Abstract
Background:
Autologous hematopoietic stem cell transplantation (AHSCT) is a therapeutic intervention for multiple sclerosis (MS) that has gained increased attention in the last decade. The impact of this intervention on the quality of life (QoL) of patients with MS remains unclear.
Objective:
The aim of this study was to investigate the impact of AHSCT on QoL in patients with MS using Multiple Sclerosis Impact Scale (MSIS-29) scores.
Methods:
In this observational retrospective cohort study, patients with relapsing-remitting MS treated with AHSCT in Sweden from 2004, when the first transplant was performed, until 31 December 2019, were considered for participation. Anonymized outcome data were extracted from the Swedish MS registry in May 2022.
Results:
Out of 213 patients assessed for eligibility in the study, 96 were included in the final analysis. After a median follow-up of 5.2 (IQR 3.2–6.8) years, 58% improved, 14% remained unchanged and 28% worsened in the physical domain of the MSIS-29. In the psychological domain, 63% improved, 18% remained unchanged and 19% worsened. Improvements in both domains occurred early, within the first year following intervention.
Conclusions:
Treatment intervention with AHSCT is associated with a clinically meaningful improvement in QoL.
Keywords
Introduction
There is growing evidence that autologous hematopoietic stem cell transplantation (AHSCT) is a safe and effective treatment alternative for patients with active relapsing-remitting multiple sclerosis (RRMS) and it continues to be a promising alternative to traditional disease-modifying therapies (DMTs). The goal of AHSCT is to achieve an immunological ‘reset’ by ablation of the immune system. Current evidence supports the use of AHSCT in RRMS while active inflammatory processes are still ongoing.1,2 The patients most likely to tolerate and benefit from AHSCT are young, ambulatory and have inflammatory multiple sclerosis (MS) activity.3,4 In a randomized clinical trial on patients with RRMS, a prolonged time to disease progression was observed in patients treated with AHSCT in comparison to DMTs. 5 An important target of therapies in RRMS is disability improvement and the maintenance of ‘no evidence of disease activity’ (NEDA), which describes the absence of relapses, absence of new MRI lesions and no worsening in disability. 6 The proportion of patients maintaining NEDA status at 5 years following AHSCT has been reported to be 73%–79%. 7 The intervention has well-documented effect on disability, with 54%–56% of patients with RRMS showing improvement.5,8,9
MS is related to a multitude of symptoms that may be difficult to assess by a physician. Patient-reported outcome measures (PROMs) can be useful in capturing their impact on patients’ lives and have seen increased use in the past decade. Several instruments have been used to assess self-reported quality of life (QoL). The Multiple Sclerosis Impact Scale (MSIS-29) is the first validated measure of disease impact specific to MS. The questionnaire consists of 29 questions of which 20 address the physical impact component and 9 the psychological impact of MS. A combined score, ranging from 0 to 100 points, can be calculated or both aspects can be reported separately. A higher score indicates worse QoL.
The implication of AHSCT on the patient’s QoL, measured with the MSIS-29, is not fully known. 10 The objective of this study was to investigate the MSIS-29 scores in patients with MS treated with AHSCT.
Methods
Patient cohort
All individuals with a diagnosis of MS and treated with AHSCT at any of the seven Swedish transplantation centres from 2004, when the first transplant was performed, and before 1 January 2020, were screened for participation in this study. A total of 213 patients were assessed and, after considering the exclusion criteria, the final study cohort was comprised of 96 patients. The following criteria served as reasons for exclusion: diagnosis of primary progressive MS or secondary progressive MS according to Lublin et al. 11 at the time of transplantation, lack of patient consent for reporting of data to the European Society for Bone and Marrow Transplantation (EBMT) registry or not fulfilling the requirements of the minimal dataset. The minimal dataset entailed data on disease course of MS at the time of transplantation and treatment date and country of transplantation; and outcomes and dates of MSIS-29 and EDSS scoring, with a minimum of one measurement pre-and post-transplantation with each instrument.
Autologous hematopoietic stem cell transplantation
The intervention consisted of AHSCT using either BEAM-antithymocyte globulin (ATG) or Cy-ATG conditioning regimens. BEAM-ATG comprised carmustine 300 mg/m2, etoposide 800 mg/m2, cytarabine arabinoside 800 mg/m2, melphalan 140 mg/m2 and ATG from rabbit (rATG) 10 mg/kg. The Cy-ATG protocol was administered for 5 days and included cyclophosphamide 200 mg/kg and rATG 6 mg/kg. For details, see Silfverberg et al. 5
Statistical methods
Statistical analysis of data was performed using GraphPad Prism (v.10.2.0). Numerical values were tested for normality using D’Agostino’s test. Data were summarized using frequencies for categorical variables, means (± standard deviation, SD) for normally distributed variables and medians (interquartile range, IQR) for variables that were not normally distributed. Correlations were described with Spearman’s r and classified according to the British Medical Journal guidelines. 12 A multivariate regression model was constructed to determine whether age, sex, disease duration, date of transplant, EDSS scores and previous treatments influenced the MSIS-29 scores. The Chi-square test was used to establish statistical significance when comparing categorical variables. Student’s t-test was used to compare normally distributed data, and Mann–Whitney test for non-parametric data. The Wilcoxon signed-rank test was used to establish statistical significance (p value < 0.05) between two time points.
Based on current literature, the threshold for a clinically meaningful change was set as a change in 8 points in the physical domain and 6 points in the psychological domain.13,14
Assessments were analysed at baseline and at years 1, 2 and 3 (±180 days) following the intervention, as well as at the latest evaluation. 15
Results
Study cohort
Data were exported from the Swedish MS registry (SMSreg) on 22 May 2022. Of the 213 considered for the study, 41 were excluded due to diagnosis other than RRMS or due to treatment outside of Sweden. A further 76 were excluded prior to the final analysis because they did not meet the criterion of at least one MSIS-29 and one EDSS measurement before and after AHSCT (Figure 1). These 76 constitute the missing data group. The final study cohort was comprised of 96 patients, 63 females and 33 males. The median age was 31 (IQR 27–36) years, with the youngest patient being 20 years and the oldest 51 years old (Table 1). The median disease duration before performing AHSCT was 4 (IQR 2.0–6.8) years. The median number of MSIS-29 assessments was 7 (IQR 5.0–10) and the median number of follow-up years was 5.2 (IQR 3.2–6.8). A total of 1118 MSIS-29 scores were analysed.
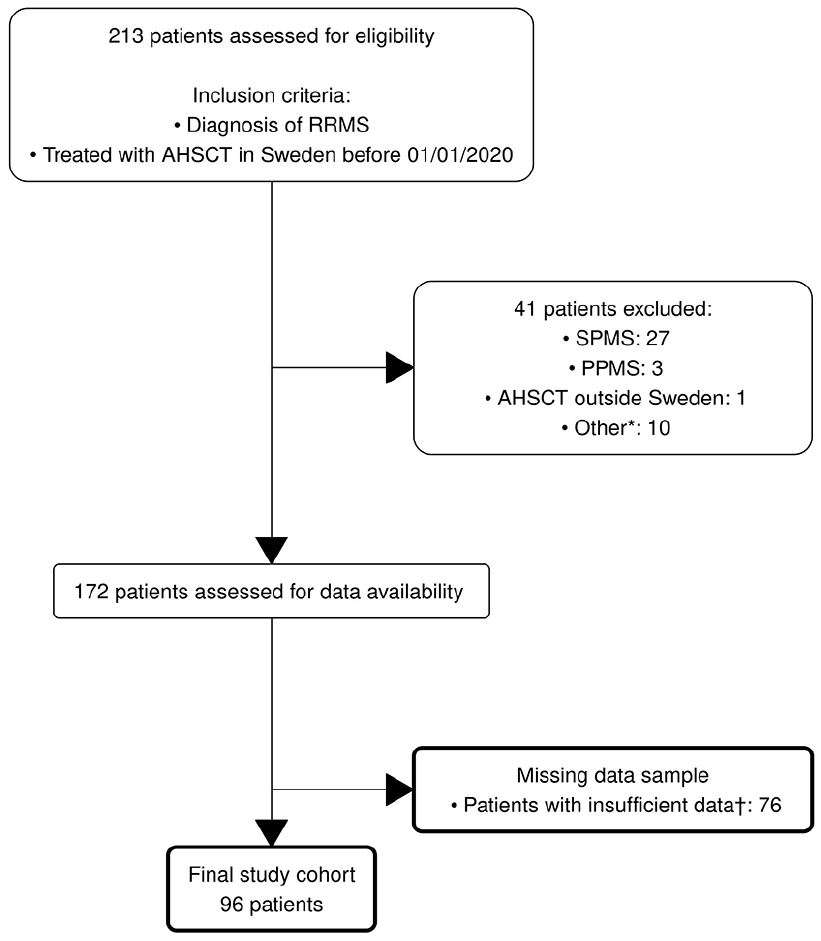
Flowchart diagram of patient inclusion and exclusion in the study.
Patient characteristics and details.
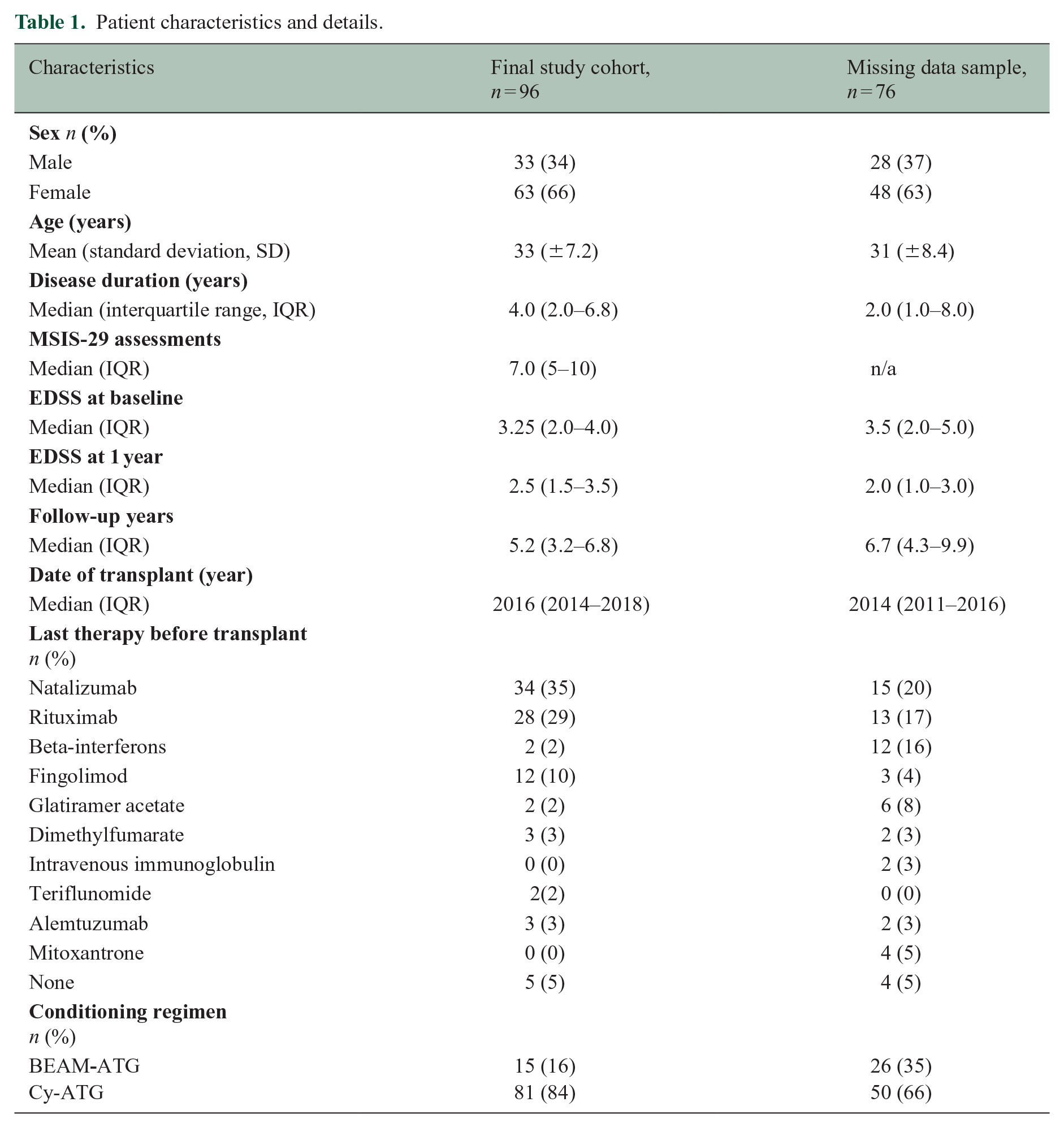
A higher degree of patients had received BEAM as a conditioning regimen in the group with the missing data. In the final study cohort, 16% had received BEAM, in contrast to 35% in the missing data group (Table 1).
Prior treatments
The median number of MS treatments prior to AHSCT was 2 (IQR 2–4). These included natalizumab (n = 63), rituximab (n = 41), beta-interferons (n = 60), intravenous immunoglobulins (n = 8), alemtuzumab (n = 3), glatiramer acetate (n = 16), teriflunomide (n = 4), fingolimod (n = 28), mitoxantrone (n = 2) and dimethylfumarate (n = 10). Some patients had received multiple therapies prior to their transplant. The most common last therapy administered prior to AHSCT was natalizumab (n = 34, n = 15). Only four patients had not used any DMT prior to AHSCT.
Analysis of the missing data sample
The main difference between the final study cohort and the group of patients with missing data was the time point for the intervention, which was considerably later in the final study cohort, median 2016 (IQR 2014–2018) versus 2014 (2011–2016). This was also reflected in the higher proportion of patients who had used high-efficacy DMTs in the final study cohort (natalizumab, 35% vs 20%, and rituximab, 29% vs 17%) and also in the proportion of patients receiving the Cy/ATG conditioning regimen (84% vs 65%).
A significant difference was observed in change in EDSS outcomes score at year 1. The missing data group experienced greater improvement than the final study cohort. The median change in EDSS score from baseline to year 1 in the final study cohort was −0.5 (n = 91, p < 0.0001) and −1.5 (n = 62, p < 0.0001) in the missing data group.
The physical and the psychological domain of the MSIS-29 correlated well with each other
The physical impact score correlated well with the psychological score at baseline (r = 0.76, p < 0.0001). Similarly, there was a strong correlation between the two at each follow-up: year 1 (r = 0.70, p < 0.0001), year 2 (r = 0.77, p < 0.0001), year 3 (r = 0.73, p < 0.0001) and at the latest assessment (r = 0.80, p < 0.0001).
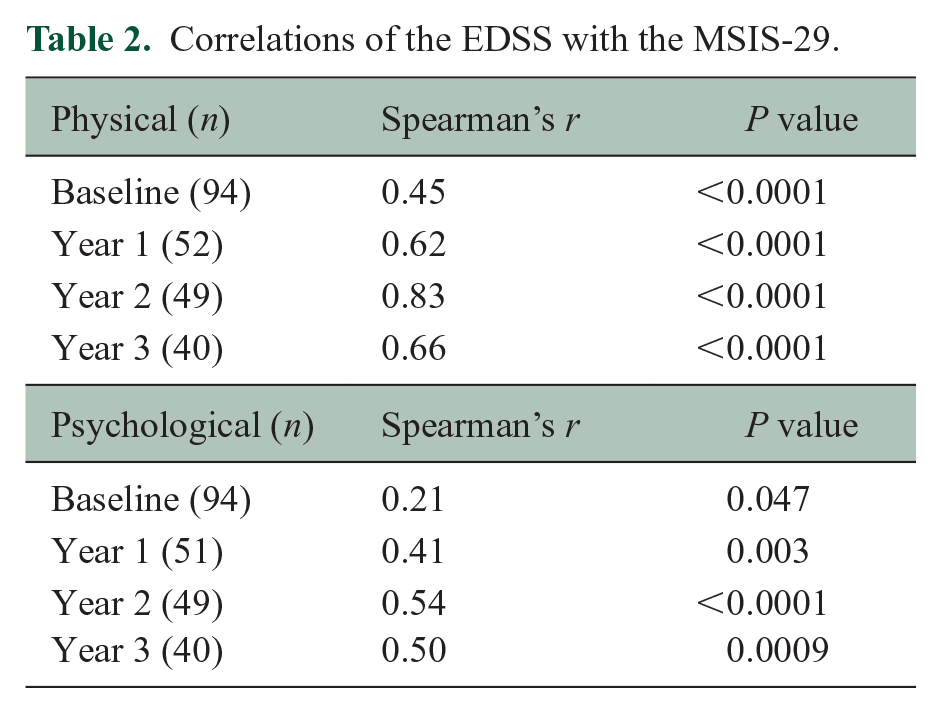
The EDSS correlated more strongly with the physical component of the MSIS-29
At baseline, the EDSS correlated moderately with the physical domain of MSIS-29, but less so with the psychological domain. At year 1, the EDSS correlated strongly with the physical component of MSIS-29, but only moderately with the psychological component. At year 2, the EDSS correlated very strongly with the physical component of MSIS-29, but only moderately with the psychological component. At year 3, the EDSS correlated strongly with the physical component of MSIS-29, but only moderately with the psychological component.
The MSIS-29 increased before intervention with AHSCT
Prior to transplant, the MSIS-29 scores increased in both domains, with a rate of 1.6 points per year in the physical and 1.9 points per year in the psychological domain (Figure 2). The mean duration until a clinically meaningful worsening occurred, following the first evaluation, was 5 years in the physical and 3 years in the psychological component. Following AHSCT, scores decreased in both domains, with a rate of 0.8 points per year in the physical and 1.6 points per year in the psychological domain.
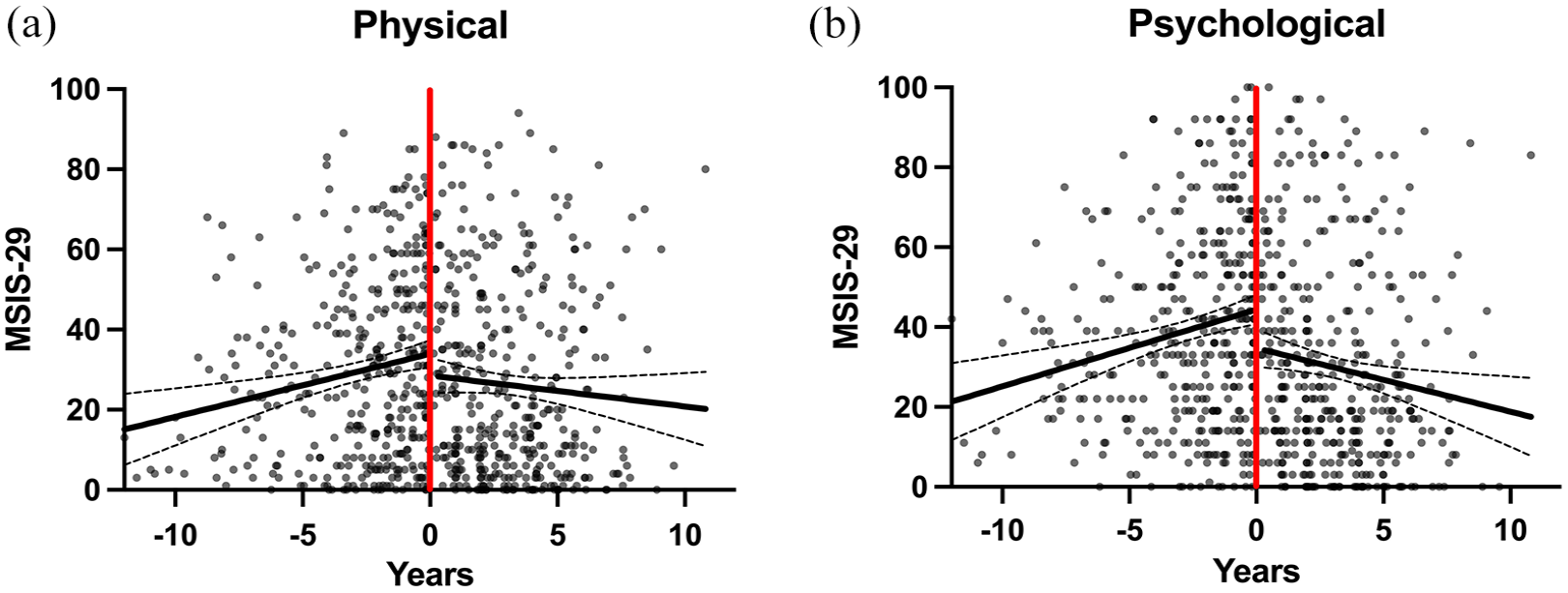
The evolution of the MSIS-29 scores over time.
The MSIS-29 score decreased after AHSCT for MS
An initial analysis of the change in physical and psychological components of the MSIS-29 over time was performed by comparing the score at baseline with the latest score (Figure 3). The median time point for the last follow-up was 4 (IQR 3–6) years from baseline. The median physical score at baseline was 34 (IQR 16–59) and at the last follow-up 13 (IQR 4–44) (p < 0.0001). The median psychological score at baseline was 42 (IQR 22–67) and at the last follow-up 19 (IQR 8–43) (p < 0.0001) (Table 2).
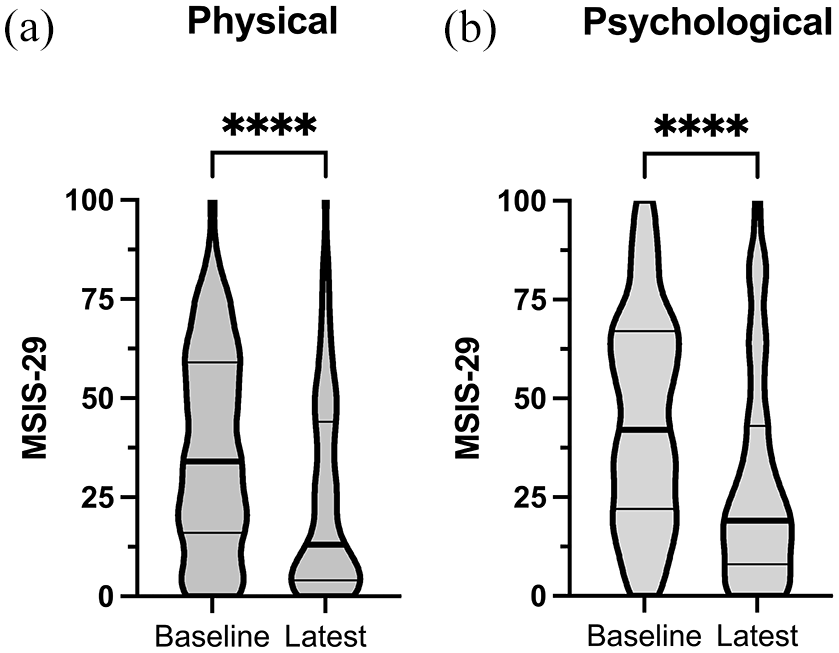
Comparison of absolute scores between baseline and latest assessment.
Correlations of the EDSS with the MSIS-29.
A majority of patients reported a clinically meaningful improvement in the physical as well as the psychological component of the MSIS-29.
Using the set definitions of the minimum clinically meaningful change, 56 patients (58%) had improved, 13 patients (14 %) remained unchanged and 27 patients (28%) had worsened at the latest assessment in the physical component of the MSIS-29. Similarly, in the psychological component, 60 patients (63%) had improved, 17 patients (18%) remained unchanged and 18 patients (19%) had worsened.
The MSIS-29 score decreased early after AHSCT
A clinically meaningful change in MSIS-29 score occurred early, within the first year following transplant. This holds true for both the physical and the psychological components.
The median physical score was 34 (IQR 16–59) at baseline, 15 (IQR 6–42) at year 1, 19 (IQR 4–48) at year 2 and 13 (IQR 3–33) at year 3 (Table 3). A statistically significant difference of −19 points was observed between baseline and year 1 in the physical domain (n = 56, p < 0.0001). There was no statistically significant difference between years 1 and 2 (n = 56, p = 0.9), or years 2 and 3 (n = 43, p = 0.4). There were fewer pairs when comparing years 1 and 2 (n = 56), as well as years 2 and 3 (n = 43). This is because assessments at these time points were not reported in all patients, as the available data varied from patient to patient.
Absolute MSIS-29 scores.
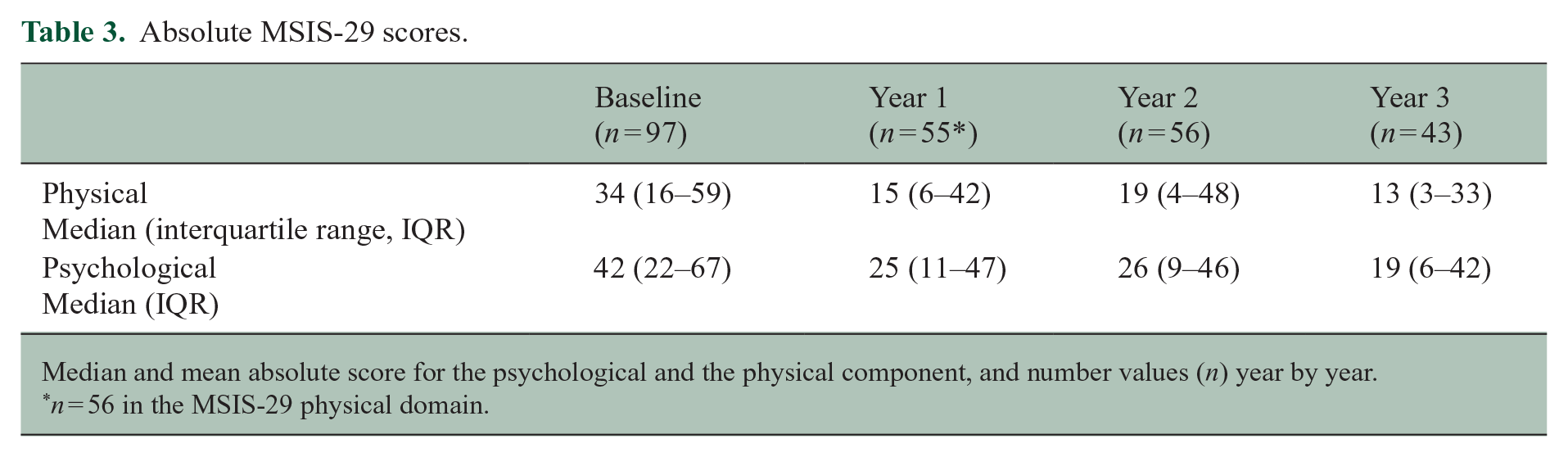
Median and mean absolute score for the psychological and the physical component, and number values (n) year by year.
n = 56 in the MSIS-29 physical domain.
The median psychological score was 42 (IQR 22–67) at baseline, 25 (IQR 11–47) at year 1 and 26 (IQR 9–46) at year 2 and 19 (IQR 6–42) at year 3. A statistically significant change of −17 points was observed between baseline and year 1 in the psychological domain (n = 55, p < 0.0001). There was no statistically significant difference between years 1 and 2 (n = 38, p = 0.4) and years 2 and 3 (n = 26, p = 0.4).
Patients with NEDA reported higher QoL
At baseline, patients maintaining NEDA throughout follow-up reported a lower MSIS-29 median physical score of 30 (IQR 15–46) than patients with EDA who reported a median physical score of 57 (IQR 27–68) (p = 0.003) (Figure 4). Similarly, at baseline in the psychological domain, patients maintaining NEDA reported a lower MSIS-29 median score of 36 (IQR 20–63) than patients with EDA reporting a median score of 59 (IQR 29–73) (p = 0.045). At year 1, patients maintaining NEDA reported a median physical score of 11 (IQR 5–29) and patients with EDA had a median physical score of 52 (IQR 29–63) (p < 0.0001). At year 1 in the psychological domain, patients maintaining NEDA reported a median score of 22 (IQR 7–39), and patients with EDA had a median score of 40 (IQR 25–53) (p = 0.02). A clinically meaningful improvement in the physical domain of MSIS-29 occurred at the 1-year follow-up in the median patient maintaining NEDA and not until the 2-year follow-up in patients exhibiting EDA. In the psychological domain, a clinically meaningful improvement occurred at the 1-year follow-up, irrespective of EDA/NEDA status.
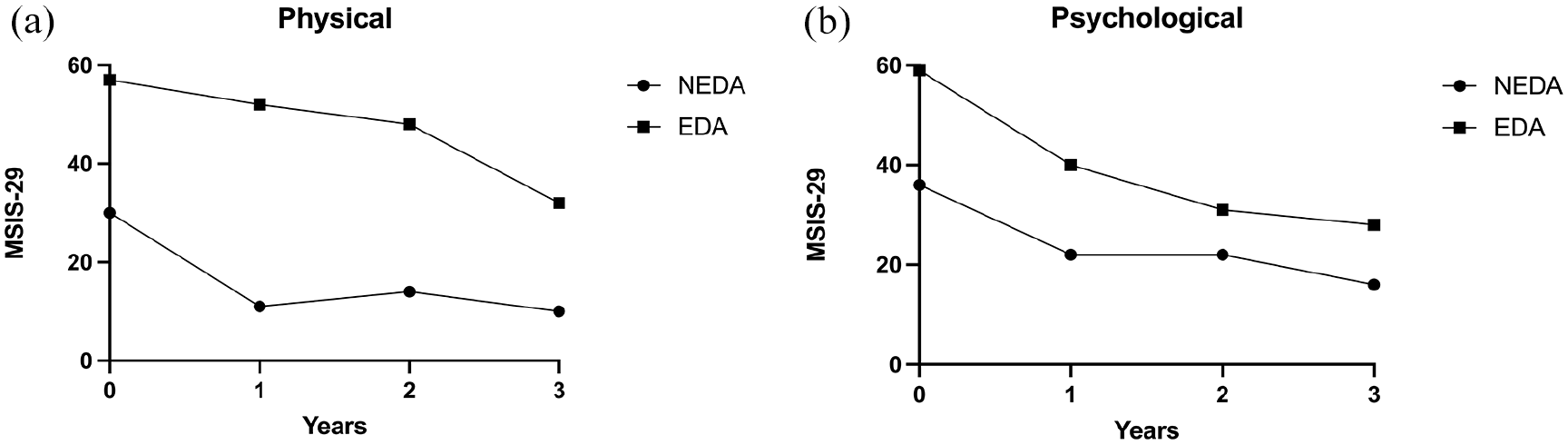
Physical/psychological absolute scores and NEDA/EDA status.
Discussion
A key finding of this study is that a majority of patients experienced an improvement in QoL following AHSCT. The greatest improvement occurred within the first year after transplant and was maintained throughout follow-up. Patients who maintained NEDA throughout the follow-up period reported lower MSIS-29 scores than those with EDA. Patients with EDA also benefitted from AHSCT, but, on average, it took longer until a clinically meaningful improvement occurred in the physical domain. These findings contribute to our understanding of how AHSCT impacts QoL.
QoL is a useful primary endpoint for interventions, as it is a multi-dimensional construct, affected both by disease-related factors and by individual factors such as perceived self-efficacy. 16 In recent years, the use of PROMs has increased in the field of MS and this has helped broaden the evaluation and definition of successful treatments. 17 These tools enable the recording of health-related data from patients, with the report coming directly from the patient. One advantage of using PROMs when evaluating disability and the well-being of patients with MS is that the questionnaires can be completed from their homes and potentially decrease the need for follow-up visits. There are, however, several factors that can influence the reliability of PROMs and that require further understanding, such as the patient’s perception of well-being which may change over time and the influence of the placebo effect. 18
The heterogeneous nature of symptoms in MS constitutes a challenge when developing PROMs. There are outcome measures that focus on specific domains and others, such as the MSIS-29, that are multi-domain measures. Unlike other instruments commonly used to evaluate QoL, such as the EQ-5D and the 36-item Short Form Health Survey (SF-36), the MSIS-29 is a disease-specific measuring tool.19,20 Beyond determining overall QoL, the MSIS-29 also measures disease-specific outcomes such as bladder dysfunction, participation in daily activities and fatigue. This enables the monitoring of multiple symptoms using a single instrument. 21 In comparison to other instruments, the physical domain of the MSIS-29 has been found to have better sensitivity to discriminate between disability groups. 22 In a British comparative study, the MSIS-29 was found to be the most responsive physical and second most responsive psychological scale. 23 An advantage with the MSIS-29 is that there exists a generally accepted minimally clinically important difference (MCID), that can be used to estimate treatment effects.13–15 In contrast, no MCID has been established for the SF-36 in patients with MS, and instead, the value of half of a standard deviation is often used. This might make it less suitable to measure changes considered clinically meaningful to patients with MS. 24 In 2022, a patient-centred standard outcome set for MS was developed, recommending the use of the MSIS-29. 25
Several studies have examined the impact of high-efficacy DMTs on QoL in patients with MS. 7 A decrease in MSIS-29 scores was observed in patients treated with natalizumab and rituximab, but the changes were relatively minor and not clinically meaningful for the average patient.5,26–28 In contrast, a clinically meaningful improvement in both domains of the MSIS-29 was seen with AHSCT in the present study. Absolute scores in both components of the MSIS-29 were decreased already at the first follow-up visit and continued to remain stable. In one previous study of 9 Italian patients with severe progressive MS, improvements in MSQOL-54 by 24 months were reported in the physical health composite as well as the mental health composite. 29 In a mixed cohort (mainly SPMS) of 34 Brazilian patients and in a report from Lithuania of 18 patients with unknown disease course, SF-36 improved at 12 months.30,31 In a larger single-centre study of a mixed cohort (mainly RRMS) of 132 patients, the total SF-36 score improved from a median of 45–64, 2 years after AHSCT. 32 These findings further support AHSCT as a viable treatment option for MS.
It is currently debated whether maintenance of NEDA is prognostic for disability in MS, and therefore we wanted to explore whether patients who maintained NEDA reported higher QoL. Somewhat unexpectedly, we found that patients with EDA during follow-up had higher MSIS-29 scores in both domains already at baseline. This probably reflects that patients with high MSIS-29 scores also have more disability and that patients with disability tend to do worse in terms of outcome after AHSCT. During follow-up, the MSIS-29 scores decreased in both domains, regardless of EDA/NEDA status, albeit slightly slower in the physical domain in patients with EDA.
This observational study is limited by the absence of a control group and therefore does not account for the potential influence of regression to the mean and the patients’ hopes and expectations. 33 There was also a fairly large proportion of patients who did not report any MSIS-29 scores – the missing data group. This was mainly due to the limited use of the MSIS-29 before 2013. The missing data group had a larger improvement in EDSS than the patients included in the study, suggesting that our study may have underestimated the effect of AHSCT on the MSIS-29 in the entire cohort. As a result of the variable follow-up of QoL, the number of reported MSIS-29 assessments varied among patients. Finally, in Sweden, AHSCT is approved by the Swedish Board of Health and Welfare for use in aggressive RRMS, whereas SPMS and PPMS are not endorsed. Therefore, our analysis was restricted to RRMS patients and the findings cannot be generalized to people with progressive forms of MS. Some notable strengths of this study are the notably high coverage of MS cases in the SMSreg, the high data density, as well as the relatively large cohort and long follow-up.
Conclusions
In this cohort study of patients with relapsing-remitting MS undergoing AHSCT, the treatment intervention was associated with a clinically meaningful improvement in QoL. This finding is significant as no current DMT has conclusively demonstrated a clinically meaningful long-term impact on QoL in patients with MS. Despite often being considered a last-resort treatment, offered only when other alternatives have failed, our findings support a more widespread use of AHSCT. This study underscores the potential of AHSCT as an effective treatment option for improving the QoL in patients with MS.
Footnotes
Acknowledgements
The authors thank all the physicians who contributed with follow-up data to this study and in particular the AutoMS-Swe investigators: Nahimi Adjmal, Erik Ahlstrand, Honar Cherif, Arta Dreimane, Sigrun Einarsdottir, Jan Fagius, Hans Hägglund, Ellen Iacobaeus, Niclas Lange, Stig Lenhoff, Per Ljungman, Jan Lycke, Johan Mellergård, Fredrik Piehl, Tomas Silfverberg, Anders Svenningsson and Andreas Tolf.
Author Contributions
A.C.M.D., T.S. and J.B. planned the study. J.B. provided funding. K.C. was part of the team that transplanted the patients. A.C.M.D. wrote the draft. Statistical analyses were performed by A.C.M.D. Graphical representations were made by A.C.M.D. and Y.N. JB provided supervision. All authors provided creative input and approved of the final version.
Data Availability Statement
The study is registered at ClinicalTrials.gov with identification number NCT05029206. De-identified individual participant data supporting the findings presented in this article, including text, tables, and figures, will be accessible alongside the study protocol for a period of 5 years, starting at the time of the article’s publication. Researchers with a sound proposal without commercial purposes can request access to this data by contacting
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: The study was funded by the Centre for Clinical Research Dalarna with grant numbers CKFUU-936620 and CKFUU-976388, the Marianne and Marcus Wallenberg Foundation, the Swedish Research Council (grant no. 2021-02814), the Swedish Society for Medical Research (grant no. 2020-02700), the Swedish Society of Medicine and Uppsala-Örebro Regional Research Council (grant no. RFR-967904).
Ethical Approval
The study received ethical approval from the Ethical Review Authority in Sweden on April 14, 2021 (reference number: 2021-01530). All participants provided written consent for their registry data to be used for research purposes.
