for example defined exposure to ocrelizumab if the last infusion was within 3 months of conception. 22 Given that here we assessed numerous infusional therapies with varying mechanisms of action, here we determined to use a consistent definition for all. Postpartum, only 50% of our study population with live births had restarted DMT use within the first 3 months of delivery. Most women resumed the use of the same DMT as they had used preconception or during pregnancy, although we did observe a low level of DMT switch. The median exposure to DMT during pregnancy was 42 days. This has increased marginally since our previous 2019 report showing a median exposure of 30 days, 26 indicating that many women still discontinue DMT upon pregnancy confirmation. However, we did see a marked increase in the duration of natalizumab exposure, median 202 days (28.9 weeks) in the present study, as compared with 24 days in our previous report. 26 This reflects our increased understanding of maternal and fetal outcomes in babies exposed to natalizumab in utero, 12 and comfort with using natalizumab through to the end of the second trimester to minimize risk of in-pregnancy disease rebound upon withdrawal.4,7
We observed pregnancy management varied among countries. Australian and Kuwaiti clinicians were most likely to manage pregnancies with monoclonal antibodies, whereas Europeans were most likely to use interferons, glatiramer acetate or dimethyl fumarate to manage pregnancy. These differences likely reflect local legislative and therapeutic agency indications for these drugs. While still relatively new, we saw the growing use of ocrelizumab, ofatumumab and subcutaneous natalizumab use perinatally.
While we did not have sufficient weight or other biometric data to analyze neonatal outcomes by DMT exposure, regardless, no overt safety signals were observed. Previous studies have not reported overt differences in outcomes of babies born to mothers with MS, and non-MS controls. 27 Furthermore, studies by other groups have not reported concerning safety signals for the newer DMT including natalizumab12,28 and ocrelizumab. 22
Few studies to-date have reported obstetric outcomes for women with NMOSD.29–31 Here we report 17 (81%) live births and one neonatal death (4.8%) for 21 completed pregnancies in women with NMOSD. Six pregnancies, including the neonatal death, were exposed to rituximab. An analysis of a 153 rituximab-exposed pregnancies reported one neonatal death at 6 weeks. 32 In the general population, across Europe, North America, and Australasia, neonatal deaths occur in 2–3 per 1000 live births. 33 Here we reported six neonatal deaths across our MS and NMOSD cohorts, marginally higher than the expected five given our cohort size. However, our per-DMT sample size is too small to draw any causal conclusions.
A key strength of our Registry is the unified, prospective, multicenter, international dataset that we have been able to accumulate, with over 2000 pregnancies from women with MS, RIS, and NMOSD recorded over a 4-year period. With time and increasing data collection, we will be able to compare pregnancy and neonatal outcomes across various regions and accumulate real-world evidence for safety signals. Our registry has further worked with the BigMS network to align key data fields, so that we will be able to collaboratively report on safety signals across registries in the future. We will therefore continue to contribute critical evidence for MS management and obstetric care.
We do, however, have several limitations. Our neonatal and pregnancy outcome data are self-reported; therefore, we are unable to confirm outcomes with obstetricians, pediatricians, or other healthcare professionals. Further, details of congenital abnormalities for 4/5 pregnancies were unavailable, and pregnancy complications were underreported. We continue to work with our network to ensure the most accurate acquisition of this important data as we move forward. Another limitation of our registry is that a large number of pregnancies, almost 20%, were lost to follow-up. We have now initiated three-monthly reminders to our network to follow up on known pregnancies, so that we can reduce the number lost to follow-up. Furthermore, the international nature of our database, differing pediatric practice, and legal frameworks means that we are unable to collect neonatal outcomes beyond 12 months of age. To better understand the impacts of DMT exposure in children born to mothers with MS, we will eagerly anticipate results from national registries such as the French RESPONSE study that will be collecting these outcomes out to 6 years. 10
Conclusion
This first report from the international MSBase pregnancy registry shows that an increasing number of pregnancies are conceived on monoclonal antibody therapies, confirming increased confidence in their use in the peri-conception period. Reassuringly our data do not show any safety issues at present. However, in an era where pregnancies are increasingly exposed to DMT use and with the growing number of DMT available, it is important to continue to collect data to monitor for fetal and maternal serious adverse events in real-world databases.
Footnotes
Acknowledgements
We would like to thank all study participants for sharing their clinical data, particularly in difficult circumstances of aborted or miscarried pregnancies or neonatal deaths. We also thank the MSBase Operations team Ms Charlotte Sartori, Ms Rein More, Ms Linda Sim, Ms Cynthia Tang, Ms Carolyn Tran, Mr Dusko Stupar, Ms Pamela Farr, and Ms Alison Le for supporting and maintaining the MSBase Registry, without which, this work could not be done.
The MSBase Study Co-Investigators and Contributors
Dr Marta Vachova, KZ a.s., Hospital Teplice, Czech Republic
Dr Jana Libertinova, Second Faculty of Medicine, Charles University and Motol University Hospital, Prague, Czech Republic
Dr Michael Barnett, Brain and Mind Centre, The University of Sydney, Australia
Dr Pavel Hradilek, Department of Neurology, Faculty of Medicine, Ostrava University, Czech Republic
Dr Pierre Duquette, CHUM MS Center and Universite de Montreal
Dr Alena Martinkova, Department of Neurology, Hospital Pardubice, Czech Republic
Dr William M Carroll, Perron Institute for Neurological and Translational Science, University of Western Australia and Centre for Molecular Medicine and Innovative Therapeutics, Murdoch University, Perth, Australia, Department of Neurology, Sir Charles Gairdner Hospital
Dr Eva Recmanova, Department of Neurology, CSSS Saint-Jérôme, Julie Prevost; Tomas Bata Hospital, Zlin, Czech Republic
Ms Louise Rath, Department of Neurology, Alfred Health, Melbourne, Australia
Dr Guillermo Izquierdo, Hospital Universitario Virgen Macarena, Sevilla, Spain
Dr Abdorreza Naser Moghadasi, Multiple Sclerosis Research Center, Neuroscience Institute, Tehran University of Medical Sciences, Tehran, Iran
Dr Mark Slee Flinders University, Adelaide, Australia
Dr Davide Maimone, Azienda Ospedaliera per l'Emergenza Cannizzaro, Catania, Italy
Dr Jiwon Oh, St. Michael’s Hospital, Toronto, Canada
Dr Murat Terzi, 19 Mayis University, Samsun, Turkey
Dr Pierre Grammond, CISSS Chaudière-Appalache, Levis, Canada
Dr Jose Luis Sanchez-Menoyo, Galdakao-Usansolo University Hospital, Osakidetza-Basque Health Service, Galdakao, Spain
Dr Emanuele D’Amico, Università di Foggia, Medical and Surgical Sciences, Foggia, Italy
Dr Beatriz Romero Ferrando, Waikato Hospital, Hamilton, New Zealand
Dr Mehmet Fatih Yetkin, Erciyes University, Neurology, Kayseri, Turkey
Dr Bassem Yamout, Harley Street Medical Centre, Abu Dhabi, United Arab Emirates
Dr Ivana Stetkarova, Third Faculty of Medicine, Charles University in Prague and Hospital Kralovske Vinohrady, Czech Republic
Dr Cristina Ramo-Tello, Hospital Germans Trias i Pujol, Badalona, Spain
Dr Koen de Gans, Groene Hart Ziekenhuis, Gouda, Netherlands
Dr Riadh Gouider and Dr Saloua Mrabet, Razi University Hospital, Tunis, Tunisia
Dr Abdullah Al-Asmi, Sultan Qaboos University, Al-Khodh, Oman
Dr Rana Karabudak, Yeditepe University, Istanbul, Turkey
Dr Aysun Soysal, Bakirkoy Education and Research Hospital for Psychiatric and Neurological Diseases, Istanbul, Turkey
Dr Samia J. Khoury, American University of Beirut Medical Center, Beirut, Lebanon
Dr Emmanuelle Lapointe, Centre Hospitalier Universitaire de Sherbrooke, Canada
Dr Simón Cárdenas-Robledo, Hospital Universitario Nacional de Colombia Bogotá, Colombia
Dr Nevin Shalaby, Cairo University, Kasr-Al-Ainy MS Clinic, Cairo, Egypt
Dr Daniele Spitaleri, Azienda Ospedaliera di Rilievo Nazionale San Giuseppe Moscati Avellino, Avellino, Italy
Dr Joana Guimarães, Unidade Local de Saúde de São João, Porto, Portugal
Dr Gregor Brecl Jakob, Department of Neurology, University Medical Centre Ljubljana, Slovenia
Dr Suzanne Hodgkinson, Immune tolerance laboratory Ingham Institute and Dept of Medicine, UNSW, Sydney, Australia
Dr Mohammad Baghbanian, Dr Baghbanian Clinic, Iran
Dr Recai Turkoglu, Haydarpasa Numune Training and Research Hospital, Istanbul, Turkey
Dr Vincent van Pesch, Cliniques Universitaires Saint-Luc, Brussels, Belgium
Dr Francois Grand’Maison, Neuro Rive-Sud, Quebec, Canada
Dr Elisabetta Cartechini, AST Macerata, Macerata, Italy
Dr Enrique Gomez-Figueroa, Civil Hospital of Guadalajara, Neurology, Guadalajara, Mexico
Dr Jennifer Massey, St Vincent’s Hospital, Sydney, Australia
Dr Chiyoko Nohara, Tokyo Metropolitan Health and Medical Treatment Corporation Ebara Hospital, Department of Neurology, Tokyo, Japan
Dr Kenzo Sakurai, St. Marianna University School of Medicine, Japan
Dr Deborah Mason, Christchurch Hospital, Christchurch, New Zealand
Dr Talal Al-Harbi, King Fahad Specialist Hospital-Dammam, Saudi Arabia
Dr Tamara Castillo-Triviño, Hospital Universitario Donostia and IIS Biodonostia, San Sebastián, Spain
Dr Fumitaka Shimizu, Yamaguchi University Graduate School of Medicine, Department of Neurology and Clinical Neuroscience, Ube, Japan
Dr Bruce Taylor, Royal Hobart Hospital, Hobart, Australia
Dr Todd A. Hardy, Dr Steve Vucic, and Dr Stephen Reddel, Concord Repatriation General Hospital, Sydney, Australia
Dr Sudarshini Ramanathan, Translational Neuroimmunology Group, Kids Neuroscience Centre and Brain and Mind Centre, Faculty of Medicine and Health, University of Sydney, Sydney, Australia
Dr Danny Decoo, AZ Alma Ziekenhuis, Sijsele—Damme, Belgium
Dr Maria Cecilia Aragon de Vecino, Hospital Moinhos de Vento, Multiple Sclerosis Unit, Porto Alegre, Brazil
Dr Yuri Nakamura, Fukuoka Central Hospital, Department of Neurology, Brain and Nerve Center, Fukuoka, Japan
Dr Norio Chihara, Kobe University Graduate School of Medicine, Division of Neurology, Kobe, Japan
Dr Eli Skromne, Hospital Angeles de las Lomas. Instituto Mexicano de Neurociencias., Huixquilucan Estado de Mexico, Mexico
Dr Oksana Kopchak, Kyiv Medical University, Ukraine
Author Contributions
Scientific committee: Jokubaitis (co-chair, scientific lead), Gray (co-chair, medical lead), van der Walt, Alroughani, Altintas, Butzkueven, Eichau, Hughes, Willekens.
Study concept and design: Jokubaitis, Gray, van der Walt, Alroughani, Altintas, Butzkueven, Eichau, Hughes, Willekens
Statistical analysis and drafting of the manuscript: Jokubaitis
Data acquisition, interpretation of the data and Critical revision of the manuscript for important intellectual content: All authors
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: VGJ Receives research fellowship support from the National Health and Medical Research Council of Australia (2025360). Her institution receives research funding support from F.Hoffmann-La Roche, the International Progressive MS Alliance, Multiple Sclerosis Australia and the Pennycook Foundation outside of this current work. She has received speaker’s honoraria from Novartis and The Limbic.
RA received honoraria as a speaker and for serving on scientific advisory boards from Bayer, Biogen, GSK, Merck, Novartis, Roche, and Sanofi Genzyme.
AA received speaker honoraria from Alexion and travel grant from Ali Raif Ilac, Merck.
SE have received speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck, Janssen, Bristol-Meyers, Bayer, Sanofi Genzyme, Roche and Teva.
SH has received unrestricted educational grants or speaking honoraria from Biogen, Merck Serono, Novartis, Roche, and Sanofi Genzyme.
BW received honoraria for acting as a member of Scientific Advisory Boards/Consultancy for Alexion, Almirall, Biogen, Celgene/BMS, Merck, Janssen, Novartis, Roche, Sandoz, Sanofi Genzyme and speaker honoraria and travel support from Biogen, Celgene/BMS, Merck, Novartis, Roche, Sanofi Genzyme; research and/or patient support grants from Biogen, Janssen, Merck, Sanofi Genzyme, Roche. Honoraria and grants were paid to the UZA/UZA Foundation. Further, B.W. received research funding from FWO-TBM, Belgian Charcot Foundation, Start2Cure Foundation, Queen Elisabeth Medical Foundation for Neurosciences, and the National MS Society USA.
DH was supported by the Charles University: Cooperation Program in Neuroscience, by the project National Institute for Neurological Research (Programme EXCELES, ID Project No. LX22NPO5107)—Funded by the European Union—Next Generation EU, and by General University Hospital in Prague project MH CZ-DRO-VFN64165. She also received compensation for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck, Bayer, Sanofi Genzyme, Roche, and Teva, as well as support for research activities from Biogen Idec.
EKH received honoraria/research support from Biogen, Merck Serono, Novars, Roche, and Teva; has been member of advisory boards for Actelion, Biogen, Celgene, Merck Serono, Novars, and Sanofi Genzyme; received honoraria/research support from Biogen, Merck Serono, Novars, Roche, and Teva; has been member of advisory boards for Actelion, Biogen, Celgene, Merck Serono, Novars, and Sanofi Genzyme; and has been supported by the Czech Ministry of Education—project Cooperation LF1, research area Neuroscience, and the project National Institute for Neurological Research (Programme EXCELES, ID project No LX22NPO5107)—funded by the European Union-Next Generation EU.
SO reports no disclosures.
CB received conference travel support from Biogen, Novartis, Bayer Schering, Merck and Teva; has participated in clinical trials by Sanofi-Aventis, Roche and Novartis.
MH Received consultation and/or speaker fees from Biogen, Merck, Novartis, Roche, AstraZeneca, Amgen.
TK Tomas Kalincik served on scientific advisory boards or as a consultant for MS International Federation and World Health Organization, Therapeutic Goods Administration, BMS, Roche, Janssen, Genzyme, Novartis, Merck and Biogen, received conference travel support and/or speaker honoraria from WebMD Global, Merck, Sandoz, Novartis, Biogen, Roche, Eisai, Genzyme, Teva and BioCSL and received research or educational event support from Biogen, Novartis, Genzyme, Roche, Celgene and Merck.
IR served on scientific advisory boards, received conference travel support and/or speaker honoraria from Roche, Novartis, Merck and Biogen. Izanne Roos is supported by MS Australia and the Trish Multiple Sclerosis Research Foundation.
ME reports no disclosures.
MP received honoraria and travel grants from the following pharmaceutical companies: Biogen, Bristol-Myers Squibb, Genzyme, Janssen-Cilag, Merck Serono, Novartis, Roche, and Teva Pharma.
JLS has received travel compensation from Novartis, Biogen, Roche and Merck. Her institution receives the honoraria for talks and advisory board commitment as well as research grants from Biogen, Merck, Roche and Novartis.
JEML Jose E. Meca-Lallana has received honoraria as a consultant, as a chairman or lecturer in meetings and has participated in clinical trials and other research projects promoted by Alexion, Almirall, Biogen, Bristol Myers Squibb, Horizon, Johnson & Johnson, Merck, Neuraxpharm, Novartis, Roche, Sandoz, Sanofi and UCB.
ZR reports no disclosures.
JH received compensations for travel and consultant fees form Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen-Cilag.
AP reports no disclosures.
MG reports no disclosures.
RA received conference travel support from Novartis, Teva, Biogen, Bayer and Merck and has participated in clinical trials by Biogen, Novartis, Teva and Actelion.
KB received speaker honoraria and/or education support from Biogen, Teva, Novartis, Genzyme-Sanofi, Roche, Merck and Alexion; has been a member of advisory boards for Merck and Biogen.
OS received honoraria and consulting fees from Bayer Schering, Novartis, Merck, Biogen and Genzyme.
NAJ is a PI on commercial MS studies sponsored by Novartis, Roche, Biogen and Sanofi. He has received speaker’s honoraria from Merck. He has had conference travel and registration reimbursement; and consultancy fees from Novartis.
AGK Served on Scientific Advisory Boards for Bayer, BioCSL, Biogen Idec, Clene Nanomedicine, Esai, Innate Immunotherapeutics, Lgpharma, Merck, Mitsubishi Tanabe Pharma, NeuroScientific Biopharmaceuticals, Novartis, Progenis, Roche, Sanofi-Aventis, Sanofi Genzyme, Teva, and View Health.
MJF-P received travel compensation from Merck.
MF received travel and meeting attendance support from Novartis, Biogen, Roche, Sanofi Genzyme and Merck.
AS received travel and meeting attendance support from Novartis, Biogen, Roche, Merck, Bristol, Sanofi Genzyme, Almirall, Piam.
YB received speaker honoraria/consulting fees from Merck, Biogen, Roche, Brystol, Novartis, Sanofi and Sandoz.
SMB has served on scientific advisory boards and have received conference travel support and/or speaker honoraria from several pharmaceutical companies including Roche, Novartis, Merck, Cinnagen, Nanoalvand and Biogen.
OG reports no disclosures.
RM or his institution has received remuneration for his speaking engagements, advisory board memberships, research and travel from Biogen, Merck, Genzyme, Bayer, Roche, Teva, Novartis, CSL, BMS, MedDay and NHMRC.
ZP received honoraria and travel grants from the following pharmaceutical companies: Biogen, Bristol-Myers Squibb, Eli Lilly, Genzyme, Janssen-Cilag, Lundbeck, Merck Serono, Novartis, Roche, and Teva Pharma.
PS received compensations for travel, speaker honoraria and consultant fees from Biogen Idec, Novartis, Merck Serono, Roche, Sanofi Genzyme, Teva and Janssen-Cilag.
PMC received speakers fees and travel grants from Novartis, Biogen, T’évalua, Sanofi
GL received travel and/or consultancy compensation from Sanofi Genzyme, Roche, Teva, Merck, Novartis, Celgene, Biogen.
HB is an employee of Monash University and has accepted travel compensation from Merck; his institution receives honoraria for talks, steering committee activities, and research grants from Roche, Merck, Biogen, Novartis, and UCB Pharma, Medical Research Future Fund Australia, NHMRC Australia, Trish MS Foundation, MS Australia and the Pennycook Foundation. He receives personal compensation for steering group activities for the Brain Health Initiative from Oxford Health Policy Forum and is funded by an NHMRC Australia Investigator Grant.
AVDW served on advisory boards and receives unrestricted research grants from Novartis, Biogen, Merck, and Roche She has received speaker’s honoraria and travel support from Novartis, Roche, and Merck. She receives grant support from the National Health and Medical Research Council of Australia and MS Research Australia.
OG received honoraria as consultant on scientific advisory boards for Genzyme, Biogen, Merck, Roche, and Novartis; has received travel grants from Biogen, Merck, Roche, and Novartis; has participated in clinical trials by Biogen and Merck. Her institution has received research grant support from Biogen.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study did not receive specific funding support. The MSBase Registry receives financial support from Biogen, Merck, Novartis and Roche.
