Abstract
Background:
Epstein–Barr virus (EBV) is thought to be a necessary causative agent in the development of multiple sclerosis (MS). Infectious mononucleosis (IM), which occurs up to 70% of adolescents and young adults with primary EBV infection, appears to be a further risk factor but few studies have been highly powered enough to explore this association by time since IM diagnosis.
Objective:
The objective was to quantify the risk of MS in individuals with IM compared with the general population, with particular focus on time since IM diagnosis.
Methods:
In this retrospective cohort study using English national Hospital Episode Statistics from 2003 to 2023, patients with a hospital diagnosis of IM were compared with the general population for MS incidence.
Results:
MS incidence in patients with IM was nearly three times higher than the general population after multivariable adjustment (adjusted hazard ratio = 2.8, 95% confidence interval (CI = 2.3–3.4), driven by strong associations at long time intervals (>5 years) between IM diagnosis and subsequent MS diagnosis.
Conclusion:
While EBV infection may be a prerequisite for MS, the disease process of IM (i.e. the body’s defective immune response to primary EBV infection) seems to be, in addition, implicated over the long term.
Introduction
Multiple sclerosis (MS) is a chronic disease of the central nervous system (CNS) in which the myelin sheath around nerve cells is destroyed. The aetiology of MS is not fully understood, but the disease is known to arise through a complex interplay of inflammatory and auto-immune mechanisms, and occurs more frequently in individuals with genetic pre-disposition. 1 Several environmental risk factors have been implicated, 2 most notably infection with Epstein–Barr virus (EBV), a gamma-herpes virus that infects more than 90% of the general population by adulthood and is virtually ubiquitous among individuals with MS.3-6 The theory that EBV is a causative risk factor for MS was first proposed more than 40 years ago. 7 Since then, strong and consistent evidence has emerged to indicate that the virus is an important, if not necessary, causative factor in the development of MS.4,5 However, EBV infection alone is far from being a sufficient factor. 8 Since 95% of the adult population have been infected with EBV (only a tiny fraction of whom will develop MS), EBV infection essentially forms part of the ‘background risk’ in the general population. The question remains as to why a tiny fraction of individuals with EBV go on to develop MS while most of the individuals with EBV do not.
One important differentiator among those infected with EBV is that the infection can either occur asymptomatically or present as infectious mononucleosis (IM), a disease characterised by fever, sore throat, enlarged lymph nodes and fatigue. 9 About 50% of the population in industrialised nations acquire primary EBV infection at age 1–5 years when the probability of developing symptomatic disease is low. 10 Another large proportion of the population acquires primary infection during adolescence and early adulthood when symptomatic disease occurs in up to 70% of individuals. 11
A previous meta-analysis of cohort and case–control studies found that IM more than doubles the risk of MS (relative risk (RR) = 2.17, 95% confidence interval (CI) = 1.97–2.39; p < 10−54). 12 Such findings are important for understanding the role of EBV in the pathogenesis of MS, since they implicate the clinical disease beyond infection alone. However, of the 18 studies included in this meta-analysis, the majority were case–control studies, often reliant upon questionnaires for ascertainment of prior IM status, and, therefore, susceptible to recall bias. Of the four cohort studies included,13-16 only two reached the threshold of statistical significance and only 129 cases of MS were found in patients with IM. There is still a need for more large-scale cohort studies to better characterise the relationship between IM and MS. In particular, few cohort studies have been highly powered enough to examine age and length of follow-up (i.e. time interval between IM diagnosis and MS diagnosis) on the strength of any association.
To help further characterise the epidemiological relationship between IM and MS, a cohort study was undertaken using English national hospitalisation data covering two decades from 2003 to 2023.
Methods
Data sources and study population
A data extract was derived from the English national Hospital Episode Statistics Admitted Patient Care (HES-APC) database with linkage to national mortality civil registrations (https://digital.nhs.uk/data-and-information/data-tools-and-services/data-services/linked-hes-ons-mortality-data). The linked data were obtained from NHS England (formerly NHS Digital). The data extract used in this study comprised all HES-APC and mortality records belonging to three categories of patients: (1) those admitted to National Health Service (NHS) hospitals between 1 April 2003 and 31 March 2023 with a diagnosis of IM (ICD10 B27.0 or B27.9), hereafter termed the ‘IM cohort’; (2) those admitted during the same time period with one of the diagnoses or procedures listed in the footnote to Supplemental Figure S1 as the primary cause of admission, hereafter termed the ‘reference cohort’; and (3) those who had MS (ICD10 G35) coded on a HES-APC record during the same time period, irrespective of whether they were also included in (1) or (2). In designing the reference cohort, a diverse range of conditions was chosen on the basis that they are common and relatively minor, so that the individuals included would be broadly representative of the general population in terms of their general health status. These conditions/procedures needed to be recorded as the primary diagnosis or operation in the hospital record to avoid selecting individuals who were coming into hospital principally for more serious or uncommon problems. The representativeness of the reference cohort in relation to MS risk was also explored in analysis, as described below. Both cohorts were restricted to individuals aged <25 years to minimise picking up prevalent MS cases at older ages and preserve the temporal relationship with MS incidence.
Study design
The study design was a retrospective cohort study. Each patient’s cohort entry date was the discharge date of their index admission, which for the IM cohort was the patient’s earliest known admission containing a diagnosis of IM, and for the reference cohort was the patient’s earliest known admission for one of the reference cohort conditions. The patients in each cohort were then ‘followed up’ through record linkage for a subsequent hospital diagnosis of MS. The null hypothesis was that the incidence of MS would be the same in both cohorts.
Statistical analysis
The individuals in each cohort were followed from date of cohort entry until the date of first known hospitalisation with MS, death or the end of the follow-up period (31 March 2023), whichever came first. Age–sex-specific annual incidence rates (IRs) of MS hospitalisation in each cohort were calculated per 100,000 person-years (censoring for death). Multivariable Cox proportional hazards regression models were used to estimate the adjusted hazard ratio (aHR) of MS in the IM cohort compared with the reference cohort, with adjustment for age (in 5-year groups), sex, year of index admission, region of residence (nine regions), patients’ index of multiple deprivation (IMD) score in quintiles, ethnicity and follow-up time (<1, 1–4, 5–9 and 10–20 years). Time from cohort entry was used as the underlying time variable. Effect modification by sex, age group and follow-up time was examined using the likelihood ratio test.
To examine how closely the reference cohort resembled the general population of England with respect to MS incidence, we compared the age–sex-specific annual incidence of MS hospitalisation in the reference cohort with the age–sex-specific annual incidence of MS hospitalisation in the whole English population. The latter was calculated by ascertaining the age–sex-specific counts of MS in a single year of national HES (2018 was chosen to represent a recent pre-pandemic year) and dividing these by the corresponding English national age–sex-specific mid-year population denominators for that year (obtained from the national Office for National Statistics), and then multiplying by 100,000. These rates were then compared with the 1-year IRs of MS which were obtained through the follow-up analysis of individuals admitted to the reference cohort in the same year, expressed per 100,000 person-years. The reason for not using the actual general population as a reference population was that the national population denominators were aggregated by age and sex only, whereas the reference cohort of hospitalised patients enabled greater internal validity with multivariable adjustment and flexibility in the analysis of follow-up time.
Results
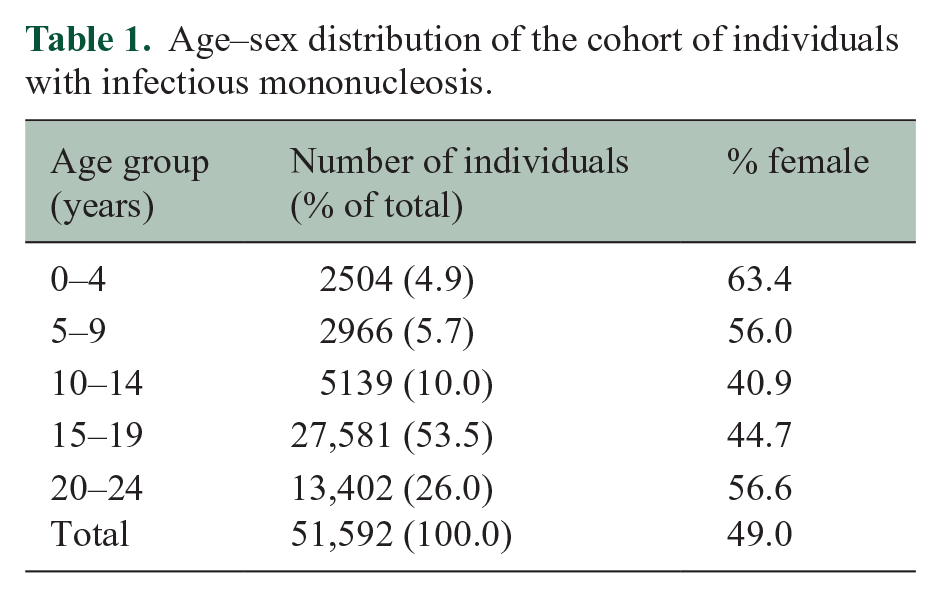
The IM and reference cohorts comprised, respectively, 51,592 and 6,711,441 individuals. Table 1 summarises the age–sex distribution of the IM cohort. The mean age of patients in the IM cohort was 17 years (standard deviation (SD) = 5); their mean follow-up time was 8.7 years (SD = 5.7); and their mean age at MS diagnosis (where it occurred) was 25 years (SD = 7). The number of IM admissions in adolescents and young adults per 100,000 population increased during the study period, save for a substantial dip during the first year of the COVID-19 pandemic (Figure 1).
Age–sex distribution of the cohort of individuals with infectious mononucleosis.
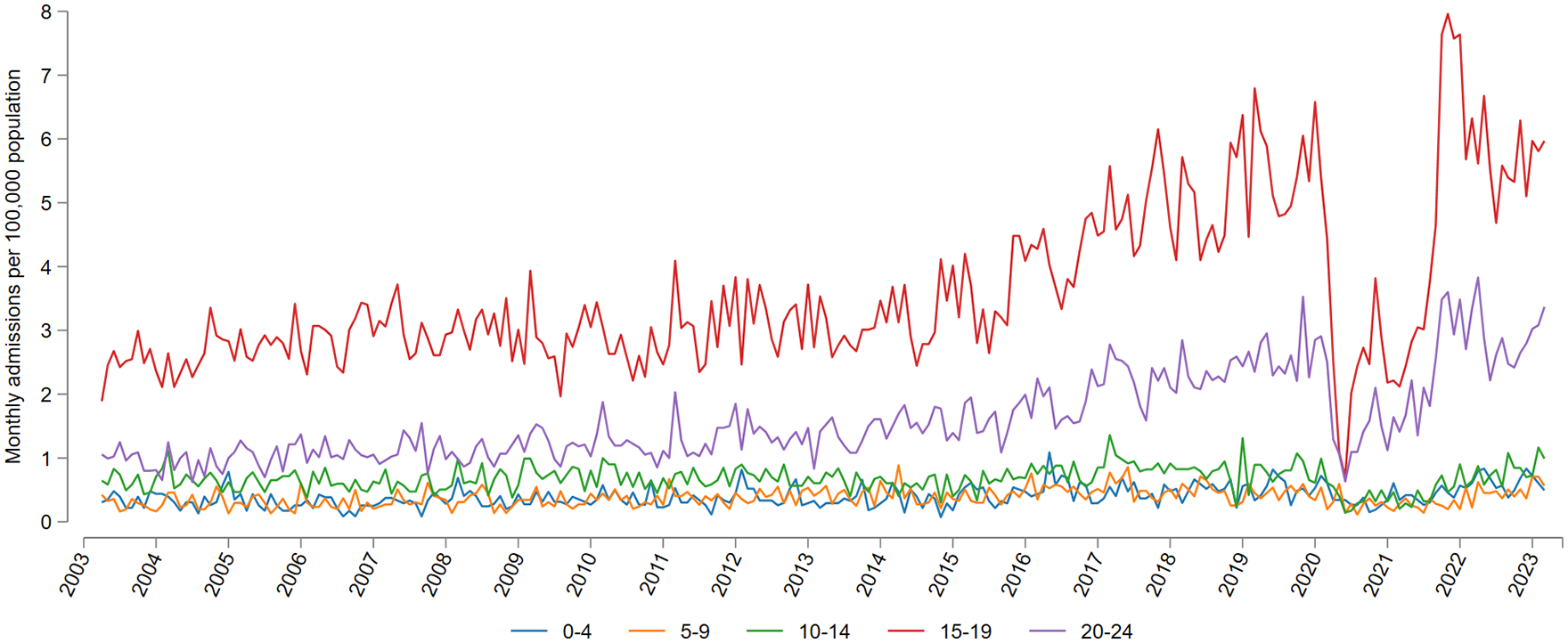
Number of individuals with a hospital diagnosis of infectious mononucleosis in England from January 2000 to December 2022, expressed per 100,000 population by age group.
The age–sex-specific incidence of MS in the reference cohort closely resembled that of the true general population (Supplemental Figure S1). Thus, the reference cohort could be regarded as equivalent to the national general population in terms of risk of MS hospitalisation.
The overall incidence of MS in the IM cohort was 24.4 per 100,000 person-years with a female:male ratio of 2.4 (34.6/14.3) (Figure 2). After multivariable adjustment, the overall incidence of MS in the IM cohort was nearly three times higher than in the reference cohort (aHR = 2.80, 95% CI = 2.31–3.39) (Figure 2). There was little evidence that age at IM diagnosis or sex modified the effect size (likelihood ratio tests for heterogeneity, p = 0.09 and p = 0.94, respectively). There was significant heterogeneity by follow-up period (likelihood ratio test for heterogeneity, p < 0.0001): the aHRs in early follow-up (<1 and 1–4 years) were not significantly elevated, whereas the aHRs in later follow-up (5–9 and 10–20 years) were significantly high (aHR = 4.31 (95% CI = 3.25–5.72) and 3.27 (2.40–4.46), respectively).
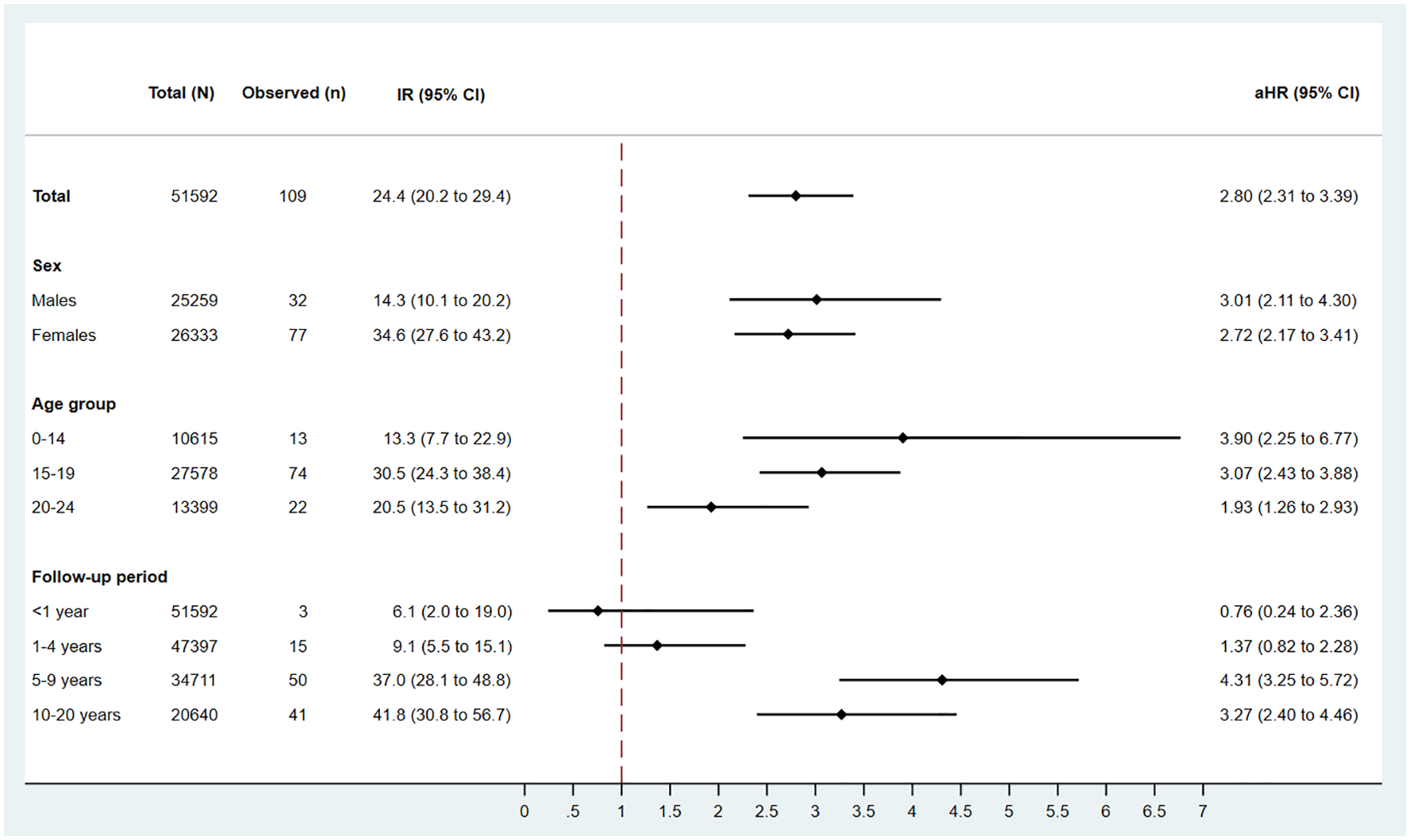
Observed number of multiple sclerosis cases in the cohort of individuals with infectious mononucleosis (n = 51,592), unadjusted incidence rate per 100,000 person-years and adjusted hazard ratios when compared with the reference cohort (n = 6,711,441).
Discussion
This population-based study provides further evidence that individuals diagnosed with IM are at significantly higher risk of developing MS than the general population. Overall incidence of MS in patients with IM was nearly three times higher than the reference cohort after multivariable adjustment (aHR = 2.80, 95% CI = 2.31–3.39). The high aHR overall was driven by strong associations at long time intervals since IM diagnosis.
The findings in this study should not be interpreted as an indication of the strength of the association between EBV infection and MS; the strength of this association has been demonstrated in other studies, which show that MS in EBV serum-negative individuals is virtually non-existent.3,4,17 Instead, these findings lend weight to the existing body of literature on the association between IM (the clinical disease manifestation of EBV) and MS.12,18,19 The comparison here is between individuals hospitalised with IM and the general population (represented by the reference cohort). Since most individuals in the general population have been infected with EBV (up to 90%–95% by the age of 20–24), the reference cohort includes many individuals who have had asymptomatic primary EBV infection, as well as some individuals who have had mildly symptomatic primary EBV infection or IM not requiring hospitalisation. The percentage of all individuals diagnosed with IM that are hospitalised varies greatly from one study to another, ranging from 7% to 85%.20-22 These estimates likely vary due to a number of factors: (1) different study designs, (2) different thresholds of admission, (3) different methods of case-ascertainment for non-hospitalised IM and (4) different case definitions for IM versus mildly symptomatic primary EBV infection.
The overall HR reported in this study is in line with a previous meta-analysis of smaller studies (pooled RR = 2.17, 95% CI = 1.97–2.39; p < 10−54). 12 In more recent studies, the magnitude of the association between IM and MS has been shown to be modified by several other risk factors including EBNA-1 antibody levels, genetics (in particular the HLA-DRB1*15:01 allele), obesity, smoking and vitamin D levels.8,23,24 The reported effect size in any one study will, therefore, be affected by the distribution of these other risk factors in the study population. This study sheds light on another effect modifier: the time interval between IM diagnosis and MS diagnosis. The association between IM and MS was not observed until at least 5 years after IM diagnosis, which suggests that any mechanism of action (if causal, as discussed below) operates over several years. The fact that the association increases with follow-up time also supports ruling out unmeasured confounding or reverse causation as potential drivers of the association. The long lag time between EBV infection and subsequent MS has been described elsewhere, 25 and this is further confirmed by the absolute rates reported in this study which are vanishingly low in the first year after IM diagnosis but increase considerably after 5 years. However, few cohort studies of IM disease and MS have been both long-term and highly powered enough to examine the strength of the association (represented in this study by the aHRs) by age and length of follow-up. One earlier cohort study found that increased risk appeared 10 years or more after mononucleosis occurred and remained increased thereafter, and in the same study, there was some evidence that the association was stronger when IM was diagnosed at younger ages (childhood and adolescence). 15 Among the case–control studies that have stratified the effect size by age at IM diagnosis, the findings have been inconsistent, with some studies reporting that the increase in MS is greater when IM is diagnosed at younger ages, 26 some reporting the opposite27,28 and others reporting no heterogeneity by age.29,30 A more recent study of population registers in Sweden, 18 in which MS outcomes were restricted to those first diagnosed after age 20, found that the multivariable aHRs were significantly high when IM was diagnosed at ages 0–10 or 11–19 years (aHR = 2.87 (95% CI = 1.44–5.74) and 3.19 (2.29–4.46), respectively) but not when IM was diagnosed at ages 20–24 (1.51 (0.82–2.76)); however, when restricting to MS outcomes first diagnosed after age 30, the HR in those aged 20–24 at IM diagnosis increased to 2.60 (1.07–6.36). This suggests that the stronger associations are driven by sufficiently long time intervals rather than by age at IM diagnosis. This concurs with the present cohort study, which found no significant heterogeneity by age at IM diagnosis but substantial heterogeneity by time interval. Since the risk of IM increases substantially in adolescents and young adults compared with young children (Figure 1), the rising hospital incidence of IM in this age group may lead to a corresponding increase in MS cases. Similarly, if an indirect effect of the COVID-19 pandemic and lockdown was the reduced circulation of infectious diseases in children, 31 this may have caused a delay in EBV transmission in younger age groups, which may too have an impact on subsequent MS incidence.
What are the key differences between symptomatic versus asymptomatic primary EBV infection that might help to explain the role of EBV, and to a greater extent IM, in the pathogenesis of MS? Current understanding suggests that IM is caused by overreaction by the immune system following EBV infection. 22 The effect of IM on MS (over and above EBV infection alone) does not seem to be explained by viral load, since EBV DNA loads in asymptomatic patients are elevated to a similar extent as seen in acute IM patients. 32 Past IM and high EBNA-1 antibody levels appear to be independent but synergistic risk factors for MS. 23 The key difference between symptomatic and asymptomatic primary EBV infection seems to lie in the size of the virus-specific CD8+ T-cell response, which is exaggerated in IM. 5 What drives the exaggerated T-cell response in IM patients is unclear, but the unique nature of the association between EBV and MS 33 suggests that the virus-specific CD8+ T-cell response may be an important factor underlying IM’s role in MS.
Strengths of this study are the size of the population, the length of follow-up and the use of a reference cohort that is demonstrably representative of the general population. The number of individuals with both IM and MS in this study (n = 109) nearly outweighs the total number from all previous cohort studies combined that were included in the most recent meta-analysis (n = 129), and, therefore, represents a powerful numeric contribution to the existing literature in this area. The study also adds new weight to the finding of stronger associations at longer time intervals between IM diagnosis and MS diagnosis. Limitations include the fact that only hospitalised cases were included in the cohort of IM patients and for the ascertainment of subsequent MS, the inability to validate the diagnoses of IM and MS in HES against a gold standard (and no such validation studies were found in the literature), the unknown EBV infection status of individuals in the reference cohort, the inability to control for factors affecting general health such as smoking and obesity (which are not reliably recorded on hospital records) and the absence of genetic or serum data. Finally, the direct effect of age at primary EBV infection on MS risk could not be examined since asymptomatic and mildly symptomatic cases of primary EBV infection were not ascertainable in the data set; therefore, notwithstanding the stratified results by age and the importance of age as a confounder, it was not possible to disentangle the effects of age at primary EBV diagnosis and clinical presentation with IM.
In conclusion, this study adds epidemiological evidence to the likelihood that, while EBV infection may be a prerequisite for MS, the disease process of IM (i.e. the body’s defective immune response to primary EBV infection) is, in addition, implicated. The increased RR conferred by IM is only apparent at long time intervals (i.e. 5 years or more after IM), which suggests that there may be a cascade effect in the immune response to EBV infection that persists long after the acute symptomatic phase of IM.
Supplemental Material
sj-tif-1-msj-10.1177_13524585241237707 – Supplemental material for Risk of multiple sclerosis in individuals with infectious mononucleosis: a national population-based cohort study using hospital records in England, 2003–2023
Supplemental material, sj-tif-1-msj-10.1177_13524585241237707 for Risk of multiple sclerosis in individuals with infectious mononucleosis: a national population-based cohort study using hospital records in England, 2003–2023 by Raphael Goldacre in Multiple Sclerosis Journal
Footnotes
Data availability statement
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The author’s salary is supported by the NIHR Oxford Biomedical Research Centre.
Ethical approval
This study involved secondary analysis of de-identified data and did not require ethical committee approval.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
