Abstract
Background:
Mobility assessment is essential for monitoring disease progression in people with multiple sclerosis (PwMS). Technologies such as wearable sensors show potential for this purpose, but consensus is needed to optimize collection and interpretation of digital measures in PwMS.
Objective:
To propose a framework for measuring and interpreting key aspects of impaired gait in PwMS using a smartphone worn at the waist level.
Methods:
The framework was developed on the basis of clinical understanding and knowledge of sensor signal processing, supported by a systematic literature review (SLR). The SLR targeted articles published after 2011 that measured gait characteristics in PwMS. Findings were used to propose standardized definitions for complementary gait domains and define digital measures that should be captured for each domain.
Results:
The resulting framework for PwMS recommends definitions for pace, rhythm, stability, symmetry, variability, smoothness, complexity and fatigability gait domains. For each domain, a set of digital measures is described with respect to their interpretability and associated caveats.
Conclusion:
This framework provides recommendations for measuring complex gait patterns in PwMS using widely available technology. This work promotes the use of standardized gait domain definitions and harmonized descriptions of associated digital measures, paving the way for future validation efforts.
Background
Walking disability is common in people with multiple sclerosis (PwMS), owing to lower limb motor function deficits caused by neurological impairment 1 and/or other mobility difficulties caused by multiple sclerosis (MS) symptoms, such as fatigue and visual and cognitive impairment.2–4 The impact of MS on walking, or gait, can manifest in various ways, including slower walking speed, instability and asymmetry, among others. 5 These impairments have a considerable negative impact on mobility, activities of daily living and quality of life.1,6 In turn, this may then place an additional burden on healthcare resources. As such, measurement of gait characteristics is an important aspect of patient management in MS; not only may such measurements be used to monitor disease activity over time 7 but they may also enable assessment of interventions to preserve walking ability in PwMS.
The standard assessment of mobility in MS is the Kurtzke Expanded Disability Status Scale (EDSS), which is a clinician-reported outcome based on patient assessment, including distance walked by the patient and their reliance on assistive devices. 8 However, this broad assessment can fail to detect more granular changes in mobility, especially in early disease.9,10 Some gait metrics may be acquired more accurately using clinical gait tests, for example, the 6-minute walk test (6MWT) and the timed 25-foot walk (T25FW) for walking speed, and the Berg Balance Scale 11 and the Timed Up and Go test 12 for balance and postural stability. However, similar to the EDSS, these tests are limited in their ability to capture subtle changes in gait.13–15 In addition, they require a clinic visit, as well as space and time to perform the test, which is not always readily available, and the tests are not intended to differentiate between specific gait presentations. 16
Mobility monitoring using digital instruments allows the collection of quantitative information and a thorough characterization of gait patterns.5,17–23 While some methods of digital mobility monitoring (e.g. electronic treadmill, video capture) may still require a clinic visit to perform the assessment,5,17–24 wearable sensors may be used to capture gait data in unsupervised settings. Although wearables such as smartwatches, motion trackers and pressure insoles are ideal for continuous unintrusive monitoring, smartphones are more effective tools for implementing remote standardized assessments because instructions can be provided to the patient via apps while collecting the data of interest. Indeed, this approach has already been explored in a variety of neurodegenerative diseases, including Parkinson’s disease, 25 spinal muscular atrophy26,27 and MS,28,29 leading to highly reproducible results from remote gait assessments.
Smartphones typically contain accelerometers, gyroscopes and magnetometers, which allow measurement of various signals during a movement, including linear accelerations, angular velocities and amplitude of magnetic field. 30 The outcome measures captured from these signals – digital measures (DMs) – can then be used to reliably characterize gait.28,31–33 In MS, studies have shown that DMs derived from a smartphone at the waist level during prescribed walking tests correlate with standard clinical measures used to quantify functional impairment and overall disability.28,29,32 Data captured from smartphones during ‘free-walking’ (i.e. real-life walking with no instruction in terms of how to walk or where to wear the monitoring device), conversely, may be affected by confounding contextual factors (e.g. crowded spaces, changes in phone positioning), requiring extensive quality control. 34 The accuracy of single inertial measurement unit (IMU)-based gait measures appears to be improved when the sensors are located close to the centre of mass, 35 which is also generally well accepted by users. 36
Despite the clear potential clinical value of gait DMs as a monitoring tool in PwMS,16,37,38 there is still a lack of agreement on which gait properties to measure, and how to use the resulting data to interpret gait impairment, and detect improvement or deterioration. This is partly due to a lack of normative data and limited evidence on the value of DMs compared with standard clinical measures. To better enable such evidence to be generated, it would be valuable to increase awareness of the essential steps associated with data collection, analysis and reporting, as well as to have a common framework for DMs. Previous efforts aimed to provide consensus in digital mobility monitoring by agreeing on definitions relevant to real-world gait, such as purposeful walking and the process of turning, 39 and by summarizing the greatest challenges and barriers to standard use of digital health technology in clinical trials.40,41 However, the literature has not yet addressed the problem of providing a thorough contextual framework to measure and interpret specific alterations in gait patterns relevant to the neurological impairment of PwMS.
With the goal of fostering the adoption of smartphones as a standard digital health tool to measure gait in PwMS, this paper aims to describe a novel framework for capturing and interpreting key aspects of impaired gait. The aims were to provide standardized definitions of relevant gait domains and to identify corresponding DMs that can be extracted from the signals recorded with a smartphone worn at the waist level and to illustrate how they can be specifically used to quantify MS gait patterns.
Overview of methodological approach
The initial stage of the process was to identify the most common gait patterns associated with the presence of MS. Subsequently, a robust systematic literature review (SLR) was conducted on articles published in the last 10 years that measured one or more gait domains to determine how clinical manifestations of MS affect different domains of gait. These domains were pre-selected for being well-established within the scientific community and were reinforced by the findings from the SLR.
The outputs arising from this process were threefold: (1) a proposal for standardized terminology, (2) an evaluation of the impact of gait domains in disease and (3) the identification of a set of DMs directly relevant to gait domains with established acceptance within the scientific community.
Gait patterns in PwMS
Typical neurological impairments that affect gait in PwMS include paresis (weakness), spastic paraparesis, cerebellar, sensory and vestibular ataxia, extrapyramidal disorder and frontal lesions.3,4,42 These can lead to a wide range of gait patterns, which can be distinctive in terms of the nature of the specific impairment. For example, gait ataxia, which is generally associated with instability, may lead to a veering gait and/or wobbly stance and sometimes a slow, stomping gait, with high lifting of feet.3,43 People with spastic paraparesis may appear to drag each leg during walking, while those with frontal gait disorders are likely to have a short step length. 3 Full descriptions of these gait patterns are provided in Table 1.
Gait patterns associated with neurological impairment in multiple sclerosis.
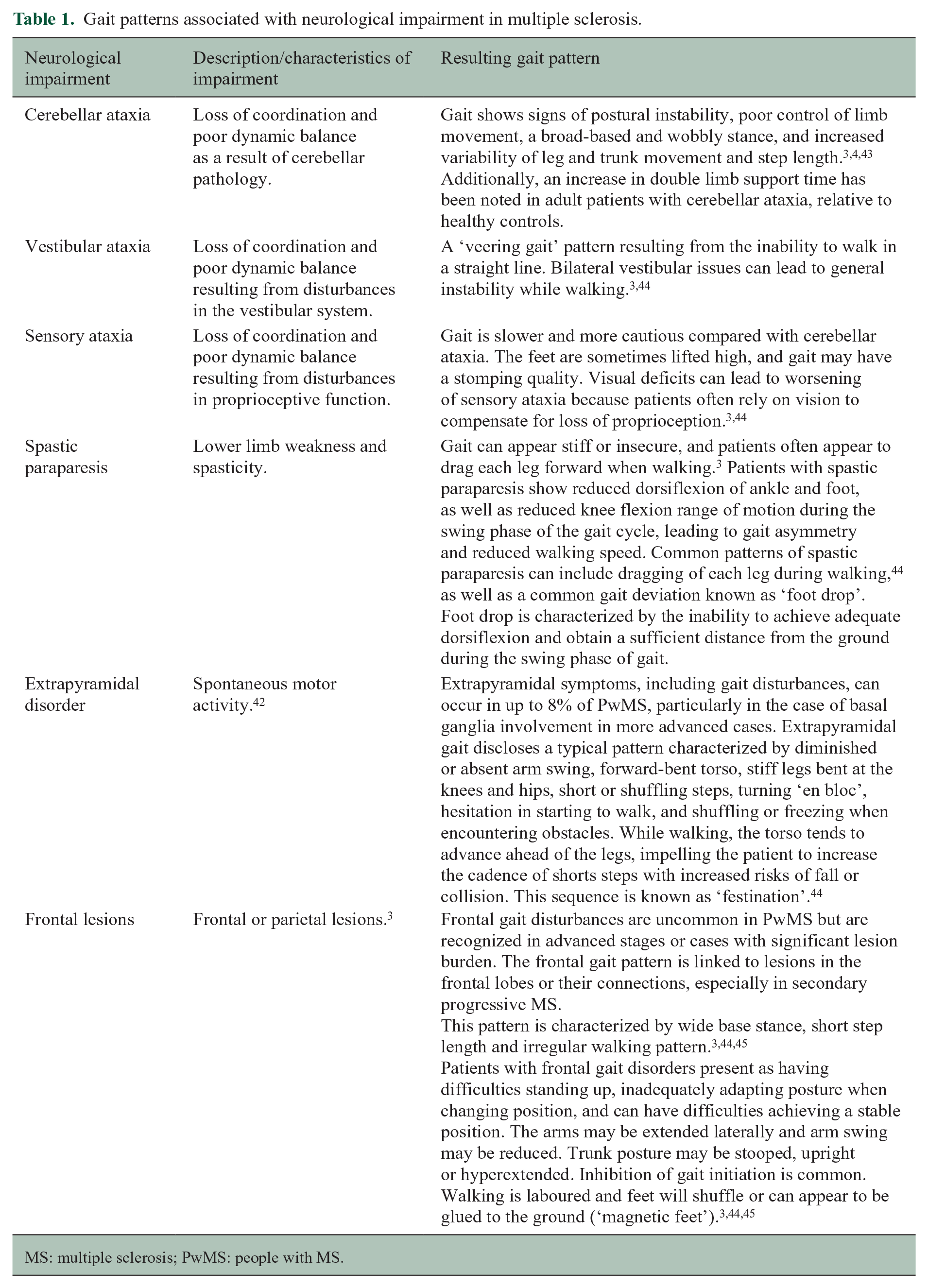
MS: multiple sclerosis; PwMS: people with MS.
From smartphone signals to gait DMs
Gait DMs can be captured from IMU signals after a series of pre-processing steps that are needed to mitigate the effects of noisy IMU measurements and transform the signal to match the expected input of the gait analysis algorithms (Figure 1). When using a smartphone, the most critical pre-processing steps (described in Supplementary Text 1) entail reducing the variability of sampling rates between different devices and calibrating the sensors to correct for inaccuracies due to their electronics or configuration (e.g. bias and misalignment). To isolate the signal content of interest, noise is filtered, and the gravity signal is removed. Finally, for certain DMs to be calculated, the signals may need to be transformed into a frame aligned with the walking direction, compensating for variations in the phone positioning on the body. 34 All these steps can critically affect the calculation of the outcomes and should be reported to allow for data comparison. After pre-processing, the next step entails detecting walking bouts (i.e. segment of the signal containing continuous walking between pauses and sharp turns) on which the gait DMs are calculated. It is important to highlight that during unsupervised testing, the reliability of DMs may be affected by the quality of the walking bout from which they are derived (e.g. whether the participants were walking along a rectilinear path or not) but also by the adherence to the measurement instructions (e.g. the measuring smartphone might be moved during the measurement to answer a phone call). Some of these issues can be automatically detected and addressed with adequate signal-processing techniques.46,47
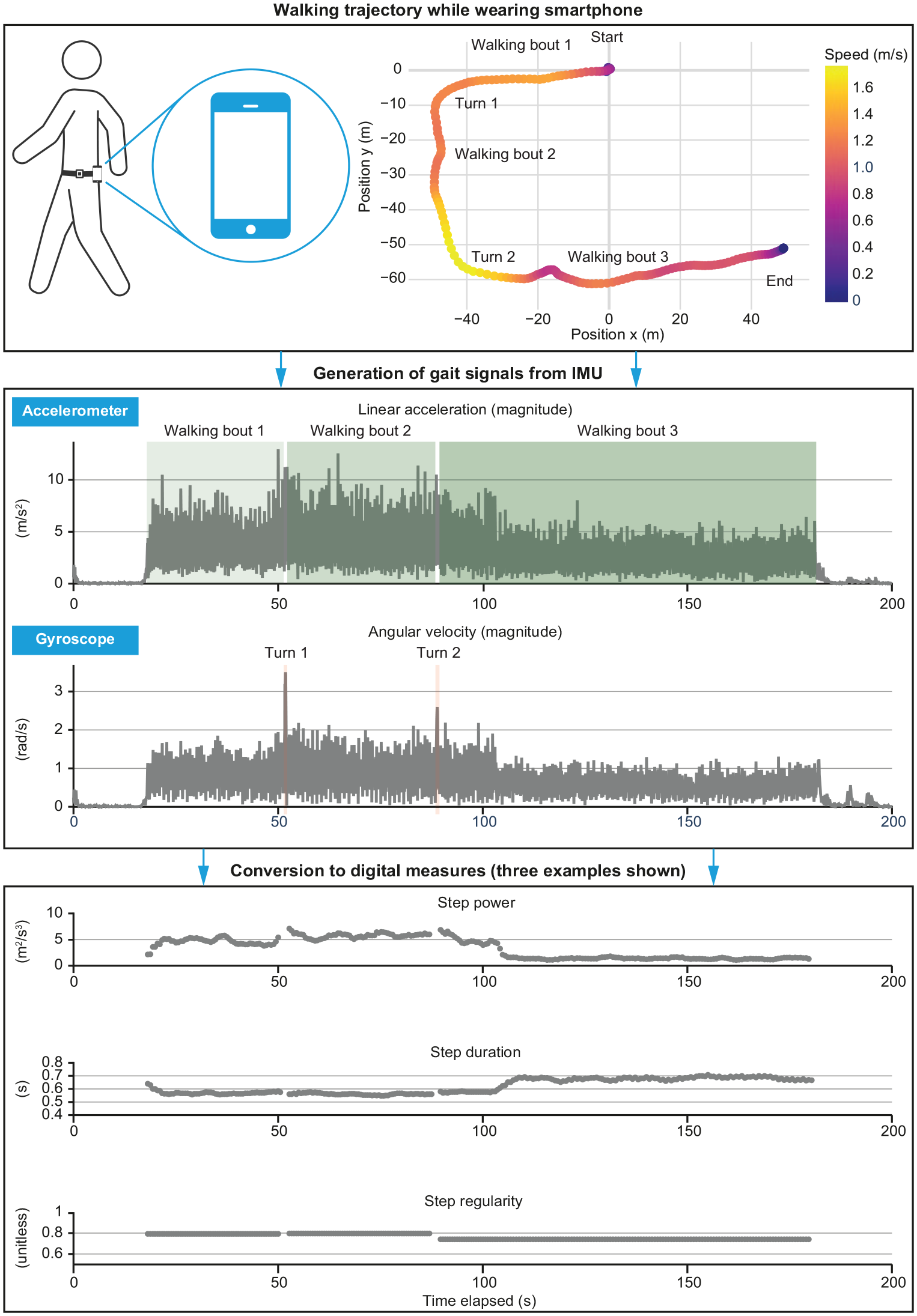
Example data processing flow from walking bout to digital measures.
Commonly adopted gait DMs range from classic spatiotemporal metrics (e.g. step/stride length, cadence, gait speed) to accelerometry-derived measures in the time and frequency domains associated with the overall quality of gait and its efficiency in terms of spent energy.16,18,19,21,22,48,49 According to their intrinsic correlation to the specific aspects of impaired gait that they intend to measure, these DMs are typically not reported in isolation but grouped into domains used to build disease-specific conceptual gait models. In recent years, such models have been proposed in MS,50,51 where domains include pace, rhythm, variability, asymmetry and forward and lateral dynamic balance. These have then been expanded to include other aspects of interest related to complexity, smoothness, endurance and fatigability when using data from longer observation, such as the 6MWT.52–54 Using this approach, it has been suggested that the variability and rhythm domains explain most of the total variance in gait performance in PwMS, with variability potentially being a clinical predictor of gait impairment and falls.2,7,55,56 This is further supported by the link between gait variability and neurological clinical presentations, such as spasticity, muscle weakness, impaired proprioception and balance, 57 motor control function, 58 a higher energetic cost of walking and fatigue. 56 The domains used in previous models, as above, were used as a basis for the SLR described in the next section.
SLR
Overview
An SLR was performed to determine the acceptance of the various gait DMs and of the associated gait domains when these are obtained from devices located at waist level (i.e. close to the centre of mass). For each domain, relevant electronic search strings were used to identify articles published between 2011 and 2021 that reported on studies using digital solutions in PwMS and containing the name of the relevant gait domain (Supplementary Tables 1a–1c). The SLR process, including date ranges and databases used, is described in Supplementary Text 2. Predefined eligibility criteria for the SLR are presented in Supplementary Table 2.
Selection of DMs
Based on the findings of the SLR, DMs of interest were selected for consideration in the proposed framework using the following inclusion criteria:
Selected DMs were required to have been described in at least one publication retrieved via the searches (except for classic spatiotemporal gait parameters, which are usually not clearly linked to a single domain, so were identified via targeted, ad hoc literature searches).
The publication describing the DM was required to contain an application in PwMS.
The digital health tool used to collect the signals from which the DMs were derived had to include a single IMU placed close to the centre of body mass.
Summary of evidence
In total, 1908 abstracts identified in the searches were screened against eligibility criteria (Supplementary Table 2). Following screening and full-text review, a few commonly used definitions of gait emerged. Specifically, 200 articles were relevant for endurance, speed, dynamic balance or symmetry of gait, 35 for complexity, smoothness or variability of gait, and 33 for cadence of gait (Supplementary Figures 1a–c).
Proposed DM framework
On the basis of findings from the SLR, we have proposed a two-part framework. Part 1 of the framework provides standardized definitions for describing key gait domains in MS (Table 2), and part 2 aims to identify specific DMs associated with each gait domain.
Proposed framework part 1: gait domains and their definitions.
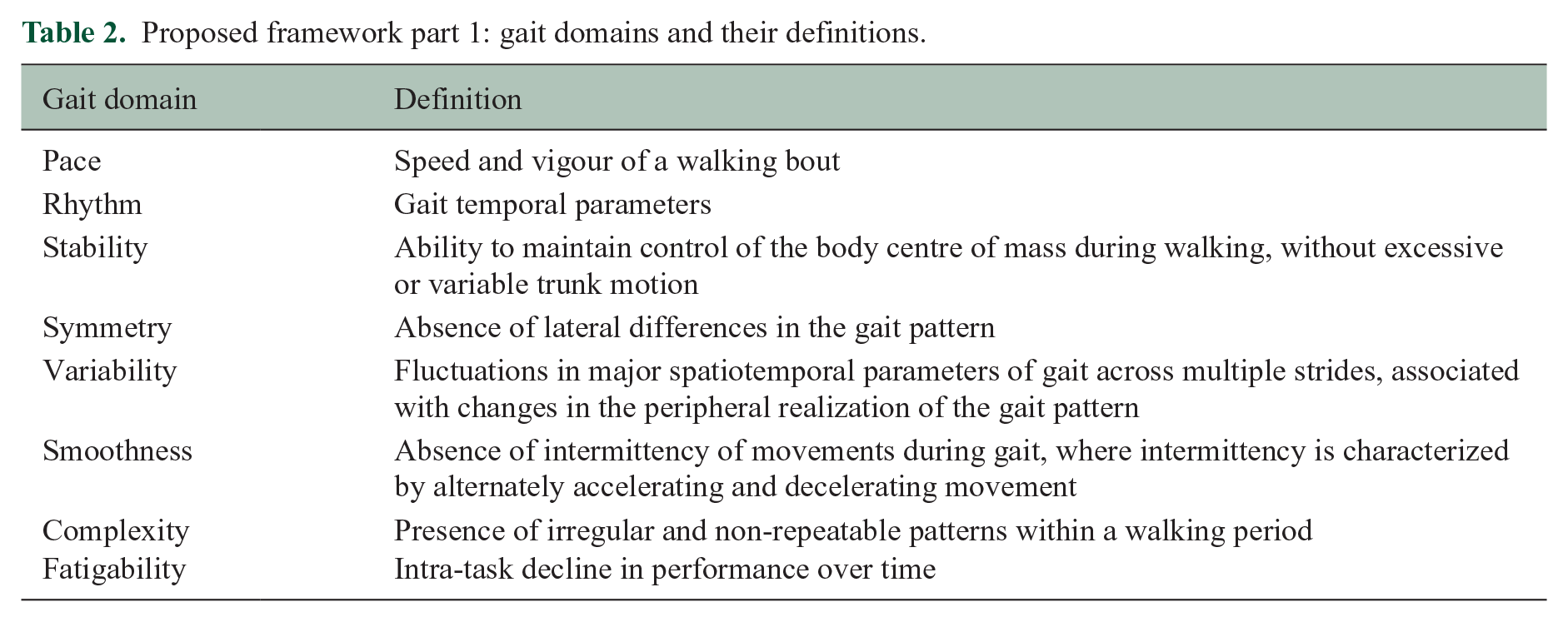
For part 1, the framework addresses pace, rhythm and stability domains that are commonly and non-quantitatively assessed during a neurological examination. 44 Performance tests such as the T25FW, 2-minute walk test (2MWT) or 6MWT are used primarily to assess the pace domain, with a time- or distance-based outcome.15,59,60 Conversely, smoothness, complexity and fatigability domains are more difficult to quantify with usual clinical examination and tests.
The findings from the literature highlighted that there is little experimental evidence on how MS affects these domains. The expected effect of MS gait impairments on selected gait domains is shown in Supplementary Table 3, together with the level of evidence that was found in the literature from each study/publication. Supplementary Table 3 shows that all types of impairment similarly affect the gait domains, suggesting that a further level of detail is needed to capture specific neurological evolution. This is expected to be provided by DMs, as described in part 2 of our proposed framework. For each gait domain, specific DMs were identified from the SLR, and the framework describes the DMs that can be used to detect impairment, explains how they are calculated and interpreted, and includes key caveats relevant to measurement and interpretation (Table 3). It is intended that multiple, complementary DMs in our framework are used to capture a fuller understanding of gait patterns, as opposed to a single DM. Note that the list has been limited to those DMs that are expected to be reliably extracted with a single IMU worn at the waist level and, as such, do not include a comprehensive set of classical spatiotemporal parameters (see Supplementary Figure 2 for an illustration of typical parameters measured during a gait cycle).
Proposed framework part 2: recommended digital measures, a by gait domain.
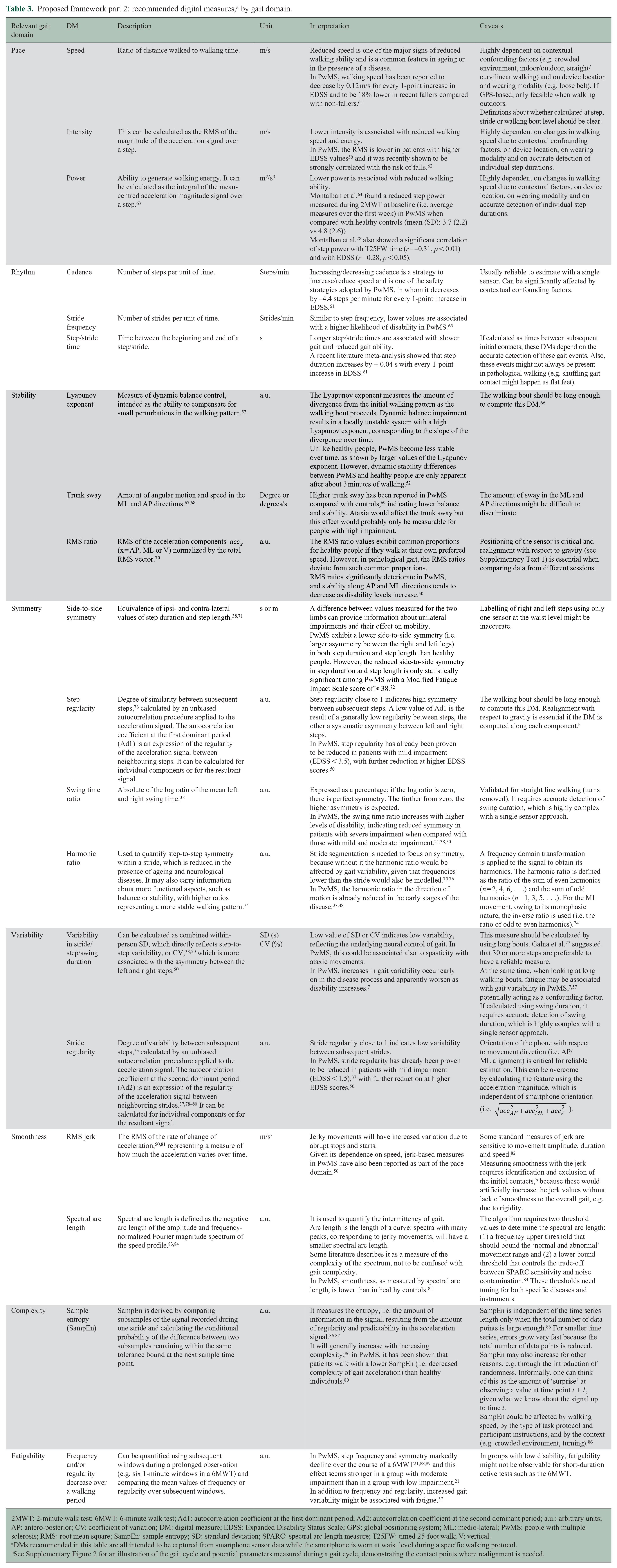
2MWT: 2-minute walk test; 6MWT: 6-minute walk test; Ad1: autocorrelation coefficient at the first dominant period; Ad2: autocorrelation coefficient at the second dominant period; a.u.: arbitrary units; AP: antero-posterior; CV: coefficient of variation; DM: digital measure; EDSS: Expanded Disability Status Scale; GPS: global positioning system; ML: medio-lateral; PwMS: people with multiple sclerosis; RMS: root mean square; SampEn: sample entropy; SD: standard deviation; SPARC: spectral arc length measure; T25FW: timed 25-foot walk; V: vertical.
DMs recommended in this table are all intended to be captured from smartphone sensor data while the smartphone is worn at waist level during a specific walking protocol.
See Supplementary Figure 2 for an illustration of the gait cycle and potential parameters measured during a gait cycle, demonstrating the contact points where realignment is needed.
Active gait testing typically involves replicating commonly adopted clinical tests (e.g. 6MWT in MS). When deployed in unsupervised remote conditions, specific recommendations emerging from the literature are associated with avoiding excessive changes in walking speed by choosing terrains and paths such that the individual can walk comfortably at a desired speed. In addition, to ensure that gait is evaluated only under the ideal and steady-state condition, the minimum duration or the minimum number of steps within such walking bouts must be taken into consideration. The definition of a walking bout, encompassing both its duration and number of steps, can be adjusted according to the DMs of interest. For example, symmetry and regularity can be reliably estimated through accelerometry-based autocorrelation analyses with a minimum of four or eight steps in a steady-state condition and a minimum of 30 or 40 steps when incorporating transitional phases of gait (e.g. initiation and termination of gait). 90
Discussion
This paper proposes a framework, developed via expert collaboration and supported by literature, which is intended to serve as a reference for those interested in using gait DMs extracted from sensor data of a smartphone device worn at the waist level during active walking tests. The framework was designed with a specific focus on assessing gait impairments in PwMS and will hopefully encourage standardization and collaboration in the field, which is needed to enable validation of DMs, consistency in clinical interpretation and comparison of data across studies. The resulting framework provides literature-based definitions for relevant gait domains as well as a set of DMs intended to capture MS-related impairment across these domains. The framework aims to promote the adoption of standard gait measures obtained via smartphone-based sensors in PwMS and drive researcher consensus on gait domain digital measurement and interpretation. Although this work is focused on measuring gait impairment in MS, similar frameworks could be developed for other neurological diseases affecting gait.
Using proposed standardized terminology, the findings from the SLR showed that different neurological impairments similarly affect the different gait domains, and as such these should be investigated simultaneously to gather a clear understanding of interactions between new and pre-existing impairments (e.g. slowly evolving lesions in the frontal region might not significantly reduce the walking speed of a patient already experiencing severe cerebellar ataxia).
The next part of establishing the framework was to identify a set of DMs directly relevant to gait domains with validity and with established acceptance within the scientific community. The use of complementary DMs, rather than focusing on a single metric, is essential to identify and monitor key clinical patterns in PwMS and to quantify various control mechanisms simultaneously utilized during gait to interact with the environment safely.
As a prerequisite for DM calculation and interpretation, we also highlighted the critical signal processing steps required to treat the signals measured by the smartphones located at waist level. This provides the necessary background and context for standardization of gait data capturing, processing and reporting. Although a wide range of solutions for quantitative assessment of gait are being explored in this field, the choice of narrowing the focus to a smartphone worn at the waist level position (rather than a multi-sensor approach for joint angle kinematic analysis, for instance) and on data captured during walking tests (as opposed to free-walking) was motivated by choosing a widely available technology and by the possibility of reducing confounding contextual factors during remote monitoring. While these might limit the number of DMs that can be reliably calculated, the integration of information coming from multiple domains is expected to meet a substantial unmet clinical need. In fact, this work defined the relationship between gait domains and MS-related gait impairment, based on findings leveraging on the proposed DMs. These relationships are consistent with results from a recent review, which showed that measurements from key gait domains can successfully discriminate between PwMS and healthy controls, and are also promising candidates in terms of convergent validity, responsiveness and ecological validity. 40 Various considerations should be noted in the context of the proposed DMs, some of which we have noted within the framework itself. For example, the main approach for measuring gait complexity is to use the sample entropy of the acceleration signal, 86 even though this is an imperfect proxy for our concept of gait complexity. We also note the potential for bias in DMs as a result of data quality issues, which might arise from differences in device hardware or their malfunctioning or from a lack of concordance with test instructions. For example, the patient may not walk consistently at the instructed speed, or they might move the belt that holds the smartphone device in a fixed location.
Several other considerations related to using smartphones for digital measurement are worth highlighting. Privacy and data security are important considerations and can be potential barriers to using mobile health technologies.91,92 Safeguards such as data encryption, use of firewalls, device configuration to allow patient-level control over data usage and regulatory oversight can all enhance data security and privacy. 93 In addition, differences in smartphone device hardware, configuration or operating system may contribute to variability in sensor data, challenges that can be mitigated with pre-processing protocols (Supplementary Text 1). Finally, using smartphones for digital measurement in free-living environments requires patients to have access to a smartphone and literacy in basic technology. Social determinants of health may impact digital health literacy and access to smartphones, which is an important consideration for integrating these technologies into research and healthcare settings. 94
Although this study has focused on smartphone-based gait measures for the longitudinal monitoring of PwMS, our approach could be extended to include other neurological diseases. This may, in turn, support the generation of further disease-specific evidence to overcome the limitations associated with the paucity of evidence linking specific measures and gait domains to individual neurological syndromes.
In conclusion, the proposed framework is expected to foster rapid advances in terms of realizing the value of digital mobility monitoring as a necessary complementary tool to standard methods of measuring disease activity in PwMS, such as the EDSS. Our framework provides recommendations for measuring complex gait patterns using widely available technology, thus encouraging standardization of outcome measurement in MS. Further development of DMs may address some of the limitations identified in our framework, and recommendations may need to be regularly updated owing to the quickly evolving landscape of digital monitoring. However, this work sets the groundwork for improving the quality of evidence in MS and, ultimately, improving patient care.
Supplemental Material
sj-docx-1-msj-10.1177_13524585251316242 – Supplemental material for Characterizing gait in people with multiple sclerosis using digital data from smartphone sensors: A proposed framework
Supplemental material, sj-docx-1-msj-10.1177_13524585251316242 for Characterizing gait in people with multiple sclerosis using digital data from smartphone sensors: A proposed framework by Angelos Karatsidis, Lorenza Angelini, Matthew Scaramozza, Emmanuel Bartholome, Susanne P Clinch, Changyu Shen, Michael Lindemann, Claudia Mazzà, Alf Scotland, Johan van Beek, Shibeshih Belachew and Licinio Craveiro in Multiple Sclerosis Journal
Footnotes
Acknowledgements
The conduct of the systematic literature review described in this article and in the ![]() was performed by Lisa Law and Gemma Carter of Oxford PharmaGenesis, Oxford, UK, and was funded by Biogen and F. Hoffmann-La Roche Ltd. Medical writing support, under the direction of the authors, was provided by Lisa Law of Oxford PharmaGenesis, Oxford, UK and funded by Biogen and F. Hoffmann-La Roche Ltd.
was performed by Lisa Law and Gemma Carter of Oxford PharmaGenesis, Oxford, UK, and was funded by Biogen and F. Hoffmann-La Roche Ltd. Medical writing support, under the direction of the authors, was provided by Lisa Law of Oxford PharmaGenesis, Oxford, UK and funded by Biogen and F. Hoffmann-La Roche Ltd.
Data Availability Statement
Data sharing not applicable to this article as no datasets were generated or analysed during the current study.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article:
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This study was funded by Biogen and F. Hoffmann-La Roche Ltd.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
