Abstract
This study evaluated the association between pediatric multiple sclerosis and vaccinations within 5 years before diagnosis using German ambulatory claims data. Children with multiple sclerosis (
Introduction
Pediatric multiple sclerosis (MS) is defined as a disease onset of MS before the age of 18. 1 The etiology of MS is multifactorial, involving genetic predisposition and environmental factors.2 –4 The potential association between childhood vaccination and the subsequent development of MS has been a subject of considerable debate and research.5,6 Given the critical role of vaccinations in preventing many infectious diseases, it is essential to rigorously assess their safety profile concerning autoimmune diseases such as MS.
We performed a population-based retrospective cohort study to investigate whether childhood vaccination could be a risk factor for the onset of pediatric MS, using three control cohorts for comparison.
Methods
We conducted a retrospective case–control study using ambulatory claims data from the Bavarian Association of Statutory Health Insurance Physicians (BASHIP) covering 2005 to 2020. BASHIP’s ambulatory claims data cover all the approximately 11 million members of Bavaria’s statutory health insurance, which accounts for about 85% of the population. It includes diagnoses coded according to the German version of the International Classification of Diseases (10th edition, ICD-10) and information about vaccinations by service records of quarterly reimbursement claims, sex, and age. Data were available for 2005 to 2020. Since vaccines are also administered in combination, we classified them into ten vaccine groups: (1) tick-borne encephalitis (TBE), (2) hepatitis A, (3) hepatitis B, (4) influenza virus, (5) meningococci, (6) MMR and VZV (MMRV), (7) pneumococci, (8)
We defined a cohort of children diagnosed with pediatric MS (
In a sensitivity analysis, we excluded persons with ICD codes of diseases that may reflect symptoms of undiagnosed MS in the 5 years before the diagnosis (Supplementary Table 1).
We conducted another analysis on a subset of patients from the main cohort who were diagnosed with secured ICD G04 (encephalitis, myelitis, encephalomyelitis, or clinically isolated syndrome (CIS)), suspected ICD G35 (multiple sclerosis, not secured), and secured ICD H46 (optic neuritis) (Supplementary Table 1). This analysis, termed the CIS analysis, focused specifically on the 5 years preceding the first neurological symptoms in patients who were later diagnosed with MS.
We present the distribution of any vaccination over the entire 5 years in both absolute and relative frequencies. We used multiple logistic regression models to investigate the association between pediatric MS and vaccinations the 5 years before diagnosis of MS or CIS. These models used MS (yes/no) as a binary outcome contrasting this cohort against each of the other cohorts, using vaccinations administered (yes = at least one/no = never) as an independent variable and further adjustment for sex and age. Results are reported using odd ratios (ORs) and corresponding 95% confidence intervals (CIs). In addition, we used Poisson regression models to compare the frequency of vaccination between the cohorts.
Hypothesis testing was performed at exploratory two-sided 5% significance levels. All analyses were conducted using R 4.4.0.
Results
We found that children with MS had lower vaccination rates (65.3%) compared to those with No AID (68.4%) in the 5 years before diagnosis (Table 1 and Supplementary Tables 1 and 2). Over the 5 years before diagnosis, the age and sex standardized relative frequencies also showed a lower vaccination in children with MS compared to No AID (68.8%), Crohn’s disease (69.0%), and psoriasis (67.4%). The annual age and sex standardized relative frequencies also indicate a lower vaccination in children with MS (Supplementary Figure 1).
Descriptive statistics of the cohorts and vaccinations.
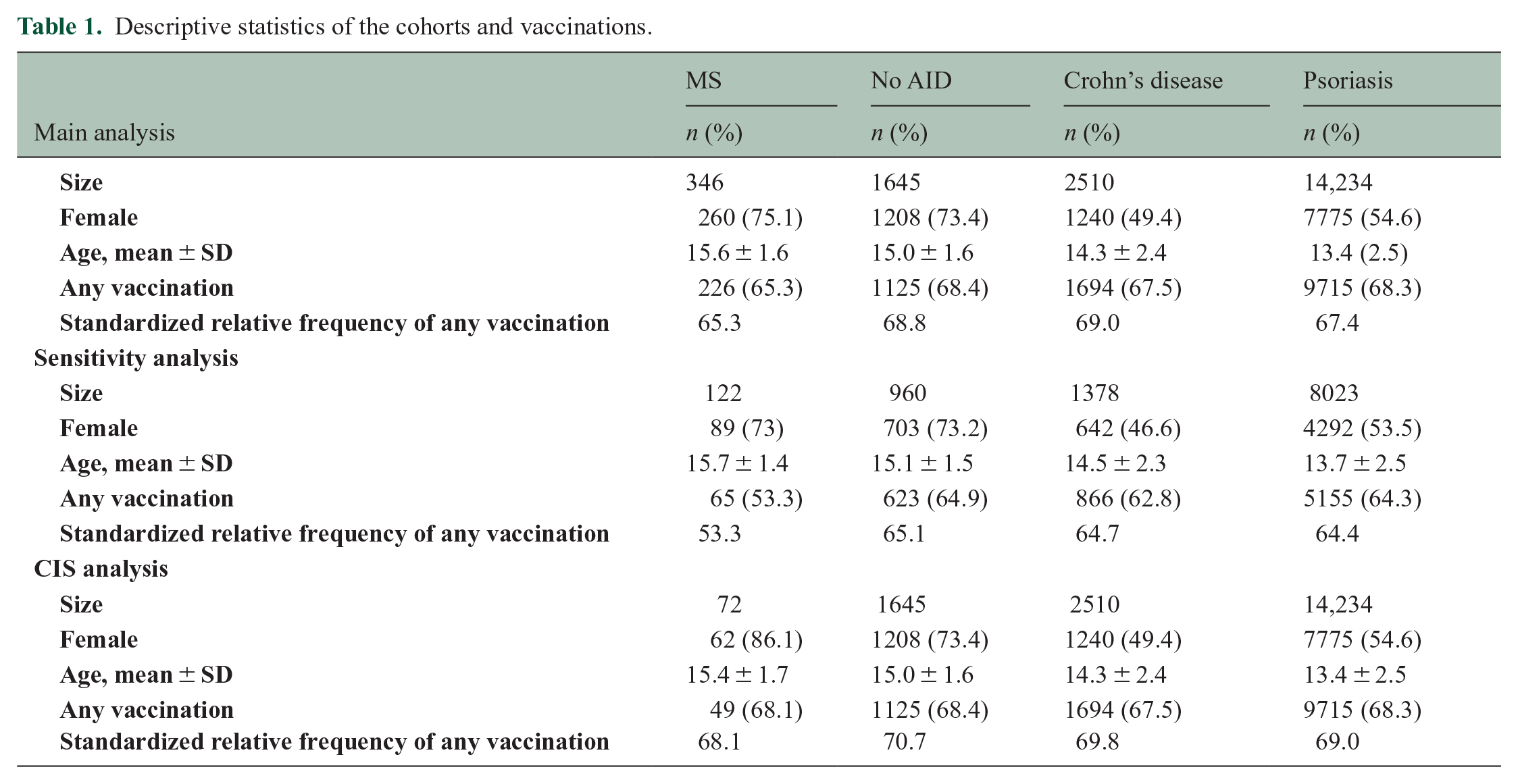
The multiple analysis by logistic regression models revealed negative and non-significant associations between vaccination records within the 5 years preceding diagnosis and the odds of diagnosing pediatric MS. Compared to children with No AID, the OR was 0.88 (
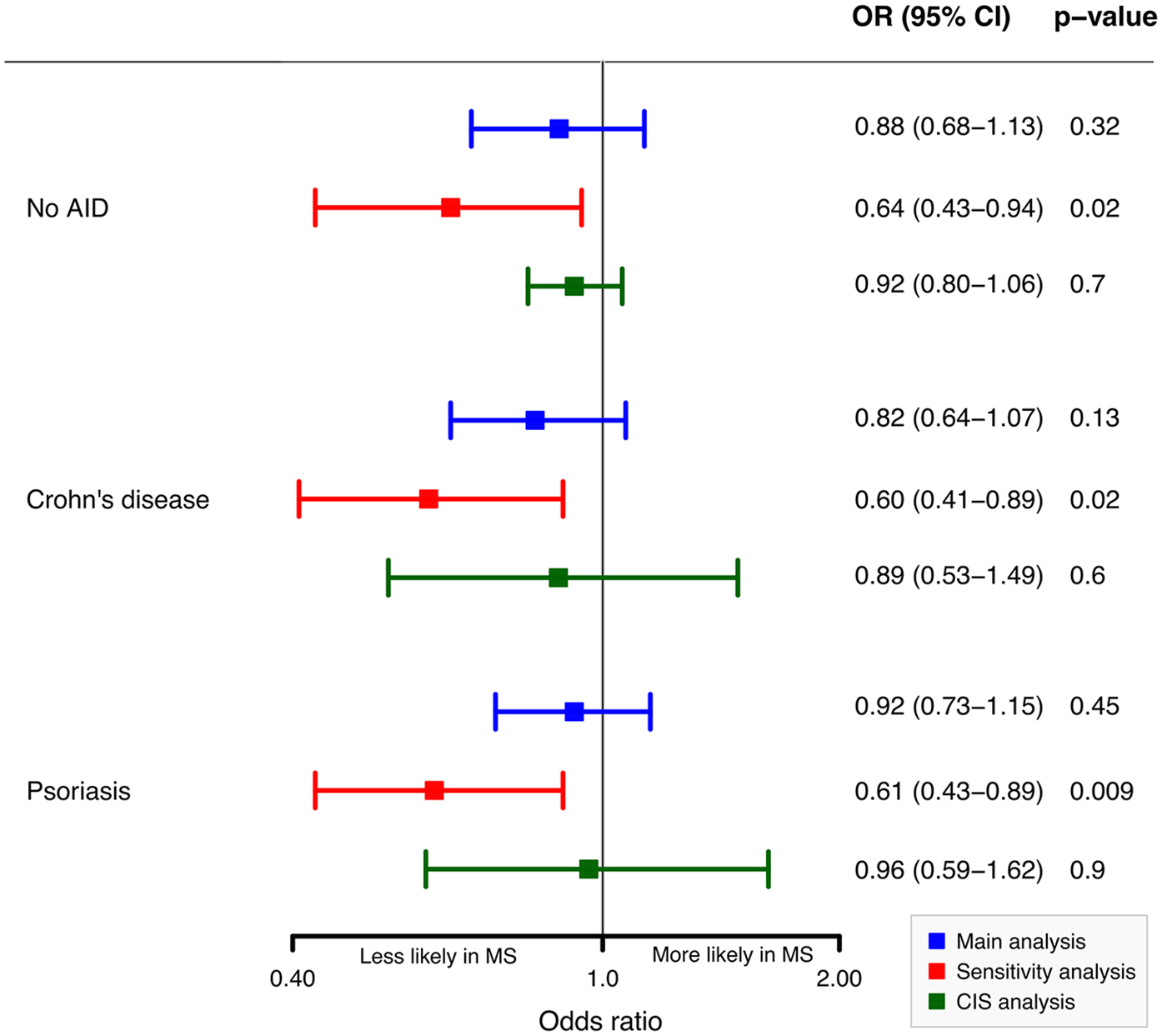
Odd ratios (ORs) of any vaccination for MS in the 5 years before the diagnosis.
The negative associations increased in a sensitivity analysis excluding children with ICD-10 codes that might reflect undiagnosed MS. In the CIS analysis, associations remained consistent (Figure 1). The Poisson regression model shows minor negative associations between MS diagnosis and vaccination counts compared to the No AID cohort (IRR = 0.96, 95% CI = 0.84–1.11), Crohn’s disease cohort (IRR = 0.97, 95% CI = 0.84–1.11), and psoriasis cohort (Incidence Rate Ratios (IRR) = 0.99, 95% CI = 0.86–1.12). Vaccination counts in the MS cohort were slightly lower compared to other cohorts (Supplementary Table 3).
Discussion
In Germany, a comprehensive childhood vaccination schedule starting at 6 weeks of age, covering vaccines for rotavirus, DPTPH (diphtheria, pertussis, tetanus, polio, and
Our findings do not indicate that childhood vaccination in the 5 years before the diagnosis is associated with an increased risk of pediatric MS. This conclusion aligns with previous studies that have found no significant link between vaccinations and the onset of MS or other autoimmune diseases. The results of a recent study from our group indicate that vaccination is also not a risk factor for MS in adults. 7 Similarly, a study in France showed that vaccination against the hepatitis B virus does not increase the risk of pediatric MS. 9
The implications of these findings are substantial for public health. Vaccinations are foundational to public health strategies, effectively preventing the transmission of infectious diseases and their severe outcomes.10,11 The negative and non-significant association observed in our study underscores the importance of maintaining trust in childhood vaccination programs.
This is the first study to cover more than 300 children with pediatric MS, including all vaccinations administered in the 5 years before diagnosis. However, it is crucial to acknowledge the limitations of our study, including potential biases related to coding practices and data completeness inherent in claims data, as well as the retrospective design, which limits causal inferences. Although vaccinations are free of charge all over Germany, we do not have information about the socioeconomic status of participants, which could influence other health-related behaviors or access to healthcare services, potentially confounding the results. In addition, our study only covered the 5 years before diagnosis, which precludes any assumptions on the effect of vaccinations more than 5 years before diagnosis. Future research should prioritize prospective cohort studies and larger population-based studies to validate our findings and further explore this association.
Conclusion
Our study findings do not indicate that vaccination during childhood is associated with an increased risk of pediatric MS in the 5 years preceding diagnosis. Vaccinations remain a critical component of public health strategies, effectively preventing the transmission of infectious diseases and their severe outcomes. These results underscore the importance of maintaining trust in childhood vaccination programs.
Supplemental Material
sj-docx-1-msj-10.1177_13524585241297003 – Supplemental material for Vaccination as a risk factor for pediatric multiple sclerosis: Insights from a retrospective case–control study
Supplemental material, sj-docx-1-msj-10.1177_13524585241297003 for Vaccination as a risk factor for pediatric multiple sclerosis: Insights from a retrospective case–control study by Sonia Darvishi, Ewan Donnachie, Christiane Gasperi, Alexander Hapfelmeier and Bernhard Hemmer in Multiple Sclerosis Journal
Footnotes
Data Availability
For data protection reasons, the authors cannot distribute the underlying data. Interested researchers may contact the corresponding author or the BASHIP to request access.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: B.H. has served on scientific advisory boards for Novartis; he has received research grants from Hoffmann La Roche for multiple sclerosis research. He has received honoraria for counseling (Gerson Lehrmann Group). He holds part of two patents; one for the detection of antibodies against KIR4.1 in a subpopulation of served as a DMSC member for AllergyCare, Sandoz, Polpharma, Biocon, and TG therapeutics; his institution patients with multiple sclerosis and one for genetic determinants of neutralizing antibodies to interferon. None of the conflicts are relevant to the topic of the study.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: B.H. received funding for the study by the European Union’s Horizon 2020 Research and Innovation Program (grants MultipleMS, EU RIA 733161; WISDOM RIA 101137154) and the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) under Germany’s Excellence Strategy within the framework of the Munich Cluster for Systems Neurology (EXC 2145 SyNergy—ID 390857198). He is associated with DIFUTURE (Data Integration for Future Medicine; BMBF 01ZZ1804[A-I]).
Consent for Publication
In our study, we analyzed anonymous claims data held by the BASHIP. Approval by an ethical standards committee on human experimentation (institutional or regional) for any experiments using human participants was not needed according to the Guidelines and Recommendations for Good Practice of Secondary Data Analysis. Approval was obtained from the responsible data protection officer of the BASHIP. Likewise, there was no need for written informed consent from participants.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
