Abstract
Background:
Many common symptoms in post-acute sequelae following severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection (PASC) overlap with those of multiple sclerosis (MS). We examined symptoms and performance of the PASC score, developed in the general population, in MS based on infection history.
Methods:
We surveyed North American Research Committee on Multiple Sclerosis (NARCOMS) registry participants regarding infections and categorized participants based on infection history. Symptoms experienced before, during, and after infection were used to identify persistent new symptoms. PASC was defined as a score ⩾ 12 based on the National Institutes of Health (NIH) study RECOVER.
Results:
Of 4787 participants surveyed, 2927 were included: 294 (10%) having recent COVID-19; 853 (29.1%) recent non-COVID-19 infection; 246 (8.4%) recent COVID-19 and non-COVID-19 infection; 1534 (52.4%) uninfected, defined as never having COVID-19 nor any infection within the past 6 months. Compared to those uninfected, infection groups reported at least a two-fold increase in fever, cough, loss of smell/taste, and shortness of breath. Based on persistent new symptoms, PASC was identified in only 1.5% of participants with COVID-19.
Conclusion:
Our study suggests lower than expected prevalence of PASC in MS and a complex association between infections and development of new persistent symptoms following infections. The similar proportions classified with PASC across infection groups shows that symptoms of PASC are common and complicate assessment of PASC in MS.
Introduction
The COVID-19 pandemic has affected millions, including those living with multiple sclerosis (MS). Some individuals experience long-term symptoms, so-called post-acute sequelae of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection (PASC), long COVID or post-COVID-19 condition. This condition is poorly understood in the general and MS population. The World Health Organization defines post-COVID-19 condition as “the illness that occurs in people who have a history of probable or confirmed SARS-CoV-2 infection; usually within three months from the onset of COVID-19, with symptoms and effects that last for at least two months . . . not explained by an alternative diagnosis.” 1
Common persistent symptoms of PASC are fatigue, post-exertional malaise, and brain fog. 2 Cough, chest pain, shortness of breath (SOB), and muscle aches may also occur. Autonomic dysfunction can also be a sequela of COVID-19 infection and may account for some of the symptoms of PASC in some individuals. 3 Many of these sequelae can be classified as neurologic and grouped into broad categories including cognition, mood and sleep disorders, exercise intolerance, and dysautonomia. Recently, the NIH RECOVER initiative defined criteria for identifying PASC based on a composite symptom score using self-reported symptoms in a cohort of participants from the general population. 4 Based on this data-driven scoring, they identified 10%–20% of infected participants with PASC and approximately 4% in those uninfected participants.
Although development of chronic symptoms following infection has been recognized previously, this issue received little attention until the recent pandemic. 5 Assessment of these post-acute infection syndromes, including PASC, may be complicated by several factors, especially in MS. First, many common PASC symptoms are nonspecific and overlap with MS symptoms, such as fatigue, impaired memory and concentration, sleep problems, and depression or anxiety. 1 Second, autonomic dysfunction is prevalent in MS, yet frequently under-recognized. 6 Thus, autonomic symptoms attributable to PASC may overlap with those from MS. Third, MS individuals have an increased risk for infections 7 ; so, it is unclear what post-infectious symptoms might be sequelae from infections in general or specifically attributable to SARS-CoV-2. To better understand post-infectious symptoms in MS, we aimed to describe persistent symptoms following COVID-19 and other infections compared to those who had no infections and characterize the performance of the PASC score in those with MS.
Methods
Study design and data source
We used cross-sectional data from the North American Research Committee on Multiple Sclerosis (NARCOMS) Registry. The NARCOMS Registry is a self-report registry for people with MS, who complete questionnaires at enrollment and semi-annually thereafter to update their health-related information. 8 At the time of this study, the registry and questionnaire were approved by the Institutional Review Board at UT Southwestern.
Infections
The spring 2023 survey, administered April–June 2023, included questions regarding infections that occurred between November 2022 and March 2023. Participants reported if they had any of the following infections in the past 6 months: influenza, common cold, gastrointestinal tract infection, pneumonia, zoster (shingles), strep throat, urinary tract infection, skin infection, osteomyelitis, and joint infection. In addition, participants reported if they had ever had a SARS-CoV-2 infection; if yes, they reported the month and year of their first and most recent infections and if the infection was confirmed using an at-home or laboratory test.
Symptoms
Participants were surveyed about the symptoms experienced in relation to infections and autonomic dysfunction. Symptom questions were harmonized with the NIH RECOVER study. 9 Participants reported the presence and timing of 29 different symptoms (see Supplemental eTable 1) by indicating if any of the following were true for each symptom: symptoms were never present (“No, I have NOT had this symptom”), present 1 year before the infection (“Yes, I DID have it in the YEAR BEFORE infection”), around the time of the infection (“Yes, I DID have it AROUND the time of infection”), and currently (“Yes, I have it NOW”). If the participant had more than one infection (COVID-19 or non-COVID-19 infection), they were instructed to use the most recent infection for symptoms they had experienced.
Using responses to these questions, we defined new persistent symptoms as those symptoms that were not present pre-infection but present at the time of infection and still present at the time of survey completion. Participants not reporting any infection, including COVID-19, were asked to answer symptom questions, too. Autonomic dysfunction was assessed using the Composite Autonomic Symptom Scale-31 (COMPASS-31) 10 questionnaire, which evaluates six domains of autonomic dysfunction, including orthostatic intolerance, vasomotor, secretomotor, gastrointestinal, bladder, and pupillomotor. 10 The COMPASS-31 generates a weighted total score ranging from 0 to 100 where higher scores indicated worse symptoms of autonomic dysfunction. Moderate or worse symptoms of autonomic dysfunction was considered as a COMPASS-31 total score ≥ 20. 10
Following the RECOVER scoring, 4 we computed the PASC score for symptoms experienced at the time of the survey by summing the number of points assigned to each symptom (loss of smell/taste (8 points), post-exertional malaise (7 points), chronic cough (4 points), brain fog (3 points), thirst (3 points), palpitations (2 points), chest pain (2 points), fatigue (1 point), sexual desire or capacity (1 point), dizziness (1 point) gastrointestinal (1 point), abnormal movements (1 point), hair loss (1 point)) and classified participants with scores ⩾ 12 as having PASC. New persistent symptoms only were scored similarly. The specific symptom of chest pain was not collected due to space limitations in the survey, so the score was calculated by substituting responses to the general pain question.
Sociodemographic and clinical characteristics
The NARCOMS enrollment questionnaire provided information regarding age, race, ethnicity, education level, gender, and age of MS symptom onset (from which we derived disease duration). The spring 2023 questionnaire provided information regarding annual household income, disability severity (measured using the Patient Determined Disease Steps, PDDS) and current clinical course. We categorized race as White, African American/Black, and other; the number of participants reporting Hispanic ethnicity was too small for analysis (< 50). Education level was categorized as high school/General Educational Development Test, and post-secondary (Post-graduate education, Associate’s, Bachelor’s, and Technical degree). Annual household income was categorized as ⩽US$50,000, >US$50,000 and “I do not wish to answer.” PDDS, a single item measure with eight response levels ranging from 0 (normal) to 8 (bedridden), was used to measure disability.11,12 This self-report measure correlates strongly with the physician-scored Expanded Disability Status Scale.13,14 Disease modifying therapy use was categorized as anti-CD20s, S1P modulators, other, and none/not answered based on their previous association with severity of COVID-19 infection. 15
Statistical analysis
We excluded survey responders if they did not have a confirmed MS diagnosis, were living outside the United States, or did not respond to the COVID-19 infection question. We divided respondents into four groups; COVID-19 infection in the last 6 months; one infection other than COVID-19 in the last 6 months; COVID-19 infection plus one infection other than COVID-19 in the last 6 months; and no infection (no COVID-19 ever or other infection in the past 6 months). We also divided participants who reported a COVID-19 infection into past (> 6 months) and recent (< 6 months) groups based on their most recent COVID-19 date (month and year). We evaluated differences in new persistent symptoms based on time from COVID-19 infection using chi-square or Fisher’s exact test, as appropriate. Participants who were at least 3 months from COVID-19 infection were evaluated for PASC based on the RECOVER threshold (⩾ 12). 4 Group differences were assessed between eligible participants who met or did not meet the PASC threshold based on new persistent symptoms using chi-square or Fisher’s exact test.
Participant characteristics were summarized using mean (standard deviation [SD]), median (25th, 75th percentile) and frequency (percent). Differences in the prevalence of current symptoms between infection groups were examined using multivariable logistic regression adjusted for age, gender, race, education, income, disease duration, and PDDS and reported the adjusted odds ratio (aOR) and 95% confidence interval (95% CI).
Results
Participants
The spring 2023 survey invitations were distributed to 7782 participants of whom 5239 (67.3%) responded. As compared to responders, non-responders were 1 year younger on average, had a lower level of education, less likely to self-identify as White, and had a higher level of disability at enrollment (see Supplemental eTable 2). After application of inclusion criteria, 4787 were eligible for analysis (see Supplemental eFigure 1).
Ever having COVID-19 was reported by 2116 (44.2%) participants, of whom 90.4% reported a confirmed COVID-19 diagnosis. Of the 2116 participants who reported a COVID-19 infection, 210 (10%) reported a recent infection within the last 3 months, 473 (22.4%) 3–6 months prior, 669 (31.6%) within the prior year, and 544 (25.7%) ⩾ 2 years before the spring 2023 survey. Participants who reported COVID-19 infections within the last 0–6 months were more likely to be older, have higher education, shorter disease duration, and higher PDDS (see Supplemental eTable 3).
Of the 2927 participants included in the analysis, 294 (10%) had a recent COVID-19 infection (< 6 months), 853 (29.1%) had one non-COVID-19 infection in the last 6 months, 246 (8.4%) had a COVID-19 infection and one non-COVID-19 infection in the last 6 months, and 1534 (52.4%) had no infection with COVID-19 ever nor a non-COVID-19 infection within the past 6 months (uninfected). The most common other non-COVID-19 infections were cold (38.3%), urinary tract infection (38.1%), pneumonia (10.1%), gastrointestinal (7.4%), and skin (5.7%) infection. Differences in demographic and clinical characteristics between groups were generally small (see Table 1).
Demographic and clinical characteristics of participants by infection group.
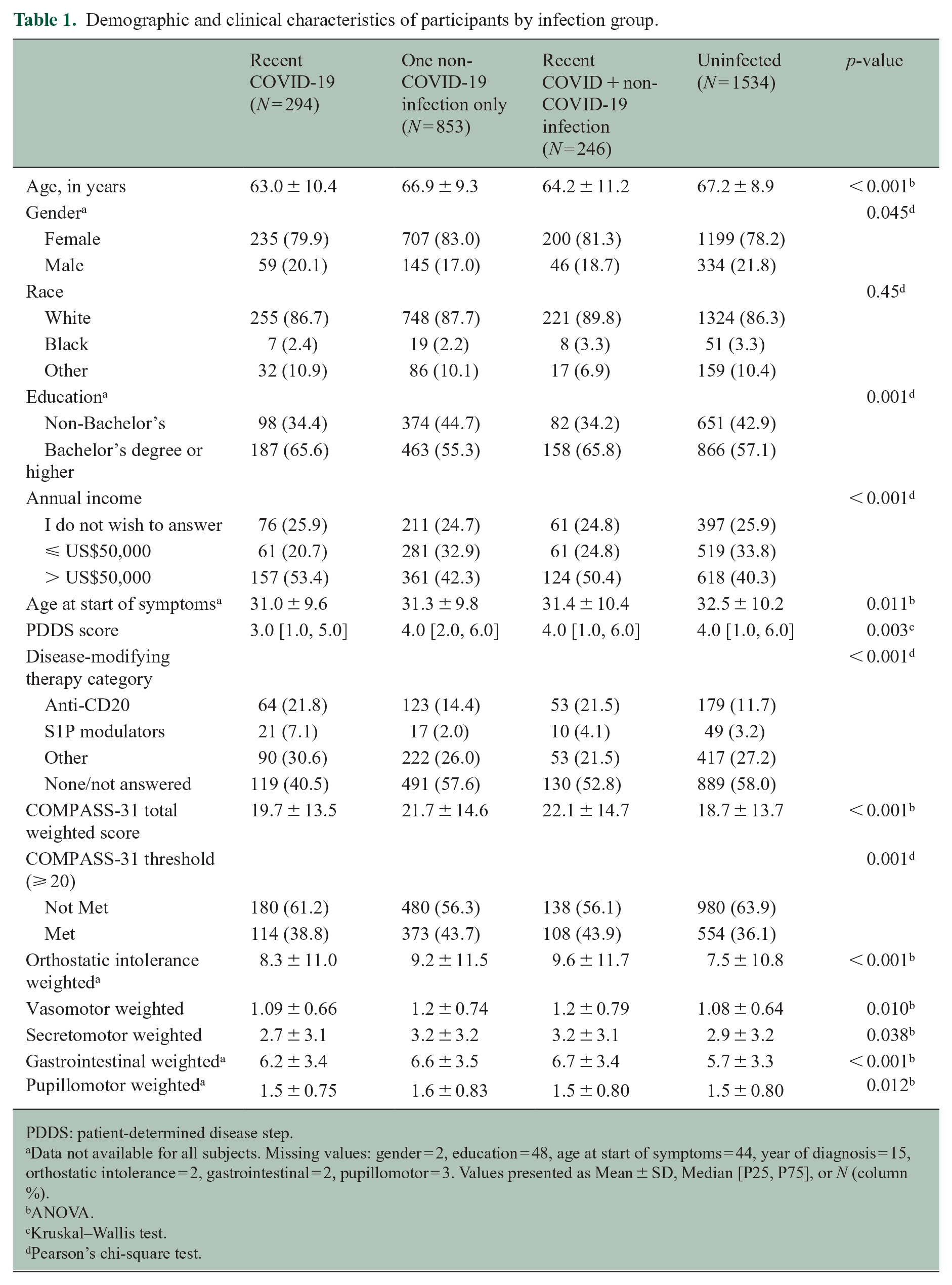
PDDS: patient-determined disease step.
Data not available for all subjects. Missing values: gender = 2, education = 48, age at start of symptoms = 44, year of diagnosis = 15, orthostatic intolerance = 2, gastrointestinal = 2, pupillomotor = 3. Values presented as Mean ± SD, Median [P25, P75], or N (column %).
ANOVA.
Kruskal–Wallis test.
Pearson’s chi-square test.
COMPASS-31 scores with recent infection
COMPASS-31 total scores showed some variation across groups (see Table 1, p < 0.001). Participants having a recent COVID-19 and non-COVID infection had higher mean total and domain scores. Differences in the proportion with symptoms suggesting moderate to severe autonomic dysfunction between groups were observed (p = 0.001) where participants with a recent COVID-19 and non-COVID-19 infection (43.9%) and participants with one non-COVID-19 infection (43.7%) were more likely to have symptoms suggesting moderate to severe autonomic dysfunction than those with only COVID-19 (38.8%) or with no infections (36.1%). After adjusting for covariates, a higher odds of symptoms suggesting moderate to severe autonomic dysfunction was observed for those having a recent COVID-19 and non-COVID-19 infection (aOR [95% CI]: 1.36 [1.028, –1.796]) and those with one non-COVID-19 infection (1.39 [1.165, –1.650]) compared to the uninfected group.
Symptoms associated with recent infection
Uninfected participants reported currently experiencing fatigue, bladder problems, weakness, pain, and vision problems. In comparison, those with a recent COVID-19 infection most frequently reported symptoms experienced at the time of infection were fatigue, fever, cough, malaise, and pain. For those with a recent non-COVID-19 infection, the most frequently reported symptoms participants reported were fatigue, fever, weakness, pain, and bladder problems. In those with a recent COVID-19 and non-COVID-19 infection, the most frequently reported symptoms participants experienced around their infections were fatigue, fever, cough, malaise, pain, and weakness.
Participants with a recent COVID-19 infection reported currently experiencing at least a two-fold increase in fever, cough, loss of taste/smell, and SOB compared to the uninfected cohort after adjusting for covariates (see Figure 1). Participants with a recent non-COVID-19 infection reported currently experiencing about a four-fold increase in fever and cough compared to the uninfected cohort after adjusting for covariates (see Figure 2). Those participants who recently had a COVID-19 and one non-COVID infection reported at least a two-fold increase in rate of cough, fever, loss of smell/taste, SOB, malaise, and fatigue compared to the uninfected group after adjusting for covariates (see Figure 3).
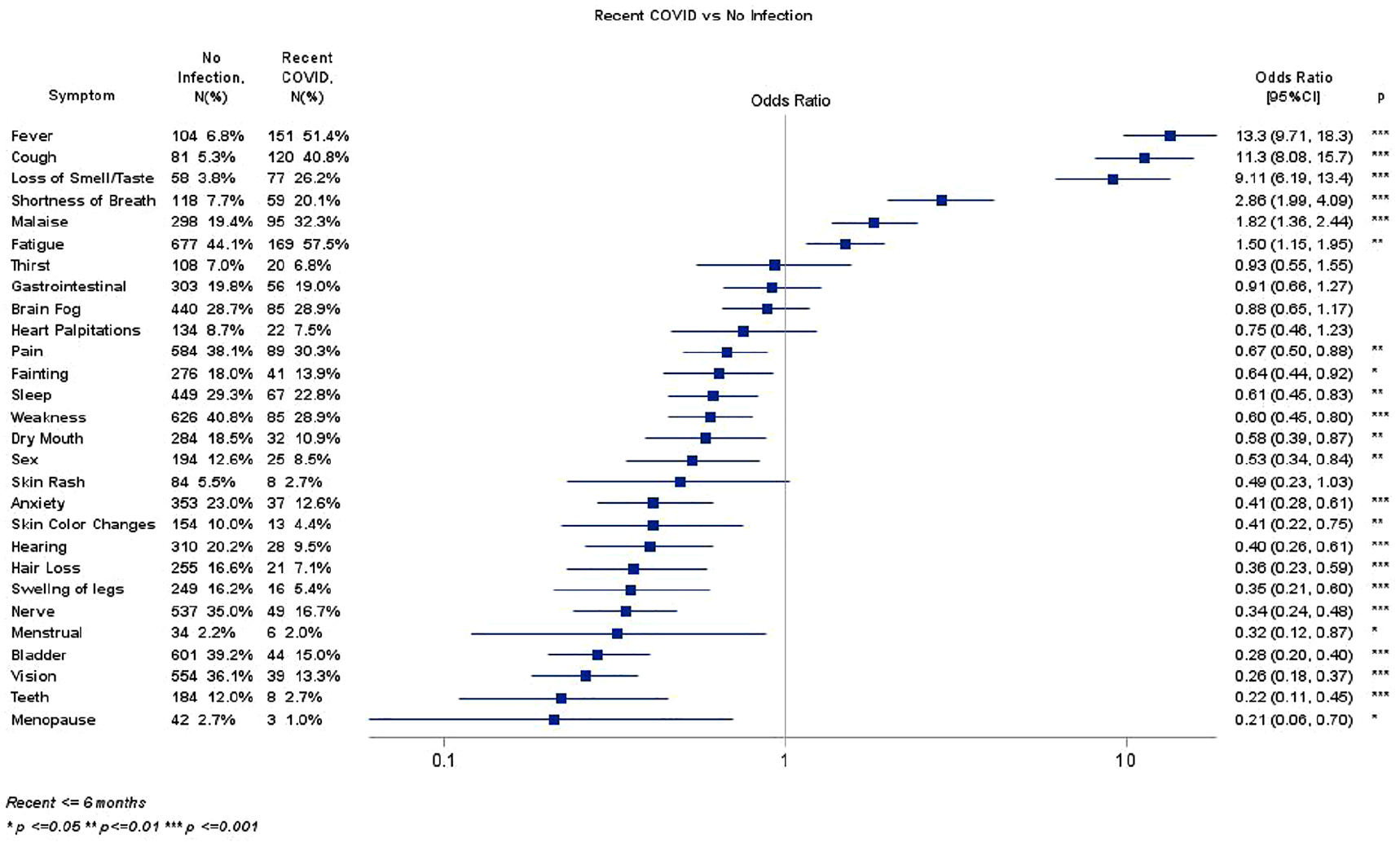
Recent COVID versus no infection.
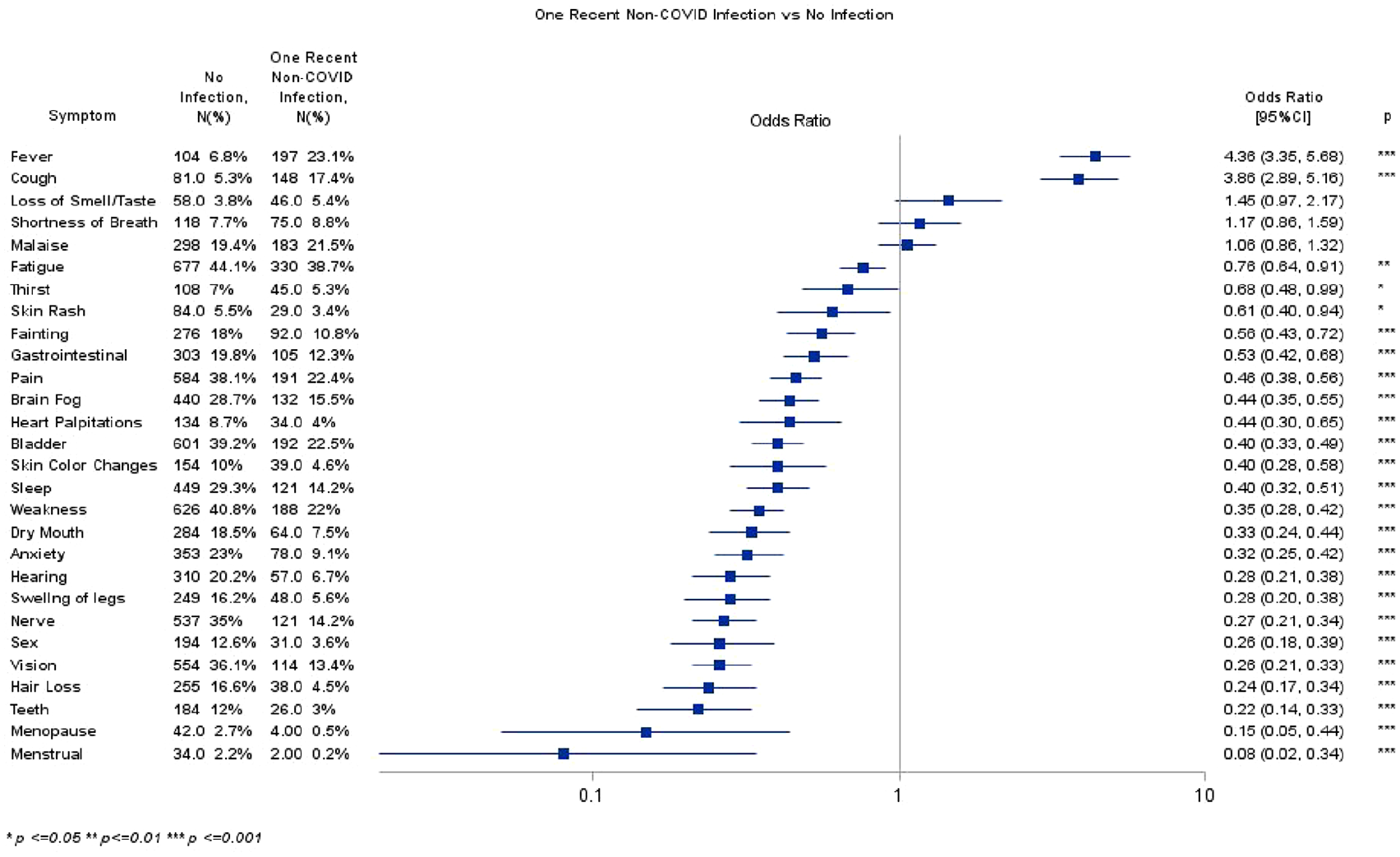
Non-COVID infection versus no infection.
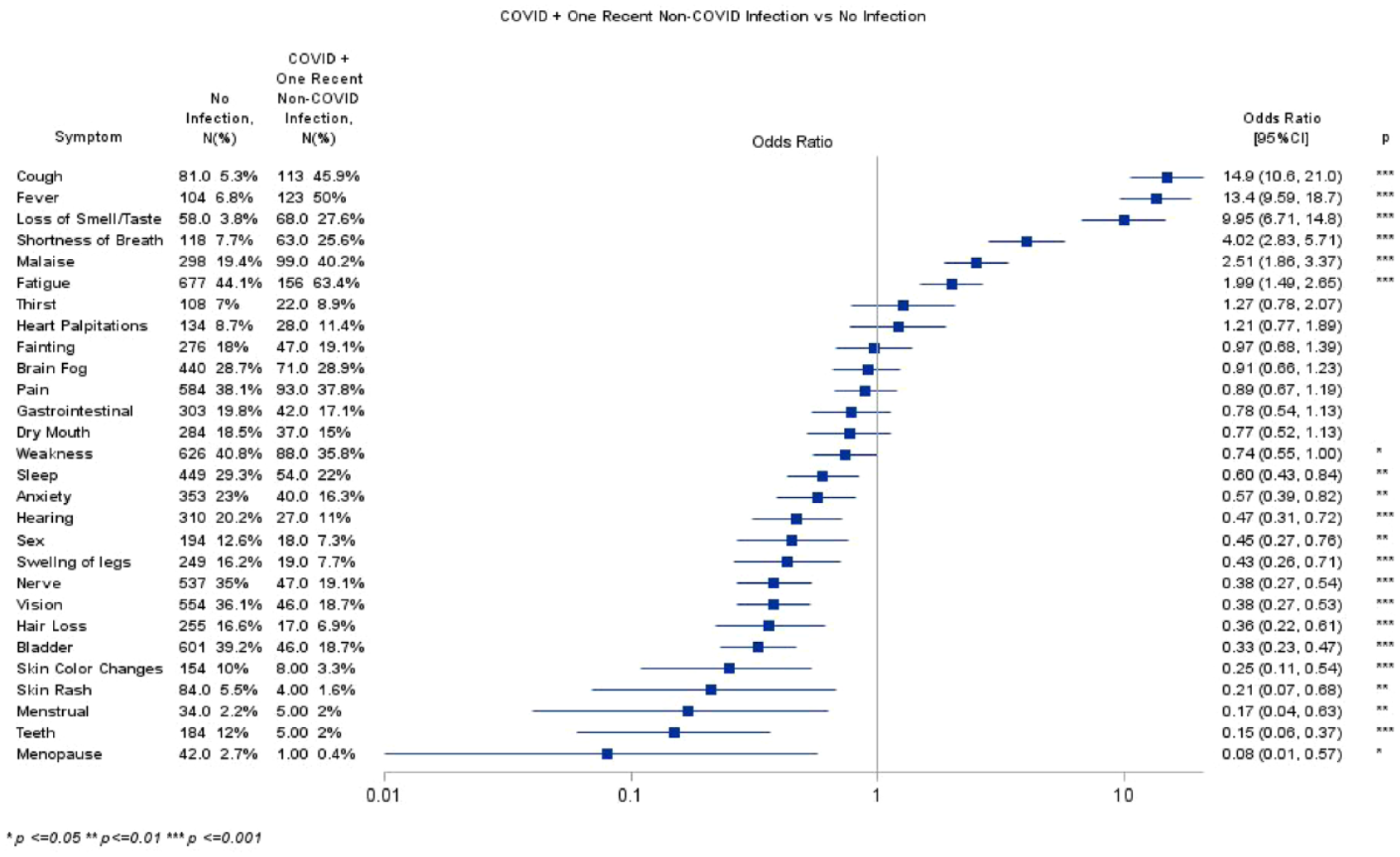
Recent COVID-19 and non-COVID-19 infection versus no infection.
Post-acute sequela of COVID-19 infection
Based on current symptoms reported in 1906 participants who had COVID-19 > 3 months prior, 303 (15.9%) met the definition for PASC. In comparison, 264 (17.2%) uninfected participants met the definition for PASC based on current symptoms (p = 0.30, see Figure 4). When considering other infection groups, a similar percentage was observed for those with both COVID-19 and non-COVID infections (16.3%) while lower percentages were seen in those with one non-COVID-19 infection (9.4%, p < 0.0001).
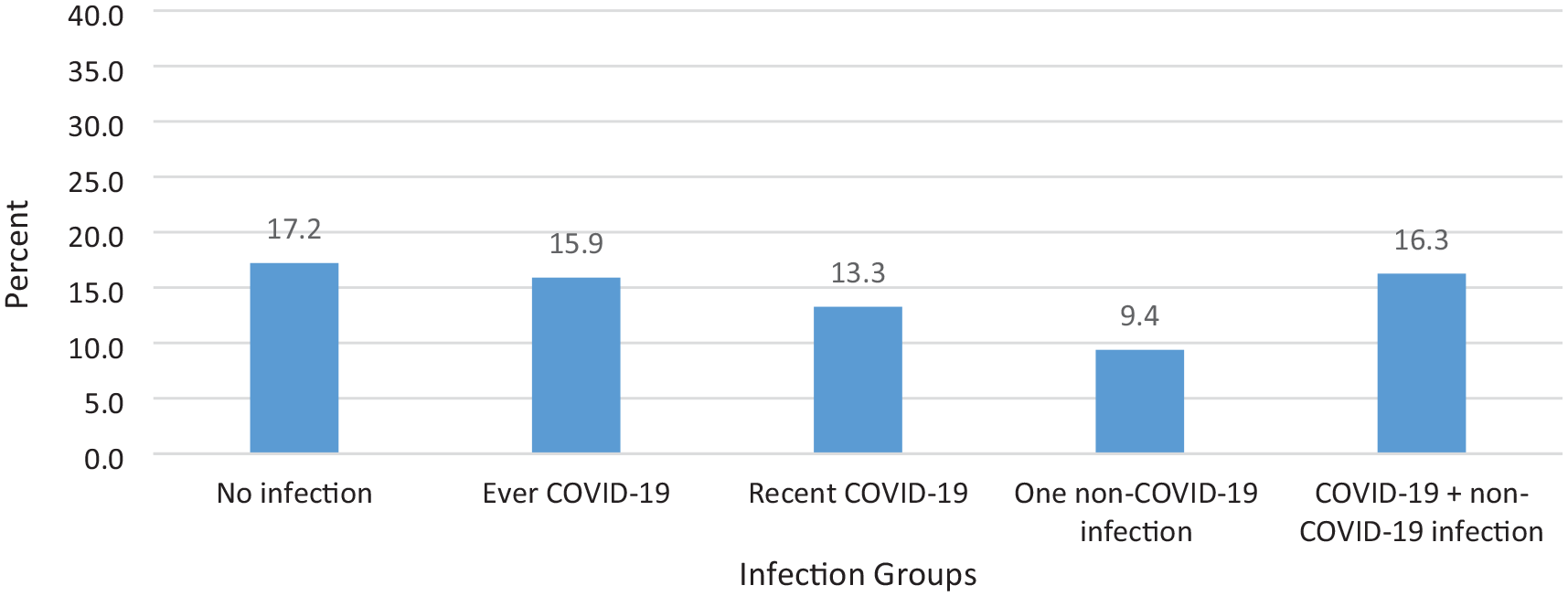
Percentage of participants in each infection group meeting the definition of PASC based on the RECOVER score.
New persistent symptoms following COVID-19 infection
Participants with COVID-19 > 3 months prior were evaluated for new persistent symptoms. Most participants did not have new persistent symptoms (84.5%), while 7.0% had one new persistent symptom, 3.4% had two and 1.8% had three or more new persistent symptoms. Most frequent new persistent symptoms reported by these participants were fatigue (3.7%), brain fog (3.6%), weakness (2.5%), malaise (2.7%), and fainting (2.3%; see Supplemental eFigure 2 and eTable 4). Restricting the RECOVER PASC score to only new persistent symptoms, 29 (1.5%) participants ever having a COVID-19 infection (among those having COVID-19 > 3 months prior) met the definition of PASC while 2 (0.23%) participants with one non-COVID infection and 8 (3.25%) with a COVID-19 and non-COVID infection (p = 0.0004).
Discussion
Our study found that both COVID-19 and non-COVID infection groups reported at least a two-fold increase in multiple symptoms such as fever, cough, loss of smell/taste, and SOB compared to those in the uninfected group. We identified a low prevalence of new persistent symptoms following an infection in persons with MS across all groups. Importantly, we evaluated the performance of the current scoring system for identifying PASC in the general population and found a similar proportion of participants currently experiencing symptoms consistent with PASC in those uninfected with COVID-19 compared to those with COVID-19. Restricting the PASC score to new persistent symptoms, PASC was identified in 1.5% of participants following COVID-19 infection and 0.23% of participants following non-COVID-19 infection. Together, these results suggest a general post-acute infectious effect in persons with MS may occur following infection, albeit at low prevalence, when considering persistent symptoms following infection.
Compared to the uninfected group, symptoms more commonly reported at the time of infection were fever, cough, and loss of smell/taste. Participants reported fatigue at a higher frequency compared to those uninfected participants in those with COVID-19 infection. The prevalence of fatigue was high even in our uninfected cohort, but this is expected given how common fatigue is reported in MS. Symptoms suggestive of moderate to severe autonomic dysfunction was prevalent in at least one-third of participants including those in the uninfected group, and participants with more than one infection had higher COMPASS-31 scores. COVID-19 was not associated with more symptoms of autonomic dysfunction compared to other infections. However, participants classified as having PASC based on new persistent symptoms were more likely to report symptoms of autonomic dysfunction, similar to what has also been reported in the general population where 66% with PASC had symptoms suggestive of moderate to severe autonomic dysfunction. 16
PASC is a concept defined by a collection of symptoms, which are quite common in people with MS.4,17,18 We found that 15.9% of MS patients with recent COVID-19 infection met the RECOVER criteria for PASC, but 17.2% of without recent COVID-19 infection also met this criteria for PASC. Perhaps because symptoms of PASC are so common in people with MS, when using persistent new symptoms in defining PASC, the proportion reporting PASC was only 1.5%, which is much lower than the general population. 4 These symptoms are also common in other conditions such as systemic autoimmune rheumatic disease and solid organ transplant recipients and together suggest the need for additional studies to identify potential objective biomarkers of PASC.19,20 Persistent new symptoms of weakness and cognitive dysfunction were also observed in a retrospective cohort study of persons with traumatic brain injury, MS, and spinal cord injury. 21 Fatigue was not identified as a symptom experienced at higher frequency compared to matched controls, 21 although Bsteh et al. and our study found fatigue to be a persistent symptom 6 months after COVID-19. 22 We also observed a higher burden of symptoms of autonomic dysfunction in participants meeting the definition of PASC in our study, although both PASC and MS are associated with these symptoms.6,16 Altogether, we illustrate the complexity of assessing PASC in MS and the similar rate of PASC suggests that the criteria for PASC—and perhaps the concept of PASC itself—needs further consideration in people with MS.
The development of post-acute infectious effect is not unique to SARS-CoV-2. Several other viral and non-viral infections have been associated with unexplained syndromes post-infection. 5 While there is a limited understanding of the mechanism by which these post-acute infection conditions occur, the symptom profiles overlap in their clinical features and are often characterized by symptoms such as fatigue, neurocognitive and sensory impairment, flu-like symptoms, myalgia/arthralgia, and other nonspecific symptoms that are often present but at variable levels of severity. With other infections, these post-acute infections may have been more difficult to detect; however, the concentration of infections during the COVID-19 pandemic may have provided an ability to better identify and study these post-acute infection events. In MS, the overlap in symptoms of post-acute infection with those commonly experienced in persons with MS complicates identifying these post-acute infectious effects, not only for COVID-19 but also for other infections in which persons with MS are more susceptible. 7
We recognize this study has several limitations. NARCOMS Registry participants voluntarily participate in the registry and may differ from the general US MS population with respect to characteristics such as age and gender. However, the 2010 peak age prevalence of MS in the United States was 55–64 years of age and female to male prevalence ratio was 2.8:1. 23 In addition, respondents with other infection categories were not reported in the analysis. Uninfected participants may have had prior asymptomatic SARS-CoV-2 infections not detected due to variations in antibody production and persistence, weakening the discriminant characteristics of the differences in symptoms. Infections and clinical status were self-reported without objective confirmation. In addition, symptom severity was not considered, but severity is not part of the PASC definition, and many symptoms of interest are difficult or impossible to assess with performance-based measures or by clinicians. In MS, information on MS clinical course and incorporating objective findings may improve defining PASC in MS.
Conclusion
Our study highlights the challenges of examining post-acute infection syndromes in MS. The proportion of participants meeting PASC criteria based on current symptoms was similar regardless of infection status, including those with no infection. Symptoms associated with PASC are nonspecific and overlap with many MS symptoms. When considering new persistent symptoms after an infection, PASC was observed in participants although at a low prevalence. With the increased infection risk in persons with MS and potential for a range of infections to cause post-acute infection syndromes, improved methods and further investigation of these events are warranted.
Supplemental Material
sj-docx-1-msj-10.1177_13524585241310104 – Supplemental material for Post-acute sequela of COVID-19 infection in individuals with multiple sclerosis
Supplemental material, sj-docx-1-msj-10.1177_13524585241310104 for Post-acute sequela of COVID-19 infection in individuals with multiple sclerosis by Amber Salter, Samantha Lancia, Gary R Cutter, Robert J Fox and Ruth Ann Marrie in Multiple Sclerosis Journal
Footnotes
Acknowledgements
NARCOMS is a project of the Consortium of Multiple Sclerosis Centers (CMSC) and the Foundation of the CMSC.
Data availability
Individual participant data that underlie the results reported in this article, after de-identification will be made available upon request. The study survey, data dictionary, and analytic code will also be made available. Data will be available beginning 3 months and ending 5 years following article publication. Proposals should be directed to
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Amber Salter receives research funding from Multiple Sclerosis Society of Canada, National Multiple Sclerosis Society, CMSC and the Department of Defense Congressionally Directed Medical Research Program and is a member of editorial board for Neurology. She serves as a consultant for Gryphon Bio, LLC, Sora Neuroscience and Abata Therapeutics. She has equity in Owl Therapeutics. She is a member of the Data and Safety Monitoring Board for Premature Infants Receiving Milking or Delayed Cord Clamping (PREMOD2), Central Vein Sign: A Diagnostic Biomarker in Multiple Sclerosis (CAVS-MS), Video Telehealth Pulmonary Rehabilitation to Reduce Hospital Readmission in Chronic Obstructive Pulmonary Disease (Tele-COPD) and Methotrexate treatment of Arthritis caused by Chikungunya virus (MARCH). She is supported (in part) by a Biostatistics/Informatics Junior Faculty Award (BI-2105-37656) from the National Multiple Sclerosis Society. She holds the Kenney Marie Dixon-Pickens Distinguished Professorship in Multiple Sclerosis Research. Samantha Lancia has nothing to disclose. Gary Cutter serves on Data and Safety Monitoring Boards for Applied Therapeutics, AI therapeutics, AMO Pharma, Astra-Zeneca, Avexis Pharmaceuticals, Biolinerx, Brainstorm Cell Therapeutics, Bristol Meyers Squibb/Celgene, CSL Behring, Galmed Pharmaceuticals, Green Valley Pharma, Horizon Pharmaceuticals, Immunic, Karuna Therapeutics, Mapi Pharmaceuticals LTD, Merck, Mitsubishi Tanabe Pharma Holdings, Opko Biologics, Prothena Biosciences, Novartis, Regeneron, Sanofi-Aventis, Reata Pharmaceuticals, Teva Pharmaceuticals, NHLBI (Protocol Review Committee), University of Texas Southwestern, University of Pennsylvania, Visioneering Technologies, Inc., Consulting or Advisory Boards for Alexion, Antisense Therapeutics, Biogen, Clinical Trial Solutions LLC, Entelexo Biotherapeutics, Inc., Genzyme, Genentech, GW Pharmaceuticals, Immunic, Immunosis Pty Ltd, Klein-Buendel Incorporated, Merck/Serono, Novartis, Perception Neurosciences, Protalix Biotherapeutics, Regeneron, Roche, SAB Biotherapeutics. He is employed by the University of Alabama at Birmingham and President of Pythagoras, Inc. a private consulting company located in Birmingham AL. Robert Fox receives personal consulting fees from AB Science, Biogen, Bristol Myers Squibb, EMD Serono, Genentech, Genzyme, Greenwich Biosciences, Immunic, INmune Bio, Janssen, Lily, Novartis, Sanofi, Siemens, and TG Therapeutics; clinical trial contract and research grant funding from Biogen, Novartis, and Sanofi. Ruth Ann Marrie receives research funding from: CIHR, MS Canada, Crohn’s and Colitis Canada, National Multiple Sclerosis Society, CMSC, the Arthritis Society, Pfizer Foundation and the US Department of Defense, and is a co-investigator on studies receiving funding from Biogen Idec and Roche Canada. She holds the Waugh Family Chair in Multiple Sclerosis and serves on the editorial board of Neurology.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This investigation was supported by a Strategic Initiatives award (SI-2209-40362) from the National Multiple Sclerosis Society. The funder had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; and decision to submit the manuscript for publication.
Ethical approval
At the time of this study, the registry and questionnaire were approved by the Institutional Review Board at UT Southwestern (STU#2021-0381). NARCOMS has a waiver of documentation of consent, so written consent of participants is not required.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
