Abstract
Background:
A rare complication of natalizumab treatment is progressive multifocal leukoencephalopathy (PML); risk can be stratified using JC virus (JCV) antibody status. Risk calculators to aid clinical decision-making use cohort data tested using the Stratify-JCV assay. Following concerns of an increased number of JCV seropositive results following the switch to biosimilar natalizumab and the associated test switch, we sought to understand the clinical implications.
Methods:
A total of 497 people with MS, who had switched from Tysabri to Tyruko with sequential results available on both assays, were included.
Results:
Of 250 patients negative on Stratify-JCV, 119 (47.6%) were subsequently positive on ImmunoWELL. Agreement was the poorest at lower JCV index, where positive/negative cutoffs inform risk stratification and pharmacovigilance protocols most significantly.
Conclusion:
These observed differences create uncertainty in how to counsel patients and best carry out PML surveillance. Specific concerns include overestimating risk leading to increased patient concern, increased monitoring burden and associated healthcare costs and lack of access to a highly effective therapy. It is incumbent on all stakeholders including pharmaceutical industry and test developers, patient groups, governmental authorities (including regulatory bodies) and clinicians to work together to find an expeditious solution.
Keywords
Introduction
Natalizumab, a monoclonal antibody against alpha-4-integrin, prevents lymphocyte migration across the blood-brain barrier and reduces inflammatory disease activity in multiple sclerosis (MS). 1 A rare but serious complication of natalizumab treatment is progressive multifocal leukoencephalopathy (PML) secondary to JC virus (JCV). PML risk in those receiving natalizumab for MS can be stratified using JCV antibody status and index (corresponding to titre). There is a significantly lower risk of PML in those who are JCV seronegative and a lower cumulative risk in those with a low versus high antibody index. 2 Studies demonstrate annualised seroconversion rates of 3%–5.8% in those receiving natalizumab over up to 6 years.3–6 Previous work has not demonstrated a link between initial antibody index and/or rate of seroconversion with regard to age, gender or disease modifying therapy (DMT).4,5
JCV testing has previously been provided using the Stratify-JCV testing system by Biogen, who manufacture originator natalizumab (Tysabri). Biosimilar natalizumab (Tyruko) is being rolled out across the United Kingdom, 7 with JCV testing provided by Medicover using the ImmunoWELL assay, supported by Sandoz.
A range of PML risk calculators to aid clinical decision-making and patient communication have been developed to support natalizumab use, based on evidence from large cohorts utilising the Stratify-JCV assay. Following early reports, 8 we sought to evaluate test agreement and potential impact on clinical care.
Methods
We sought to pool routine clinical data capturing the two different JCV assays across four MS centres in London and Sheffield, UK, in order to understand the clinical implications of assay change in terms of patient communication and clinical decision-making based on JCV serostatus. Bloods were sent for analysis according to the natalizumab product used at the time of testing as per usual clinical care, at an approximate 6-month interval as per Summary of Product Characteristics (SmPC) recommendations. 9
This study was approved at each centre as a service evaluation with a focus on the implications for risk stratification and monitoring and patient counselling within routine clinical practice.
Results
A total of 497 people with MS switched from Tysabri to Tyruko with results available on both assays were included. The mean age at the ImmunoWELL test was 44.5 (range = 19–79), 378 (76%) were female. The Expanded Disability Status Scale (EDSS) was available for 444 (range = 0–7.5, mode 1.5). Tests were performed a median of 151 days apart (interquartile range = 151–182 days).
Of 250 patients reported to be JCV negative on Stratify-JCV, 119 (47.6%) were subsequently positive on ImmunoWELL; 111 were low positive (JCV index < 1.5) and 8 were high positive (JCV index > 1.5). Of 140 patients who were low positive on Stratify-JCV, 31 (22.1%) were now high positive (Figure 1(a)).
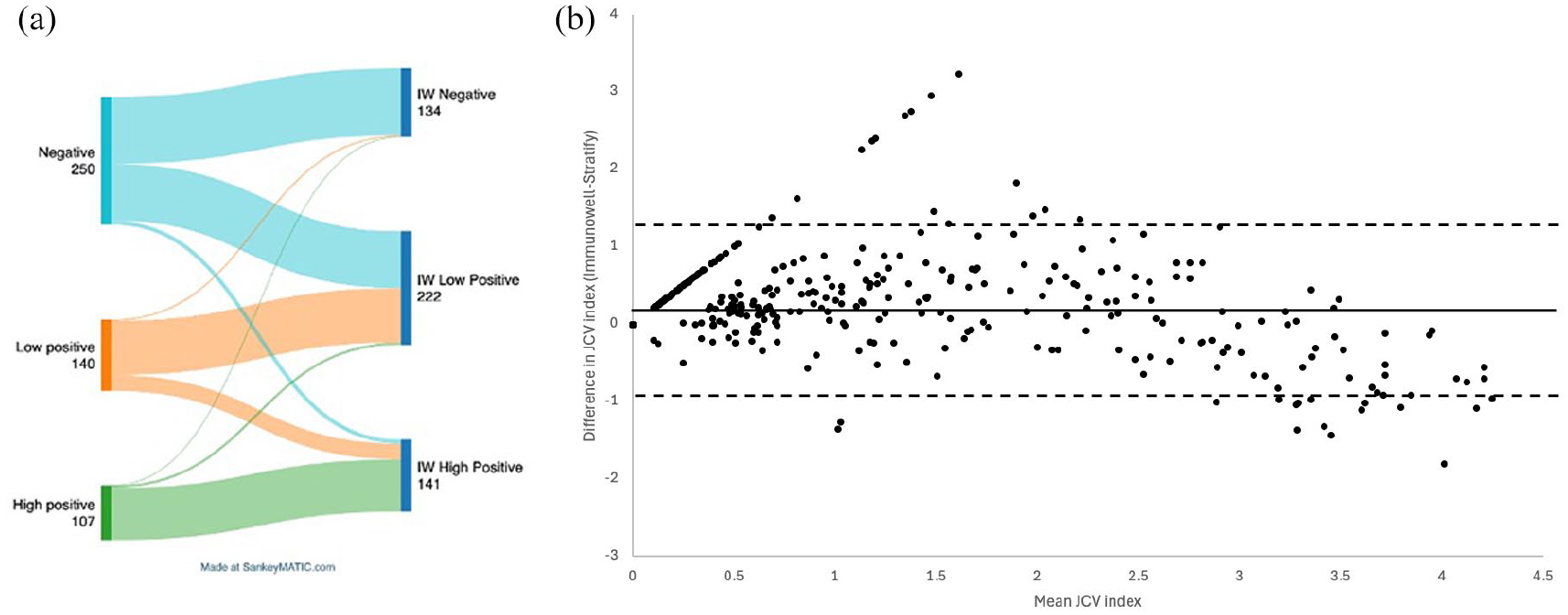
(a) Sankey plot of the JCV index stratification of patients via the Stratify assay on the left and how this relates to ImmunoWELL stratification on the right. Created using SankeyMATIC.com. (b) Bland–Altman plot of agreement between the two tests across the JCV index – with the poorest agreement seen at the lower levels of JCV testing. The mean (solid line) and upper and lower bounds of the 95% confidence interval (2 standard deviations) are also indicated.
Taking Stratify-JCV as the reference (based on existing practice), the performance of ImmunoWELL testing demonstrated sensitivity of 98.8% (95% confidence interval (CI) = 96.5%–99.7%), specificity of 52.4% (95% CI = 46.2%–58.5%), positive predictive value (PPV) of 67.3 (95% CI = 62.2%–71.8%) and negative predictive value (NPV) of 97.8% (95% CI = 93.6%–99.4%). The agreement between the two tests was poorest at lower JCV index, where positive/negative cutoffs inform risk stratification and subsequent pharmacovigilance protocols most significantly (Figure 1(b)).
The clinical implication of this was that 47.6% of patients previously thought to be low risk were potentially subject to additional risk monitoring procedures including personalised risk discussion, enhanced magnetic resonance imaging (MRI) monitoring and consideration of treatment switching. Of those previously thought to be moderate risk, 22.1% would be judged high risk and likely recommended to switch DMT. Within our cohort, an additional 76 outpatient clinic visits were required for patients now classified as JCV positive to discuss results and clinical implications. All who had exceeded 2 years of therapy (n = 105) were subject to enhanced MRI monitoring (3–6 monthly rather than annually). In total, 52 patients switched treatment as a result of their change in serostatus, of whom most (44) reverted to the originator product, 2 to fingolimod and 6 to anti-CD20 medications.
Discussion
While the overall correlation between tests was reasonable, the low specificity of the ImmunoWELL test (close to 50%) along with the relatively low positive predictive value substantially limits the utility of this test. Clinical decisions around perceived PML risk are usually based on positive/negative JCV serostatus. While we do not have simultaneous paired testing due to manufacturers only providing access to testing for those receiving their product, this reflects routine clinical practice. The high proportion (47.6%) of those negative on Stratify subsequently positive on ImmunoWELL over a 6-month period is markedly different from the previously observed annual seroconversion rate of 3%–5.8%.3–6 While we were not able to Stratify by demographic or disease characteristic, age does not correlate with change in JCV index over time; 3 previous work has not demonstrated any association with demographic characteristic.4,5 While we did not have access to serial testing using the ImmunoWELL assay to evaluate test–retest reliability, this has been assessed by the regulatory authorities and found to be satisfactory.
In terms of immediate implications for clinical practice, in our cohort, a significant number of additional clinical appointments were required, in addition to over 100 additional MRI appointments per year. Those patients who switched therapy potentially risk relapse or disease reactivation during the switching process. A rational management strategy is thus urgently needed until more definite risk stratification related to ImmunoWELL is available – data which will take years to derive. Potential immediate solutions to secure patient care include centralisation of JCV testing and/or harmonisation of techniques through reagent sharing and cooperation between companies. Due to the nonlinearity, we demonstrate at low JCV indexes, where the crucial distinction between negatives and low positives is made, a simple mathematical correction is not possible. Importantly, at present, safety in terms of under-monitoring for PML does not appear to be a concern; however, redrawing cutoffs for positivity risks reducing assay sensitivity, leading to underestimation of PML risk. In the current situation, potential negative implications of overestimating risk include increased patient anxiety and increased monitoring burden. At worst, there is a risk that a proportion of patients will be inappropriately identified as being at high risk of PML and therefore discount the option of natalizumab or undergo unnecessary burdensome monitoring and anxiety.
Where patients have a negative JCV index on Stratify but low positive on ImmunoWELL, it is likely that the biological factor being measured is stable, while the assay results differ. However, given the previously documented rate of seroconversion,3–6 patients should be advised that should their JCV index remain positive on repeat testing at 6 months, treatment switching/derisking should be considered, typically 12–24 months after the first positive test. This should be based on an individual risk–benefit discussion with their treating team, considering prior MS disease activity, clinical and radiological response to natalizumab, prior treatment exposure and monitoring burden.
Where patients are known to be JCV positive at low titre and subsequently test high positive on ImmunoWELL, they should be counselled that their overall risk may have increased, and they should consider treatment switching/derisking, based on reasonable agreement between assays towards the middle and upper ranges of the testable range.
The cost of this increased monitoring burden is not insubstantial; doubling the number of MRI scans required along with the neuroradiological time to adequately review the increased scan number, along with increased patient and clinician anxiety. This issue has been observed in early reports from Norway; 5 our work provides a large and systematic evaluation of this observation.
It is important for healthcare systems to seek maximum cost-effectiveness by using biosimilars; however, clinicians must be alert to unintended consequences. It is incumbent on all MS stakeholders including pharmaceutical industry and test developers, patient groups, governmental authorities (including regulatory bodies) and clinicians to work together to find an expeditious solution.
Footnotes
Data Availability Statement
Data would need to be requested from each trust via the Caldicott guardian given current approvals.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: J.V. has received reimbursement for advisory boards, conference attendance and educational events from Merck, Novartis and Roche. R.B. has no acknowledgements. R.N. has received reimbursement for advisory boards from Roche and Novartis. R.D. has received reimbursement for advisory boards, conference attendance and educational events for Sanofi, Merck, Biogen, Novartis, Janssen and Roche. W.B. has received speaker honoraria and/or acted as a consultant for Biogen, Janssen, Merck, Novartis, Roche, Sandoz, Sanofi and Viatris. D.P. is principal investigator for commercial trials funded by Novartis, Merck, Janssen Pharmaceuticals and Roche; chief investigator for commercial trials funded by Novartis; has an investigator grant from Sanofi Genzyme; he has previously received advisory board/consultancy and speaker’s fees from Biogen, Celgene, Janssen, MedDay, Merck, Novartis, Roche and Sandoz. J.M. has Biogen, Janssen, Merck, Neurax, Novartis, Roche, Sandoz and Sanofi. RD has received honoraria for speaking and/or travelling from Biogen, Eisai, Merck, Roche and Janssen. Advisory boards for Roche, Biogen, Sandoz, Novartis and Merck in the past 2 years. All honoraria for speaking, travelling and advisory boards were paid into an institutional account and used to support research, open access publications and training for research group members. She has received grant support from Biogen, Merck and Celgene.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
